Abstract
Background
Atrial fibrillation (AF) after acute pulmonary embolism (PE) may lead to a poor prognosis. We conducted this study to explore influencing factors of new-onset AF in patients after acute PE.
Methods
Patients with objectively confirmed acute PE at the China-Japan Friendship Hospital from first January 2015 to 31st May 2022 were retrospectively included in the study, and patients with new-onset AF confirmed by electrocardiography were defined as the case group. The control group was obtained from the above PE patients without new-onset AF in age matching. Patients with a history of AF, pulmonary hypertension, acute myocardial infarction, valvular heart disease and hyperthyroidism were excluded. Logistic regression was conducted to identify potential influencing factors for the development of new-onset AF in patients with acute PE. To assess the prediction value of potential variables, receiver operating characteristic curves were plotted.
Results
Among 853 patients diagnosed with acute PE, 732 patients met the including criteria, and 29 patients with new-onset AF were identified. The median age of all patients was 74 years, with 77.6% being male. No statistically significant differences were observed between the case and control groups regarding demographic characteristics. Patients with new-onset AF had significantly enlarged right atrium, right ventricle and left atrium in echocardiography compared with control group, but no significant differences were observed in the diameter of the left ventricle and the proportion of left ventricular ejection fraction (LVEF) ≤ 40%. Right atrial enlargement (OR, 4.19; 95%CI, 1.24-15.09; P = 0.023), left atrial enlargement (OR, 14.76; 95%CI, 4.79–57.28; P < 0.001) and the simplified pulmonary embolism severity index (sPESI) (OR, 2.04; 95%CI, 1.19–3.67; P = 0.012) were associated with increased risk of new-onset AF.
Conclusions
Both severity of acute PE and enlargement of left and right atrium were associated with an increased risk of new-onset AF in patients with acute PE. In patients with high-risk acute PE, greater vigilance is needed for the occurrence of new-onset AF.
Keywords
Background
Pulmonary embolism (PE) is the third most common cause of cardiovascular death around the world. 1 When patients are diagnosed with PE, they are at risk of short-term and long-term adverse events. A notable concern among these is atrial fibrillation (AF). The development of AF in patients with PE could be both a cause and a consequence of PE. Large population-based studies have provided substantial evidence of the role of PE in the subsequent development of AF.2,3 Prior studies have evaluated the incidence of AF after PE. In 2014, Tromsø study described the incidence of AF after PE as 2630 people/100,000 person-years. 2 RIETE registry study in 2021 identified 445 cases of new-onset AF among 16,497 patients with PE within 2 days of acute PE. 3 In spite of the relatively low incidence of new-onset AF, the increased risk of AF after PE has potentially significant clinical implications in population level. In PE patients, both preexisting AF and incident AF were associated with significantly increased risk of adverse clinical outcomes. 4
Despite clear evidence linking PE and AF,5,6 relatively few studies have focused on identifying influencing factors for new-onset AF after PE. The clinical characteristics and influencing factors associated with new-onset AF in PE patients remain unclear. Given the importance of early identification and treatment of AF after PE, investigating a better method to identify patients at risk of AF is vital to guide clinical practice. There are several possible mechanisms for PE to cause AF, including elevated right atrial pressure, electrical remodeling, stress-induced increased sympathetic tone, and hypoxemia.7.8 We hypothesized that AF may be associated with right ventricular dysfunction based on the pathophysiological mechanism. Therefore, we proposed to conduct a case-control study to explore the potential influencing factors of new-onset AF among patients with acute PE.
Methods
Study Design
Patients diagnosed with PE by computed tomography pulmonary angiogram (CTPA) or chest contrast-enhanced computed tomography at the China-Japan Friendship Hospital from January first, 2015 to May 31st, 2022 were included in the study. The patients were excluded in following criteria: (1) patients younger than 18 years old, (2) patients with previous history of AF, (3) patients with pre-existing pulmonary hypertension, acute myocardial infarction, valvular heart disease, and hyperthyroidism.
The diagnosis of acute PE was confirmed in accordance with the 2019 European Society of Cardiology (ESC) guidelines. 1 Pulmonary artery dilatation was defined as the ratio of pulmonary artery diameter to aortic diameter (PA/AA) more than 1. Echocardiograms were performed promptly by a specialized echocardiography team following the diagnosis of acute PE. Recorded parameters included the diameters of the left atrium, right atrium, right ventricle, left ventricle, and the left ventricular ejection fraction. The enlargement of cardiac chamber was evaluated based on recommendations for Cardiac Chamber Quantification by Echocardiography in Adults (2015 edition). 9 Left atrial enlargement was defined as left atrial diameter (LAD) ≥ 38 mm. Right atrial enlargement was defined as right atrial diameter (RAD) ≥ 44 mm. Right ventricular enlargement was defined as right ventricular diameter (RVD) ≥ 41 mm. Left ventricular enlargement was defined as left ventricular end diastolic diameter (LVEDD) ≥ 55 mm in men and ≥ 50 mm in women. Patients received standard care for acute PE following diagnosis.
Among the included patients, those diagnosed with AF confirmed by electrocardiography (ECG) following the diagnosis of PE were included in case group (Group A). ECG was conducted when patients were admitted to the hospital and when clinicians considered the presence of a change in disease condition, and echocardiograms were conducted for severity stratification after the diagnosis of PE. In acute PE patients without new-onset AF during the same period, age matching was applied to match controls in a 1:4 ratio (Group B). An additional age-matched control group was included to compare the size of left heart morphology in AF patients without PE (Group C).
We gathered factors potentially associated with new-onset AF through clinical practice insights and a review of existing literature. Demographic and clinical data were collected from electronic medical records, including medical history, comorbidities, laboratory test results, echocardiographic findings, radiographic examination results, and the anticoagulation prescribed. In addition, CHA2DS2-VASc and the simplified pulmonary embolism severity index (sPESI) were calculated retrospectively.10,11 The sPESI included age > 80 years, history of cancer, heart rate ≥ 110 beats per minute, systolic blood pressure < 100 mm Hg, oxygen saturation < 90%, and history of chronic cardiopulmonary disease. 10 The CHA2DS2-VASc score included congestive heart failure, hypertension, age, female, diabetes mellitus, stroke or transient ischemic attack or thromboembolism, and vascular disease. 11 The Ethics Committee of China-Japan Friendship Hospital approved this study with the approval number (2016-SSW-7).
Statistical Analysis
Continuous variables were assessed using histograms and Shapiro-Wilk test presented as means and 95% confidence interval (CI) for normally distributed data and median and interquartile range (IQR) for data with a skewed distribution. Kruskal-Wallis test (qualitative data) and t-test or Mann-Whitney U test (quantitative data) were used for groups’ comparison. Categorical data were expressed as counts and proportions, and the between group comparisons were made using Chi-squared or Fisher's exact tests. The frequency and proportion of missing data for indicators were reported.
Age matching was performed to ensure age equity between the two groups. Factors potentially associated with AF were compared between the case and control groups. Those that showed significant differences were further included in univariate and multivariate logistic regression, with consideration of previous literature. Univariable and multivariable logistic regression were performed to explore potential influencing factors for the development of new-onset AF in patients with acute PE. Tolerance and variance inflation factor were calculated to assess potential multicollinearity in multivariable regression models. To assess the prediction value of potential variables, receiver operating characteristic curves (ROC) were plotted, and area under the curve (AUC), sensitivity, specificity, and 95% confidence interval (CI) were also calculated. Appropriate threshold values were determined based on the Youden index and AUC. To improve the prediction performance, variables combining for prediction and evaluation were conducted. All P values were set two-sided and P values < 0.05 were considered statistically significant. All analysis was performed with the R (version 4.2.3).
Results
A total of 853 patients were diagnosed with acute PE at China-Japan Friendship Hospital, of which 732 were included in the final analysis (Figure 1). 4.0% (29/732) of patients were diagnosed with AF during their hospitalization, with a median time of 2 days from the diagnosis of PE to the onset of AF. After age-matching, 96 patients were included in the control group. The median age of all patients was 74 years, with 77.6% (568/732) being male. No significant difference was observed in age or sex between the case and control group. The proportions of patients aged over 75 years in both groups were similar (55.2% [16/29] VS. 47.9% [46/96], P = 0.636).

Flow chart of population selection. The control group was age-matched in a ratio of 1:4 from pulmonary embolism patients without new-onset AF.Abbreviations: AF = atrial fibrillation.
Body mass index (BMI), smoking history, history of venous thromboembolism, immobility ≥ 3 days, and history of surgery or trauma in 1 month were comparable between the two groups. The median sPESI was higher in patients with new-onset AF (2 VS. 1, P = 0.002), but there were no significant differences in NYHA cardiac function class, CHA2DS2-VASc score, or incidence of in-hospital deaths. No significant differences were seen between the two groups in terms of comorbidities, including hypertension, diabetes mellitus, coronary heart disease, chronic lung disease, lower extremity deep venous thrombosis (DVT). The proportion of prophylactic anticoagulation was higher in the control group than in the case group without statistically significant difference (7.3% [7/96] VS. 0% [0/29], P = 0.30), and it was noticeable that no patients in the case group underwent prophylactic anticoagulation.
Despite electrolyte levels being generally within the normal range, serum sodium and chloride levels were higher in the case group, while calcium levels were lower. Additionally, an elevated level of the higher N-terminal fragment of B-type natriuretic peptide precursor (NT-proBNP) was observed in case group than control group. There were no statistically significant differences in D-dimer, troponin, or oxygenation index in arterial blood gas analysis (Table 1).
Baseline Characteristics of Patients with New-Onset Atrial Fibrillation and Patients Without New-Onset Atrial Fibrillation.

*Pulmonary artery dilatation was defined as the ratio of PA to AA more than 1. Left atrial enlargement was defined as LAD ≥ 38 mm. Right atrial enlargement was defined as RAD ≥ 44 mm. Right ventricular enlargement was defined as RVD ≥ 41 mm. Left ventricular enlargement was defined as LVEDD ≥ 55 mm in men and ≥ 50 mm in women. Continuous variables are presented as mean ± SD and median with 25th and 75th percentile. Categorical variables are described as counts and proportions.
Abbreviations: BMI = body mass index, NYHA = New York Heart Association functional classification, sPESI = simplified pulmonary embolism severity index, VTE = venous thromboembolism, pH = potential of Hydrogen, pO2 = partial pressure of oxygen, pCO2 = partial pressure of carbon dioxide, SaO2 = arterial oxygen saturation, eGFR = estimated glomerular filtration rate, BUN = blood urea nitrogen, LDL-C = low-density lipoprotein cholesterol, CHO = total cholesterol, HDL = high-density lipoprotein, PT = prothrombin time, c-TnT = cardiac troponin T, NT-proBNP = N-terminal fragment of B-type natriuretic peptide precursor, PA = pulmonary artery diameter, AA = aortic artery diameter, PA/AA = the ratio of pulmonary artery diameter to aortic diameter, LAD = left atrial diameter, RAD = right atrial diameter, RVD = right ventricular diameter, LVEDD = left ventricular end diastolic diameter, LVEF = left ventricular ejection fraction.
With regard to CTPA indicators, the case group exhibited a higher percentage of widened pulmonary artery diameters (27.6% [8/29] VS. 13.5% [13/96], P = 0.026). In the echocardiographic parameters, LAD (44 mm VS. 35 mm, P < 0.001), RAD (42 mm VS. 37 mm, P = 0.011), and RVD (37 mm VS. 35 mm, P = 0.032) in the case group were significantly larger than those in the control group (Figure 2). Left ventricular ejection fraction (LVEF) was significantly lower in the case group (62% VS. 65%, P = 0.028) but remained within the normal range. Analysis of the proportion of patients with LVEF < 40% revealed no significant difference between the two groups. Another age-matched control group (Group C), consisting of 93 patients, was added to compare the size of left heart morphology in AF patients without PE. Detailed information comparison between patients with new-onset AF and matched AF patients without PE was revealed in Supplementary Table 1. Although there were no significant differences, the diameter of left atrium in the case group was larger than that in the age-matched group of AF patients without PE (44 mm VS. 41 mm, P = 0.44).

Comparison of the diameters of left atrium (A), right atrium (B), right ventricle (C), left ventricle (D) between acute pulmonary embolism patients with and without new-onset atrial fibrillation. Comparison of the Diameters of Left Atrium (E), Left Ventricle (F) between Acute Pulmonary Embolism Patients with New-Onset Atrial Fibrillation and Atrial Fibrillation Patients without Pulmonary Embolism after Age Matching. Group A was defined as acute pulmonary embolism (PE) patients with new-onset atrial fibrillation (AF). Group B was defined as acute PE patients without new-onset AF. Group C was defined with AF patients without PE. The diameters of the atrium and ventricle were measured using echocardiography. P values were determined by using unpaired, two-sided Mann-Whitney U-test and X2 test.Abbreviations: LAD = left atrial diameter, RAD = right atrial diameter, RVD = right ventricular diameter, LVEDD = left ventricular end diastolic diameter.
In the univariable logistic regression analysis, several factors were associated with an increased risk of new-onset AF in patients with acute PE: the sPESI (OR, 2.04; 95%CI, 1.30–3.33; P = 0.003), left atrial enlargement (OR, 17.75; 95%CI, 6.18–64.85; P < 0.001), right atrial enlargement (OR, 6.82; 95%CI, 2.52–19.28; P < 0.001), left ventricular enlargement (OR, 7.32; 95%CI, 2.03–30.06; P = 0.002), pulmonary artery dilatation (OR, 3.86; 95%CI, 1.28–11.53; P = 0.014) (Table 2).
Univariable Logistic Regression for New-Onset Atrial Fibrillation in Acute Pulmonary Embolism.

*Pulmonary artery dilatation was defined as the ratio of PA to AA more than 1. Left atrial enlargement was defined as LAD ≥ 38 mm. Right atrial enlargement was defined as RAD ≥ 44 mm. Right ventricular enlargement was defined as RVD ≥ 41 mm. Left ventricular enlargement was defined as LVEDD ≥ 55 mm in men and ≥ 50 mm in women.
Abbreviations: sPESI = simplified pulmonary embolism severity index, PT = prothrombin time, NT-proBNP = N-terminal fragment of B-type natriuretic peptide precursor, PA = pulmonary artery diameter, AA = aortic artery diameter,. LAD = left atrial diameter, RAD = right atrial diameter, RVD = right ventricular diameter, LVEDD = left ventricular end diastolic diameter, LVEF = left ventricular ejection fraction.
In subsequent multivariable logistic regression, left atrial enlargement (OR, 14.76; 95%CI, 4.79–57.28; P < 0.001), right atrial enlargement (OR, 4.19; 95%CI, 1.24–15.09; P = 0.023), and the sPESI (OR, 2.04; 95%CI, 1.19–3.67; P = 0.012) were identified as significant predictors of new-onset AF in Model 1. In consideration of the potential relationship between right heart enlargement and pulmonary dilation or left ventricular enlargement, we further refined Models 2 and 3. In model 2, left atrial enlargement (OR, 12.78; 95%CI, 4.19–48.46; P < 0.001), right atrial enlargement (OR, 3.86; 95%CI, 1.18–13.23; P = 0.026), and left ventricular enlargement (OR, 2.11; 95%CI, 0.47–10.48; P = 0.335) were analyzed. In model 3, left atrial enlargement (OR, 17.38; 95%CI, 5.76–66.76; P < 0.001), the sPESI (OR, 2.02; 95%CI, 1.19–3.60; P = 0.012), and pulmonary artery dilatation (OR, 1.38; 95%CI, 0.37–5.05; P = 0.625) were assessed (Figure 3). The multicollinearity among variables in all three models was determined to be weak. It is noteworthy that the PA/AA ratio and left ventricular enlargement was not identified as a risk factor for new-onset AF when right atrium or right ventricle enlargement were incorporated into the multivariable logistic regression model.

Forest plot for new-onset atrial fibrillation in acute pulmonary embolism. Model 1 included left atrial enlargement, right atrial enlargement, and sPESI score. Model 2 included eft atrial enlargement, right atrial enlargement, and left ventricular enlargement. Model 3 included left atrial enlargement, pulmonary artery dilatation, and sPESI score. Left atrial enlargement was defined as LAD ≥ 38 mm. Right atrial enlargement was defined as RAD ≥ 44 mm. Left ventricular enlargement was defined as LVEDD ≥ 55 mm in men and ≥ 50 mm in women. The diameters of the atrium and ventricle were measured using echocardiography. Pulmonary artery dilatation was defined as the ratio of PA to AA more than 1 in computed tomography pulmonary angiogram. Group A was defined as acute PE patients with new-onset AF. Group B was defined as acute PE patients without new-onset AF. Multivariable logistic regression was conducted to analyze the data. sPESI is ordinal variable presenting with mean, the rest are described in counts and proportions, along with their odds ratios, 95% confidence interval (95% CI) and P values. Abbreviations: LAD = left atrial diameter, RAD = right atrial diameer, LVEDD = left ventricular end diastolic diameter, PA = pulmonary artery diameter, AA = aortic artery diameter, sPESI = simplified pulmonary embolism severity index.
ROC curves for left atrial diameter, right atrial diameter, left ventricle diameter, PA/AA, NT-proBNP, and sPESI were plotted. The continuous variables and categorical variables of left atrial diameter and right atrial diameter were also analyzed (Figure 4). The best cut-off values of LAD and RAD were 38.5 mm and 40.5 mm. With a LAD threshold set at ≥ 38 mm, the model demonstrated the highest AUC of 0.80, with a sensitivity of 0.86 and a specificity of 0.74. Given that the sPESI was calculated as an integer, a cut-off value of 2 was determined to be optimal, resulting in a sensitivity of 0.72 and a specificity of 0.57 (Table 3).
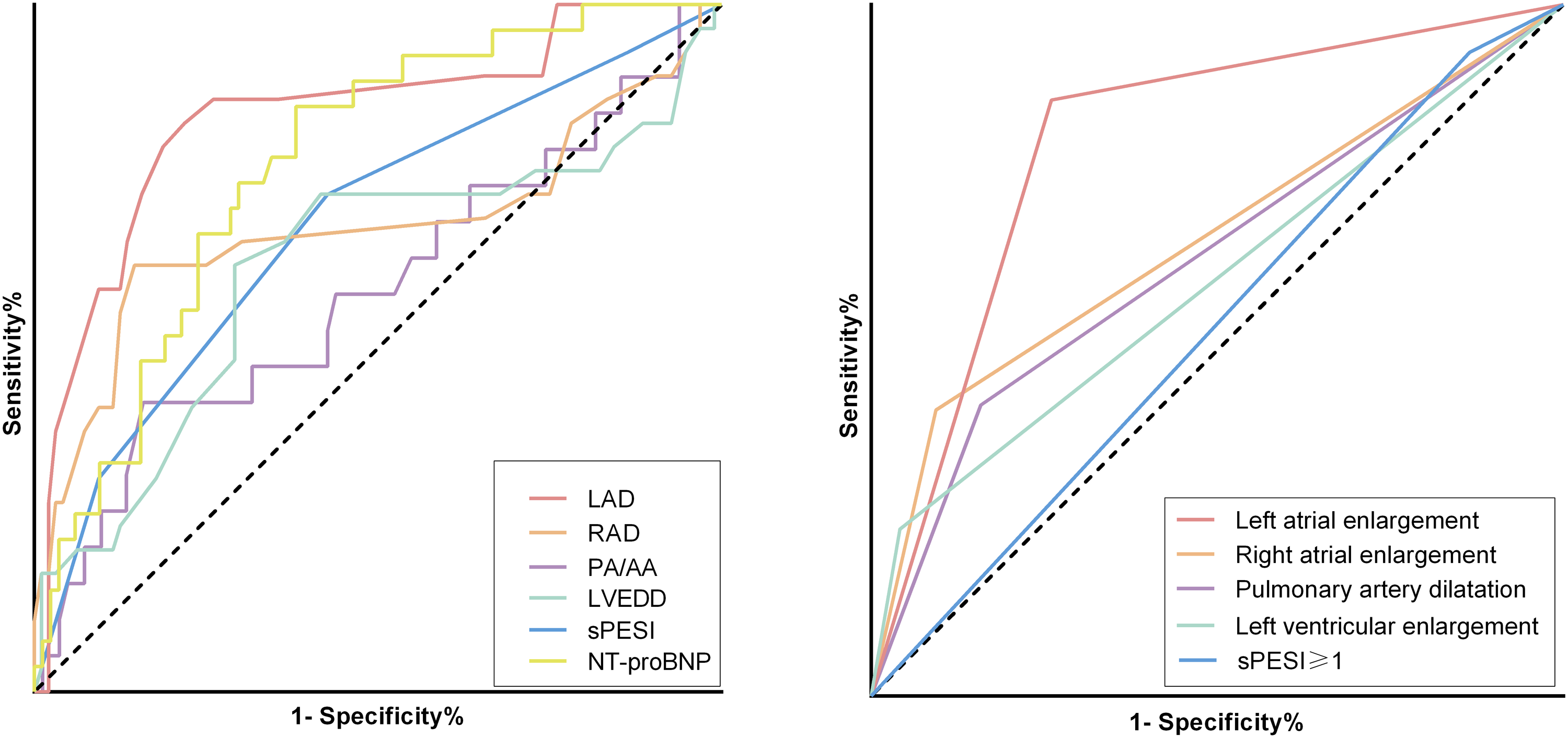
Receiver operating characteristic curve for predicting new-onset atrial fibrillation in patients with acute pulmonary embolism. (A) LAD, RAD, PA/AA, LVEDD, sPESI and NT-proBNP in the prediction of new-onset AF (continuous variable). (B) Left atrial enlargement, right atrial enlargement, left ventricular enlargement, pulmonary artery dilatation and sPESI ≥ 1 points in the prediction of new-onset AF (categorical variable). Left atrial enlargement was defined as LAD ≥ 38 mm. Right atrial enlargement was defined as RAD ≥ 44 mm. Left ventricular enlargement was defined as LVEDD ≥ 55 mm in men and ≥ 50 mm in women. The diameters of the atrium and ventricle were measured using echocardiography. Pulmonary artery dilatation was defined as the ratio of PA to AA more than 1 in computed tomography pulmonary angiogram. Abbreviations: LAD = left atrial diameter, RAD = right atrial diameter, PA = pulmonary artery diameter, AA = aortic artery diameter, PA/AA = the ratio of pulmonary artery diameter to aortic diameter, LVEDD = left ventricular end diastolic diameter, sPESI = simplified pulmonary embolism severity index, NT-porBNP = N-terminal fragment of B-type natriuretic peptide precursor.
Performance of Receiver of LAD, RAD, LVEDD, PA/AA, sPESI, NT-ProBNP, Left Atrial Enlargement, Right Atrial Enlargement, Left Ventricular Enlargement, Pulmonary Artery Dilatation, and sPESI ≥ 1 for Predicting New-Onset Atrial Fibrillation in Patients with Acute Pulmonary Embolism.

*Left atrial enlargement was defined as LAD ≥ 38 mm. Right atrial enlargement was defined as RAD ≥ 44 mm. Left ventricular enlargement was defined as LVEDD ≥ 55 mm in men and ≥ 50 mm in women. The diameters of the atrium and ventricle were measured using echocardiography. Pulmonary artery dilatation was defined as the ratio of PA to AA more than 1 in computed tomography pulmonary angiogram.
Abbreviations: LAD = left atrial diameter, RAD = right atrial diameter, LVEDD = left ventricular end diastolic diameter, PA = pulmonary artery diameter, AA = aortic artery diameter, PA/AA = the ratio of pulmonary artery diameter to aortic diameter, sPESI = simplified pulmonary embolism severity index, NT-porBNP = N-terminal fragment of B-type natriuretic peptide precursor.
Discussion
We conducted a case-control study to explore potential risk factors of new-onset AF after acute PE. The results support that left and right atrium enlargement are associated with an increased risk of new-onset AF in patients with acute PE, which might be helpful in improving the clinical management and prognosis of patients with PE. In addition to risk stratification, morphological examinations, including echocardiography and CTPA, play crucial roles in the assessment of PE. They also have the potential to predict the onset of AF following PE, thereby reinforcing the importance of morphological assessment. Our study provides new insights into the association between PE and AF.
The phenomenon of combining AF at the time of diagnosis of PE in population raises the concern about the association between these two diseases. Several studies have found that the occurrence of AF in patients with PE is linked to a poor prognosis, highlighting the necessity to investigate the role of AF in patients with PE.12,13 A large prospective registry study reported that AF was associated with increased mortality in patients with PE. 14 In patients with acute symptomatic PE, both pre-existing AF and new-onset AF were associated with poor outcomes. 4 In-hospital mortality was lower in sinus rhythm (5%) and paroxysmal AF (6.5%) compared with the permanent AF group (25%) (P < 0.001). 15 AF is not only associated with stroke, but also contributes to the poor long-term prognosis in PE patients by some unknown mechanism. 16 Another study found that sPESI is an independent predictor of poor hospitalization outcomes, and new-onset AF may enhance the predictive value of sPESI in patients who have experienced acute PE. 17 A study from China revealed that newly diagnosed AF was an independent risk predictor for the occurrence of in-hospital adverse events. Furthermore, the study found that newly diagnosed AF combined with sPESI ≥ 2 had a high predictive value for the occurrence of in-hospital adverse events. 18 The above illustrate the importance of early identification of risk factors for the development of AF to improve the prognosis of PE patients. In our study, no significant difference in in-hospital mortality was observed between the two groups, possibly due to the limited sample size.
The understanding of AF occurrence has been enriched by additional studies investigating the risk factors associated with new-onset AF following pulmonary embolism. A study enrolled 590 patients with acute PE, of whom 54 (9.2%) were diagnosed with new-onset AF, whose results indicated massive PE, ischemic heart disease and congestive heart failure were risk factors for the development of new-onset AF. 19 Another study found potential independent predictors of new-onset AF after acute PE included age (Hazard Ratio [HR], 1.06; 95%CI, 1.04–1.08; P < 0.001), history of congestive heart failure (HR, 1.88; 95%CI, 1.12–3.16; P = 0.02), diabetes mellitus (HR, 1.72; 95%CI, 1.07–2.77; P = 0.02), obstructive sleep apnea (HR, 4.83; 95%CI, 1.48–15.8; P = 0.009), and serum sodium level on day 1 during admission (HR, 0.94; 95%CI, 0.90–0.98; P = 0.002). 20 In addition, left ventricle end diastolic diameter, LAD, uric acid, bilirubin, albumin and troponin levels, and PESI score to be univariate predictors of new-onset AF were found. Meanwhile, they found a positive association between PESI score and new-onset AF. A PESI score greater than 82.50 may help predict new-onset AF in these patients. 21 In terms of risk stratification, the performance of PESI may be overestimated due to the coexistence of AF. According to the revised Geneva score and sPESI, patients with AF were more frequently classified as a high-risk group than patients with sinus rhythm. The addition of paroxysmal AF to the ESC risk model did not improve patient risk reclassification for all-cause mortality prediction but may have enhanced the ability of existing models to discriminate. 13 Therefore, for the specific population of patients with PE combined with AF, there is a need to develop and validate a new risk stratification model. The development of AF subsequent to PE may be attributed to several factors, including increased cardiac load due to obstruction of the pulmonary arteries, aberrant electrical activity, activation of the autonomic nervous system, and an inflammatory response that releases vasoconstrictors and inflammatory cytokines.8,22 Previous studies have proposed that right atrial enlargement is associated with an increased risk of developing AF.23,24 The right atrium, a specific indicator closely related to PE, deserves more attention. A recent meta-analysis found a high rate of recognition of right heart dysfunction after PE, which may persist for months after onset. 26 The greater impairment of parameters reflecting right ventricular (RV) afterload may indicate an association between PE severity and the onset of paroxysmal AF. 15 And the combined echocardiographic ratio of RV function to afterload is confirmed to be a useful predictor of poor outcome in acute PE. 25 Previous studies have found a correlation between right heart and prognosis, 26 but there is a lack of studies of echocardiographic indices and AF correlation in PE patients.
Current guidelines do not provide specific guidance on anticoagulation for PE in the context of AF, 27 but the identification of AF may influence the prolonged duration of anticoagulation and the prognosis of patients with PE. This study demonstrates the potential value of incorporating morphological examination into the assessment of PE, with the aim of facilitating the early identification of AF. Furthermore, dynamic ECG during hospitalization in PE patients could be essential rather than once at admission, especially in patients with enlargement of left or right atrium. When new-onset AF is identified in a patient with PE, it is crucial to evaluate the need for pharmacological or electrical cardioversion in accordance with established guidelines. 27 In addition, anticoagulation therapy should be initiated promptly if the patient's bleeding risk is assessed to be low. For PE patients combined with AF, the duration of anticoagulation therapy is typically extended. However, there is no definitive guideline on whether lifelong anticoagulation is required in these patients, leaving this decision to clinical judgment based on individual risk factors and patient-specific considerations.
There are also limitations in our study. This was a single-center, retrospective study with small sample size. Based on the definition of new-onset AF in this study, patients may simply have been previously asymptomatic and there may be an underestimation of outcomes. Our study had only in-hospital outcome and lacked detailed information on long-term outcome. Nevertheless, the observation of a robust association in a limited number of observational studies may also provide some viewpoints. Moreover, the participants included in the study were predominantly older, so the applicability of these findings to the general population warrants further investigation and validation. Compared to younger patients, older individuals showed a higher incidence of both AF and PE, highlighting the need for increased attention to this population. Future multicenter, large sample, prospective studies are needed to confirm the findings of this study with systematic follow-up.
Conclusions
Both severity of acute PE and enlargement of left or right atrium were associated with an increased risk of new-onset AF in patients with acute PE. This sheds light on the potential early detection of new-onset AF.
Supplemental Material
sj-docx-1-cat-10.1177_10760296251322779 - Supplemental material for Clinical Characteristics and Influencing Factors of New-Onset Atrial Fibrillation in Patients with Acute Pulmonary Embolism: A Case-Control Study
Supplemental material, sj-docx-1-cat-10.1177_10760296251322779 for Clinical Characteristics and Influencing Factors of New-Onset Atrial Fibrillation in Patients with Acute Pulmonary Embolism: A Case-Control Study by Rui Liang, Dong Liu, Yinong Chen, Ying Tang, Qian Gao, Wanmu Xie, Yihong Sun and Zhenguo Zhai in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Acknowledgments
We would like to express our gratitude to Kevin I Duan from the University of British Columbia for his valuable contributions to the study design. We also wish to thank the ATS MECOR program for providing clinical methodology training.
Data Availability Statement
Data described in the manuscript, code book, and analytic code will be obtained by contacting the corresponding author.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval
This study involving human participants were reviewed and approved by the Ethics Committee of China-Japan Friendship Hospital (2016-SSW-7).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the Fund of the National Key Research and Development Program of China (grant number No. 2023YFC2507200), National High Level Hospital Clinical Research Funding (grant number 2023-NHLHCRF-YGJH-03), and Chinese Academy of Medical Sciences Innovation Fund for Medical Sciences (grant numbers 2021-I2M-1-049 and 2021-I2M-1-061).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
