Abstract
Background
Hemophilia A (HA) occurs due to the deficiency of factor VIII (FVIII). Individuals with HA generally present with elevated activated partial thromboplastin time (aPTT) and normal prothrombin time (PT). The only possible treatment for this bleeding condition is factor concentrate.
Aim
The aim of this study is to compare the effect of recombinant factor VIII (rFVIII) and recombinant factor VII (rFVII) on prothrombin time (PT), activated partial thromboplastin time (aPTT), FVIII and FVII in severe HA.
Methodology
A mixing study was conducted on 30 samples of severe HA patients to assess the correction of PT, aPTT, FVIII, and FVII values using biosimilars of rFVIII (NovoEight and Kogenate FS) and rFVII (NovoSeven and AryoSeven) using a fully automated coagulation analyser ‘Ceveron alpha’.
Results
All the four drugs demonstrated a significant alteration for both PT (P < .0001) and aPTT (P < .0001) values. A significant, notable increase of FVIII levels were observed for FVIII biosimilars. Further investigation into the effect of rFVII biosimilars on inhibitor-positive patients revealed a significant alteration in PT (P < .0001) and aPTT (P < .001) values.
Conclusion
To the best of our knowledge, this is the first study to compare the effect of all four drugs on PT, aPTT, FVIII, and FVII. Two different groups of biosimilars were found to have a high potential to alter the PT and aPTT values. The FVIII biosimilars are efficient in increasing the FVIII levels.
Introduction
Patients with congenital hemophilia having defective coagulation processes, experience a high risk for severe bleeding complications. Hemophilia A (HA) is characterized by a deficiency or absence of factor VIII (FVIII) in the blood. 1 The severity of HA is classified based on the residual activity levels of the coagulation factor, which is largely influenced by genetic modifications. Severe, moderate and mild hemophilias are characterized by factor levels of below 1 IU/dL, between 1 and 5 IU/dL, and more than 5 IU/dL but less than 40 IU/dL respectively. 2 Diagnostic evaluation for hemophilia is undertaken in cases of known family history, excessive bleeding to minor injuries, or an elevated activated partial thromboplastin time (aPTT). If the complete blood count (CBC) and prothrombin time (PT) are normal but the aPTT is elevated, hemophilia is strongly suspected, necessitating coagulation factor testing. 3 The primary bleeding issue in hemophilia is hemarthrosis, which predominantly occurs in severe cases and most frequently affects the elbows, knees, and ankles. 4 Repeated episodes of hemarthrosis can lead to the development of hemophilic arthropathy, leading to pain, disability, and decreased quality of life. 5 Bleeding episodes are managed by replacing the deficient coagulation factor FVIII to reach levels sufficient for effective hemostasis.
Routine prophylactic treatment with FVIII is the major current therapeutic approach to treat bleeding episodes and prevent joint damage in patients with severe HA. 6 With FVIII's short half-life of about 12 h, prophylaxis for severe HA demands frequent intravenous infusions, posing a considerable burden on patients. The development of recombinant FVIII (rFVIII), having an extended half-life, improved patient management by reducing the doses and minimizing the risk of blood-borne pathogens.7,8 Kogenate-FS, a second-generation full length rFVIII, is the human rFVIII protein produced from the genetically engineered baby hamster kidney (BHK) cells, and NovoEight is manufactured by the Chinese hamster ovary (CHO) cell line without the use of animal plasma belongs to the fourth generation FVIII concentrate with a truncated B domain.9,10
Although recombinant protein replacement therapy improves the quality of life, in approximately 25%-30% of severe HA cases, inhibitors develop against FVIII, complicating the management of bleeding episodes. 11 Administering FVIII concentrate is ineffective in these patients because their antibodies neutralize the infused coagulation factors. Bypassing agents (BPAs) are the main treatment options currently available for patients with inhibitors. BPAs achieve hemostasis by using pathways that bypass the need for FVIII to produce thrombin. 12 The currently available BPAs for the HA patients with inhibitors are activated recombinant factor VII (rFVIIa) and activated prothrombin complex concentrate (aPCC). Both rfVIIa and aPCC have shown approximately 80% hemostatic efficacy in patients with hemophilia and inhibitors in various clinical situations. 13 rFVIIa, marketed as NovoSeven and AryoSeven is approved in many countries globally for treating spontaneous and surgical bleeding in patients with hemophilia and inhibitors, as they are structurally similar to plasma-derived activated factor VII (pdFVIIa). 14 The increased activated factor X (FXa) production on the platelet surface, which results from the hemostatic effectiveness of exogenous rFVIIa, occurs regardless of the presence of FVIII. This increase in FXa leads to higher thrombin production, which in turn enhances platelet aggregation, fully activates thrombin-activatable fibrinolysis inhibitor (TAFI) and factor XIII and forms a stable fibrin clot.15–17 rFVIIa is produced by introducing the human factor VII (FVII) gene into BHK cells and cultured in bovine albumin media. As a recombinant product, it is not sourced from human or animal plasma, eliminating the risk of transmitting blood-borne diseases, and it is reported to have a shorter half-life (∼2 h). AryoSeven is a biosimilar to NovoSeven with comparable efficacy and safety profiles. 17
Previous studies reported that rFVIIa can directly activate FX on a negatively charged phospholipid surface without the presence of tissue factor (TF). 18 However, following studies demonstrated that rFVIIa competes with the inactive form of FVII for binding to TF. This competition enhances its procoagulant activity by forming TF–FVIIa complexes rather than TF–FVII complexes.19,20 A mechanism suggested by Monroe et al involving the surface of activated platelets, where rFVIIa binds, could explain how rFVIIa's activity is localized to the site of injury. 20
The current study was designed to compare the effects of biosimilars of rFVIIIa (Kogenate FS and NovoEight) and biosimilars of rFVIIa (NovoSeven and AryoSeven) on PT, aPTT, FVII, and FVIII in severe HA patients.
Materials and Methods
Study Subjects
This study involved 30 patients with severe HA whose diagnosis was confirmed through coagulation factor assays indicating FVIII levels below 1%. Patients with FVIII levels greater than 1% were excluded from the study. Laboratory findings demonstrated normal platelet counts and PT, but a prolonged aPTT. All participants provided written informed consent, and sample collection adhered to the principles of the Declaration of Helsinki. Study protocols were thoroughly reviewed and approved by the Human Ethics Committee of the All India Institute of Medical Sciences (AIIMS), New Delhi (EC No. IEC 426).
Sample Collection
Peripheral blood samples (10 ml each) were collected from all the participants using BD vacutainer tubes containing 3.2% sodium citrate (Becton Dickinson and Company, USA). Clinical characteristics including age, sex, onset, and frequency of bleeding episodes, family history as well as transfusion frequency, were documented for each patient. The samples were centrifuged at 80×g for 10 min at room temperature to obtain platelet-rich plasma (PRP), and the remaining samples were again centrifuged at 800×g for 10 min at room temperature to obtain platelet-poor plasma (PPP). Processing of all samples was completed within 4 h of collection. The resulting supernatants were rapidly frozen at −80 °C until needed for further use.
Reagents
NovoEight and NovoSeven were purchased from Novo Nordisk, Denmark; AryoSeven was acquired from AryoGen, Iran, and Kogenate FS was purchased from Bayer, USA. According to the product insert, NovoSeven and AryoSeven contained 1.2 mg and 1.0 mg of rFVIIa, respectively. However, NovoEight and Kogenate FS have 2000 IU of rFVIIIa. Both rFVIIa and rFVIIIa drugs were diluted in the given diluent to obtain a desirable concentration. Further dilutions were made using saline to prepare working concentrations of 10 ug/mL (NovoSeven and. AryoSeven) and 10 IU/ml (NovoEight and Kogenate FS). Table 1 represents the reconstitution details of all the drugs. All other chemicals used were of molecular biology grade.
Reconstitution of Biosimilars of rFVIII and rFVII. [rFVIII: Recombinant Factor VIII; rFVII: Recombinant Factor VII].
Measurement of PT, aPTT, FVIII and FVII
The samples were analyzed using a fully automated coagulation analyzer, the Ceveron Alpha (Technoclone, Austria), to measure PT, aPTT, FVIII, and FVII levels after the addition of the drug solutions. PT is the duration, measured in seconds (s), for plasma to clot after thromboplastin (a combination of TF, calcium, and phospholipid) is added to plasma samples. It is one of the most valuable diagnostic tests used to evaluate the coagulation status of patients. In healthy conditions, PT value ranges from 9 to 13 s.21,22 aPTT identifies the activity of coagulation factors involved in the intrinsic and common pathways of the coagulation cascade. Coagulation is triggered by incubating plasma with a contact activator, phospholipid, and calcium, and the time for clot formation is recorded in s. In normal conditions, the aPTT value ranges from 25 to 35 s. 23 FVIII plays a crucial role in promoting and maintaining hemostasis following an injury, as it is essential to the coagulation cascade. It acts as a vital cofactor, significantly enhancing the production of FXa and, subsequently, thrombin. 24 FVII is a coagulation factor that takes part in the extrinsic coagulation cascade along with TF. In the presence of TF and calcium ions, activated factor VII (FVIIa) activates FX to FXa and factor IX (F1X) to factor IXa (FIXa). 25
Statistical Analysis
The data was analysed using IBM SPSS Statistics Version 22.0 (SPSS Inc, Chicago, IL, USA) and summarized according to their statistical parameters. Quantitative data were presented as mean ± standard deviation (SD), while qualitative data were shown as frequency and percentage. To compare quantitative variables across study groups, a one-way ANOVA test was applied for data with normal distribution, and a Kruskal-Walli's test was used for non-normally (non-parametric) distributed data. Pearson's correlation was used to assess relationships between variables with a normal distribution, while Spearman's rank correlation was applied for variables with a non-parametric data. Statistical significance was indicated by *P < .05, **P < .01, ***P < .001, and ****P < .0001.
Results
Subjects Characteristics
In this comparative study, 30 HA patients were included. The mean age was 21.4 ± 9.9 years (range: 7-44 years). Twenty-two patients were reported a family history of the condition. Although most were diagnosed early, three patients showed symptoms after the age of five, and five HA patients developed inhibitors. All patients had normal platelet counts and PT, but their aPTT was prolonged (Table 2).
Characteristics of Severe HA Patients. [HA: Hemophilia A; N: Number; FVIII: Factor Eight; s: Seconds; SD: Standard Deviation; PT: Prothrombin Time; aPTT: Activated Partial Thromboplastin Time; %: Percentage].

Results of PT, aPTT, FVIII and FVII in Drug-treated Samples
The mean PT value at the time of sample collection was found to be 13.9 ± 1.2 s. The NovoEight and Kogenate FS-treated samples displayed average PT values of 15.8 ± 1.8 s and 15.6 ± 1.8 s, respectively. Whereas the NovoSeven and AryoSeven,-treated samples displayed average PT values of 8.5 ± 0.73 s and 8.7 ± 0.81 s, respectively. During the sample collection, the mean aPTT value was 91.4 ± 12.1 s. After the addition of NovoEight and Kogenate FS, the mean aPTT values were 34.3 ± 13.8 s and 36.9 ± 15.5 s, respectively. The NovoSeven and AryoSeven-treated samples displayed the mean aPTT values of 58.66 ± 14.9 s and 55.1 ± 15.8 s, respectively. The FVIII levels of all the patients, at the time of sample collection, were below 1%. Following the treatment with drug, the mean FVIII level was increased to 146.3 ± 59.6% and 93.8 ± 36.01% for NovoEight and Kogenate FS, respectively. The FVIII levels in NovoSeven and AryoSeven- treated samples displayed 16.61 ± 25.1% and 20.8 ± 31.9%, respectively. The FVII levels were not recorded at the time of sample collection. However, after treating the plasma samples with the four drugs, FVII levels were measured. Both NovoEight and Kogenate FS produced average FVII values of 16.6 ± 17.1% and 88.8 ± 70.9%, respectively. In contrast, rFVII products resulted in average FVII levels of 491.4%.
Discussion
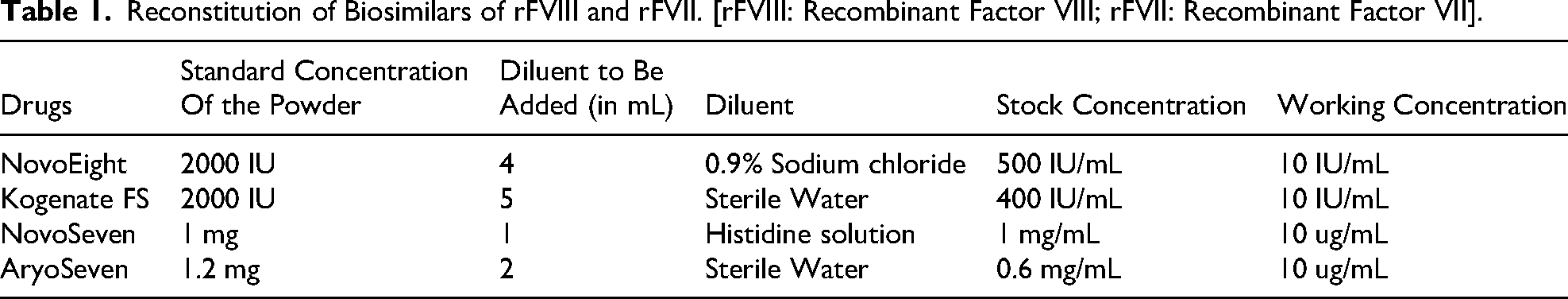
Severe HA is a genetic, recessive bleeding disorder that leads to a high risk of severe bleeding and is typically managed with infusions of either plasma-derived factor VIII (pdFVIII) or rFVIII. The development of rFVIII products aimed to address long-standing concerns over pathogen risks associated with blood-derived products. This study aims to examine the effects of the biosimilars on coagulation parameters, specifically PT, aPTT, FVIII, and FVII. NovoEight and Kogenate FS are widely recognized recombinant FVIII products globally, used to prevent and treat severe HA. We compared the impact of NovoEight and Kogenate FS on these coagulation parameters and observed significant changes in aPTT (P < .0001) and FVIII (P < .0001) values, along with a notable change in PT value (P < .0001) (Figure 1). The aPTT values were significantly shortened and the FVIII value was significantly increased upon adding both the rFVIII biosimilars. The increase of PT from 13.9 s in untreated samples to 15.8 s or 15.6 s in rFVIII-treated samples could be due to confounding factors, as it is known that rFVIII does not prolong the PT. The previous studies demonstrated that NovoEight and Kogenate FS are efficient in hemostatic response in controlling blood loss in severe HA patients undergoing surgery.26,27 In the current study, based on the observation in the drug-treated samples, we find that both the NovoEight and Kogenate FS are efficient in shortening the aPTT and increasing the level of FVIII in severe HA.
Comparative effect of NovoEight and Kogenate FS on PT, aPTT, and FVIII in severe HA. [PT: Prothrombin Time; aPTT: Activated Partial Thromboplastin Time; FVIII: Factor VIII; s: Seconds; %: Percentage; ****: P < .0001HA: Hemophilia A].
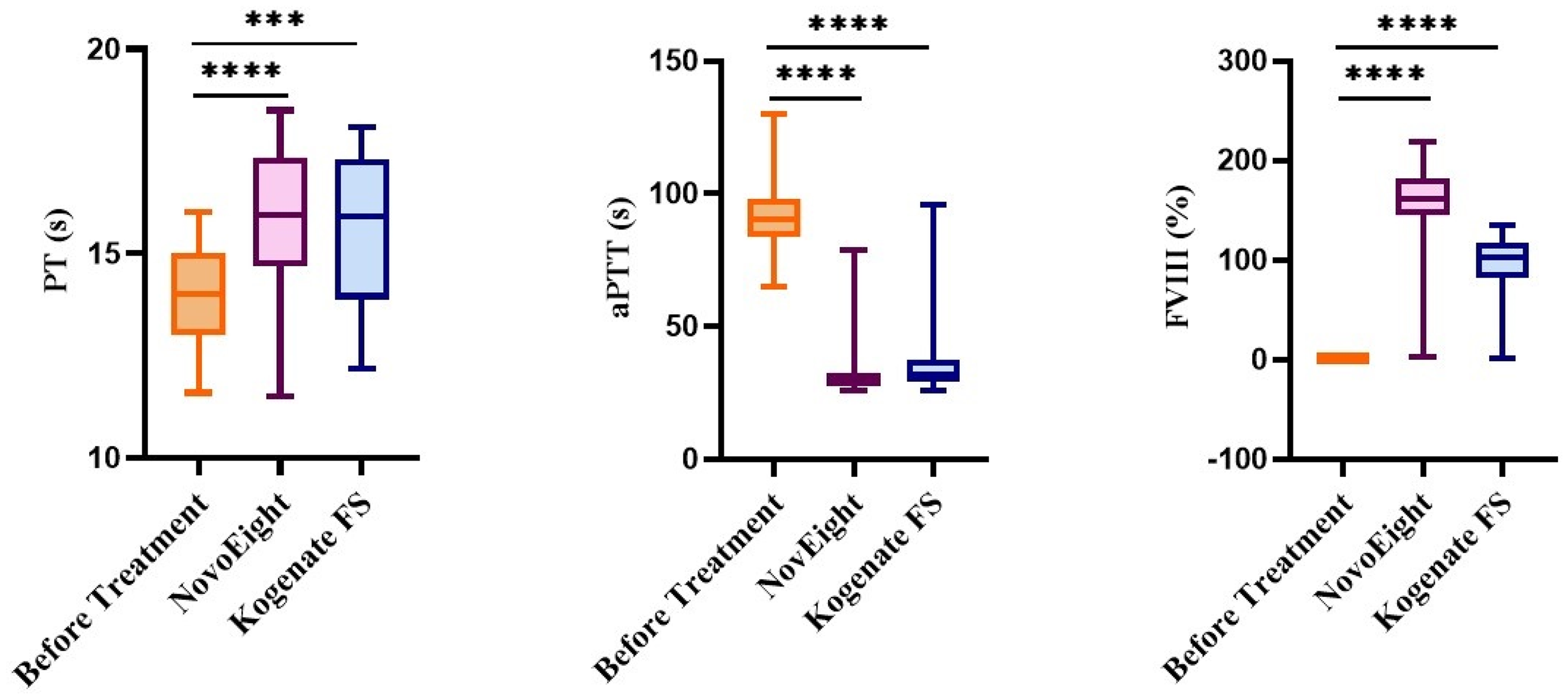
Though the rFVIII biosimilars improve the quality of life in severe HA patients, about one-third of patients with severe HA produce neutralizing alloantibodies, known as inhibitors, targeting rFVIII. Thus, the use of rFVII (NovoSeven and AryoSeven) for the treatment of severe HA patients with inhibitors has introduced a new approach. We observed shortened PT (P < .0001) and aPTT (P < .0001) values in NovoSeven and AryoSeven- treated samples (Figure 2). It is known that rFVII do not directly increase the FVIII level as it bypasses the need for FVIII in coagulation cascade. In our study population, we have observed a significant, but very minimal changes for FVIII levels in rFVII-treated samples. When compared to the rFVIII, the rFVII had very minimal impact on the FVIII level.

Comparative effect of NovoSeven and AryoSeven on PT, aPTT, and FVIII in severe HA. [PT: Prothrombin Time; aPTT: Activated Partial Thromboplastin Time; FVIII: Factor VIII; s: Seconds; %: Percentage; ****: P < .0001; HA: Hemophilia A].
Also, we specifically wanted to understand the potential changes of coagulation parameters following rFVII treatment in inhibitor-positive HA patients. Notably, significant alterations were observed in PT (P < .0001) and aPTT (P < .001) values. No significant changes were observed in FVIII level for both the NovoSeven and AryoSeven (Figure 3).
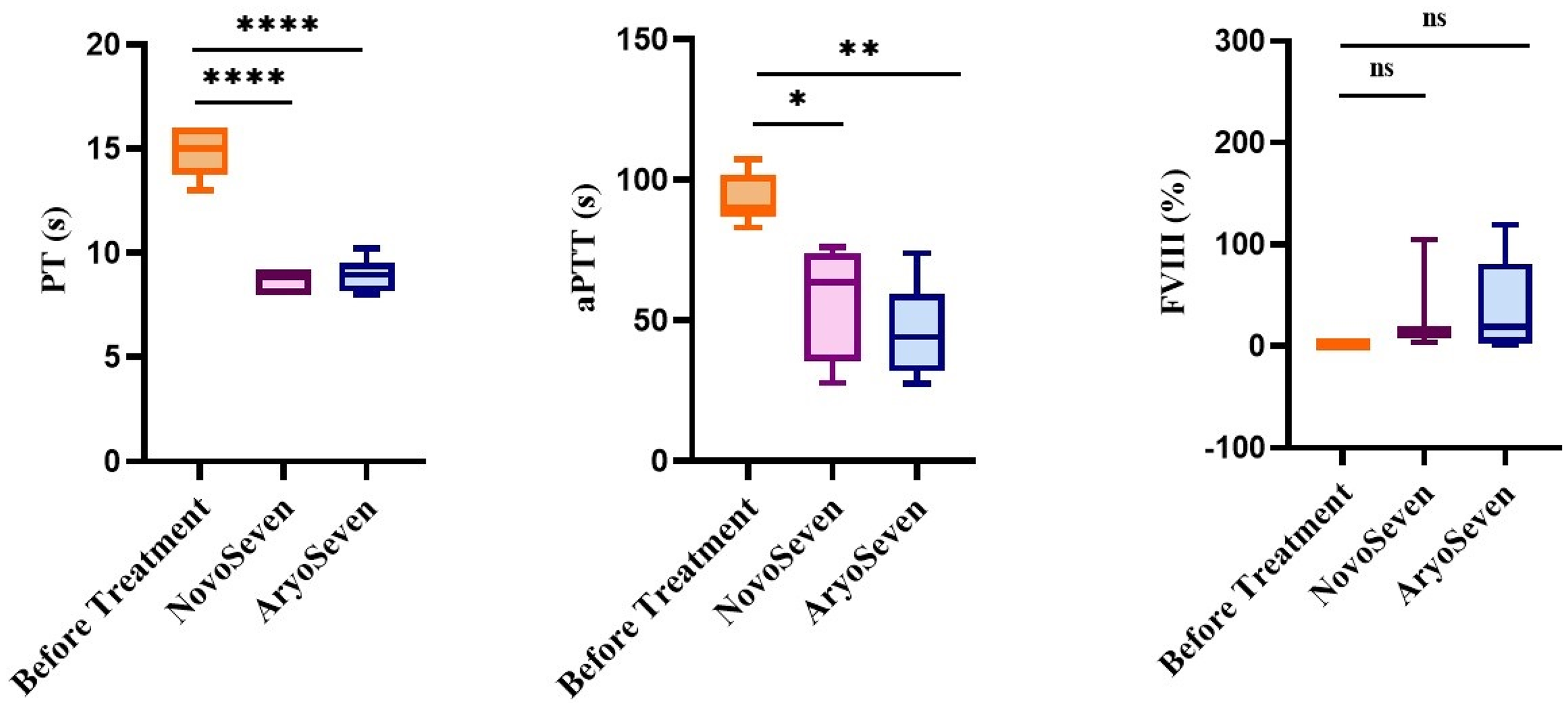
Comparative effect of NovoSeven and AryoSeven on PT, aPTT, and FVIII in severe HA patients with inhibitor. [PT: Prothrombin Time; aPTT: Activated Partial Thromboplastin Time; FVIII: Factor VIII; s: Seconds; %: Percentage; **: P < .01; ****: P < .0001; HA: Hemophilia A].
Additionally, we performed a correlation analysis to assess the relationship between coagulation parameters in severe HA patients treated with the four investigational drugs. For both the NovoSeven (r = −0.373) and AryoSeven (r = −0.042), we observed a negative correlation between aPTT and FVIII levels. With NovoEight, negative correlations were identified between FVIII and aPTT (r = −0.865). Similarly, Kogenate FS showed a negative correlation between FVIII and and aPTT (r = −0.898). No significant differences in efficacy were observed when comparing the NovoEight and Kogenate FS, nor among the rFVII biosmilars.
Conclusion
Our study demonstrates that both rFVIII biosimilars, NovoEight and Kogenate FS as well as rFVII biosimilars, NovoSeven and AryoSeven, effectively reduced aPTT values in severe HA patients. No significant differences in efficacy were observed between NovoEight and Kogenate FS, nor between NovoSeven and AryoSeven. To our knowledge, this study is the first to directly compare the effects of these four drugs on coagulation parameters, specifically PT, aPTT, FVIII, and FVII. Further research with larger cohorts is warranted to validate these results and to provide more detailed insights into the relative efficacy and safety profiles of these treatments.
Footnotes
Abbreviations
Acknowledgments
The authors thank the technical staff of the Central University of Tamil Nadu. We are also thankful to Ms. Erin Healy-Erickson for her skilful assistance in this manuscript
Authors’ Contributions
MK, MM, RS and JF designed the study. CLR and RR collected samples, performed experiments and collected data. CLR drafted the manuscript. PK revised the manuscript for important intellectual content. RS and MM supported the clinical data, interpreted and validated the manuscript. MK and JF supervised the project, interpreted the data and wrote the manuscript. All authors have approved the final article.
Consent to Participate
Informed consent had been taken from the participants of this study.
Consent for Publication
Consent for publication had been taken from all the contributing authors.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
All participants provided written informed consent and sample collection adhered to the principles of the Declaration of Helsinki. Study protocols were thoroughly reviewed and approved by the Human Ethics Committee of the All India Institute of Medical Sciences (AIIMS), New Delhi (EC No. IEC 426).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: MK is supported by the Department of Biotechnology (DBT), Government of India, India (Grant ID # BT/PR24078/MED/12/813/2018). CLR is supported by Indian Council of Medical Research (ICMR) (ID # 45/02/2022-HAE/BMS).
