Abstract
Objective
This study aimed to investigate the relationship between the transient increase in D-dimer following anticoagulant therapy and thrombolysis at 30 days in patients with pulmonary embolism (PE).
Method
Patients diagnosed with PE at our hospital were included in the study. CT pulmonary angiography (CTPA) was performed 7-10 days after starting anticoagulant therapy. Patients were divided into two groups: the change group and the non-change group, based on whether the thrombus had broken into smaller clots and/or dissolved compared to baseline. Plasma D-dimer levels were measured 1-10 days after anticoagulant therapy to observe any transient increase. The correlation between the transient D-dimer increase and thrombolysis at 30 days in PE patients was analyzed.
Results
A total of 172 patients with PE were included. The rate of thrombus change was 63.4% (75/172) at 7-10 days after anticoagulant therapy. The proportion of thrombolysis at 30 days was 68.6% (118/172). Spearman correlation analysis showed a significant correlation between the transient increase in D-dimer and thrombus changes (Rs = 0.482, P < .001), between thrombus changes and thrombolysis at 30 days (Rs = 0.413, P < .001), and between the transient increase in D-dimer and thrombolysis at 30 days (Rs = 0.540, P < .001). ROC curve analysis indicated that the transient increase in D-dimer predicted thrombus changes (AUC: 0.750, 95%CI: 0.673-0.827, P < .001), and predicted thrombolysis at 30 days (AUC: 0.786, 95%CI: 0.714-0.858, P < .001). Thrombus changes also predicted thrombolysis at 30 days (AUC: 0.712, 95%CI: 0.626-0.797, P = .001).
Conclusion
After anticoagulant therapy for PE, D-dimer levels may transiently increase. The rate of thrombolysis at 30 days was higher, and a transient increase in D-dimer indicated a higher likelihood of thrombolysis at 30 days.
Keywords
Introduction
Pulmonary embolism (PE) is the third leading cause of death from cardiovascular disease, following myocardial infarction and stroke.1,2 In clinical practice, the simplified Wells score or the revised Geneva score is commonly used for the initial assessment of suspected acute PE, followed by D-dimer testing to further screen for acute PE.3-6 However, while D-dimer has high sensitivity, its specificity is low.7,8 When D-dimer levels are ≤0.5 mg/L, it has a high negative predictive value for patients with low or moderate risk of PE, but its usefulness is limited in patients with a high suspicion of PE.9-12 An increase in D-dimer during or after anticoagulant therapy in PE patients may suggest recurrent or new pulmonary thrombi.13,14 As a result, elevated D-dimer levels are often associated with a poor prognosis.
In clinical practice, we have observed that some PE patients undergoing anticoagulant therapy experience an initial decrease in D-dimer, followed by a brief increase, and then a subsequent decline to normal levels. During this period, imaging often shows that large thrombi have broken into multiple smaller thrombi or have dissolved and reduced in size. This raises the question: is there a correlation between the transient increase in D-dimer and imaging changes of pulmonary thrombosis? Furthermore, is the transient elevation of D-dimer associated with thrombolysis? Currently, few studies have explored this topic.
In this study, we conducted a retrospective case-control analysis to investigate the correlation between the transient increase in D-dimer after anticoagulant therapy and thrombolysis at 30 days in PE patients. We also aimed to explore the feasibility of using early-stage D-dimer changes during anticoagulation to predict pulmonary thrombolysis.
Methods
Patients
A total of 172 patients with PE, including 88 males and 84 females aged 31 to 94 years, diagnosed through computed tomography pulmonary angiography (CTPA) at the Department of Respiratory and Critical Care Medicine of Shanghai Sixth People's Hospital between January 2018 and December 2023, were enrolled in this study.
The inclusion criteria used in the present investigation were based on the diagnostic criteria of “Guidelines for the diagnosis, treatment, and prevention of pulmonary thromboembolism” published by the Chinese Society of Thoracic Society (CTS) in 2018. 15
Inclusion criteria: Age ≥18 years old. CTPA diagnosed PE for the first time. Course of disease <30 days. No contraindications of anticoagulation therapy. Patients had to complete 90 days of treatment and follow-up. D-dimer levels and arterial blood gas were measured at various times before and after anticoagulant therapy. CTPA was performed before anticoagulant therapy, and repeated at 7-10 days, 30 days, and 90 days after treatment.
Exclusion criteria: Patients who were initially diagnosed with CTPA in other hospitals and then referred to our hospital for further treatment, those with incomplete medical records lacking essential research information, individuals unable to undergo CPTA examination due to factors such as obesity, severe renal insufficiency, or contrast allergy, those with chronic pulmonary embolism were excluded. Additionally, patients with contraindications to direct oral anticoagulants (DOACs) treatment such as chronic kidney disease with creatinine clearance <30 mg/dL, cirrhosis (Child-Pugh grade B or C), alanine aminotransferase levels more than twice the upper limit of normal, younger than 18 years at the time of diagnosis, known to be pregnant, were also excluded.
Definition of pulmonary thrombosis breakdown: Thrombus in the main pulmonary artery or the left and right pulmonary arteries was considered broken if, during anticoagulation therapy, the thrombus fragmented into smaller clots that moved into different segments or subsegments of the pulmonary arteries compared to the previous imaging.
Transient increase in D-dimer: A temporary rise in D-dimer was defined as a D-dimer level measured within 1-10 days after starting anticoagulation therapy that was higher than the previous day's result, followed by a subsequent decrease from that elevated level upon retesting.
This study was approved by the Ethics Committee of Shanghai Sixth People's Hospital, Shanghai Jiao Tong University. All procedures involving human participants were conducted in accordance with the ethical standards of the institutional and national research committee and adhered to the principles outlined in the 1964 Helsinki Declaration and its subsequent amendments or comparable ethical standards, and informed consent was waived due to its retrospective design (No.: 2023-KY-138(K)).
Treatment Methods
Following PE diagnosis, anticoagulant therapy was initiated. Low-molecular-weight heparin (100 IU/kg every 12 h) was subcutaneously administered to 69 patients. Warfarin (2.5 mg) was introduced orally 24-48 h after the start of low-molecular-weight heparin. Both low-molecular-weight heparin and warfarin were used concurrently for 4-5 days. Once prothrombin time (PT) and international normalized ratio (INR) reached 2.0-3.0 for two consecutive days, low-molecular-weight heparin was discontinued, but warfarin was continued orally. When INR was less than 2.0, warfarin doses was increased 0.625 mg. When INR was more than 3.0, warfarin doses should be reduced or even discontinued.
In 103 patients, rivaroxaban was used (15 mg orally twice daily for 3 weeks, followed by 20 mg orally once daily for 9 weeks). All patients received anticoagulant therapy for 3 months .None of the patients experienced adverse events such as subcutaneous hematoma, gastrointestinal bleeding, or intracranial hemorrhage.
Data Collection
The following factors were retrospectively recorded and comprehensively analyzed: gender, age, body mass index (BMI), smoking history, underlying diseases (hypertension, coronary heart disease, stroke, diabetes, fractures within the past 4 weeks, recent fixation or surgery, malignancy), major symptoms (dyspnea, chest pain, shortness of breath, etc), (arterial blood gas analysis, plasma D-dimer (D-D), B-type natriuretic peptide (BNP), Troponin (TNI)) were performed within 4 h of PE diagnosis. Ultrasound examination of lower extremity veins, CTPA results, Pulmonary artery obstruction index (PVOI, 16 At the time of PE diagnosis, after 7-10 days anticoagulation therapy, after 30 days of anticoagulant therapy, and after 3 months anticoagulation therapy), treatment modalities, and outcomes.
Statistical Analysis
Statistical analysis was conducted using SPSS version 17.0. The Mann-Whitney U test was applied for comparisons between groups, while normally distributed continuous data were expressed as mean ± standard deviation (Mean ± SD) and compared using the t-test. Categorical data were presented as frequencies (%) and compared using the χ² test. The analysis focused on the following variables: changes in thrombi (±) as observed on CTPA 7-10 days after anticoagulant therapy, transient increases in D-dimer (±) after anticoagulant therapy, and complete dissolution of thrombi (±) as observed on CTPA 30 days after therapy. Spearman's correlation coefficient was used to evaluate the association between thrombus changes at 7-10 days, transient D-dimer elevations, and thrombus dissolution at 30 days. ROC curve analysis assessed the predictive value of transient D-dimer increases (±), thrombus changes at 7-10 days (±), and thrombolysis at 30 days (±) following anticoagulant therapy. A P-value < .05 was considered statistically significant.
Results
Basic Information
A total of 172 patients met the inclusion criteria, consisting of 88 males and 84 females. All patients received anticoagulant therapy, survived, and were discharged. CTPA was repeated 7-10 days after the start of anticoagulant therapy. Based on whether the thrombi had fragmented or dissolved and absorbed more than before, the patients were categorized into the change group (109 cases) and the non-change group (63 cases). In terms of treatment, 69 patients who received warfarin anticoagulant therapy were further divided into the change group (42 cases) and non-change group (27 cases), while 103 patients who received rivaroxaban were categorized into the change group (67 cases) and non-change group (36 cases). Gingival bleeding occurred in 8 patients during warfarin therapy and in 10 patients during rivaroxaban therapy. There were no cases of hemoptysis, subcutaneous hematoma, gastrointestinal bleeding, or intracranial hemorrhage. Detailed results are provided in Tables 1, 2, and 3.
Comparison of Risk Factors (Mean ± SD) (n, %).

Note: * P < 0.05.TNI, troponin I; BNP, B-type natriuretic peptide; PVOI, pulmonary vascular obstruction index; PaCO2, partial pressure of carbon dioxide; PaO2, oxygen partial pressure; DVT, deep vein thrombosis; BMI, body mass index.
Comparison of Risk Factors in Warfarin Treatment Group (Mean ± SD) (n, %).

Note: * P < 0.05.TNI, troponin I; BNP, B-type natriuretic peptide; PVOI, pulmonary vascular obstruction index; PaCO2, partial pressure of carbon dioxide; PaO2, oxygen partial pressure; DVT, deep vein thrombosis; BMI, body mass index.
Comparison of Risk Factors in Rivaroxaban Treatment Group (Mean ± SD) (n, %).

Note: * P < 0.05.TNI, troponin I; BNP, B-type natriuretic peptide; PVOI, pulmonary vascular obstruction index; PaCO2, partial pressure of carbon dioxide; PaO2, oxygen partial pressure; DVT, deep vein thrombosis; BMI, body mass index.
Thrombosis borken rate from 7 to 10 days: The overall thrombus change rate at 7-10 days was 63.4% (109/172). In the warfarin group, the change rate was 60.8% (42/69), while in the rivaroxaban group, it was 65.0% (67/103). There was no significant difference in the thrombus change rate between the two groups at 7-10 days (χ² = 0.311, P = .577).
The overall thrombolysis disappearance rate at 30 days was 68.6% (118/172), with 68.1% (47/69) in the warfarin group and 68.9% (71/103) in the rivaroxaban group. There was no significant difference in thrombus dissolution at 30 days between the two groups (χ² = 0.013, P = .920).
Proportion of thrombus dissolution and disappearance at 90d: At 90 days, CTPA showed complete thrombus absorption in all patients in both groups.
Spearman Correlation Analysis
A positive correlation was found between the transient increase in D-dimer and thrombus changes at 7-10 days after treatment in PE patients (Rs = 0.482, P < .001). Additionally, a positive correlation was observed between thrombus changes at 7-10 days and thrombus disappearance at 30 days (Rs = 0.413, P < .001). The transient increase in D-dimer after treatment was also positively correlated with thrombus disappearance at 30 days (Rs = 0.540, P < .001).
In the warfarin group, a positive correlation was noted between the transient D-dimer increase and thrombus changes at 7-10 days (Rs = 0.435, P < .001), and between thrombus changes at 7-10 days and thrombus disappearance at 30 days (Rs = 0.407, P < .001). However, the correlation between the transient increase in D-dimer and thrombolysis disappearance at 30 days was weaker (Rs = 0.404, P = .001).
In the rivaroxaban group, a positive correlation was noted between the transient D-dimer increase and thrombus changes at 7-10 days (Rs = 0.522, P < .001), and between thrombus changes at 7-10 days and thrombus disappearance at 30 days (Rs = 0.420, P < .001). And between the transient D-dimer increase and thrombus disappearance at 30 days (Rs = 0.629, P < .001).
ROC Curve Analysis
ROC curve analysis demonstrated that a transient increase in D-dimer predicted thrombus breakdown after anticoagulant therapy (AUC: 0.750, 95%CI: 0.673-0.827, P < .001). Additionally, a transient D-dimer increase predicted thrombolysis in PE patients at 30 days post-treatment (AUC: 0.786, 95%CI: 0.714-0.858, P < .001). Thrombus breakdown also predicted thrombolysis at 30 days (AUC: 0.712, 95%CI: 0.626-0.797, P = .001) (Figures 1-3).

The Transient Increase in D-dimer Predicted Thrombus Changes.

The Transient Increase in D-dimer Predicted Thrombolysis at 30 Days.

Thrombus Changes also Predicted Thrombolysis at 30 Days.
In the warfarin group, the transient D-dimer increase predicted thrombus breakdown (AUC: 0.721, 95%CI: 0.594-0.848, P < 0.001). It also predicted thrombolysis at 30 days (AUC: 0.715, 95%CI: 0.582-0.847, P = .004). Thrombolysis at 30 days was predicted by thrombus breakdown (AUC: 0.713, 95%CI: 0.579-0.848, P = .005) (Figures 4-6).
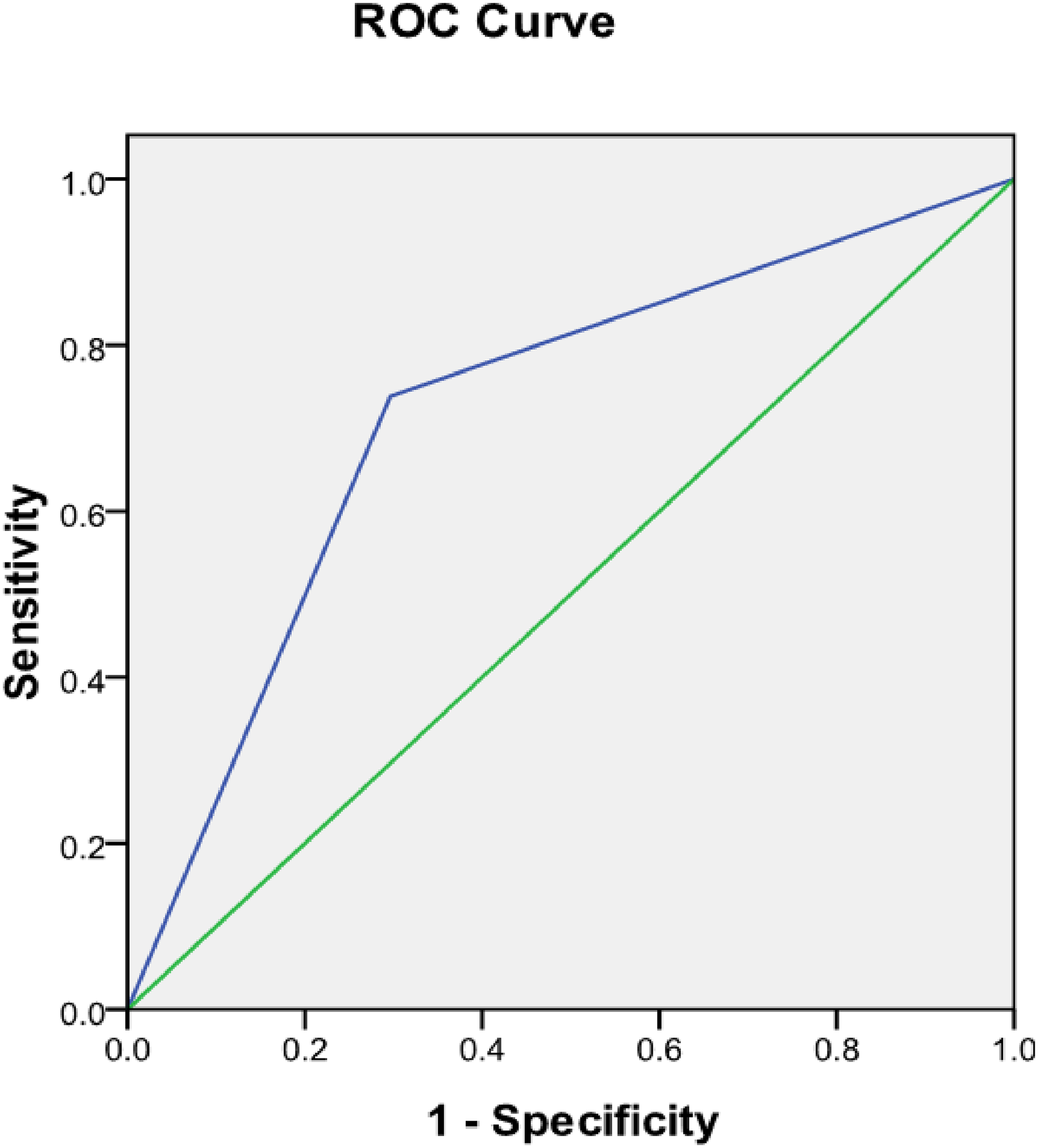
The Transient Increase in D-dimer Predicted Thrombus Changes.
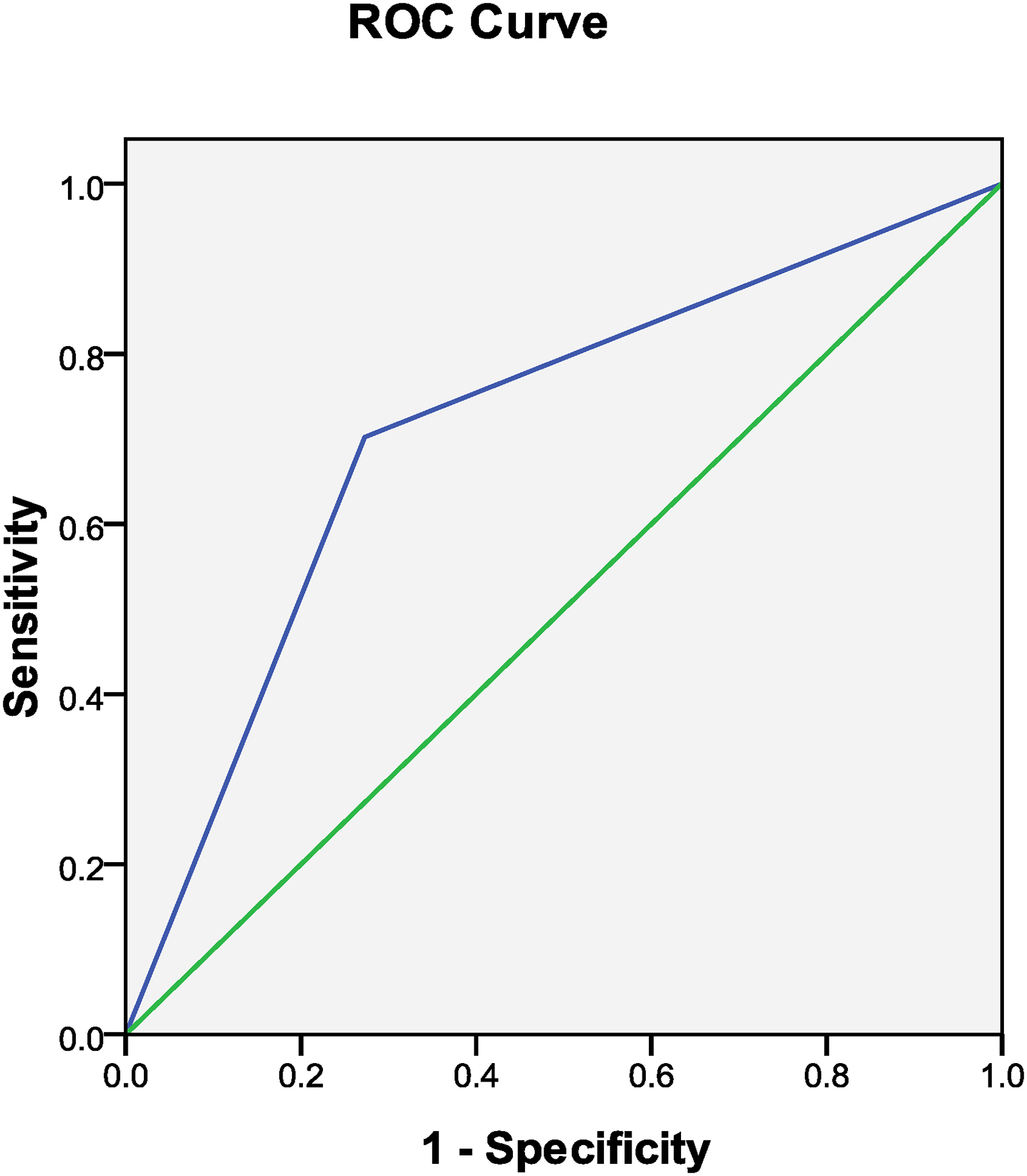
The Transient Increase in D-dimer Predicted Thrombolysis at 30 Days.

Thrombus Changes also Predicted Thrombolysis at 30 Days.
In the rivaroxaban group, the transient D-dimer increase predicted thrombus breakdown (AUC: 0.774, 95%CI: 0.680-0.868, P < .001). It also predicted thrombolysis at 30 days (AUC: 0.832, 95%CI: 0.751-0.912, P < .001). Thrombolysis at 30 days was predicted by thrombus breakdown (AUC: 0.711, 95%CI: 0.601-0.821, P < .001). Figures 7-9.

D-dimer Transient Increases -Thrombi Changes.

The Transient Increase in D-dimer Predicted Thrombolysis at 30 Days.

Thrombus Changes also Predicted Thrombolysis at 30 Days.
Discussion
The CTS guidelines for the diagnosis and management of PE (2018) strongly recommend anticoagulant therapy for three months, after which the decision to extend treatment should be based on the degree of thrombolysis and absorption. 15 In this study, 68.6% of patients achieved complete thrombus dissolution and absorption at 30 days post-anticoagulant therapy, and all PE patients experienced complete thrombus absorption by 90 days. Therefore, for PE patients with transient VTE risk factors, while treating the primary disease and correcting the risk factors for thrombosis, further studies are needed to determine if anticoagulant therapy can be shortened when CTPA at 30 days shows complete thrombosis absorption.
The data also showed no significant difference in the rate of thrombotic changes between patients receiving rivaroxaban and those treated with warfarin, and there was no difference in thrombus absorption after 30 days of anticoagulant therapy. This suggests that the anticoagulant effects of both drugs are similar, consistent with current research findings.17,18
The data indicated that the proportion of transient D-dimer increases was higher in the group with thrombus changes compared to the non-change group. Spearman correlation analysis showed a relationship between thrombus changes and transient D-dimer increases. In both warfarin treatment group and rivaroxaban treatment group, our results also showed that thrombus changes were associated with transient D-dimer increases. This may be because D-dimer is a degradation product of cross-linked fibrin, produced by the action of the fibrinolytic system, and it has early diagnostic value for thrombotic diseases. After anticoagulant therapy, coagulation and fibrinolytic activity is enhanced, causing D-dimer levels to decrease. However, in some PE patients, due to the short duration of thrombosis and the loose internal structure of the thrombus, anticoagulant therapy can break the thrombus into smaller fragments. This increases the surface area of the pulmonary thrombus, allowing anticoagulant drugs to penetrate deeper into the thrombus, further activating the coagulation and fibrinolysis processes, and causing a temporary increase in D-dimer. As anticoagulant therapy continues, the thrombus burden decreases, leading to a gradual reduction in D-dimer levels. Similar results were seen in warfarin treatment group and rivaroxaban treatment group. Previous studies have shown a positive correlation between D-dimer levels and thrombus volume.19-21
D-dimer is increased in many diseases. When D-dimer is increased, it is often as a factor in disease progression. We observed that the rate of thrombolysis and thrombus disappearance at 30 days was higher in patients who experienced thrombus changes during the first 7-10 days of anticoagulant therapy compared to those without changes. Spearman correlation analysis indicated that transient increases in D-dimer and thrombus changes were associated with thrombus disappearance at 30 days. Similar results were observed in warfarin treatment group and rivaroxaban treatment group. This may occur because large thrombi in the main pulmonary arteries break down into smaller clots, increasing the surface area for fibrinolytic factors to act on the fibrin within the thrombus, thereby accelerating its dissolution. Additionally, if the thrombus has formed recently, its structure is relatively fresh and loose, making it easier to fragment and dissolve. Mansueto et al 22 found that platelet aggregation is observed histologically within the first hour of thrombosis in pulmonary thrombi, and the presence of lymphocytes occurs between 1 and 24 h. Fibroblasts appear around vascular tissues between 48 and 72 h, and after 72 h, a large number of fibroblasts and collagen fibers become visible in the thrombus. Thus, earlier initiation of anticoagulation therapy and thrombus changes post-treatment lead to better thrombolysis outcomes. In this study, ROC curve analysis showed that transient increases in D-dimer after anticoagulation therapy could predict thrombus dissolution at 30 days in PE patients. Therefore, after earlier initiation of anticoagulation therapy in some PE patients, transient D-dimer increases may not be an indicator of disease progression, but may be an indicator of thrombus changes, indicating that the thrombus is easy to thrombolysis and thrombus disappearance.
In this study, some PE patients did not have a transient D-dimer increase and break the thrombus into smaller fragments, and the rate of thrombolysis and thrombus disappearance at 30 days was lower in patients who experienced thrombus changes during the first 7-10 days of anticoagulant therapy compared to the non-change patients. The reason may be related to the long onset time. large thrombus, and complicated with deep vein thrombosis (DVT). In addition, some patients were in bed after PE, which would also affect thrombolysis and thrombus disappearance.
However, this study has certain limitations. First, the retrospective design conducted at a single center limits the representativeness of the sample. Therefore, further research involving a larger, prospective patient cohort from multiple institutions, including comprehensive medical histories and long-term follow-up, is necessary to validate these preliminary findings. Second, high-risk PE patients were excluded because they required thrombolysis therapy after diagnosis, which may have influenced changes in coagulation and fibrinolysis function. Third, the timing of the transient D-dimer increase after anticoagulant therapy was not fixed, preventing us from analyzing D-dimer values consistently across different time points, and making it impossible to construct a dynamic change curve for D-dimer. Additionally, the degree of thrombus occlusion was measured using the PVOI method, and thrombus volume was not quantified, potentially leading to bias in detecting slight changes in thrombus size.
Conclusions
After initiation of anticoagulant therapy in some PE patients, large thrombi in the main pulmonary arteries break down into smaller clots and enter each branch of pulmonary artery. At the same time, D-dimer of these patients would also show a transient increase with thrombus changes, and the rate of thrombolysis and thrombus disappearance at 30 days was higher compared to the non-change patients. Therefore, transient D-dimer increase in the early stage after anticoagulant therapy could predict thrombus dissolution at 30 days in PE patients.
Footnotes
Acknowledgments
This study received support from the Shanghai Sixth People's Hospital Affiliated to Jiao Tong University School of Medicine Hospital-level project Fund (grant number: ynhg202317).
Ethical Considerations
The study received approval from the Medical Ethics Committee of Shanghai Sixth People's Hospital, Shanghai Jiao Tong University (No.: 2023-KY-138(K)). This study conforms to the principles of the Declaration of Helsinki. Informed consent was not required owing to the retrospective nature of the study.
Author Contributions/CRediT
Danrong Yang: Conceptualization, Design, Investigation Resources, Writing-original draft. Yi Liu: Investigation, Data curation, Software, Funding acquisition,Supervision, Writing-review & editing, Validation. Yan Zhang: Investigation, Data curation, Software, Funding acquisition,Supervision, Writing-review & editing, Validation. All authors read and gave final approval of the version to be published.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study received support from the Shanghai Sixth People's Hospital Affiliated to Jiao Tong University School of Medicine Hospital-level project Fund (grant number: ynhg202317). This study was approved by the Ethics Committee of Shanghai Sixth People's Hospital, Shanghai Jiao Tong University. All procedures involving human participants were conducted in accordance with the ethical standards of the institutional and national research committee and adhered to the principles outlined in the 1964 Helsinki Declaration and its subsequent amendments or comparable ethical standards, and informed consent was waived due to its retrospective design.
Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
The data and materials supporting the results or analyses presented in this paper are available upon reasonable request.
