Abstract
Background
Oesophagectomy is a major oncological surgical procedure. Previous studies have shown a wide range of bleeding during and after surgery, and it is unknown if perioperative bleeding associated with oesophagectomy is purely surgical in nature, or if it is exacerbated by impaired haemostasis. We aimed to perform a detailed investigation of the perioperative coagulation in patients undergoing oesophagectomy due to cancer.
Methods
The study was a prospective study including adult patients with adeno- or squamous cell carcinoma referred for intended curative oesophagectomy. Operative bleeding volume and blood transfusions were recorded. Blood samples were collected at three timepoints: before, at the end of surgery, and on postoperative day one. Dynamic global haemostasis was investigated employing thromboelastometry (ROTEM®). Platelet aggregation was analysed with a Multiplate Analyzer®, and routine coagulation parameters were analysed.
Results
We included 87 patients. Patients bled a median of 300 mL during surgery. One patient bled 1830 mL, while the remaining patients bled ≤1000 mL. Blood transfusions were administered to 14 (16%) patients. Median platelet aggregation was within the reference ranges at all time points. Platelet aggregation increased during surgery and normalised within 24 h. ROTEM® analyses showed no perioperative significantly decrease of clot formation or clot strength. Routine coagulation parameters were overall normal.
Conclusions
Severe perioperative bleeding was rare, and transfusions of blood products were used sparingly. Patients undergoing oesophagectomy due to cancer had an intact haemostasis with no sign of impaired haemostasis.
Clinical trial registration
The trial was registered prior to initiation at www.clinicaltrials.gov (identification number NCT05067153).
Introduction
Oesophagectomy is the current cornerstone of intended curative treatment for cancer of the oesophagus.1,2 Around half of patients experience complications, and procedure-related mortality is approximately 3%.2–4
Blood transfusions impact long-term outcome and mortality after oesophagectomy.4–7 The rates of transfusions vary, but most studies report transfusions in more than 30% of patients.7–9 Despite this, only two studies examined perioperative coagulation with a focus on bleeding.10,11 The 2021 study by Ren et al examined if CO2 artificial pneumothorax during thoracoscopic esophagectomy affected coagulation and fibrinolysis. 10 Although they found decreased, slower clot formation measured with thromboelastography (TEG) in the intervention group, no difference was found in blood loss. 10 Song et al examined once versus twice-daily administration of low molecular weight heparin (LMWH) following oesophagectomy and found no difference in red blood cell counts in the chest drainage production between the two groups, despite prolonged TEG clotting times in the twice-daily group. 11 Other studies that measure markers of coagulation focus on potential markers of thrombosis and prognosis and do not investigate in detail whether haemostasis is impaired.12–18
We aimed to examine the perioperative coagulation of oesophageal cancer patients undergoing intended curative surgery. We hypothesised that coagulation was impaired during and in the first 24 h after surgery.
Materials and Methods
Study Design
Patients were included at Aarhus University Hospital, a tertiary referral hospital in Aarhus, Denmark, between 01 September 2021-01 April 2024. The study population was part of a randomised, clinical trial investigating the efficacy of prolonged, one-month thromboprophylaxis after oesophagectomy and had the same in- and exclusion criteria.19,20 Inclusion criteria were 1) age > 18 years, 2) presence of oesophageal adeno- or squamous cell carcinoma and 3) referred for intended curative oesophagectomy. We excluded patients with known inherited bleeding disorders, patients unable to provide informed consent, patients with arterial or venous thromboembolic events within the last three months and patients receiving anticoagulant treatment. Furthermore, patients were not eligible for inclusion if they had a recent (< three months) or current pregnancy or were allergic to Dalteparin. All patients received thromboprophylaxis with 5000 IU (International Units) of LMWH (Dalteparin®) from the evening before surgery until discharge from hospital. The observation period of the present study ended on day 10, before the randomisation for the clinical trial took effect.
Patients were scanned with ultrasound of the lower extremities prior to surgery to exclude presence of deep venous thrombosis. If thrombosis was present, the patient was excluded from the study.
Patients were operated with open oesophagectomy, hybrid or minimally invasive oesophagectomy at the surgeons’ discretion. The open oesophagectomy was a hemi or full thoracotomy in combination with a laparotomy. The hybrid procedure was a laparoscopic abdominal procedure in combination with a hemi thoracotomy to establish the intrathoracic anastomosis. The minimally invasive procedure was fully laparoscopic and thoracoscopic.
Clinical Data
Clinical data were recorded from the patients’ medical records before, during, and the morning after surgery. Furthermore, a follow-up was performed ten days after surgery where mortality and surgical complications were recorded. Major bleeding was defined according to the International Society on Thrombosis (ISTH) recommendations as leading to transfusion of 2 or more units of whole blood or packed red cells, decrease in haemoglobin of 2 g/L (1.24 mmol/L), bleeding in critical sites, defined as: spinal, epidural, intraocular, intracranial, pericardial, retroperitoneal, or leading to death. 21
Blood Sampling and Analyses
Blood samples were taken at three timepoints: before induction of anaesthesia, when surgical incisions were closed, and the morning after surgery. Samples for analysis of thromboelastometry (ROTEM®) were taken in 3.2% sodium citrate tubes (Vacuette®, Greiner Bio-One International GmbH, Kremsmünster, Austria). Samples rested for 30 min before ROTEM® analysis (Werfen GmbH, Munich, Germany). The EXTEM, INTEM, FIBTEM and HEPTEM assays were performed according to manufacturer's instructions, and clotting time (CT), maximum velocity of clot formation (MaxVel), time to MaxVel (tMaxVel), lysis at 30 min (Ly30) and maximum clot firmness (MCF) were recorded. Reference ranges were established in a previous study. 22
Samples for measurement of platelet aggregation were collected in 1.6 mL hirudin tubes (Sarstedt AG & Co. KG, Nürnbrecht, Germany). Samples rested for 30 min and were then analysed employing the Multiplate® Analyzer (Roche, Mannheim, Germany) using adenosine diphosphate (ADPtest 6.5 μM), arachidonic acid (ASPItest 1.0 mM) and thrombin receptor activating peptide (TRAPtest 32 μM) as agonists. Platelet aggregation was determined as the mean of duplicate areas under the curve (AU × min). The reference ranges were published previously.23,24
Samples for measurement of International Normalised Ratio (INR), activated partial thrombin time (aPTT), antithrombin (functional), thrombin time, fibrinogen (functional, Clauss method), and fibrin d-dimer were obtained in 3.2% sodium citrate tubes (Vacuette®, Greiner Bio-One International GmbH, Kremsmünster, Austria) for analysis on CS 5100i (Sysmex, Kobe, Japan). BD Vacutainer® K2 ethylenediamine tetraacetic acid (EDTA) tubes was used for measurement of haemoglobin, platelet and immature platelet count and fraction, using the Sysmex XN-9000. BD Vacutainer® lithium heparin tubes were used for C-reactive protein (CRP), alanine transaminase (ALT) and estimated glomerular filtration rate (eGFR) measurement on Atellica (Siemens Healthcare GmbH, Erlangen, Germany).
Sample Size
Sample size was calculated for the primary endpoint of the randomised, clinical trial. 25 As the present study was exploratory in nature, no sample size calculation was performed.
Statistical Methods
Data were visually examined for Gaussian distribution with QQ-plots and histograms. Mean and 95% confidence intervals (CI) were calculated for data following a Gaussian distribution and median with interquartile range were calculated for data not following a Gaussian distribution. To calculate mean or median of some of the analytes, data was recoded as follows: For eGFR, the value >90 mL/min was recoded to 91. For CRP, < 4.0 mg/L was recoded to 3.9. For aPTT, < 20 s was recoded to 19 and for fibrin d-dimer <0.25 mg/L fibrinogen equivalent units (FEU) was recoded to 0.24 and >20 mg/L FEU to 21. For ALT, < 9 U/L was recoded to 8, and for INR <1.0 was recoded to 0.9. Student's t-tests and Wilcoxon Signed Rank tests were performed on data with two time points as applicable. Repeated measurements analyses were tested with a one-way analysis of variance (ANOVA). A Tukey's test was carried out on significant results from ANOVA analyses to identify the specific differences between groups. Spearman's rank correlation coefficient was calculated to investigate correlations. All analyses were carried out in R, version 4.2.3.
Results
We included 87 patients (Figure 1). Preoperative clinical data is shown in Table 1. Most patients (79%) were between 60 and 80 years of age, and 74% were male. The most frequent cancer type was adenocarcinoma (80%), and 72% had cancer situated in the gastroesophageal junction. Median T stage was 3. No patients had metastasis to solid organs. Most patients (78%) underwent preoperative chemotherapy, and 42 patients (48%) underwent radiotherapy. The most common comorbidities were hypertension (44%), cerebrovascular disease (14%) and chronic pulmonary disease (13%).

Flowchart of patient screening, inclusion and exclusion. aTwo patients had thrombosis on their central venous catheter, which was discovered when placing the central venous line prior to surgery. These patients were prescribed therapeutic levels of low molecular weight heparin and were excluded from the study. Abbreviations: DVT: Deep venous thrombosis, VTE: Venous thromboembolic event(s).
Preoperative Characteristics of 87 Patients Undergoing Oesophagectomy due to Cancer.

Abbreviations: AFLI: Atrial fibrillation, AFLA: Atrial flutter, BMI: Body Mass Index, IQR: Interquartile range, WHO: World Health Organisation.
Table 2 shows the peri- and postoperative clinical data. More than half (67%) of patients were operated with a minimally invasive approach, and only three patients (3%) required conversion to open procedures or cervical incisions. The mean duration of surgery from induction of anaesthesia to incision closure was 7 hours.
Peri- and Postoperative Data of 87 Patients following Oesophagectomy due to Cancer.

Three procedures started as minimally invasive but were converted to open. These are included under “minimally invasive”.
Each patient received 1000 mg of tranexamic acid.
Major bleeding was defined as leading to transfusion of 2 or more units of whole blood or packed red cells, decrease in haemoglobin of 2 g/L, bleeding in critical sites, defined as: spinal, epidural, intraocular, intracranial, pericardial, retroperitoneal, or leading to death. Abbreviations: ASA: Acetylsalicylic acid, IQR: Interquartile range, NSAID: Nonsteroidal anti-inflammatory drugs, PE: Pulmonary embolism.
Patients bled a median of 300 mL during surgery. The largest recorded bleeding volume was 1830 mL, while the remaining patients bled 1000 mL or less during surgery. In total, 14 (16%) patients received a blood transfusion. Of these, ten patients (71%) received ≤ two units red blood cells. The largest transfusion was five units of red blood cells. Only one patient received fresh frozen plasma (three units), and no patients received platelet transfusions. Three patients (3%) met the requirements for a major bleeding.
The most common postoperative complication was pneumonia (nine patients, 10%). Five patients (6%) had leakage of the anastomosis and three patients (3%) developed pulmonary embolism. No patients died during surgery or in the following ten days.
Routine Biochemistry
Table 3 shows results of routine biochemical analyses. Haemoglobin was below reference range at all sample time points, and a significant decrease in mean haemoglobin was observed from the preoperative to the postoperative day 1 sample (P < .0001). Mean platelet count, immature platelet count and fraction were within reference ranges throughout all time points. Thrombin time was less than 21 s for all patients at all sample times and INR and aPTT were likewise within reference ranges, except for one patient who had an aPTT of 44 s, an INR of 1.4 and a thrombin time of 35 s on postoperative day 1. The patient had normal values of the parameters in the preoperative and end of surgery samples. Median fibrinogen stayed within reference range, but a gradual increase in median fibrin d-dimer was observed (P < .0001).
Routine Laboratory Analyses in 87 Patients Undergoing Esophageal Cancer Surgery.

Data presented as median with interquartile range or mean with 95% confidence interval as applicable. a Reference interval for males. b Reference range for 65-75 years. Abbreviations: ALT: Alanine transaminase, aPTT: Activated partial thromboplastin clotting time, CRP: C-reactive protein, eGFR: Estimated glomerular filtration rate, EVF: Erythrocyte volume fraction, INR: International Normalised Ratio, IPC: Immature platelet count, IPF: Immature platelet fraction, MPV: Mean platelet volume.
Platelet Aggregation
Figure 2 shows platelet aggregation before, at end of surgery and on postoperative day 1. Median platelet aggregation was within the reference ranges at all time points.
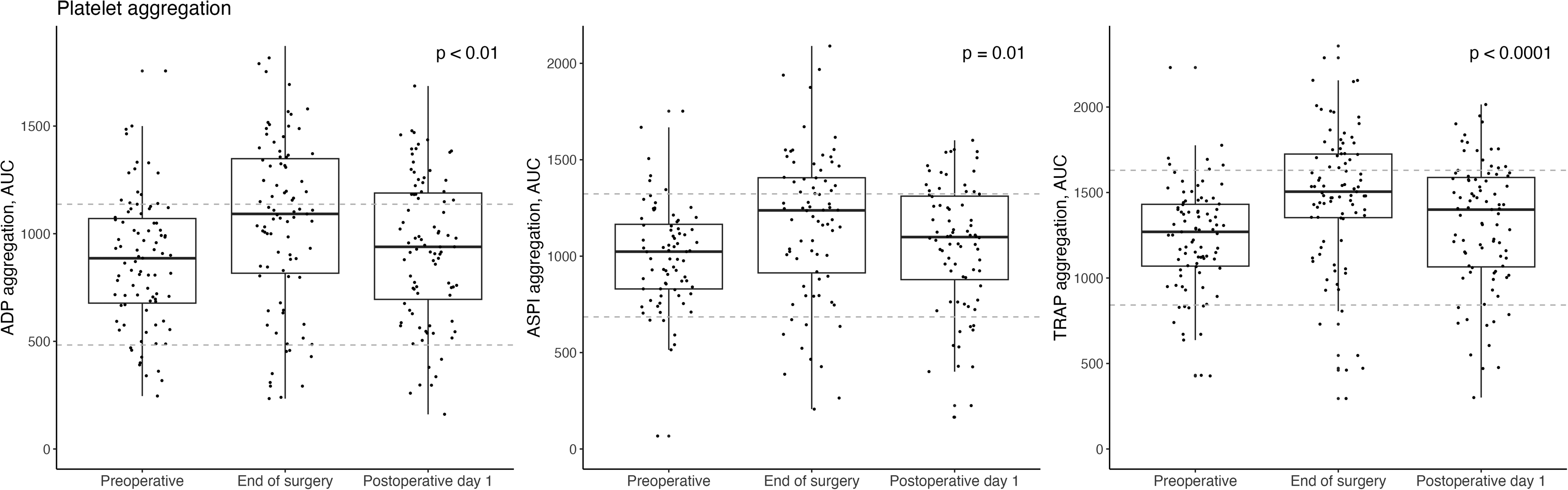
Platelet aggregation in 87 patients preoperatively, at end of surgery and one day after oesophageal cancer surgery. The agonists ADP, ASPI and TRAP were used. Boxplots denote median, interquartile range and range. Reference ranges are depicted with dotted lines, and P-values for the ANOVA tests are shown. Patients receiving acetylsalicylic acid or nonsteroidal anti-inflammatory drugs were excluded for ASPI analyses (n remaining = 76).
A statistically significant change in platelet aggregation was found for all three agonists (ANOVA P-values ≤ .01). For all agonists platelet aggregation increased during surgery (P-values < .01), while no significant difference was demonstrated between the preoperative sample and the first postoperative day (P-values ≥ .34).
Clot Initiation and Development (ROTEM®)
CT, MaxVel and tMaxVel for the EXTEM and INTEM ROTEM® assays are depicted in Figure 3a and 3b. ANOVA analyses revealed significant differences in CT and MaxVel for EXTEM (Figure 3a, P values ≤ .02). For INTEM, significant differences were found in CT and tMaxVel (Figure 3b, P values < .0001). When examining the significant ANOVA findings with Tukey's test, the significance was mainly created by the decrease in CT, MaxVel and tMaxVel at the end of surgery compared to the other sample times, whereas the difference between the preoperative and postoperative day 1 samples was only significant in INTEM CT and tMaxVel (Tukey P-values ≥ .003).
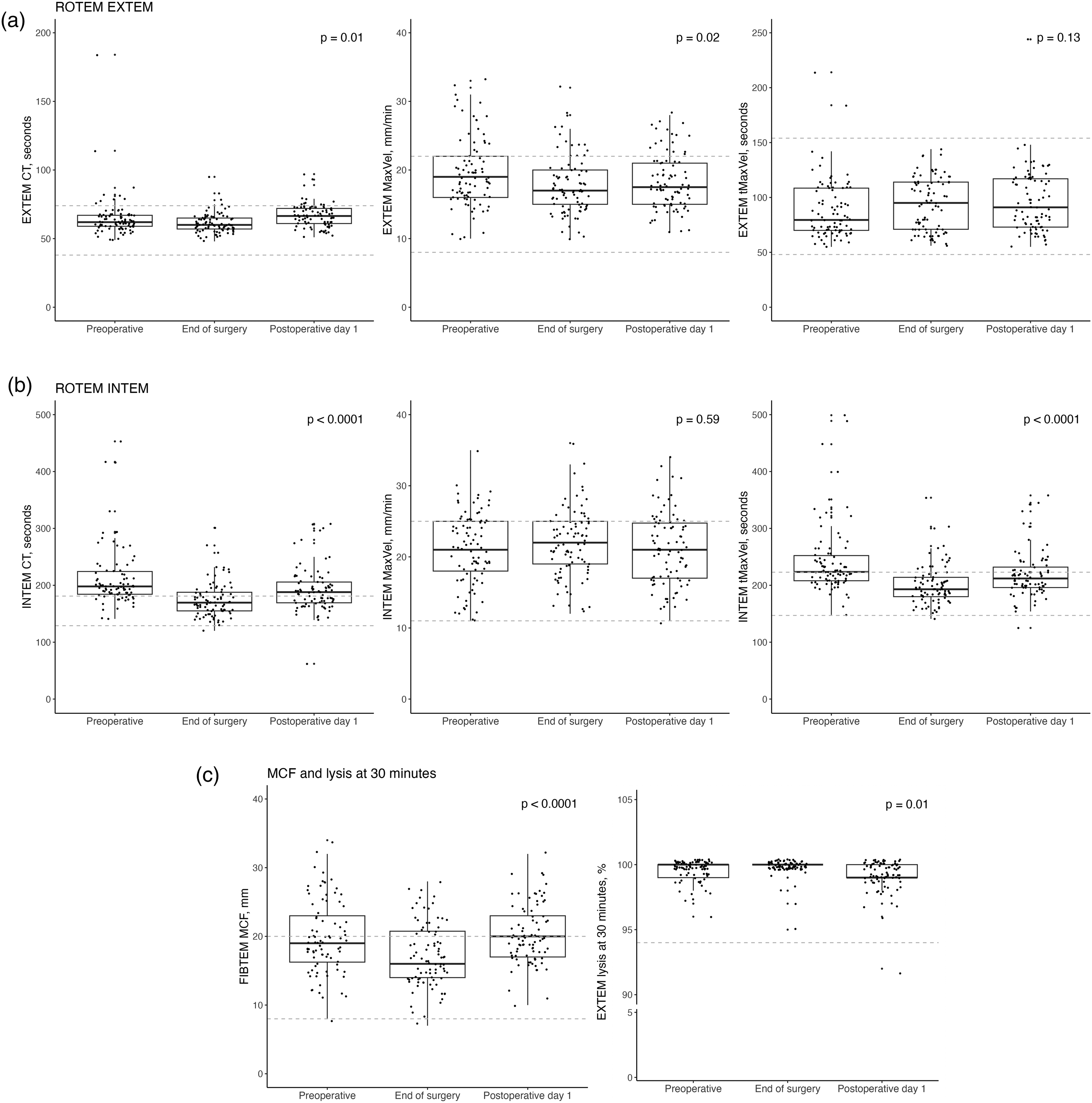
Clot initiation and development investigated by thromboelastometry (ROTEM®) in 87 patients undergoing oesophageal cancer surgery. Boxplots denote median, interquartile range and range of the preoperative, end of surgery and one day after surgery samples. Reference ranges are depicted with dotted lines, and P-values for the ANOVA tests are shown. Values have been slightly jittered to improve legibility. Abbreviations: CT: Clotting time, MaxVel: Maximum velocity of clot formation, MCF: Maximum clot firmness, tMaxVel: Time to MaxVel.
While most values for both INTEM and EXTEM parameters were within reference ranges, INTEM had several outliers with prolonged CT and tMaxVel at the preoperative sample. A total of 15 patients (17%) had INTEM values > 240 s. The prolongation in CT was less pronounced in HEPTEM, indicating that heparin was the likely cause. This was, however, not evident in the thrombin time analyses. When examining preoperative INTEM CT and the recorded bleeding volume for the patients during surgery, a weak correlation was found, rho = 0.22 and P = .05. No correlation was found for EXTEM CT, P = .85.
Clot Strength and lysis (ROTEM®)
FIBTEM MCF and EXTEM lysis at 30 min (Ly30) are depicted in Figure 3c. The upper quartile MCF levels were above reference range before and one day after surgery. MCF levels were significantly lower at end of surgery compared to the preoperative and the postoperative day 1 sample, both P < .0001. EXTEM Ly30 values were within reference cutoff across sample times.
Discussion
The present study demonstrated no impaired haemostasis in patients undergoing oesophageal cancer surgery based on performance of dynamic global analyses, platelet aggregation analyses and routine coagulation analyses. This finding is in accordance with an overall low bleeding volume during surgery.
We found evidence of increased platelet aggregation and slower clot formation during surgery for oesophageal cancer when compared to preoperative samples. While significant, these changes occurred mainly within reference ranges and were likely exacerbated due to a heparin-induced prolonged clot initiation in the preoperative sample. 22 Approximately 25% of patients had an increased clot strength preoperatively, which decreased during surgery and increased again on postoperative day 1. Fibrin d-dimer was also elevated throughout the study period. These findings are in line with a previous study by Van Haren et al. 13 Overall, the changes in coagulation induced by surgery were normalised one day after surgery.
Median operative bleeding volume was only 300 mL, indicating an overall intact haemostatic function and controlled surgical bleeding. This was reflected in a lower frequency of blood transfusions (16%) than reported in the literature.7–9 The highest incidence was found in a 2017 study on 565 patients, where 84% received a blood transfusion. 7 In another, recent study Alwatari et al found that 44% of patients undergoing emergent oesophagectomy received blood transfusions. 8 Likewise, Sakamoto et al reported that 22%-34% of patients received blood transfusions, depending on whether surgical approach was minimally invasive or open. 9 The lowest incidence was found in study from 2019 by Ohkura et al, where only 9% of patients received blood transfusions. 4 The relatively limited use of transfusions observed in our cohort may be both due to non-impaired haemostasis and due to advances in surgical techniques leading to an increase in minimally invasive procedures and less bleeding as well as a gradually more restrictive transfusion strategy in the later years.9,26
LMWH overall did not significantly impact haemostasis as reflected by the normal aPTT and thrombin times in the cohort, likely due to the fact that dosages were prophylactic and not therapeutic, and the short half-life (3-5 h) of dalteparin. 27
Strength and Limitations
The prospective design of the study, the systematic data collection and comprehensive and dynamic investigations of coagulation strengthen our findings.
The present study was exploratory in nature, and no sample size calculation was performed. However, the cohort is of a significant size and the findings are consistent, leading us to believe that the results of our study are valid, and applicable to similar cohorts of oesophageal cancer patients undergoing intended curative surgery.
The measurement and registration of operative bleeding have pitfalls. Cumulative drain production may include other fluids such as serous liquid and chylos. One strategy to circumvent this is the use of proxy measurements for bleeding, such as Song et al, who measured red blood cells in chest drain production from placement to 72 h post-surgery. 11 We chose to restrict our measurement to operative bleeding volume, ie the volume collected via suction and drains during surgery, ending when the patient was transferred from theatre to the intensive care ward. While this may underestimate total bleeding volume slightly, we believe it to be a feasible and reproducible way of evaluating procedure-related bleeding, especially when combined with haemoglobin and erythrocyte volume fraction (EVF) measurements.
Conclusion
We found only discrete changes in global haemostasis with preserved clot strength and no impairment of platelet aggregation. Bleeding was low to modest in most patients. Thus, impaired haemostasis does not challenge patients undergoing oesophageal cancer surgery.
Footnotes
Acknowledgements
The authors wish to thank M. S. Nielsen for kindly assisting with extraction of ROTEM® data.
Consent to Participate
All patients gave verbal and written informed consent prior to participation in the study.
Data Availability
The statistical code used to analyse the dataset is available from the corresponding author on reasonable request. Danish law and GDPR prohibits sharing of raw data, as it contains confidential health information.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Thomas Decker Christensen has been on the speaker bureaus for AstraZeneca, Chiesi and GlaxoSmithKline and has been in an Advisory Board for Sanofi and AstraZeneca. The other authors declared no potential conflict of interests with respect to the research, authorship and/or publication of this article.
Ethical Considerations
Prior to study initiation, the experimental protocol was approved by the regional Scientific Committee (case number 1-10-72-226-21) and the Danish Data Protection Agency. The study was conducted in accordance with the Declaration of Helsinki and applicable local regulatory requirements.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Aarhus Universitetshospital, Sundhedsvidenskabelige Fakultet, Aarhus Universitet, Oda og Hans Svenningsens Fond,
