Abstract
Objective
Recurrent deep venous thrombosis (RDVT) is a relatively frequent phenomenon; however, scarce literature is available on late RDVT (>2 years). This study aimed to investigate the incidence, associated risk factors, and characteristics of late RDVT.
Methods
We conducted a retrospective analysis of patients diagnosed with symptomatic lower extremity deep venous thrombosis (DVT) between April 2017 and April 2022. The incidence and mortality rates of late RDVT, as well as the demographic data, risk factors, and characteristics, were assessed. Additionally, a comprehensive Cox regression analysis was performed to evaluate 14 potential risk factors.
Results
Of the 638 patients with DVT, 89 (13.9%) experienced recurrence during a mean follow-up period of 30.10 ± 15.23 months. The RDVT group comprised 50 males and 39 females (the male-to-female ratio: 50/39), with a mean age of 64.08 ± 13.23 years (the age range: 26-88 years). Among them, early recurrence was observed in 57 (64%) patients, while late recurrence occurred in 32 (36%) after their initial symptomatic DVT episode. Late RDVT predominantly manifested in the contralateral lower extremity. Multivariate Cox regression analysis further identified proximal DVT, immobility, non-retrieved inferior vena cava filter (IVCF), and ≥50% stent extension into the inferior vena cava (IVC) as significant risk factors for late RDVT. Moreover, mortality among patients with late RDVT was low, with 2 (6.25%) dying during the follow-up period.
Conclusions
Late RDVT is relatively infrequent within the RDVT cohort and primarily manifests in the contralateral lower extremity. Furthermore, patients with late RDVT have a low mortality rate. Proximal DVT, immobility, non-retrieved IVCF, and ≥50% stent extension into the IVC are strongly correlated with the development of late RDVT.
Introduction
Recurrence after the first episode of symptomatic deep venous thrombosis (DVT) is common following anticoagulation cessation, especially in patients with unprovoked DVT. 1 RDVT has been reported to have a cumulative prevalence of 16% (13%-19%) at 2 years after treatment, 25% (21%-29%) at 5 years, and 36% (28%-45%) at 10 years, with 4% of these RDVT events resulting in death in the unprovoked DVT population. 2 Although extended anticoagulation is highly effective at reducing RDVT risk, 3 it can increase the risk of bleeding and elevate health care costs. A previous study reported that direct oral anticoagulant use led to a clinical bleeding incidence of 6.60%-10.24% and a major bleeding incidence of 1.1%-4.0%. 4
Furthermore, RDVT prevalence was found to increase gradually after anticoagulation discontinuation, even in patients with provoked DVT. Moreover, relapse incidence in provoked DVT after discontinued anticoagulation was shown to be 3.3% per patient-year up to 2 years; however, no definite data have been published on late RDVT. Several risk factors for RDVT have been identified, such as malignancy, inherited thrombophilia, poor anticoagulation compliance, and residual venous obstruction. 5 These risk factors appear to be associated with early RDVT, with limited related information on late RDVT. Therefore, this study aimed to investigate the incidence, characteristics, and risk factors of late RDVT.
Materials and Methods
Study Design and Population
This study was approved by our institutional review board, and informed consent was obtained from all included patients. All consecutive patients with symptomatic lower-extremity DVT who received anticoagulant therapy, thrombolysis, or percutaneous mechanical thrombectomy in our institution between April 2017 and April 2022 were initially identified. Anticoagulant therapy, thrombolysis, or percutaneous mechanical thrombectomy for all patients with DVT is determined according to the guidelines of the American College of Chest Physicians (ACCP). Patients with mental disease, conscious disorders, or hearing/speech impairments who could not accurately describe their thrombosis history and those lost to follow-up were excluded. The study flowchart is illustrated in Figure 1. Data was primarily collected from a centralized electronic medical record system containing information on every outpatient and inpatient visit, along with findings from imaging modalities (including ultrasound, computed tomography, and magnetic resonance imaging) and laboratory tests. The demographic characteristics and comorbidities of the patients, such as age, sex, course, interval to recurrence, diabetes, hypertension, peripheral artery disease, and congestive heart disease, were also collected. The following variables were retrospectively analyzed to identify the risk factors of late RDVT: malignancy; inherited thrombophilia; autoimmune disease (including systemic lupus erythematosus, rheumatoid arthritis, or nephrotic syndrome); poor anticoagulation compliance (details provided below); preexisting inferior vena cava filter (IVCF); previous stent position (categorization information described below); untreated iliac vein compression syndrome (IVCS); and immobilization. According to the established guidelines,6,7 thrombophilia tested (including antithrombin deficiency, protein C or S deficiency, factor V Leiden mutation, prothrombin 20210A mutation, and methylenetetrahydrofolate reductase (MTHFR) C677 T mutation) is recommended for patients with a history of venous thromboembolism (VTE), those under 40 years old, or patients with a first episode of VTE associated with pregnancy or oral contraceptive use. Lastly, information on the site of index DVT, treatment modalities, and a composite of death from venous thromboembolism-related causes or other causes was also obtained.

Study Flow Chart. DVT, Deep Venous Thrombosis.
Follow-up
All patients underwent scheduled follow-up, including physical and ultrasound examinations, at 3 and 6 months, with subsequent annual follow-ups. Additionally, all patients were followed up through outpatient visits or telephone calls at study initiation. A total of 96 patients were excluded from the cohort because of the lack of complete follow-up of at least 2 years.
Definitions
RDVT was primarily diagnosed via ultrasonography, with the diagnostic criterion being the detection of a new noncompressible vein in either lower limb or at least a 4-mm increase in the diameter of a previously affected vein segment.8,9 Residual venous obstruction (RVO) was characterized by the persistence of a thrombus occupying at least 40% of the lumen after 3 months of anticoagulation therapy, as well as untreated iliac vein stenosis. 10 Patients with poor anticoagulation compliance were categorized as those with provoked DVT receiving anticoagulation therapy <3 months, those with unprovoked DVT who required extended anticoagulation therapy and were undergoing anticoagulation therapy for <6 months, and those with International Normalized Ratio (INR) values out of the therapeutic range (2-3) at least twice and receiving warfarin anticoagulation and irregular medication during anticoagulation. 6 Stent extension into the IVC was defined as the extension of one side of the common iliac vein stent to some extent into the IVC to cover the contralateral common iliac vein outflow. 11 Proximal DVT was established as a thrombus in the popliteal vein and more proximal locations, while distal DVT was classified as a thrombus in the veins distal to the popliteal vein. 12
Stent Position Categorization
According to venography, stent extension into the IVC was classified into the following three categories: 11 (1) confined to the ipsilateral iliac vein, (2) extended to the IVC with 0%-50% coverage, and (3) extended to the IVC with 50%-100% coverage. A diagram illustrating the three categories of stent extension into the IVC is provided in Figure 2.

Diagram Illustrating the Following Categorization of the Stent. (a) Confined to the Ipsilateral Iliac Vein. (b) 0-50% Coverage. (c) 50%-100% Coverage.
Statistical Analysis
Statistical analysis was performed using SPSS version 22.0, and p < 0.05 was considered statistically significant for all comparisons. Risk factors of late-phase recurrence were investigated. Patient data were censored at the time of death or the last available follow-up. Categorical data were presented as absolute numbers and percentages. Univariate and multivariate Cox regression analyses were conducted to identify the risk factors for late RDVT.
Results
Basic Demographics
A total of 744 patients were diagnosed with lower-extremity symptomatic DVT between April 2017 and April 2022 in our hospital, but patients with mental disease (n = 4), conscious disorders (n = 4), or hearing/speech impairments (n = 2) who could not accurately describe their thrombosis history and those lost to follow-up (n = 96) were excluded. Of the 638 recruited patients with a first episode of symptomatic DVT, RDVT was observed in 89 (13.9%) during a median follow-up of 30.10 ± 15.23 months (range: 2-58 months). The baseline demographic and clinical characteristics of this study population are summarized in Table 1. The RDVT group comprised 50 males and 39 females (the male-to-female ratio: 50/39). Of the 89 patients with RDVT, 57 (64%) and 32 (36%) experienced early and late RDVT, respectively, after the initial episode of symptomatic DVT. Late RDVT was relatively rare compared to early RDVT (p < 0.05). Table 2 summarizes the demographics and clinical characteristics of the early and late RDVT groups. Late RDVT was frequently observed in the contralateral lower extremity (P < 0.05).
The Baseline Demographic and Clinical Characteristic of Study Population.

PAD, peripheral artery disease; CHD, congestive heart disease; DVT, deep venous thrombosis; IVCS, iliac vein compression syndrome; CDT, catheter-directed thrombolysis;IVCF, inferior vena cava filter; NOACs, new oral anticoagulants; RVO, residual venous obstruction.
Demographic Data and Clinical Characteristics in the Early and Late Recurrence Groups.

DVT, deep venous thrombosis; PAD, peripheral artery disease; CHD, congestive heart disease.
Risk Factors Associated with Late RDVT
A total of 14 risk factors underwent Cox regression analysis, and the results are presented in Table 3. The univariate analysis showed that the risk factors of late RDVT were proximal DVT (P < 0.05), diabetes (P = 0.012), inherited thrombophilia (P = 0.029), non-retrieved IVCF (P < 0.05), stent extension (≥50%) (P = 0.024), poor coagulation compliance (P = 0.002), and immobility (P = 0.033). Conversely, age, gender, left lower extremity, malignancy, autoimmune disease, RVO, and IVCS were not significant risk factors for late RDVT (P > 0.05). The previously mentioned seven variables with statistical significance during univariate analysis were further subjected to multivariate Cox regression analysis to identify the statistically significant risk factors for late RDVT. The results revealed independent associations between late RDVT and the following four out of the seven variables: proximal DVT (HR, 0.304; 95% CI, 0.146-0.633; p = 0.001), immobility (HR, 0.319; 95% CI, 0.118-0.864; p = 0.025), non-retrieved IVCF (HR, 4.600; 95% CI, 2.140-9.887; p < 0.05), and stent extension (≥50%) (HR, 3.526; 95% CI, 1.228-8.634; p = 0.018).
Univariate and Multivariate Cox Regression for Risk Factors Associated with Late Recurrent Deep Venous Thrombosis.
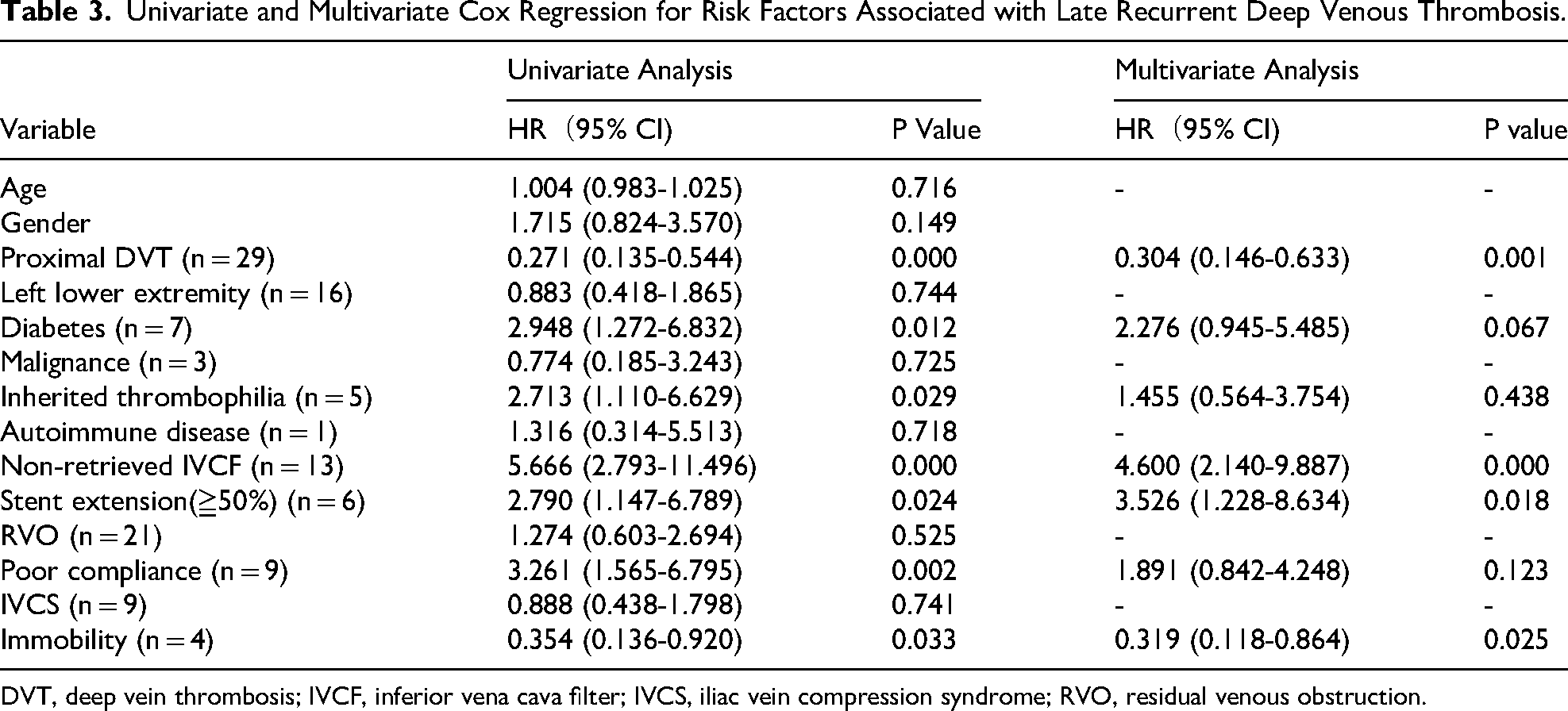
DVT, deep vein thrombosis; IVCF, inferior vena cava filter; IVCS, iliac vein compression syndrome; RVO, residual venous obstruction.
Mortality
Of the 32 patients with late RDVT, two died during the follow-up period, which resulted in a mortality rate of 6.25%. Heart failure was the cause of death in one patient, while trauma was the cause in the other. However, neither patient died from pulmonary embolism (PE) or anticoagulation-associated bleeding.
Discussion
In our study, the recurrence rate after the first episode of symptomatic DVT was 13.9%, aligning with the results from previous studies.2,13 Among the 89 patients with RDVT, 32 (36%) exhibited late RDVT. This relatively small proportion of late RDVT was similar to that reported in a recent individual patient data meta-analysis of 7515 patients. 2
The present study also revealed that late RDVT was frequently localized on the contralateral side, potentially due to the high incidence of stent extension (≥50%) in this cohort. Stent extension (≥50%) has been shown to be associated with a greater tendency toward the development of late RDVT compared with early RDVT (HR, 2.790 and 1.510, respectively). 14 Furthermore, our multivariate Cox regression analysis confirmed that stent extension (≥50%) was an independent risk factor for late RDVT. This finding can be attributed to the “jailing” phenomenon, which is a venous flow disturbance in the contralateral vein owing to an overextended stent that has been found to be a risk factor for RDVT. 15 Stent extension into the contralateral iliac vein wall or IVC wall can induce intimal hyperplasia at the contact point, which then impairs the contralateral venous flow. 15 Moreover, the continuous growth of venous intimal hyperplasia typically requires considerable time to reach the level needed to hinder contralateral venous flow. This factor may explain the late occurrence of contralateral RDVT. These findings further highlight the pivotal role of venous flow disorders in late contralateral RDVT following the overextension of iliac vein stent placement.
A previous study reported a 4.8-fold higher recurrence in patients with proximal DVT than in those with isolated distal DVT. 16 Similarly, our study revealed that patients with late RDVT had a significantly higher previous history of proximal deep veins than distal deep veins, suggesting that proximal DVT was a risk factor for late RDVT. Relevant literature has also demonstrated a higher risk of proximal thrombosis extension and post-thrombotic syndrome in proximal DVT than in distal DVT. 17 Proximal thrombosis extension and post-thrombotic syndrome may damage the venous valve and block venous return, leading to RDVT; however, this process may occur over a lengthy duration. These results underline the strong link between proximal DVT and late RDVT.
Although DVT treatment using IVCF implantation can effectively prevent PE occurrence, 18 a permanent IVCF or long-term non-retrieval of IVCF may increase thrombosis risk in the lower limb veins and the IVC. 19 Furthermore, the retrieval rate of IVCF has been reported to be as low as 8.5%. 18 Another study also showed that the recurrence rate of DVT in patients with permanent IVCF implantation or long-term non-retrieval of IVCF was higher than that in the retrieval group. 20 Long-term IVCF placement may cause numerous problems, such as damage to the IVC wall and vena cava narrowing after thrombus capture by the IVCF, which all affect venous return in the lower limbs. 13 The obstruction of venous return and eventual thrombosis recurrence in the lower limbs caused by the long-term use of the IVCF is a process that requires considerable time, thus emphasizing the link between the non-retrieval of IVCF and late RDVT.
Immobility is a prominent risk factor for DVT, 21 with previous literature highlighting its strong association with DVT recurrence in the lower extremity. 22 In our study, 4 (12.5%) patients with late RDVT experienced immobility, which was in line with the results of Prandoni et al in 2007. 23 Our multivariate regression analysis further demonstrated that immobility was strongly associated with late RDVT (P < 0.05).
A prior study established that patients with RDVT have a higher mortality risk (HR, 2.42; 95% CI, 1.75-3.36) than those with non-recurrent DVT. 24 However, only 2 (6.25%) patients with late RDVT died during our study period, which was a significantly lower mortality rate than that observed among those with RDVT in previous studies. 22 This outcome could be because previous RDVT studies did not distinguish between early and late RDVT, whereas our investigation focused solely on late RDVT. Malignancy has been shown to be an independent risk factor for DVT relapse within 6 months of the initial event, 13 underscoring the strong association between malignancy and early RDVT. Concurrently, an earlier study showed that patients with DVT and malignancy have a relatively higher mortality. 25 All these findings suggest that malignancy is a major contributor to increased mortality risk in patients with early RDVT. However, our study had an extremely limited number of patients with late RDVT accompanied by malignancy, and our univariate Cox regression analysis indicated no significant association between late RDVT and malignancy (HR, 0.774; 95% CI, 0.185-3.243; p = 0.725). This result may explain the low mortality rate in patients with late RDVT.
The main strength of this study was that this research is the first to elucidate the incidence, risk factors, and clinical characteristics of late RDVT in the general population. However, the current study has several limitations that should be considered. First, this study was a single-center retrospective analysis, and the number of included patients with RDVT was small. Second, some patients were inadequately followed up, thus hampering the precise determination of the time to recurrence. Third, the large number of patients lost to follow - up may have led to the mortality rate of late RDVT in our research results being lower than the actual rate. Finally, the patients with DVT received different anticoagulant drugs; however, we did not analyze the influence of varied anticoagulant drugs on recurrent DVT.
Conclusions
Late RDVT is relatively infrequent within the RDVT cohort and more likely to occur in the contralateral lower extremity. Moreover, patients with late RDVT have a low mortality rate. Proximal DVT, immobility, non-retrieved IVCF, and ≥50% stent extension into the IVC are independent risk factors for late RDVT. Lastly, the optimal duration of anticoagulation therapy requires further investigation according to the risk factors and clinical characteristics of late RDVT.
Footnotes
Acknowledgements
No Acknowledgement.
Ethical Considerations
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
Author Contributions/CRediT
Lili Zhang conceived and designed the studies; Haiyan Yan, Jianming Sun and Haiyang Wang analyzed the data; Haoyuan Wang prepared the tables; Bo Wu and Jian Fu co-wrote the paper. All authors reviewed and approved the manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Natural Science Foundation of Chongqing, China (grant number CSTB2023NSCQ-MSX0723).
Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
