Abstract
Recurrent spontaneous abortion (RSA) is a multifactorial condition influenced by genetic, hormonal, immunological, and anatomical factors. Thrombophilia, characterized by a heightened propensity for blood clotting, is a significant contributor to RSA. This review examines the mechanisms connecting thrombosis and RSA, focusing on hypercoagulable states, placental thrombosis, inflammation, and endothelial dysfunction. Genetic and acquired thrombophilic factors, such as factor V Leiden mutation, prothrombin gene mutation, protein C and S deficiencies, antithrombin III deficiency, antiphospholipid syndrome, and hyperhomocysteinemia, are discussed in detail. The diagnosis of thrombophilia in RSA entails a comprehensive clinical evaluation, including the assessment of physical examination, medical history and laboratory investigations, although there is still debate over the need for universal screening. Therapeutic strategies, including anticoagulant and antiplatelet therapies, as well as lifestyle modifications, are tailored to individual risk factors and disease severity. Although anticoagulant therapy demonstrates potential in lowering the risk of miscarriage, additional research is necessary to refine treatment protocols and assess long-term outcomes. This review highlights the need for a nuanced approach to managing thrombophilia-associated RSA, balancing diagnostic precision with therapeutic efficacy to improve reproductive outcomes.
Keywords
Introduction
Recurrent spontaneous abortion (RSA), also referred to as recurrent pregnancy loss (RPL) or recurrent miscarriage (RM), is a reproductive condition defined by the occurrence of at least two pregnancy failures prior to the 20th week of gestation.1,2 RSA is categorized into primary and secondary types based on the history of successful deliveries; in primary RSA, women have never had a live-born child. 3 The causes of RSA are multifaceted, encompassing factors such as maternal age, nutritional habits, genetic and anatomical anomalies, endocrine and immune system disturbances, infections, and coagulopathies. Additionally, some studies have indicated that molecular abnormalities in males may also play a role.4,5 Unfortunately, there exists a clear correlation between advancing age and the likelihood of experiencing pregnancy loss. Furthermore, research indicates that the risk escalates with each subsequent miscarriage. 3 Previous investigations have established a relationship between insufficient levels of vitamin D and the occurrence of miscarriage.6,7 The genetic predisposition is shaped by familial history and factors during the first trimester of pregnancy. Additionally, chromosomal and cytogenetic abnormalities, along with single-gene mutations, are recognized as potential causative factors. 3 Aneuploidy, a chromosomal abnormality, is associated with advanced maternal age and is commonly observed in cases of pregnancy loss. 8 Both congenital and acquired abnormalities of the uterus are responsible for about 15% of cases of miscarriage. Among these, polyps of the endometrium are recognized as the most prevalent type of acquired uterine abnormalities.5,8 Dysfunction in various endocrine organs, including the thyroid, pancreas, and ovaries, can result in conditions such as luteal phase defect, polycystic ovary syndrome (PCOS), and either hyperthyroidism or hypothyroidism. These disorders can negatively impact pregnancy outcomes. Prolactin and progesterone are crucial for the successful progression of pregnancy; thus, irregularities in the secretion of these hormones may pose significant risks to gestation. Furthermore, obesity is recognized as a contributing factor to recurrent miscarriages.5,9 It is notable that elevated levels of thyroid-stimulating hormone (TSH) produced by the pituitary gland are involved in 60% of pregnancy losses. 10 Besides, autoimmune or alloimmune responses arising from incompatibilities between the mother and fetus play a crucial role in RPL. In this context, the major histocompatibility complex (MHC) is central to the process. The expression of specific antigens within the HLA system, such as HLA-C, HLA-G, HLA-DR, and HLA-DQ, as well as the function of uterine natural killer (uNK) cells, the release of cytokines, and the production of antinuclear antibodies associated with systemic lupus erythematosus (SLE), have all been implicated in this situation.5,11 Bacterial and viral pathogens are recognized as potential contributors to RSA. However, the precise mechanisms underlying this association remain undetermined. Several studies suggest that endometritis may play a role in the etiology of RSA. It has been observed that preventing dysbiosis of the normal endometrial flora can be beneficial for sustaining pregnancy in certain instances.5,12 An additional contributing factor to miscarriage is the existence of hematologic disorders, especially thrombophilia. Thrombophilia is defined by an elevated tendency for the formation of abnormal blood clots in blood vessels, particularly venous thromboembolism (VTE) being the predominant manifestation of this condition. 13 VTE is mainly caused by the G20210A mutation in prothrombin and the presence of Factor V Leiden (FVL). 14 In addition to certain physiological alterations in the blood vessels that arise from the expansion of the uterus, platelet aggregation—whether stemming from genetic predispositions or acquired factors—may hinder blood flow and lead to complications during pregnancy.15,16 Platelets, minute cellular fragments derived from megakaryocytes, are essential in the coagulation process owing to their surface molecules, such as integrins, selectins, and various receptor types. 17 Following the activation of platelets, phosphatidylserine molecules translocate to the exterior of the platelet membrane. These molecules subsequently interact with coagulation factors IXa and VIIIa, thereby initiating the clotting process. 18 Antiphospholipid syndrome (APS) is defined by the existence of antiphospholipid antibodies associated with thrombosis, pregnancy loss, or related complications. 19 In fact, these antibodies interfere with normal pregnancy, increasing the risk of miscarriage. 20 Pregnant women exhibit an increased susceptibility to thrombotic conditions that promote the formation of clots. This phenomenon is observed more frequently in arteries compared to veins. This imbalance of homeostasis may also result from a deficiency in anticoagulant factors.15,21,22 Despite prior interventions, over 50% of RSA cases remain with an unclear etiology. The majority of these idiopathic instances are believed to be immune-related. Consequently, this category, referred to as unexplained recurrent spontaneous abortion (URSA), necessitates focused attention.2,23,24 According to current estimates, this kind of loss affects 1% to 5% of pregnant women. Furthermore, up to 80% of individuals with RSA endure miscarriage. 2 In light of the extensive research conducted on the pathology of RPL, the essential roles played by components of the clotting and fibrinolytic systems—including platelets, endothelial cells, blood coagulation factors, and plasma proteins—have not been thoroughly explored. Therefore, this review aims to explore the hematological dimensions of thrombosis-related RPL, emphasizing diagnostic approaches, management techniques, and treatment options.
Mechanisms Linking Thrombosis and Recurrent Spontaneous Abortion
Multiple factors contribute to the occurrence of RSA (Figure 1). This discussion delves into the fundamental elements within the field of hematology, with a specific focus on coagulation processes as well as placental and endothelial characteristics.
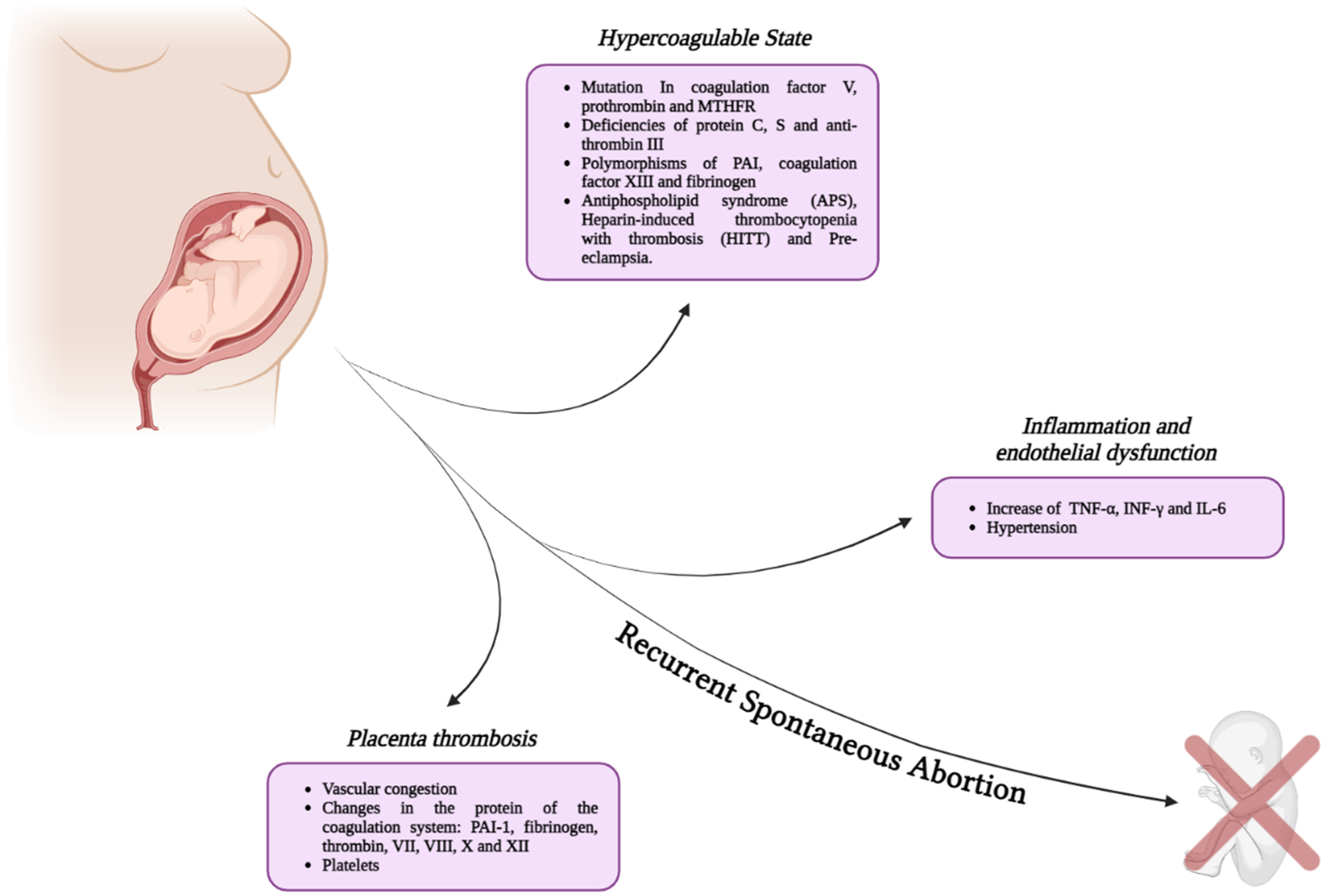
Mechanisms linking thrombosis and recurrent spontaneous abortion. Thrombotic factors, such as hypercoagulable states during pregnancy, placental thrombosis, inflammation, and endothelial dysfunction, contribute to the risk of RSA in pregnant women.
Hypercoagulable States
Hypercoagulopathy can manifest in two primary forms: hereditary and acquired. Hereditary hypercoagulopathy may result from various genetic mutations, with the Factor V Leiden (FVL) and prothrombin gene mutation being the most prevalent among them. The discourse surrounding genetic mutations remains a subject of considerable debate. Certain studies support the notion that MTHFR mutations contribute to thrombosis, whereas others dispute any association between this mutation and conditions such as thrombophilia or pregnancy loss. Similarly, in examining deficiencies of antithrombin III (AT III), proteins C and S, as well as various polymorphisms like plasminogen activator inhibitor (PAI), factor XIII, and β-fibrinogen, some research indicates a potential link to RPL. However, the majority of studies fail to establish a significant correlation.25–29
Activated protein C serves as an anticoagulant by cleaving factor V, thus impeding its activation. However, a mutation known as G1691A, which occurs in factor V Leiden, provides resistance to the action of protein C, resulting in a hypercoagulable state. 30 Furthermore, the G20210A mutation in the prothrombin gene is associated with RPL due to its role in increasing prothrombin production. 31 While the validity of information regarding MTHFR remains subject to scrutiny, certain variants have been identified that lead to diminished enzyme activation. This decrease can result in hyperhomocysteinemia, a condition associated with a hypercoagulable state in patients.31–33 Any condition that reduces the efficacy or levels of anticoagulant proteins can result in a hypercoagulable state. Proteins C and S exemplify factors that are affected by vitamin K levels. There is a greater body of evidence indicating a deficiency in protein S compared to protein C.34–36 Additionally, a particular nucleotide repeat consisting of guanine has the potential to elevate PAI-1 levels, resulting in a reduction of fibrinolytic activity and subsequent coagulation processes. 31
Nowadays, acquired types are receiving increased attention. Various factors contribute to these complications, including infections, trauma, malignancies, and pharmaceutical agents. The inflammatory responses associated with these conditions are characterized by the expression of surface adhesion molecules, which may initiate a hypercoagulation process.37,38 APS and heparin-induced thrombocytopenia and thrombosis (HITT) are key conditions in this category, while there is a well-documented association between APS and pregnancy complications.37–39 In addition, preeclampsia is mainly associated with endothelium or placenta dysfunctions and is a major risk factor for abortion, particularly in thrombophilia cases.40,41
Interestingly, coagulation proteases such as thrombin, activated protein C (APC), factor Xa (FXa), and factor VIIa (FVIIa) play crucial roles in the release and function of extracellular vesicles (EVs), particularly in the context of thrombosis during pregnancy. Thrombin, a key protease, activates protease-activated receptors (PARs) on various cell types (particularly PAR-1 and PAR-4), including endothelial cells, placental cells and platelets, leading to the release of EVs that carry pro-coagulant factors and inflammatory mediators. PAR-1 activation by thrombin on endothelial cells involving ROCK-II signaling and platelets contributes to platelet aggregation, fibrin deposition, and vasoconstriction, all of which are hallmarks of thrombosis.42,43 During pregnancy, the hypercoagulable state is also marked by elevated coagulation factors and decreased anticoagulant activity, potentially leading to complications like preeclampsia and intrauterine growth restriction (IUGR). 44 In this setting, excessive thrombin generation and PAR activation can promote placental thrombosis, impairing blood flow to the fetus. 45 EVs released from activated platelets and placental cells can amplify coagulation and inflammation, contributing to these adverse outcomes. 46 In summary, the interplay between coagulation proteases and EVs is also critical in the pathophysiology of pregnancy-related thrombosis and associated complications.
Placental Thrombosis
Blood, along with its constituents like oxygen and essential nutrients, creates an optimal environment for the development of fetal tissues. Nonetheless, the formation of clots is acknowledged as a possible factor that may disrupt this developmental process. 40 Several pregnancy-related factors contribute to thrombosis, including vascular congestion, which disrupts blood flow in maternal veins, and changes in coagulation and fibrinolytic proteins that heighten the risk of miscarriage. Plasminogen activator inhibitor-1 (PAI-1), along with fibrinogen, thrombin, and various coagulation factors such as VII, VIII, X, and XII, are recognized for exhibiting elevated levels during pregnancy. During the hypercoagulable state of pregnancy, platelets additionally undergo both morphological and quantitative changes. A decrease in platelet count is an early gestational response linked to elevated levels of von Willebrand factor. Platelets are essential for forming placental vasculature, and impaired regulation of platelet activation can lead to serious conditions such as HELLP syndrome.41,47–49
Inflammation and Endothelial Dysfunction
Inflammation is a protective mechanism of the innate immune system and is vital for maintaining pregnancy. However, disturbances in the immune system can hinder implantation. Uncontrolled increases in specific cytokines, such as tumor necrosis factor-alpha (TNF-α), interferon-gamma (IFN-γ), and interleukin-6 (IL-6), can disrupt this process by reducing trophoblast invasion and proper implantation.50–52 Inflammatory mediators can indirectly affect the endothelial lining of blood vessels, one of the factors in Virchow's triad. There is growing recognition of the roles of inflammation and the immune system in thrombosis, with thrombo-inflammation conditions arising from excessive activation of both immune and coagulation factors. In this process, platelets serve as key mediators.37,53,54 Endothelial cells, along with platelets and white blood cells, establish an environment that promotes the coagulation process. 53 Studies suggest a possible link between endothelial dysfunction and hypertension as complications during pregnancy. It is proposed that inadequate blood flow to the placenta may lead to increased maternal blood pressure, subsequently initiating an inflammatory response.55,56
Risk Factors for Thrombosis and Miscarriage
The risk of thrombosis needs to be evaluated in conjunction with other causative factors, including cancers, hepatic disorders, and infectious diseases.5,57–59 Identifying genetic factors linked to thrombophilia in pregnant women experiencing recurrent miscarriage could greatly enhance clinical management by facilitating the adoption of personalized treatments, including anticoagulant prophylaxis. These targeted interventions may improve pregnancy outcomes by diminishing the risk of complications such as thrombosis, which may affect up to 50% of cases, contingent upon the study population.60,61 Figure 2 illustrates the thrombosis-related risk factors in RSA occurrence.

Risk factors related to thrombosis and miscarriage. Multiple inherited or acquired factors, encompassing maternal, genetic, and environmental factors, are believed to significantly influence the occurrence of RSA.
Maternal Factors
In many cases, women may not fully understand the underlying causes of miscarriage and its recurrence. The condition is often perceived as a relatively minor physical health issue, with few immediate health consequences. However, the emotional and psychological impact of experiencing a miscarriage is far more profound and lasting than the physical symptoms. 62 Furthermore, in many societies, women's status is often evaluated through the lens of their ability to conceive and bear children, with cultural and traditional beliefs playing a significant role in this assessment. 63 Considering the impact of various recognized risk factors for miscarriage, 30% to 50% of cases occur due to an unidentified etiology. However, prior obstetrical history, chromosomal abnormalities, reproductive tract abnormalities, immune dysfunction, genital tract infections, and cervical insufficiency are among the documented risk factors for RPL. The risk is multifaceted, and although certain risk factors are identified, it remains challenging to determine definitive predictors of future pregnancy loss. 64 Research indicates that inflammation is a significant factor contributing to RSA. Key lifestyle factors, including smoking, obesity, and an imbalance in immune cell function, are closely associated with an increased risk of pregnancy failure. 65
Maternal age correlates with an increased probability of miscarriage, attributed to diminished egg quality and chromosomal abnormalities. One or both parents may occasionally possess an unpredictable genetic abnormality; however, the children may be severely affected, potentially resulting in miscarriage. Studies demonstrate that the probability of miscarriage escalates in couples if the woman exceeds 35 years of age and the man surpasses 40 years of age. 63 Advanced paternal age is also becoming acknowledged as a risk factor for thrombosis, with aging believed to induce an acquired thrombophilic condition. 66 Research indicates that elevations in fibrinogen, factors VIII and IX, and other coagulation proteins, without a corresponding increase in anticoagulant factors, heighten the likelihood of thrombus formation. 67 This disruption in the coagulation system underscores the necessity for reproductive specialists to recognize the effects of advanced paternal age, both for thrombophilic disorders and reproductive results. 68 Studies have shown that paternal age correlates with genetic abnormalities, chromosomal aneuploidies, and epigenetic alterations, which might diminish sperm quality and elevate the likelihood of negative pregnancy outcomes. 66 The implications of prolonged paternal age extend to reproductive health, underscoring the necessity for thorough evaluations in clinical practice. 68 Reproductive physicians must prioritize understanding the thrombophilic risks linked to aging and their implications for fertility and offspring health. 67 Promising therapeutic strategies, such as targeted interventions to fix coagulation imbalances and genetic counseling, could reduce these risks. 66 Collaborative research initiatives are crucial for formulating evidence-based guidelines that incorporate thrombophilic and reproductive factors, guaranteeing comprehensive therapy for affected couples. 68
A study conducted in Ireland revealed that recurrent miscarriage is associated with a higher risk of adverse perinatal outcomes in subsequent pregnancies. These outcomes include premature delivery, extremely premature delivery, and perinatal death. 63 S. Jivraj et al discovered that women with a history of recurrent miscarriage exhibit a markedly elevated risk of premature delivery, delivering small for gestational age (SGA) infants, and necessitating cesarean section, in contrast to women without a history of pregnancy loss. The findings indicate that recurrent miscarriage may elevate the risk of delivery complications. 64 The heightened risk of venous thromboembolism (VTE) during pregnancy is affected by various physiological factors, including progesterone-induced venous dilation and uterine compression of the inferior vena cava and iliac veins. Moreover, factors such as age, obesity, comorbidities, cesarean delivery, postpartum infections, and a personal or familial history of VTE significantly increase the risk of VTE during pregnancy and the postpartum phase. 69
APS, the most prevalent form of acquired thrombophilia, is a systemic autoimmune disorder and is marked by pregnancy-related complications that arise in the context of antiphospholipid antibodies.70,71 Recurrent miscarriage has been linked to the presence of these antiphospholipid antibodies, including lupus anticoagulant, anticardiolipin antibodies, anti-β2-glycoprotein I antibodies, and antiphosphatidylserine antibodies. 20 These antibodies trigger an inflammatory reaction in the vascular endothelium, thereby facilitating the development of thrombosis. In vitro studies show that antiphospholipid antibodies (aPLs) activate endothelial cells, increasing the expression of adhesion molecules. Thrombosis in APS is additionally influenced by neutrophils, monocytes, and the complement system. Key mechanisms include the formation of neutrophil extracellular traps (NETs) triggered by aPLs and the heightened production of tissue factor and vascular endothelial growth factor (VEGF) by activated monocytes.5,72 Antibodies can induce thrombosis in multiple organ systems, with venous thrombosis most commonly occurring in the lower limbs and arterial thrombosis primarily affecting the cerebral circulation. Thrombosis can affect any organ, and in rare cases, catastrophic antiphospholipid syndrome (CAPS) may occur, representing a severe complication associated with significantly higher mortality rates. 19 APS accounts for 90% of recurrent miscarriages in the absence of treatment 20 and may not always manifest significant symptoms, and some cases experience nonspecific symptoms similar to those of multiple sclerosis (MS), including fatigue or paresthesia in various body regions. 73
Genetic Factors
In addition to maternal factors, genetic factors are also involved. Factors associated with thrombophilia include factor V Leiden (FVL) mutation, activated protein C resistance (APCR), prothrombin G20210A gene mutation, factor XIII polymorphism, protein C (PC), protein S (PS), and antithrombin III (AT III) deficiency, as mentioned.74,75 However, the low prevalence of detectable thrombophilia in recurrent miscarriage cases and the uncertain benefits of anticoagulation and other therapies have led some studies to argue against routine screening,73,76,77 while others suggest that thrombophilia, among other correlated factors, can be identified and treated to reduce the risk of recurrent miscarriages.78,79 Additionally, several mutations and polymorphisms, including MTHFR A1298C, MTHFR C677 T, β-Fibrinogen 455 G > A, Factor VII R353Q, and FII prothrombin, have been reported as contributing risk factors of pregnancy loss and miscarriage.26,80–84
Factor V Leiden Mutation
The Factor V Leiden mutation, identified as Factor V Leiden 1691 G > A, is a genetic alteration in the factor V gene that could result in a coagulation disorder. 85 Factor V Leiden is a point mutation of factor V that eliminates the cleavage site in factor V and factor Va. An elevated risk of thrombosis is linked to this genetic anomaly, particularly in individuals with homozygous or pseudo-homozygous factor V Leiden mutations. 85 However, numerous studies conducted globally have demonstrated a strong correlation between FVL and RPL. 86 Research indicates that carriers of heterozygous and/or homozygous FVL gene mutations experience significantly higher rates of pregnancy loss compared to individuals without these mutations. 87 This circumstance increases the likelihood of early spontaneous RPL, thereby reinforcing the hypothesis that placental microthrombosis contributes to its pathophysiology. 88 Hence, even in the absence of a thrombosis history and with prior successful pregnancies, evaluation for FVL mutation may be warranted in healthy women experiencing unexplained RPL. 89
Activated Protein C Resistance (APCR)
APCR refers to a hemostatic disorder that leads to an increased risk of VT, encompassing pulmonary embolism and deep vein thrombosis (DVT). This hypercoagulable condition is marked by an absence of response to APC, which typically functions to inhibit excessive blood clotting. 75 APCR could be either hereditary or acquired, and conflicting reports exist regarding the correlation between APCR and RPL. 90 The pathogenesis of RPL entails a complex interplay of factors. Resistance to the anticoagulant effects of activated protein C (APC) is frequently attributable to the factor V gene mutation, which is strongly linked to complications during pregnancy. 91
Prothrombin G20210A Gene Mutation
The prothrombin G20210A gene mutation, the second most common inherited thrombophilia after Factor V Leiden (FVL), is associated with an increased risk of thromboembolic events, such as DVT and pulmonary embolism.92,93 The prothrombin G20210A mutation results in increased plasma thrombin levels and enhances clot formation in heterozygous individuals, who exhibit a twofold elevated risk of thrombosis relative to non-carriers. 74 Besides, this mutation significantly elevates the VT risk, potentially leading to placental thrombosis and increasing the likelihood of miscarriage due to inadequate utero-placental perfusion.93,94 This mutation elevates the likelihood of obstetric complications by 4-fold and increases pregnant women's risk of thrombosis by nearly 10-fold. 95
Protein C and Protein S Deficiency
Protein C (PC) deficiency, whether inherited or acquired, is a significant risk factor for thrombophilia, with symptoms ranging from asymptomatic cases to venous thromboembolism and, in severe instances, neonatal purpura fulminans, a life-threatening condition. PC activation occurs on endothelial cell surfaces and requires two receptors: the endothelial PC receptor and thrombomodulin. Thrombomodulin binding to thrombin enhances PC activation by 3- to 4-fold, while the endothelial PC receptor accelerates activation to activated protein C (APC) by 20-fold. APC then inhibits thrombin generation by inactivating factors V and VIII through limited proteolysis. 96 Protein S (PS) is primarily secreted by endothelial cells, hepatocytes, and Leydig cells. About 2.5% of circulating PS exists within platelet α-granules; platelet PS is derived exclusively from expression within megakaryocytes. Research indicates that during early pregnancy, the PS activity decreases dramatically, with a mean level reported at approximately 46% of non-pregnant values. This decline can complicate the diagnosis of true PS deficiency, which is crucial for maintaining coagulation balance. PS deficiency has been associated with adverse pregnancy outcomes, including thrombosis and complications affecting placental function. 97 PS deficiency may be either hereditary or acquired. Vitamin K deficiency, nephrotic syndrome, or hepatic disease are the most common causes of acquired deficiencies. Hereditary protein S deficiency is an autosomal dominant condition, and thrombosis occurs in both heterozygous and homozygous deficiencies. 58 PC and PS deficiencies require particular attention, especially during pregnancy, due to the physiological hypercoagulable state. This situation is characterized by elevated procoagulants, reduced fibrinolysis, and diminished anticoagulants to sustain placental hemostasis throughout gestation. 98 During normal pregnancy, levels of functional PC and PS typically remain stable. However, a deficiency in antithrombotic factors, such as PS, can lead to hypercoagulability, causing placental thrombosis and hypoperfusion. PS deficiency is more commonly identified than PC deficiency, resulting in a 15-fold heightened RPL risk. 99
Plasminogen Activity Inhibitor 1 (PAI-1) 4G/5G and 4G/4G Genotypes
PAI-1 is a protein that helps clot formation by inhibiting tissue plasminogen activator (TPA). Deletion or duplication of guanine nucleotides (4G and 5G) in the PAI-1 gene promoter affects its production rate by affecting the transcription process. Studies show that PAI-1 levels are higher in people who have two 4G alleles (4G/4G) than in other people (4G/5G or 5G/5G). Therefore, the 4G/4G genotype seems to increase the risk of thrombosis by increasing clot production. 75 Regarding RPL, the 4G/5G and 4G/4G genotypes contribute to elevated PAI-1 expression, thereby inhibiting fibrin lysis and placental thrombosis, resulting in abortion. 100 The presence of the 4G allele in the PAI-1 4G/5G polymorphism has been related to an elevated risk of pregnancy-related complications, particularly those linked to thrombotic events. This underscores the importance of genetic screening and personalized management of thrombotic risks in pregnant women. 101
FXIII V34L Gene Variant
While the FXIII V34L variant may play a role in pregnancy complications when combined with other genetic factors, its isolated effect on pregnancy thrombosis and abortion remains inconclusive and warrants further investigation.31,102
Antithrombin III Deficiency
Antithrombin is a plasma glycoprotein crucial for regulating the coagulation process during bleeding. It binds specifically to serine proteases such as factor II (thrombin), factor IXa, and factor Xa, thereby inhibiting the blood clotting mechanisms within the coagulation cascade. 103 A person with congenital antithrombin III deficiency, an autosomal dominant disorder, inherits one copy of the gene SERPINC1, which codes for antithrombin III. This condition increases the risk of venous and arterial thrombosis. 104 Women with AT deficiencies are more likely to experience VTE during pregnancy and the postpartum phase, and this risk is particularly elevated in individuals who have previously experienced VTE. 105 Additionally, women with AT deficiencies are more likely to experience obstetric complications, such as embryo-fetal losses caused by VTE.106,107
Environmental Factors in Thrombosis Miscarriages
Hereditary thrombophilia is not sufficient to ensure the occurrence of thrombosis on its own. The clinical condition known as thromboembolic syndromes typically involves the presence of one or more hereditary thrombophilia factors, in addition to one or more acquired thrombophilia factors. These acquired thrombophilia factors include pregnancy, immobility, surgical procedures, and the use of oral contraceptives. Environmental factors contributing to recurrent abortions additionally encompass exposure to organic solvents, pharmaceuticals, ionizing radiation, environmental toxins, and three particular substances: tobacco, alcohol, and caffeine. Proofed evidence indicates that exposure to certain environmental pollutants, such as volatile organic compounds, heavy metals, and specific chemicals, may negatively impact pregnancy outcomes. 5
Diagnosis of Thrombosis and Recurrent Spontaneous Abortion
Clinical Evaluation
Clinical evaluation for thrombophilia should be conducted to achieve a more accurate diagnosis in patients with VTE. 108 These situations include women with a history of pre-eclampsia, recurrent miscarriage, fetal growth restriction, first- or second-trimester loss, placental abruption, or intrauterine death. Family and personal medical history, as well as a history of thrombosis or related medical conditions, are key factors in this area.109,110 A comprehensive clinical examination is advised, with particular focus on the skin to identify potential signs of coagulation problems and thrombosis, such as petechiae and purpura. 111 Additionally, assessment of the lymphatic and peripheral veins, the cardiorespiratory system, the abdomen, the urinary system, and the neurological system is required. 112 Taking a comprehensive history is crucial; this includes previous abortions, maternal age, alcohol and cigarette consumption, caffeine and sedative intake, BMI, and the examination of thyroid disease history, all of which can assist in identifying the condition and the etiology of the abortion. 113 Therapeutic decisions are influenced by the distinction between idiopathic-unprovoked and secondary-provoked VTE. In the secondary-provoked kind, transitory conditions are changeable concerning small and major risk factors, but idiopathic-unprovoked conditions are typically regarded as irreversible and permanent.109,111 These instances necessitate investigation within the thrombophilia field to facilitate prophylaxis when one or more risk factors are present, including age, gender, oral contraceptive use, hormone therapy, pregnancy, prolonged travel, trauma, surgery, immobilization, and malignancy. 108 Research indicates that 1 in 1000 pregnancies is susceptible to VTE, particularly post-delivery. 114 The primary symptoms of DVT encompass dyspnea, tachycardia, groin discomfort, unilateral pain, and unilateral leg edema. Many of these symptoms are typical physiological changes during pregnancy, and while most women experiencing them will not develop DVT, the risk of VTE is increased tenfold. As a result, women with a history of recurrent miscarriage should be assessed for thrombophilic factors to ensure proper management during pregnancy. 114 Thrombophilia linked to early pregnancy loss elevates the risk of VTE, which is a significant contributor to maternal death and newborn complications. Pregnancy is a risk factor that elevates the incidence of VTE by 5-fold, which escalates to 12-fold postpartum.108,115 Consequently, identifying the risk factors associated with hereditary thrombophilia is crucial for managing pregnancy, ensuring safe delivery, and preventing complications for both the mother and infant (Figure 3).108,116

Classification of thrombophilia risk in clinical evaluation. Recognizing the thrombophilia-related risk factors in clinical assessment is essential for managing pregnancy, facilitating safe delivery, and averting complications affecting the mother and infant.
Laboratory Tests
Despite the correlation between hereditary thrombophilia and pregnancy loss, ESHRE and ASRM do not support routine screening for MTHFR, factor V Leiden, and other thrombophilia-related mutations in cases of recurrent miscarriage117–119; testing for hereditary thrombophilia is recommended only if there is a personal history of VTE, with or without additional risk factors like prolonged immobility, or if a first-degree relative has a history of a high-risk inherited thrombophilia. 116 The assessment of plasma coagulation markers is not recommended during pregnancy for women with a history of recurrent miscarriages. These markers should not be used as a criterion for starting medication for miscarriage prevention, according to the DGGG (German Society for Gynecology and Obstetrics)/ÖGGG (Austrian Society for Gynecology and Obstetrics)/SGGG (Swiss Society for Gynecology and Obstetrics) guideline.117–119 In the situation of APS, a kind of acquired thrombophilia, the diagnostic criteria are definitive, necessitating the evaluation of lupus anticoagulant antibodies, anticardiolipin antibodies, and anti-β2 glycoprotein antibodies.117,118 The APS screening has been accepted by various guidelines for identifying the etiology of recurrent miscarriages. This screening should be conducted not just to identify the cause but also to minimize complications during pregnancy, including pre-eclampsia and neonatal death.113,120 DGGG/ÖGGG/SGGG/RCOG (Royal College of Obstetricians and Gynecologists) and ASRM (American Society for Reproductive Medicine) guidelines for APS screening recommend evaluation after three recurrent unexplained miscarriages prior to 10 weeks, or one unexplained fetal death after 10 weeks of gestation, or one preterm delivery before 34 weeks due to eclampsia, severe pre-eclampsia, or placental insufficiency. On the other hand, ESHRE (The European Society of Human Reproduction and Embryology) advises that the antiphospholipid antibody screening should be conducted following the second miscarriage, irrespective of gestational age.113,116,119,121 In the context of hereditary thrombophilia, all guidelines, with the exception of the RCOG, believe that routine screening is not advisable solely for the prevention of miscarriage due to an unclear correlation with recurrent miscarriage and the demonstrated efficacy of anticoagulants in enhancing live birth rates in women with these mutations. 113 The RCOG advises screening for hereditary thrombophilia, such as factor V Leiden, prothrombin gene variations, and protein S deficiency, after a second-trimester abortion. This advice is derived from a meta-analysis encompassing 16 case-control studies, which demonstrated a significant correlation between second-trimester abortion and these complications.86,113 A 2022 retrospective cohort research and systematic review analyzed 1155 women having a history of three or more recurrent first-trimester losses. The prevalence of thrombophilia was 9.2% (106/1155), with 5.1% (94/1155) attributed to hereditary cases and 1% (12/1155) to acquired cases. Among women with hereditary thrombophilia, 4.8% (34 out of 1155) were found to have factor V Leiden. The findings indicate that the prevalence of hereditary thrombophilia among the examined women is comparable to that of the general population. The identified type showed a significantly low incidence of recurrent early miscarriage, so the use of aspirin or LMWH for prophylaxis is not recommended unless backed by strong laboratory and clinical evidence. 122 Despite contrary opinions suggesting that LMWH may be beneficial in this context, the Ottilia study demonstrated its efficacy in decreasing fetal loss among thrombophilic women. 123 Therapeutic approaches and routine tests to evaluate thrombophilia in pregnancy are summarized in Figure 4.
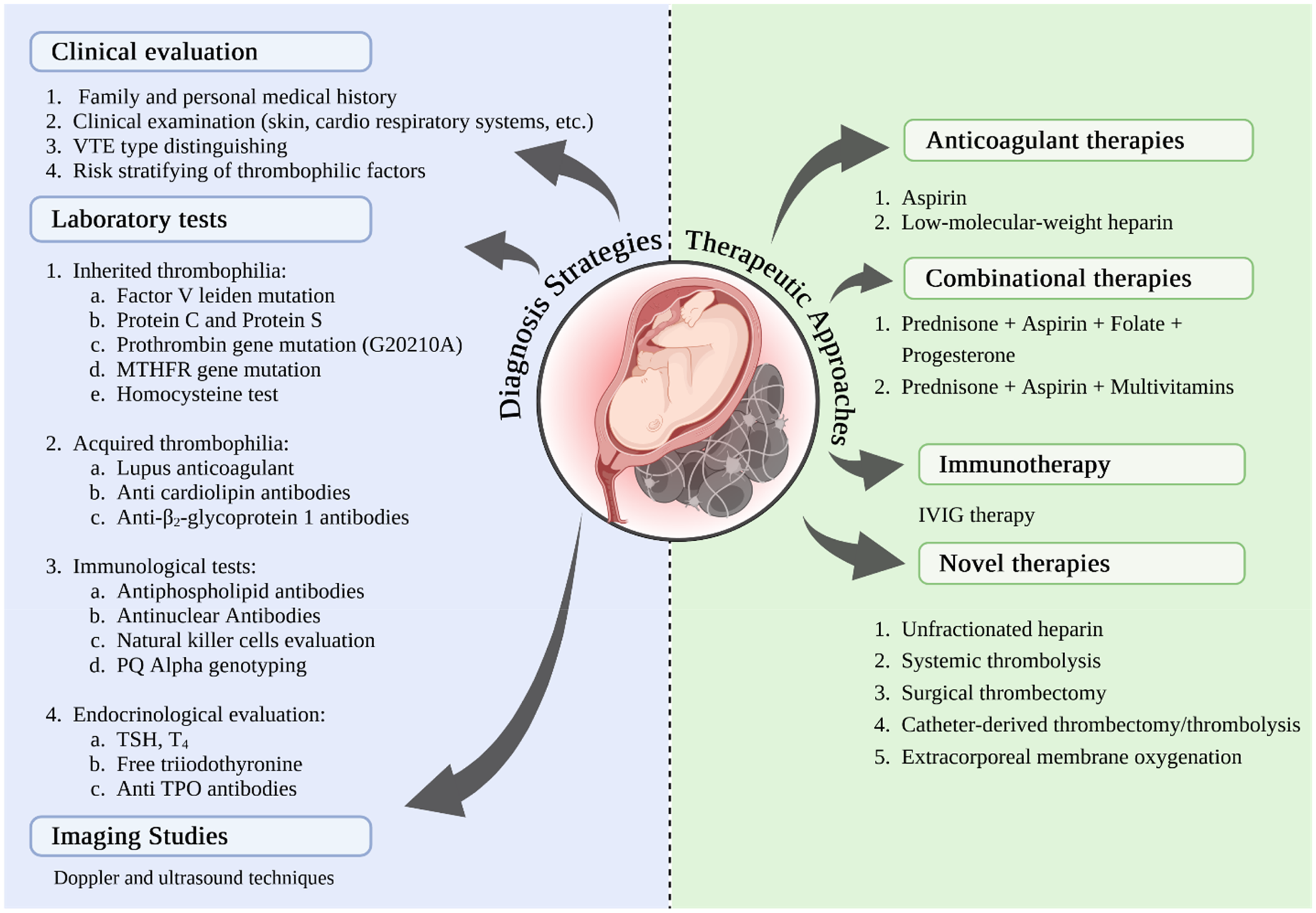
Diagnostic and therapeutic approaches for thrombosis-related RSA. Clinical evaluation and laboratory screening tests for thrombophilia, including factor V Leiden, prothrombin gene mutations, and protein S deficiency, serve as essential diagnostic methods to attain a more precise diagnosis in patients with venous thromboembolism. In order to assess placental blood flow and identify potential thrombotic lesions, imaging studies, especially ultrasound and Doppler techniques, are crucial. In terms of treatment, anticoagulant medications, such as LMWH and aspirin, prevent thrombosis in patients with RSA, and combination therapies can improve the outcomes of pregnancy. Immunotherapy and other targeted treatments could assist with handling recurrent miscarriages. Additionally, new surgical techniques and other innovative therapeutic approaches have recently been developed to mitigate the adverse consequences of thrombotic events during pregnancy.
Imaging Studies
Imaging studies, particularly ultrasound and Doppler techniques, are crucial for evaluating placental blood flow and detecting possible thrombotic lesions. Ultrasound refers to sound waves with frequencies exceeding the upper limit of human hearing. It is used in various applications, including medical imaging and industrial testing, due to its non-invasive nature and ability to provide real-time feedback.124,125 Ultrasound is utilized to evaluate placental morphology, positioning, and any structural anomalies, and is capable of identifying placental masses, abnormal implantation, and various conditions that may impact placental function.124,125 Doppler ultrasound refers to a diagnostic imaging technique that utilizes the Doppler effect to assess blood flow and movement within the body. This method is commonly employed in various medical applications, including cardiology and obstetrics, to evaluate vascular conditions and fetal health. 124 Doppler ultrasound assesses blood flow within the placenta and umbilical cord and differentiates between the maternal and fetal components of placental circulation, offering critical insights into placental health and functionality.124,125 The anatomical distribution of DVT in pregnant women differs from other populations, often affecting the proximal iliac vessels. For pregnant women with suspected DVT, leg ultrasound, pressure maneuvers, and vascular Doppler evaluations are typically sufficient for diagnosis and exclusion. If symptoms persist or isolated iliac DVT is suspected, serial evaluations over a 7-day period may be helpful.114,126 Also, Doppler ultrasound evaluates the hemodynamics of the uterine arteries responsible for placental perfusion. Abnormal flow patterns may signify conditions like preeclampsia or IUGR. 114 Doppler techniques can be used to identify regions of reduced or absent placental blood flow, which may suggest the presence of thrombotic lesions or infarction. Recent advancements in ultrasound technology, including superimposed microvascular imaging (SMI), are also developing and enabling more comprehensive evaluations of placental microvasculature. These techniques enhance the detection of both adequate and diminished blood flow, offering improved insights into placental health. 127 In addition, low-dose perfusion scanning (LDQ) and CT pulmonary angiography (CTPA) are effective techniques, with LDQ offering reduced radiation to both the mother and fetus. Therefore, LDQ is a suitable first-choice imaging option for suspected pulmonary embolism in pregnant women with a negative chest radiograph. 128
Management of Thrombosis and Recurrent Spontaneous Abortion
Anticoagulant Therapy
The majority of individuals pursuing abortions are in normal condition. However, healthcare providers may encounter cases involving various medical conditions, such as bleeding disorders, thrombophilia, a history of VTE, or those on anticoagulation therapy for other medical reasons. 129 In terms of this issue, anticoagulant medications, such as LMWH and aspirin, prevent thrombosis in patients with RSA. Moreover, aspirin and LMWH are often used for cases experiencing unexplained repeated miscarriages or those with recurrent miscarriages and inherited thrombophilia. 130 Recent pregnancy loss may be linked to genetic blood clotting disorders that affect placental blood flow. Recurrent miscarriage can also occur without an identified cause. However, these anticoagulants can assist women with clotting disorders, preventing complications like nosebleeds and hematomas in the mother without harming the baby. 32 Low-dose aspirin is utilized in the pregnancy period in conditions like recurrent miscarriage, pre-eclampsia, fetal growth restriction, and infertility. However, the mechanisms through which aspirin produces its antithrombotic effects have not been entirely clarified. Also, it remains uncertain if anticoagulant therapy can enhance the live birth rate across all scenarios. Further, a 2020 study found that aspirin is dose-dependent, with its interaction partially enhancing its antithrombotic effects while also limiting other effects. 131 It is still unclear whether anticoagulant therapy can improve the live birth rate in all cases. However, recent findings also indicate that a combination of low-dose aspirin and heparin is effective in preventing RPL among women diagnosed with antiphospholipid syndrome. 132 It is worth noting that 70% of repeated miscarriages are unknown, although anticoagulant treatment is beneficial in preserving pregnancy in the absence of antiphospholipid antibody syndrome. 133 It refers to Tzafettas et al's research that indicated a favorable impact associated with low molecular weight heparin therapy in around 50%–75% of instances with an unknown cause. 134 Tulppala et al found that low-dose aspirin had a positive biochemical effect, but it did not enhance pregnancy outcomes for women with recurrent miscarriage, whether or not they had detectable anticardiolipin antibodies. 135 Additionally, Ogasawara and colleagues found that low-dose aspirin is beneficial for treating repeat miscarriage in individuals with factor XII deficiency.136–138
Lifestyle Modifications
Pregnancy represents a significant period of transformation in a woman's life and serves as a crucial opportunity to impact not only the health behaviors of women but also those of their family members. Physiological modifications during pregnancy elevate the likelihood of thrombosis by heightened coagulation factors, reduced fibrinolysis, trauma, and venous stasis. Additionally, factors associated with pregnancy, such as advanced maternal age, obesity, multiple gestations, and cesarean sections, further contribute to the increased risk of thrombosis.139,140 A lack of physical activity during pregnancy is related to a significant health risk of DVT, posing dangers to both the mother and the infant. Over time, a sedentary lifestyle may result in obesity or, at the very least, play a significant role in the maintenance of chronic obesity. 141 An increase in cardiovascular comorbidities, including gestational diabetes, pregnancy-induced hypertension, pre-eclampsia, and heart disease, can occur when there is excessive weight and obesity during pregnancy. Maternal multimorbidity directly affects the fetus, notably increasing its vulnerability to cardiovascular problems. Importantly, engaging in physical activity during pregnancy can significantly mitigate the effects of these comorbidities on women's health. Significantly, physical activity advantages during pregnancy include enhancements in cardiovascular health, increased well-being, and improved sleep quality.142–146
Supportive Care
Pregnancy is a usual health state that requires ongoing and attentive care to reduce the risk of adverse outcomes. Although there have been advancements in reducing maternal and neonatal mortality rates, disparities persist between developing and developed countries or regions. Hemorrhage is the primary reason for maternal death.147,148 Further, during pregnancy, women face a higher chance of developing venous and arterial thromboembolism. Other factors that can increase the risk include a background of thrombosis, thrombophilia, specific health issues, and certain pregnancy-related issues. Although the risk of thrombosis is higher during and after pregnancy, most women do not require anticoagulation. However, those at high risk may need it. Anticoagulation during pregnancy must consider both maternal and fetal safety, with heparin being the preferred option. Prior to delivery, surgery, or invasive procedures, anticoagulation should be adjusted to minimize bleeding risks while still preventing thrombosis.149,150
Therapeutic Strategies for Thrombosis and Recurrent Spontaneous Abortion
In a therapeutic setting, LMWH is just as effective as unfractionated heparin, but they are easier to use and have fewer side effects. In terms of UFH, recent advancements in the field of anticoagulation therapy have introduced new insights that may influence the future application of UFH. This will encapsulate crucial aspects that highlight the controversies associated with UFH, providing a nuanced understanding of its role in clinical practice. In addition, laboratory assessments for monitoring have notable limitations. Established therapeutic ranges lack comprehensive evidence, leading to potential inconsistencies in clinical use. Furthermore, the effect of antithrombin levels on UFH's anticoagulant efficacy is not well understood, raising concerns about the reliability of current monitoring practices.151–153
Targeted Therapies
The immune system plays a vital role in sustaining pregnancy and embryo implantation, involving different immune cells, cytokines, and regulatory mechanisms that establish the essential immune equilibrium. As well as different therapeutic methods, such as immunomodulatory medications, immune adsorption treatments, and immune cell therapies, are continuously being investigated and advanced. Immunotherapy might contribute to treating recurrent miscarriages; however, several ongoing controversial issues remain, and this needs additional research for confirmation.154,155 Immunoglobulin therapy, which involves altering the immune system's activity through the delivery of immunoglobulins, may serve as a possible treatment for RSA. Intravenous immunoglobulin (IVIG) therapy markedly decreases the count of Th1 cells and significantly boosts the number of Th2 cells in women experiencing RSA. This coincides with reduced levels of pro-inflammatory cytokines IFN-γ and TNF-α, along with a notable rise in the anti-inflammatory cytokine IL-10, ultimately enhancing the pregnancy outcomes for patients with RSA. 156 The use of IVIG for treating RSA notably reduces the levels of IL-17 and RORγt mRNA in peripheral blood while also leading to a significant rise in the anti-inflammatory cytokines TGF-β and IL-10. This observation shows a decline in the Th17/Treg ratio, resulting in an expansion of the Treg population and a reduction in the Th17 response. 157 Tregitopes are IgG-derived epitopes capable of binding to MHCII, activating natural regulatory T cells (nTregs), and fostering tolerance. The administration of Tregitopes promotes the growth of regulatory T cells (Tregs), boosts interleukin-10 (IL-10) production from Tregs and Bregs, modifies the co-stimulatory response of antigen-presenting cells, preserves the tolerance phenotype of these cells, and contributes to decreased fetal mortality.157,158 Immunoglobulin therapy could be beneficial in certain instances, but its effects may be limited or uncertain in others. However, the treatment process should prioritize qualified doctors who tailor care to the patient's specific needs.
Combination Therapies
Pregnancy consequences in women with RSA can be enhanced through treatment strategies that include a combination of low-dose aspirin and heparin or heparin separately. On the other hand, aspirin on its own appeared less effective than other treatments in securing a higher number of live births. Low-dose aspirin combined with heparin and heparin-only treatment showed a notable positive impact on live births. Nonetheless, no advantage of low-dose aspirin monotherapy was observed in live birth. However, several observational studies offer valuable insights into the use of LMWH for prophylaxis, exploring its potential to improve live birth rates. Also, exploring LMWHs as a prophylactic treatment remains a significant area of interest. Continued research is essential to understand better the potential role of these anticoagulants in improving reproductive outcomes and to establish more robust evidence that can guide clinical practice. 123 Meanwhile, the combination of low-dose aspirin and heparin did not significantly reduce the risk of recurring placenta-related pregnancy complications, such as preterm birth, IUGR, gestational diabetes, or minor bleeding. However, heparin monotherapy was shown to have a positive therapeutic impact in preventing preterm birth. Additionally, the combination of aspirin and heparin significantly reduced the likelihood of preeclampsia. 159 Aspirin is classified as a non-steroidal anti-inflammatory medication. It functions by blocking platelet cyclooxygenase to decrease the production of prostaglandins. This may reduce the likelihood of thrombosis in patients, aiding embryo implantation and lowering the chance of repeated miscarriage.160,161 The current research disclosed that the prevalence of URSA in women experiencing early RSA was 47.2%, and the combination therapy of aspirin and prednisone enhanced the success rate of treatment in women with URSA. Furthermore, using a combination of aspirin, prednisone, and multivitamins could lower the risk of miscarriage and enhance pregnancy results for women with URSA. In a similar manner, an earlier study indicated that a blend of prednisone, aspirin, folate, and progesterone enhances birth results for women experiencing idiopathic recurrent abortions. Also, the current retrospective study revealed that the combination therapy of aspirin and prednisone enhanced the success rate of treatment in comparison to the control therapy. In fact, aspirin's anticoagulation property may have lowered the risk of thrombosis, while prednisone dampened the maternal autoimmune reaction to the zygote and facilitated successful implantation. Multivitamins created a favorable nutritional environment for women, decreasing the chances of fetal malformation and thus enhancing the success rate of treatment.162–166
Future Directions
Research Priorities
There is still disagreement over preventative measures, despite the fact that there are many guidelines regarding the management and treatment of thrombophilia during pregnancy and recurrent miscarriages. 122 Conversely, novel discoveries like microRNAs are subjected to ongoing investigation and identification daily. MicroRNAs (miRNAs) significantly contribute to thrombophilia, a disorder marked by atypical expression of post-transcriptional target genes. Over a thousand human miRNAs have been identified, each regulating numerous target genes, including those involved in placental development. MiRNAs are crucial regulators of placental development during pregnancy. 108 A 2020 study identified 16 miRNAs of significance in families with idiopathic thrombosis. Four of these miRNAs exhibited differential expression in patients with VTE, signifying a substantial correlation with VTE and possible prognostic indicators of this condition. The human placenta harbors numerous miRNAs that are crucial to its developmental and peri-implantation processes, such as hsa-miR-126-3p, hsa-miR-885-5p, hsa-miR-194-5p, and hsa-miR-192-5p. 167 The results are insufficient for a definitive assessment, indicating that this subject is a research priority in the discipline.
Besides, experts increasingly acknowledge the male role in RPL as a key contributor to pregnancy outcomes. Nevertheless, robust suggestions about particular diagnostic and treatment strategies are still absent, underscoring a vital domain for additional inquiry and a research imperative, especially for reproductive specialists. Current treatments frequently emphasize routine semen analysis and karyotyping; yet, these methods may inadequately elucidate the male factors to RPL. Recent studies suggest that sperm DNA fragmentation, chromatin abnormalities, and oxidative stress are critical determinants of early embryonic development and pregnancy outcomes. Addressing these deficiencies through focused research initiatives is crucial for strengthening diagnostic precision and therapeutic strategies, hence improving the overall management of RPL. Future research should aim to develop evidence-based protocols for assessing and managing male factors in RPL. Advancing molecular research to identify genetic, epigenetic, and proteomic markers will be key. Collaboration among reproductive physicians, geneticists, and molecular biologists could lead to new diagnostic tools and treatments targeting the male factor in RPL. Focusing on this aspect will deepen the understanding of RPL and ensure comprehensive care for affected couples. This approach could improve treatment outcomes and address a significant unmet need in reproductive medicine.68,168
Novel Therapeutic Strategies
Nearly all applicable guidelines concur that anticoagulants should not be used in patients with recurrent miscarriage and genetic thrombophilia only for the purpose of preventing subsequent miscarriages. Anticoagulants should be administered solely in the presence of significant risk factors for VTE. 113 The predominant anticoagulants are UFH and LMWH. To prevent complications associated with LMWH, regular assessments of D-dimer and anti-Xa levels should be conducted. The ideal dosage remains uncertain, constituting a significant challenge in the administration of anticoagulants during pregnancy. Anticoagulants should be provided prophylactically following the completion of testing and clinical diagnosis; however, their usage during pregnancy may present complications, including hemorrhage risk and potential for abortion. The predominant anticoagulants are UFH and LMWH. The ideal dosage remains uncertain, constituting a significant challenge in the administration of anticoagulants during pregnancy. 169 UFH can cause thrombocytopenia, osteoporosis, and maternal hemorrhage, making LMWH the preferred choice. Although anticoagulants and thrombolytics help manage complications in pregnant women with hereditary thrombophilia, this condition remains a bleeding risk, especially during delivery. However, this risk is manageable.108,170 Antiplatelet medications pass the placenta; hence, their usage is not advised. The sole exception in this context is aspirin, which is generally well tolerated and not linked to the risk of fetal demise; however, any aspect of it must be analyzed.108,171 Anticoagulant therapy is adequate for the majority of VTE cases; however, in instances of extensive pulmonary embolism, additional interventions are required, such as systemic thrombolysis, surgical thrombectomy, catheter-directed thrombectomy/thrombolysis, or extracorporeal membrane oxygenation (ECMO). 172
Oxidative stress (OS), defined by an excess of reactive oxygen species (ROS), is a recognized contributor to numerous clinical diseases, including hematological illnesses. 173 Excessive ROS may damage biomolecules, including DNA, proteins, and lipids, resulting in cellular malfunction and apoptosis. 174 This sequence of events not only adds to systemic disorders but also significantly impacts reproductive health. In the context of RPL and RSA, OS has been associated with an impairment of sperm and oocyte quality, in addition to restricting early embryonic development. 175 The buildup of oxidative damage might initiate inflammatory responses and thrombus formation, hence worsening pregnancy problems. 173 These findings highlight the essential requirement for thorough evaluation and specific therapies to alleviate OS in impacted persons. 174 Preserving redox equilibrium is crucial for cellular function and overall health. 175 Promising therapeutic techniques, including the application of antioxidants, have been suggested to mitigate the adverse consequences of oxidative stress. 173 Antioxidants neutralize ROS, therefore reducing cellular damage and restoring stability in the redox state. 174 Research has emphasized the promise of molecular-level therapies to enhance reproductive outcomes in couples facing RPL. 175 Additionally, coordinated research initiatives are essential to establish evidence-based protocols for the therapeutic utilization of antioxidants and other redox-modulating treatments. 174 By targeting OS and promoting redox homeostasis, researchers and clinicians can facilitate the development of novel therapies that improve fertility and alleviate the challenges of RPL. 175
Conclusion
RSA is a complex condition with various causes, including thrombophilia. Understanding the pathogenesis of RSA, particularly its thrombotic origins, and identifying inherited and acquired risk factors is crucial. This review highlights the relationship between thrombosis, genetic and acquired factors, inflammation, and endothelial dysfunction in RSA. However, further research is needed to explore the relationship between this complex interplay, as well as the paternal role in RSA. As highlighted in this review, managing thrombophilia can improve pregnancy outcomes, but uncertainties remain regarding effective diagnostics and treatment, particularly with anticoagulants. Despite existing guidelines, significant disagreement persists over the best preventative and therapeutic approaches for thrombophilia in pregnancy and recurrent miscarriages. This underscores the need for more evidence-based recommendations. Comprehensive, forward-looking case-control studies are essential to provide deeper insights and refine strategies for better outcomes. It is also important to balance the benefits of screening and treatment with the risks of unnecessary interventions. Advances in this field will lead to more effective diagnostic tools, novel biomarkers like microRNAs, and innovative therapies, including immunotherapies, based on RSA pathogenesis. These advancements aim to improve reproductive outcomes for women with RSA, offering hope for more successful pregnancies and better care.
Footnotes
Abbreviations
Acknowledgments
Although the authors received no financial support, they would like to express their gratitude to the researchers whose articles were used in this study.
ORCID iDs
Author Contributions
Hamed Soleimani Samarkhazan were involved in the conception of the study, data analysis, manuscript preparation, and monitoring. Moreover, Ahmed Hussein, Amin Solouki, Niloofar Pilehvari, Fatemeh Studeh, Hanieh Noormohamadi, Parvaneh Abbasi Sourki, Athena Behforouz contributed to the search for relevant manuscripts and the preparation of the manuscript. Hamed Soleimani Samarkhazan contributed to the development of the search strategy, article search, and manuscript preparation. Finally, all the authors reviewed and approved the manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
No datasets were generated or analysed during the current study.
