Abstract
Background
Intravenous thrombolytics are essential for achieving timely reperfusion in acute ischemic stroke (AIS), with alteplase historically serving as the standard of care. Emerging alternatives like recombinant human prourokinase (rhPro-UK), reteplase, and tenecteplase offer potential improvements in efficacy, safety, and convenience, necessitating a comparative analysis.
Methods
Electronic databases, including PubMed, ScienceDirect, and Cochrane Central, were comprehensively searched from inception till December 2024 for relevant studies. A frequentist network meta-analysis was performed using R software version 4.2.3, and the “netmeta” package was employed. Alteplase 0.9 mg served as the reference group, with P-scores employed to determine the relative rankings of various interventions. The risk of publication bias was evaluated through funnel plots and Egger's regression test.
Results
Eighteen trials with 12,950 participants were included in the final analysis. Compared to alteplase 0.9 mg, excellent functional outcome (mRS 0-1) was significantly improved by Reteplase 18 + 18 mg (RR = 1.13, p < 0.01) and Tenecteplase (TNK) 0.25 mg (RR = 1.05, p < 0.01). For a good functional outcome (mRS 0-2), Reteplase 18 + 18 mg (RR = 1.06, p < 0.01) and TNK 0.32 mg (RR = 1.30, p < 0.01) were significantly more effective than alteplase. Safety outcomes, symptomatic intracranial hemorrhage (sICH), and mortality were not significantly different between alteplase and other thrombolytics. According to P-scores, Reteplase 18 + 18 mg ranked the best for excellent functional outcome (P-score = 0.89) and TNK 0.32 mg for good functional outcome (P-score = 0.99), while rhPro-UK 35 mg ranked the best for sICH (P-score = 0.89).
Conclusion
Reteplase 18 + 18 mg and TNK 0.32 mg demonstrated superior functional outcomes compared to alteplase, while rhPro-UK 35 mg showed the best safety profile with the lowest sICH risk.
Introduction
Stroke is one of the principal drivers of long-term disability and healthcare resource utilization while also representing the second most prevalent cause of mortality globally.1–3 Ischemic stroke constitutes approximately 87% of all stroke incidences. 4 Acute ischemic stroke (AIS), characterized by sudden neurologic dysfunction due to focal brain ischemia with imaging evidence of acute infarction, affects approximately 700,000 individuals in the United States.5,6 The attainment of reperfusion via intravenous thrombolysis is the most efficacious intervention in the settings of AIS.7–9 Administration of thrombolytic medicines within a few hours of stroke onset facilitates thrombus resolution, thereby safeguarding optimal nerve function and cerebral perfusion. 10
The guidelines set forth by the American Heart Association (AHA)/ American Stroke Association (ASA) recommend the administration of intravenous thrombolysis with alteplase within 4.5 h of symptom onset in eligible patients with AIS. 11 Alteplase, an intravenous recombinant tissue plasminogen activator (IV rt-PA), is the standard Food and Drug Administration (FDA) approved thrombolytic for patients with AIS. 11 It constitutes a contracted incidence of bleeding complications owing to its ability to selective plasminogen activation.12,13 However, utilization of alteplase is impeded due to its limitations, including limited plasma half-life and complex dosage regimen that involves the administration of an hour-long perfusion following the initial bolus. 14 Alteplase also constitutes a restrained fibrinolytic potential, which is why recanalization is achieved in less than half of the patients, with only 50% attaining the desired outcome within 2 h of treatment. 13
Due to alteplase's stated limitations, using tenecteplase as an alternative and a third-generation version of alteplase gained traction. 15 The various amino acid substitutions enable tenecteplase to harbor a 14-times higher fibrin specificity, 80-times higher resistance to plasminogen activator inhibitor-1 (PAI-1), and longer plasma half-life, eventually permitting a single bolus dosing regimen in contrast to alteplase. 16 Tenecteplase is an FDA-approved medication for acute ST-segment elevation myocardial infarction, with a lower incidence of systemic hemorrhage and a similar mortality incidence to alteplase. 17 The utilization of tenecteplase among patients with AIS is not a licensed thrombolytic therapy, but various studies have demonstrated non-inferiority of functional outcomes with tenecteplase18,19 compared to alteplase. In contrasting evidence, tenecteplase has been reported to be associated with a higher risk of symptomatic intracranial hemorrhage (sICH). 20
The evidence with varying conclusions regarding the comparative efficacy of tenecteplase in contrast to alteplase 21 mandated the manufacturing of reteplase, which is an alternative, cost-effective, non-glycosylated rt-PA variant of alteplase. Due to its structural modifications, including the absence of N-terminal fibronectin, kringle 1 domain, and epidermal growth factor, reteplase features an increased half-life and allows a double-bolus approach with a fixed dosage. 18 It constitutes effective penetration into the thrombus, albeit with inferior fibrin specificity compared to alteplase. 22 It is conventionally used for the treatment of acute myocardial infarction (AMI). 23 A phase 2 randomized trial that employed reteplase as an off-label thrombolytic demonstrated similar efficacy of reteplase compared to alteplase. In addition, the higher dosing criterion of reteplase was not associated with an elevated risk of fatal hemorrhagic complications. 9 In congruence, a phase 3 randomized trial comparing reteplase and alteplase illustrated that the treatment group receiving reteplase was associated with a higher incidence of excellent functional outcomes. However, the incidence of any intracranial hemorrhage (ICH) was elevated with reteplase in comparison to alteplase. 23
Amidst the inconclusive establishment of clinical superiority of the thrombolytics mentioned above, the recombinant human prourokinase (rhPro-UK) therapy confers a reduced risk of haemorrhagic complications as compared to other thrombolytic agents.24–26 This is primarily due to its capacity to avoid forming covalent complexes with serum protease inhibitors.24,25 Prourokinase introduces thrombolysis primarily in the thromboembolic thrombus and exhibits no influence on hemostatic thrombus. 27 The PROACT II trial that included patients with AIS due to the middle cerebral artery (MCA) occlusion illustrated that intra-arterial administration of prourokinase within −6 h of symptom onset was associated with elevated incidence of favorable functional outcomes compared to placebo. However, it also showed an increased incidence of intracranial hemorrhage and worsening neurological outcomes. 28 In contrast, the DUMAS trial demonstrated that prourokinase with small bolus alteplase was not superior to alteplase administered as monotherapy in patients with AIS. 29 The most recent PROST −2 trial illustrated that prourokinase administered within 4·5 h of stroke onset corresponded with excellent functional outcomes and lower bleeding complications, including sICH. However, no difference in mortality was observed between the two treatment groups. 30
To synthesize well-powered estimates, we conducted a systematic review and network meta-analysis to evaluate the comparative efficacy of alteplase, tenecteplase, reteplase, and prourokinase in patients with AIS. We synthesized cumulative evidence by constructing pragmatic rankings of the mentioned thrombolytic agents, potentially enhancing functional outcomes in the clinical management of AIS. While highlighting the gaps in the current research paradigm, we enumerated a review of existing literature and highlighted future research avenues to augment translational impact. By establishing direct and indirect evidence, this paper aims to mitigate physicians, clinicians, and policymakers to incorporate evidence-based management protocols, thereby optimizing the healthcare models concerning stroke management and expanding the incidence of favourable patient-related outcomes.
Methods
This systematic review and network meta-analysis were conducted according to the Preferred reporting items for systematic reviews and meta-analysis (PRISMA)
31
and the
Literature Search:
We comprehensively search electronic medical databases, including PubMed, ScienceDirect, and Cochrane Central, from inception till December 2024. In addition, we searched Clinicaltrials.gov for any unpublished trials. The following medical subject headings (Mesh) terms and keywords were used: “Alteplase,” “Tenecteplase,” “Reteplase,” “stroke,” “Prourokinase,” and “ischemic stroke.” The bibliographies of the included studies and related review articles were also searched manually for any additional studies. The detailed search strategy is provided in Supplementary Table S1
Study Selection and Eligibility Criteria
Following a thorough database search, all retrieved articles were imported into EndNote version 20, and any duplicate studies were eliminated. Two independent reviewers screened the titles and abstracts of the remaining articles against pre-determined eligibility criteria. The full-text screening of the remaining articles followed this. Any discrepancies were resolved by discussing them with a third author. The inclusion criteria were: (1) Population: Patients with acute ischaemic stroke; (2) Interventions: Studies involving alteplase (tPA), reteplase (rPA), tenecteplase (TNK), and recombinant human prourokinase (rhPro-UK) at various doses; (3) Study design: Randomized controlled trials. The following studies were excluded: (1) studies assessing thrombolytic agents after a 4.5-h window; (2) articles in a language other than English; (3) case-control studies, cohorts, case reports, letters, and review articles.
Data Extraction and Outcomes
Two independent authors extracted data on the variables of interest from the included studies using a pre-designed Microsoft Excel sheet. Any disagreements were settled through discussions involving a third author. Data extracted included the study characteristics (First author name, year of publications, sample size, study location or country, and study design), patient characteristics (age, gender, thrombolytic regimen, and different comorbidities including diabetes, hypertension, smoking status, and baseline NIHSS score), and outcomes of interest. The primary and secondary outcomes of interest were excellent functional outcome (mRS 0-1) at 3 months, symptomatic intracranial haemorrhage (sICH), good functional outcome (mRS 0-2) at 3 months, poor functional outcome (mRS 5-6) at 3 months, mortality at 3 months, and major neurological improvement within 72 h. The functional outcomes were defined using the modified Rankin scale (mRS), a clinical assessment tool ranging from 0 to 6, which was used to measure the degree of neurological impairment following a stroke or other neurological disorder. The outcome of sICH was determined based on the definitions set forth by the individual trials.
Quality Assessment
The risk of bias in the included studies was assessed using the Cochrane risk of bias tool for randomized clinical trials (RoB 2.0) 33 by two independent authors. Any disagreements were resolved by a third author. The RoB 2 tool assesses the quality of the included trials in five domains: (1) bias arising from the randomization process, (2) bias due to deviations from intended interventions, (3) bias due to missing outcome data, (4) bias in the measurement of the outcome, and (5) bias in the selection of the reported result. The risk of bias in the included studies is graded as low, high, or uncertain for each domain.
Statistical Analysis
A frequentist network meta-analysis employing the random effects model was performed using the
Results
Search Results
Searching different electronic databases, including PubMed (n = 710), ScienceDirect (n = 2372), and Cochrane Central (n = 474), retrieved 3556 articles. After removing the duplicates (n = 450), the remaining articles underwent title and abstract screening. This was followed by a full-text screening, resulting in 18 trials being included in the final analysis. The details of the study selection process are provided in the PRISMA diagram Figure 1.

PRISMA Diagram for the Process of Study Selection.
Characteristics of the Included Studies:
Eighteen RCTs with 12,950 patients were included in the final analysis. The studies were conducted between 2010 and 2024, with sample sizes varying from 104 to 1777. The mean age of the population ranged from 60 to 73 years. Five trials took place in China, three in Australia, two in Norway, one in Canada, the UK, the USA, and Scotland, while one was multicentre. The doses of tenecteplase administered were TNK 0.1 mg, TNK 0.25 mg, TNK 0.32 mg, and TNK 0.4 mg, with 0.25 mg being the most frequently used. Alteplase 0.9 mg was the most commonly used dose of alteplase and served as the reference group. The detailed characteristics of the included studies are presented in Table 1.
Characteristics of the Included Studies.

Risk of Bias Assessment
The Cochrane Risk of Bias tool (RoB 2.0) was utilized to assess the quality of the included randomized clinical trials. Most of the studies included demonstrated a low risk of bias. One of the included studies showed some concern for the domain of bias in the measurement of the outcome, and three studies9,34,35 showed some concern for deviations from the intended intervention. Overall, most of the included studies were considered good quality. Supplementary Figures S1 and S2
Outcomes
Excellent Functional Outcome (mRS 0-1)
Excellent functional outcome at 3 months (mRS 0-1) was reported in 18 studies with 12,808 patients. Compared to Alteplase, excellent functional outcome was significantly improved with Reteplase 18 + 18 mg (RR = 1.13, 95% CI: [1.06, 1.20]; p < 0.01) and TNK 0.25 mg (RR = 1.05, 95% CI: [1.01, 1.10]; p < 0.01). No significant difference was observed for the other treatments (Figures 2A and 3A). The network diagram is shown in
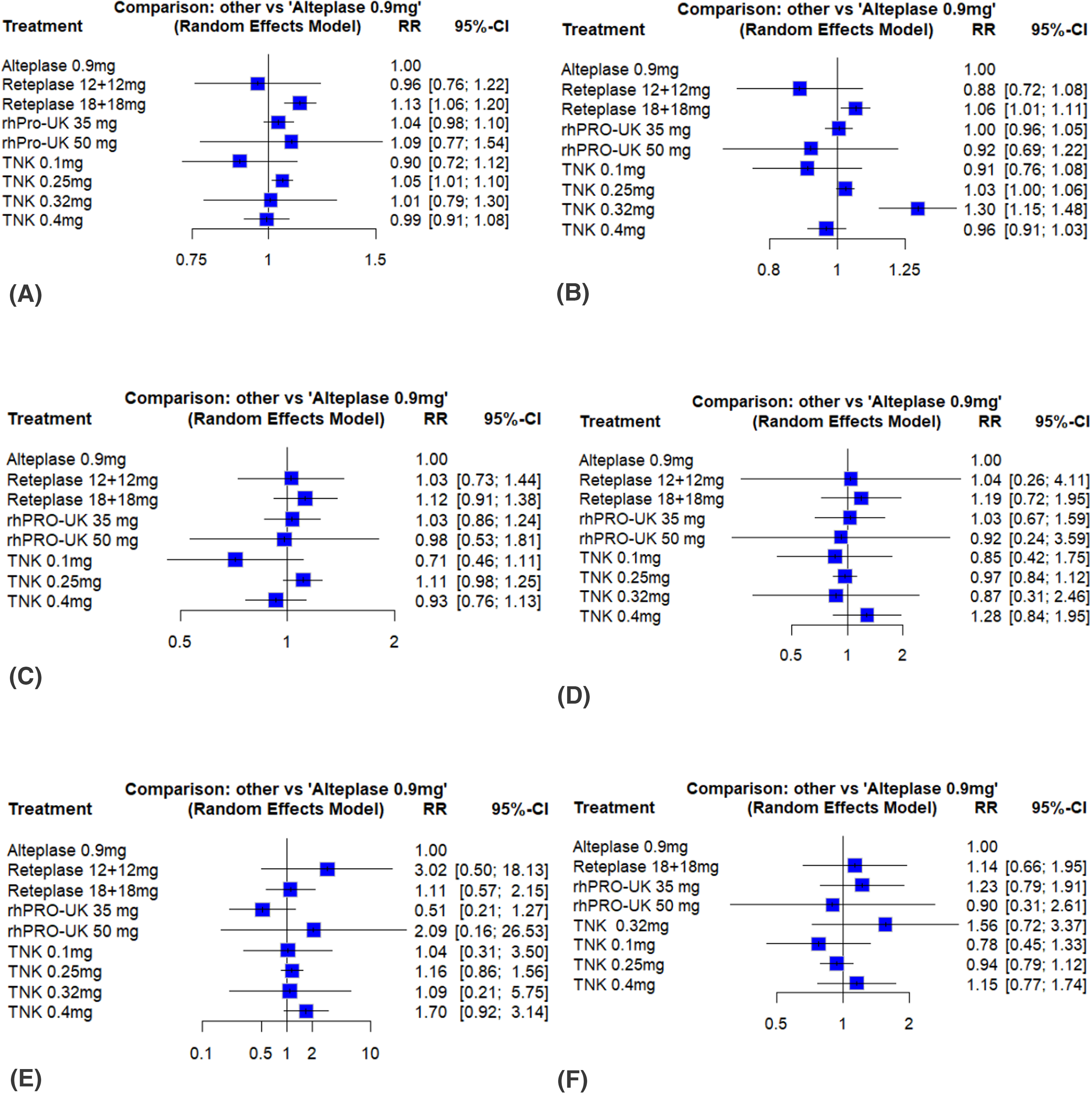
Forest Plots

League Tables for Network Estimates
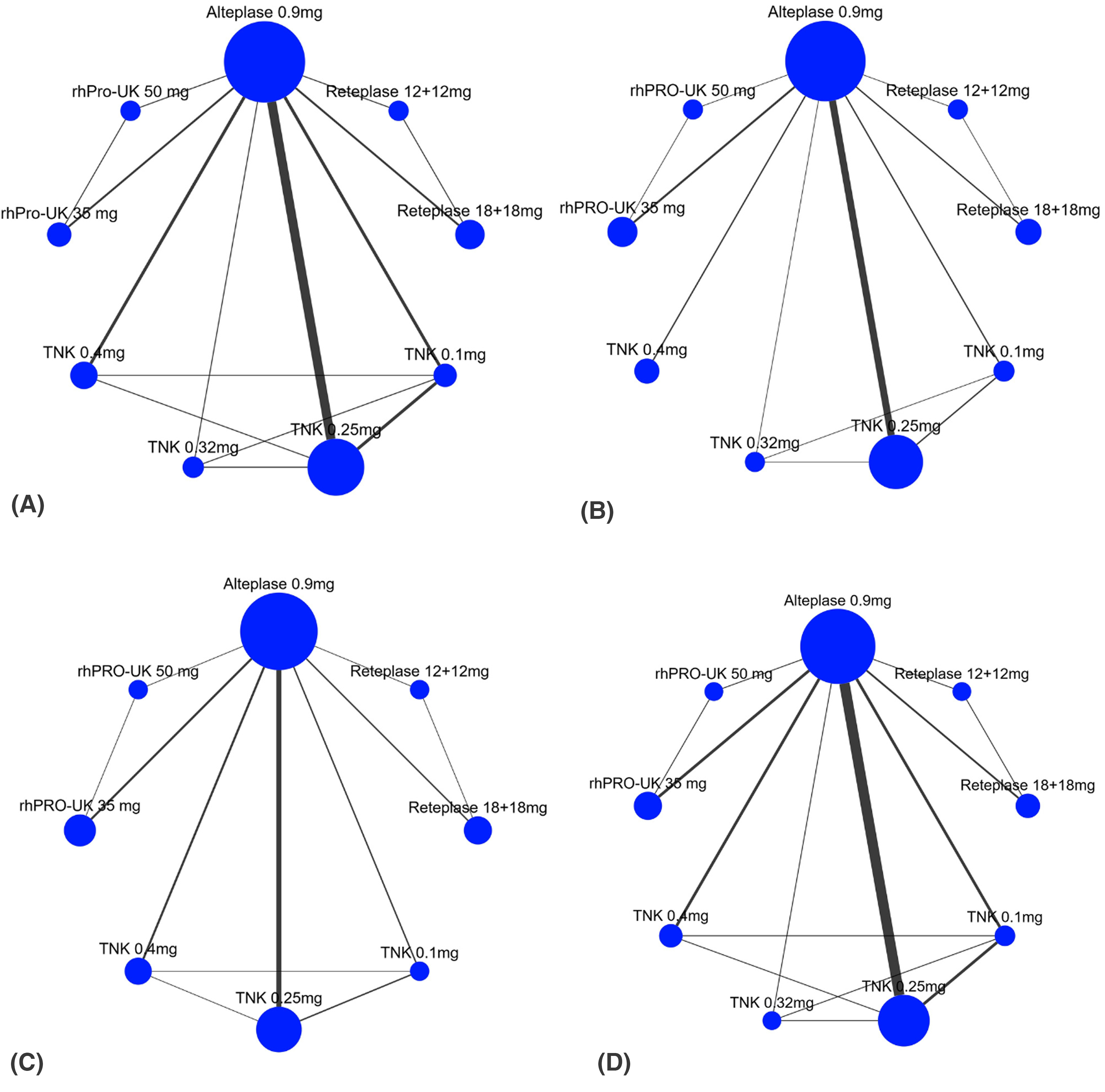
Network Evidence Plots
The P-Score Ranking of Different Interventions.

Good Functional Outcome (mRS 0-2)
Seventeen trials with 12,679 patients and nine treatments reported good functional outcome at 3 months (mRS 0-2). Compared to alteplase, good functional outcome was significantly better with Reteplase 18 + 18 mg (RR = 1.06, 95% CI: [1.01, 1.11], p = 0.01) and TNK 0.32 mg (RR = 1.30, 95% CI: [1.15, 1.48]; p < 0.01). The remaining interventions showed no significant difference (Figures 2B and 3B). The network plot is shown in Figure 4B. The heterogeneity between the studies was low (tau < 0.0001, I2 = 30%). Among the nine interventions, TNK 0.32 mg was ranked the best (P-score = 0.99), and Reteplase 12 + 12 mg was ranked the worst (P-score = 0.17) for improving good functional outcomes (Table 2).
Major Neurological Improvement Within 72hrs
In 14 trials with 9,57 patients, major neurological improvement within 72 h was reported. The network meta-analysis showed no significant difference between the treatments (Figures 2C and 3C). The network evidence diagram is shown in Figure 4C. The heterogeneity between the studies was moderate (I2 = 52%). Among the eight treatments, the probability of major neurological improvement was highest with Reteplase 18 + 18 mg (P-score = 0.92) and lowest with TNK 0.1 mg (P-score = 0.03) (Table 2).
All-Cause Mortality
All-cause mortality at 3 months was reported in 18 trials with 12,881 patients. Compared to alteplase, none of the interventions showed any statistically significant difference (Figures 2D and 5A). The network diagram is shown in Figure 4D. The between-study heterogeneity was low (I2 = 24%). TNK 0.1 mg was ranked as the best treatment (P-score = 0.65), and TNK 0.4 mg as the worst (P-score = 0.25) (Table 2).

League Tables for Network Estimates
Symptomatic Intracranial Hemorrhage
Seventeen trials analyzed the sICH at 3 months involving 12,827 patients. The network meta-analysis showed no significant difference between the various thrombolytic agents for sICH (Figures 2E and 5B). The network evidence plot is shown in Figure 6A. The heterogeneity between the studies was low (I2 = 0%). According to the P-score ranking, rhPRO-UK 35 mg had the lowest sICH risk (P-score = 0.89), while Reteplase 12 + 12 mg had the highest risk (P-score = 0.18) (Table 2).

Network Evidence Plots
Poor Functional Outcome (mRS 5-6)
Poor functional outcome at 3 months was reported in 17 trials with 12,633 patients. Compared to alteplase, the risk of a poor functional outcome was not significantly different with any interventions (Figures 2F and 5C). The network evidence diagram is shown in Figure 6B. The heterogeneity between the studies was moderate (I2 = 37%). Among the eight interventions, the risk of poor functional outcome was lowest with TNK 0.1 mg (P-score = 0.82) and highest with TNK 0.32 mg (P-score = 0.15) (Table 2).
Publication Bias and Inconsistency
The risk of publication bias was assessed visually through funnel plots and statistically through Egger's regression test. Upon inspecting the funnel plots visually, publication bias was found to be non-significant for all outcomes. Egger's regression test further confirmed this finding. Details are provided in the Supplementary Figure S3-S8
Discussion
To our knowledge, it is the first and most extensive network meta-analysis that systematically evaluates and compares the prourokinase, reteplase, TNK, and ALT for their efficacy and safety in AIS. Prourokinase (rhPro-UK), Reteplase, and Tenecteplase (TNK) offer several advantages over Alteplase, including greater fibrin specificity and reduced systemic bleeding risks.36,37 Their longer half-lives allow bolus administration instead of continuous infusion, improving convenience. Also, their long-shelf half-lives make their storage relatively easy. Additionally, TNK and Reteplase resist plasminogen activator inhibitor-1 (PAI-1) and may achieve better reperfusion rates and functional outcomes. 38 An additional benefit of Prourokinase is its price, which is half the price of r-tPA. 39
This network metanalysis included 18 trials comparing the clinical outcomes among doses of four thrombolytics (Prourokinase, Reteplase, Tenecteplase, and Alteplase). The network meta-analysis showed Reteplase 18 + 18 mg and TNK 0.32 mg as the most effective treatments for functional outcomes (mRS 0-1) at 3 months compared to alteplase. Reteplase 18 + 18 mg showed the highest probability of achieving excellent and good functional outcomes, while TNK 0.32 mg significantly improved good outcomes. No significant differences were observed for poor functional outcomes, all-cause mortality, symptomatic intracranial hemorrhage, or major neurological improvement within 72 h. rhPro-UK 35 mg had the lowest risk of sICH, and TNK 0.1 mg showed the lowest risk of poor outcomes and mortality.
Our network meta-analysis showed that functional outcomes at 3 months were significantly higher in the Reteplase 18 + 18 and Tenecteplase 0.25 mg (excellent functional outcome) and Reteplase 18 + 18 and Tenecteplase 0.32 mg (good functional outcome). The findings are consistent with the recently published network meta-analysis. 40 Although prourokinase did not show significantly higher functional outcomes compared to reteplase and TNK, multiple trials have demonstrated the non-inferiority of prourokinase compared to alteplase in achieving functional outcomes.9,28,39 Contrary to one of the meta-analyses, the major neurological improvement within 72 h showed that Reteplase 18 + 18 and Tenecteplase 0.1 mg showed significantly higher efficacy than other regimens. 41 Regarding symptomatic intracerebral haemorrhage, our network meta-analysis showed no significant difference between all the options. Similar findings were reported by the latest network meta 40 and other meta-analyses42,43 to the P-score ranking, rhPRO-UK 35 mg had the lowest sICH risk, while Reteplase 12 + 12 mg had the highest risk. This contradicts the latest published network meta, which linked 0.1 mg/kg TNK to higher S-ICH risk. 40 In contrast, Wu et al found no increased risk, and Srisurapanont et al noted higher TNK doses increased S-ICH and parenchymal haemorrhage risk.42,44 Our study showed no significant difference considering all-cause mortality among these different thrombolytics. Other meta-analyses reported similar findings.45,46
Future Direction and Recommendations:
Future research should focus on large-scale, multicenter randomized controlled trials to confirm this network meta-analysis's findings and address the identified knowledge gaps. Specifically, further studies are needed to explore the optimal dosing strategies for recombinant human prourokinase (rhPro-UK), reteplase, and tenecteplase, as well as to assess their long-term safety profiles, particularly concerning sICH and parenchymal hemorrhage. Furthermore, head-to-head trials that directly compare these thrombolytics in varied populations and real-world settings could yield more substantial evidence to support clinical decision-making. Special attention should be given to assessing these agents in environments with logistical challenges to ascertain their practical advantages, such as bolus administration and storage stability. Moreover, cost-effectiveness analyses should be emphasized to investigate the economic implications of using rhPro-UK as a lower-cost alternative to alteplase, particularly in healthcare settings with limited resource systems. Ultimately, incorporating advanced imaging techniques in future studies could aid in identifying patients who are most likely to benefit from specific thrombolytics, thereby further personalizing stroke treatment management.
Limitations
While this network meta-analysis offers valuable insights into the comparative efficacy and safety of various thrombolytic agents, several limitations must be recognized. Firstly, the study is based on data from a range of randomized controlled trials (RCTs) that exhibit variations in population characteristics, stroke severity, and thrombolytic administration protocols, potentially introducing heterogeneity despite statistical adjustments. Secondly, the analysis encompasses a limited number of studies for specific thrombolytic regimens, such as higher doses of tenecteplase and prourokinase, which may impact the robustness of the findings. Furthermore, the study's exclusion of non-English articles and non-RCT designs may have omitted pertinent data. Lastly, the focus on short-term outcomes, such as functional recovery at three months and major neurological improvement within 72 h, constrains the generalizability of the findings to long-term stroke management outcomes. Future research addressing these gaps through large-scale, standardized trials and long-term follow-up is essential to validate and expand on these conclusions.
Conclusion
Reteplase 18 + 18 mg showed significant improvement in the efficacy outcomes, such as excellent functional outcome (mRS 0-1) and good functional outcome (mRS 0-2). TNK 0.32 mg also significantly improved good functional outcomes compared to alteplase. All-cause mortality, major neurological improvement, and poor functional outcome (mRS 5-6) were comparable between different interventions. For the safety outcome of sICH, rhPro-UK 35 mg was ranked as the best treatment. More high-quality randomized clinical trials are required to confirm these findings.
Supplemental Material
sj-docx-1-cat-10.1177_10760296251334563 - Supplemental material for Head-to-Head: Recombinant Human Prourokinase Versus Intravenous Thrombolytics in Acute Ischemic Stroke Within 4.5 Hours – A Systematic Review and Network Meta-Analysis of Randomized Clinical Trials
Supplemental material, sj-docx-1-cat-10.1177_10760296251334563 for Head-to-Head: Recombinant Human Prourokinase Versus Intravenous Thrombolytics in Acute Ischemic Stroke Within 4.5 Hours – A Systematic Review and Network Meta-Analysis of Randomized Clinical Trials by Muhammad Hassan Waseem, Zain ul Abideen, Areeba Shoaib, Muhammad Osama, Muhammad Abdullah Ali, Sania Aimen, Muhammad Wajih Ansari, Rowaid Ahmad, Muhammad Arslan Tariq, Ameer Haider Cheema, Aleeza Afzal and Pawan Kumar Thada in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
