Abstract
Introduction:
In hemophilia A, factor activity usually correlates with clinical severity; however, there are patients with severe hemophilia who have bleeding less than expected.
Aim:
The aim of this study is to evaluate the impact of prothrombotic mutations on annual factor consumption in children with hemophilia.
Methods:
Factor V Leiden (FVL) G1691A, Prothrombin (PT) G20210A and methylenetetrahydrofolate reductase (MTHFR) C677T and A128C mutations were evaluated in children with moderate–severe hemophilia A (n = 51) and controls (n = 25).
Results:
None of the cases and controls carried the FVL and PT G20210A in homozygous state. There was no difference in factor consumption between carriers of FVL, PT mutations, and noncarriers. Patients who were homozygous for MTHFR C677T were found to have increased factor consumption compared to noncarriers, and this was a negative association. No decrease in factor consumption was noted in patients with hemophilia having MTHFR A1298C mutation.
Conclusion:
We could not demonstrate a significant decrease in factor concentrate consumption in children with hemophilia having prothrombotic mutations.
Introduction
Hemophilia A is an X-linked hemorrhagic disorder resulting from factor VIII (FVIII) deficiency. Patients with FVIII levels <1% of normal are severely affected and may present with spontaneous hemorrhages or bleeding after trauma or surgery. Other patients with FVIII levels between 1% and 40% have usually a mild-to-moderate disease. However, the clinical severity of bleeding in patients with hemophilia may vary among patients with the same level of factor and even the same genetic mutation. 1 It is not clear why some patients with severe hemophilia may experience a milder bleeding tendency and other patients with moderate FVIII levels have a severe phenotype. A possible explanation for this phenotypic heterogeneity may be the coinheritance of prothrombotic gene mutations, however there are conflicting results in published studies so far. 1 –6
Factor V Leiden (FVL G1691A) and prothrombin G20210A (PT G20210A) mutations are the most common genetic deep vein thrombosis risk factors in Turkish population with frequencies of 9.8% and 2.7%, respectively. 7,8 Among Turkish patients with thromboembolism, FVL G1691A was detected in 21.5% and PT G20210A mutation in 9.2%. 9 The risk of thrombosis was found to be increased when these 2 mutations coexist. A common mutation in methylenetetrahydrofolate reductase (MTHFR C677T) has been shown to cause a predisposition to thrombosis by having synergy with FVL but has little effect on its own. 10
The aim of this study was to analyze the clinical impact of prothrombotic mutations (FVL G1691A, MTHFR C677T and A128C, and PT G20210A) on factor consumption in children with hemophilia A. Our hypothesis is that children with hemophilia having prothrombotic mutations would have decreased factor consumption due to decreased bleeding.
Patients and Methods
Study Population
Children with hemophilia A were recruited in the study, between January and December 2009. In all, 34 patients with severe hemophilia A (FVIII <1%) and 17 with moderate hemophilia A (FVIII 1%-5%) followed up at Cerrahpasa Medical Faculty, Department of Pediatric Hematology/Oncology and The Hemophilia Society of Turkey were included in the study. Table 1 shows the main demographic and clinical characteristics of the patients and controls. Prothrombotic mutations were screened in patients (n = 51) and controls (n = 25). The mean age at evaluation was 11.2 ± 5.4 years in patients and 11.2 ± 3.5 years in controls (P = .95). Patients' medical records and factor prescription booklets were retrospectively reviewed to obtain data on factor utilization. Average annual factor consumption per kilogram of body weight of the last 24 months was assessed. Factors used for elective interventional and surgical procedures were not taken into account. None of the patients or controls had a history of thromboembolic event. Twenty-two patients with hemophilia (43.1%) had a history of cardiovascular disease or thrombotic event in the family. There was no difference in prothrombotic mutation positivity between cases with a positive and negative family history of thrombotic event. Seven cases on prophylaxis had a history of central venous catheter; none of them had catheter thrombosis. All participating patients and parents gave written consent, and the protocol was approved by the local ethics committee.
Main Demographic and Clinical Characteristics of the Patients.
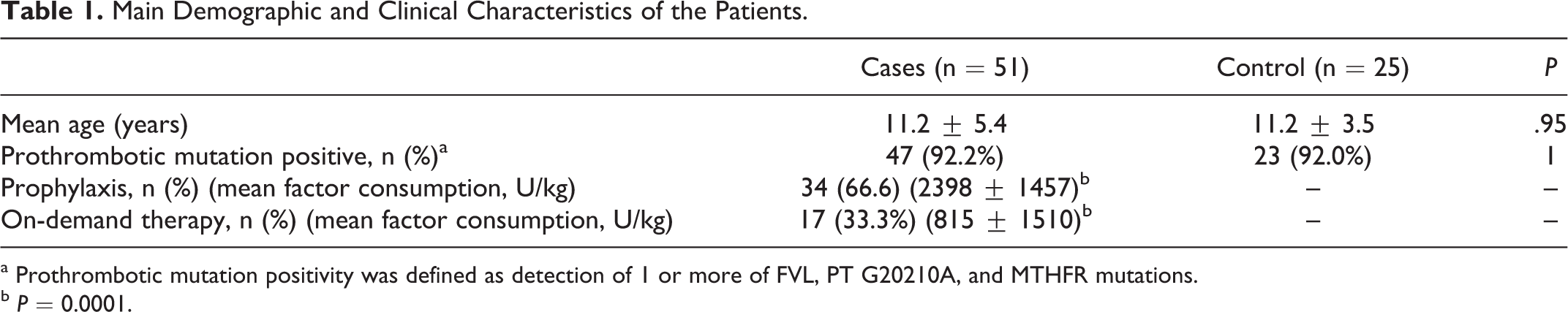
a Prothrombotic mutation positivity was defined as detection of 1 or more of FVL, PT G20210A, and MTHFR mutations.
b P = 0.0001.
Laboratory Analysis
Two milliliters of venous blood was obtained in ethylene diamine tetraacetic acid (EDTA)-containing sample tubes from each patient during outpatient clinic follow-up. Using standard techniques as previously described, DNA was extracted from blood samples and stored at 70°C. 11 Polymerase chain reaction (PCR) amplification for the detection of FVL, PT G20210A, and MTHFR mutations and visualization of the PCR products was carried out using a commercial assay (CVD StripASSAY, Vienna Lab, Vienna, Austria).
Statistical Analysis
All descriptive and comparative statistical analyses were carried out using SPSS 11 for Windows. According to the distribution of data, median (interquartile range [IQR]) and mean (standard deviation [SD]) were used for descriptive analysis. Differences in dichotomous variables were analyzed by Pearson chi-square test or Fisher exact test, and differences in continuous variables were analyzed by independent samples t test and Mann-Whitney U test. Wilcoxon signed ranks test was used for repeated comparisons. P < .05 was set as the level of statistical significance.
Results
Frequency of prothrombotic mutations in patients and controls are summarized in Table 1. None of the 51 cases and 25 controls carried the FVL and PT G20210A in homozygous state. FVL, PT G20210A, MTHFR C677T, and MTHFR A1298C heterozygote mutations were detected in 13.7% (n = 7), 3.9% (n = 2), 43.1% (n = 22), and 41.2% (n = 21) of the 51 patients with hemophilia, respectively. MTHFR C677T homozygote mutation was found in 11.8% (n = 6) and MTHFR A1298C homozygote mutation in 19.6% (n = 10; Table 2 ).
Prothrombotic Mutations in Cases and Controls.a
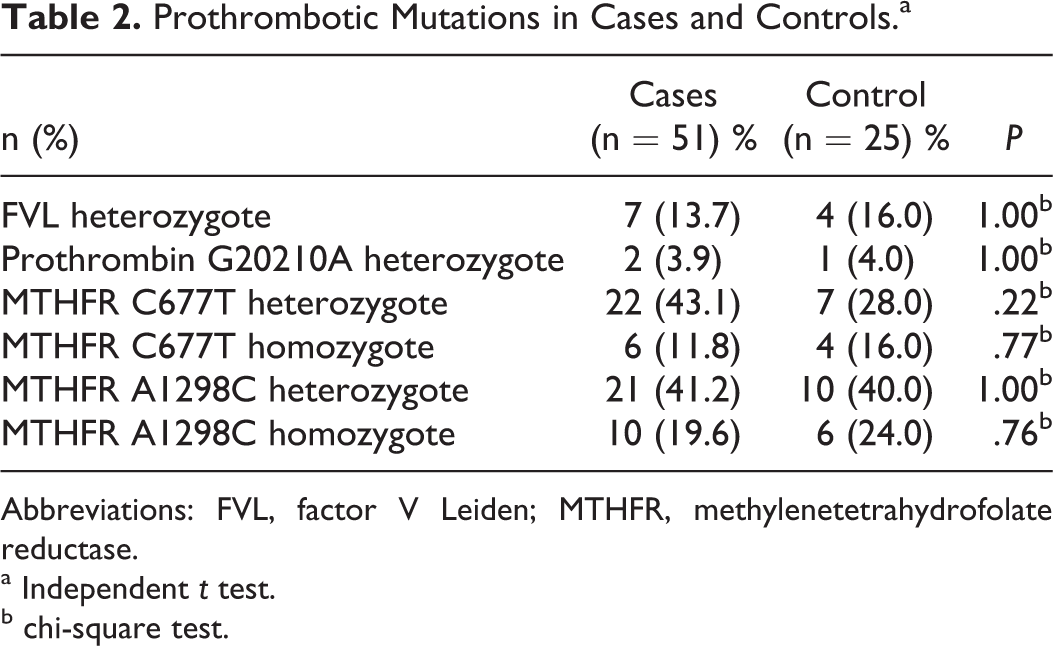
Abbreviations: FVL, factor V Leiden; MTHFR, methylenetetrahydrofolate reductase.
a Independent t test.
b chi-square test.
The mean annual factor concentrate consumption of hemophilia patients with and without a prothrombotic mutation was compared for each mutation (Table 3 ). There was only statistical difference in factor consumption between carriers of MTHFR C677T in homozygous state and noncarriers, but it was reverse of what was expected. Patients who were homozygous for MTHFR C677T were found to have increased factor consumption compared to noncarriers (P = .015).
The Mean Annual Factor Concentrate Consumption of Hemophilia Patients With and Without a Prothrombotic Mutation.

Abbreviations: FVL, factor V Leiden; MTHFR, methylenetetrahydrofolate reductase.
a chi-square test.
b Mann-Whitney U test.
Discussion
The assessment of clinical impact of prothrombotic mutations on hemophilia phenotype has been studied for different thrombophilic genetic factors. Factor V Leiden is the most commonly studied modifier. In individuals with FVL, there is a mutation in the FV gene (Arg 506 to Gln), which increases the thrombin levels leading to a procoagulant state. 12 Initially, it has been suggested that FVL may be an important modulator of disease severity in patients with hemophilia A. 4 Lee et al confirmed this data by showing a reduced factor concentrate use in carries of FVL mutation compared to noncarriers. 13 Ettingshausen et al investigated 124 children with hemophilia A and demonstrated that the first symptomatic bleeding onset occurred significantly later in life in patients carrying the FVL mutation than in noncarriers. 14 Later, these clinical observations were supported by the finding that FVL has the ability to improve hemophilia phenotype in mice. 15 Hemophilic mice heterozygous/homozygous for FVL was shown to have the ability to form clots at injured sites in contrast to normal hemophilic mice where no clot was observed. Another in vitro study showed that there is a 3- to 7-fold increased thrombin generation in FVIII-depleted plasma supplemented with FVL plasma. 16 In contrast, other groups failed to demonstrate the protective effect of FVL mutation in hemophilia. 17 –19 Chan et al could not demonstrate an association between FVL mutation and hemophilia phenotype. 17 In a study of 18 patients with severe hemophilia A but a milder bleeding phenotype, none of them was found to carry FVL mutation. 18 Ahmed et al could not show an increase in FVL mutations in patients with severe hemophilia having a milder phenotype. 19
The prevalence of other prothrombotic risk factors is less than FVL mutation, and there are a number of studies that investigated the effect of these mutations on bleeding phenotype. Kurnik et al found that carriers of FV and FII mutations had a lower annual bleeding frequency compared to noncarriers. 20 Shetty et al studied the common prothrombotic markers in the anticoagulant and fibrinolytic cascade like protein C, protein S, antithrombin III, PT G20210A, and MTHFR C677T and showed a higher prevalence of 1 or more of these factors in patients with severe hemophilia having a mild phenotype. 21 Tizzano et al investigated PT G20210A in patients with hemophilia A having intron 22 inversion, the carriers had significantly less spontaneous bleeding, FVIII utilization, and hemophilic arthropathy. 5 Ar et al studied the impact of FVL, PT G20210A, and MTHFR mutations on clinical phenotype of Turkish adults with hemophilia, they could not find an association of these mutations with clinical phenotype. 2 Recently, López-Jiménez et al determined the thrombotic mutations (FVL, PT G20210A, and MTHFR) in 257 Mexican patients with hemophilia. 22 Of the 7 patients positive for investigated mutations, 5 demonstrated attenuation in hemophilia-related bleeding manifestations mostly as decreased bleeding frequency. However, in individuals heterozygous for FVL or PT G20210A, there was no additional effect of MTHFR 677TT and C677T/A1298C genotypes on bleeding phenotype.
In Turkey, after government regulations on factor concentrate usage in patients with hemophilia, prophylaxis is the preferred treatment of choice for the last 6 to 7 years. In our center, since 2005, we use an early low-dose, low-frequency regimen for the newly diagnosed patients to decrease the cost without morbidity. 23 Patients who are already on demand therapy are switched to secondary prophylaxis when more than 2 bleedings occur in the same joint or more than 3 joint bleedings occur within a 6-month period. In this study, the factor consumption of the patients on prophylaxis was higher than the patients on demand therapy as it was expected because they are usually the patients who are bleeding more. The patients' prothrombotic risk was not known when prophylaxis or on-demand therapy was chosen. However, our survey is still limited by the inclusion of a heterogeneous group of patients. We found that patients who were homozygous for MTHFR C677T were found to have increased factor consumption compared to noncarriers. This was the reverse of what was expected, however 5 of 6 of MTHFR C677T carriers were on prophylaxis compared to 29 of 45 of noncarriers. So a negative association may be the result of anticipated high factor consumption in patients on prophylaxis compared to ones on demand therapy. Also, factors like different FVIII mutations and platelet hyperresponsiveness which we did not investigate might be effective. The other point is none of the patients carried the FVL and PT G20210A in homozygous state. Most of the patients had only heterozygosity for prothrombotic mutations which were shown to be less effective on thrombosis risk. Therefore, the results should be interpreted with caution. A prospective large-scale study including increased number of children with the same mutation and the same level of factor and who are on prophylaxis may give further insight into this association.
Our results did not show any impact of prothrombotic mutations on clinical phenotype of hemophilia in children. This may be due to the heterogeneous character of the study group. Large-scale studies are needed for conclusion.
Footnotes
Acknowledgment
This research was funded by Istanbul University Scientific Research Projects (Project number 4207).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
