Abstract
Rivaroxaban was FDA-approved in 2019 for venous thromboembolism (VTE) prophylaxis in acutely ill hospitalized patients. Little to no published data is available to determine the level of correlation between rivaroxaban drug concentration and UFH/LMWH calibrated anti-Xa assays at VTE prophylactic doses of rivaroxaban 10 mg daily. This study aimed to assess the anticoagulant effects of rivaroxaban prophylactic doses using LMWH calibrated anti-Xa levels at the University of Colorado Hospital (UCH). This prospective cohort study evaluated seventy-three hospitalized patients at UCH taking rivaroxaban 10 mg daily for VTE prophylaxis from June 2023 to April 2024. Patients were enrolled if they were between the ages of 18-89 years old, received rivaroxaban 10 mg daily, and had active orders for coagulation studies. A linear regression model and coefficient of determination was used to evaluate the primary outcome assessing the relationship between rivaroxaban drug concentrations and anti-Xa levels. The LMWH calibrated anti-Xa assays were strongly correlated to rivaroxaban concentrations ranging from (1-59 ng/ml) in patients receiving rivaroxaban 10 mg daily, r2 = 0.99 (P < .00001). Our data suggests that LMWH calibrated anti-Xa levels less than 1.40 IU/ml may indicate minimal anticoagulation effects for rivaroxaban 10 mg daily. The secondary outcomes assessing the relationship between rivaroxaban drug concentrations and time since administration, r2 = 0.16 (P = .049), as well as time since administration of rivaroxaban and anti-Xa activity, r2 = 0.15 (P = .066), were weakly correlated and showed a trend. Characterizing the high correlation between anti-Xa levels and rivaroxaban plasma concentrations at 10 mg daily doses, provides additional insight to rivaroxaban's anticoagulant effects in clinical practice. This can be beneficial in various clinical scenarios and has the potential to reduce the waiting time for clinical procedures.
Introduction
Hospital-acquired venous thromboembolism (VTE), defined as a VTE event occurring during or within 3 months of hospitalization, account for more than 50% of all VTE events in the United States. 1 These preventable events have been linked to increased morbidity, mortality, and hospital length of stay. Approximately 12% of all VTE prophylaxis is not administered during hospitalization, with 30%-40% of missed doses attributed to patient refusal of injectable medications.2,3 Further exploration of general medication administrations showed that subcutaneous or injectable medications are more likely to be missed when compared to oral medications. 4
Historically, only subcutaneous injections, unfractionated heparin (UFH) and low-molecular-weight (LMWH) heparin, were Food and Drug Administration (FDA)-approved for VTE prophylaxis in medically ill patients. In 2011, following the RECORD 1-4 trials, rivaroxaban 10 mg daily was approved for VTE prophylaxis in the setting of total hip and knee replacements.5–8 In 2019, rivaroxaban 10 mg was approved for the indication of VTE prophylaxis in acutely ill hospitalized patients following the results of the MAGELLAN and MARINER trials.9–11 These studies showed that rivaroxaban 10 mg daily had favorable efficacy outcomes when compared to enoxaparin while exhibiting a similar safety profile.
Oral factor Xa inhibitors, like rivaroxaban, have been favored as anticoagulants due to their predictable pharmacokinetic and anticoagulation effect. 12 Rivaroxaban is expected to reach peak concentrations within 2-4 h of administration in adult patients.12,13 Steady-state peak concentrations vary by administered dose, with median concentrations expected to be around 125 ng/ml (91-196 ng/ml) for rivaroxaban 10 mg daily used for VTE prevention after total hip replacement, 229 ng/ml (178-313 ng/ml) for 15 mg once daily for stroke prevention in patients with atrial fibrillation and creatinine clearance of 30-49 ml/min, and 270 ng/ml (189-419 ng/ml) for 20 mg once daily dosing used for DVT treatment. 14 In addition, median trough concentrations are expected to be around 9 ng/ml (1-38 ng/ml), 57 ng/ml (18-136 ng/ml), and 26 ng/ml (6-87 ng/ml), respectively. 14 Consequently, rivaroxaban drug concentrations of 50 ng/ml or less are considered to have undetectable or minimal anticoagulation effect.15–17 These predictable effects have allowed for the use of factor Xa inhibitors without routine monitoring. However, there are clinical situations where monitoring the anticoagulant effect may be beneficial, such as: perioperative monitoring of bleeding risk, acute bleeding events, renal insufficiency, recurrent thrombosis, overdose, concern for malabsorption, or with concomitant CYP3A4 and P-glycoprotein inducers or inhibitors. Rivaroxaban drug concentrations have correlated with prolongation of prothrombin time (PT) and have minimal effect on activated partial thromboplastin time (aPTT) in plasma. 12 However, use of PT to monitor factor Xa inhibitor pharmacodynamic effects in clinical practice is not reliable due to variations in patient conditions and assays used. The international normalized ratio (INR), a standardized version of PT, is also associated with elevated values in the setting of factor Xa inhibitors, although, INR values for measuring anticoagulation effect of factor Xa inhibitors is not routinely recommended due to similar issues of high variability. 18
Anti-Xa monitoring is frequently used in clinical practice to measure anticoagulation effects of UFH and LMWH. Currently, there are no FDA approved or readily available direct oral anticoagulant (DOAC) calibrated anti-Xa assays. 19 Such DOAC calibrated anti-Xa assays are only available for research purposes. Studies have showcased a linear association between rivaroxaban plasma drug concentrations and UFH/LMWH calibrated anti-Xa assays. 20 Such data suggests that heparin-calibrated anti-Xa assay of 0.96 IU/ml correlated to 50 ng/ml drug concentrations of treatment doses of rivaroxaban 20 mg daily. 21 Nevertheless, these studies included data from patients receiving treatment doses of rivaroxaban. The question remains on whether the same high correlation between rivaroxaban plasma drug concentration and UFH/LMWH calibrated anti-Xa assays will hold true for low-dose rivaroxaban 10 mg daily. This study aimed to assess the correlation between LMWH calibrated anti-Xa levels and drug concentrations for rivaroxaban 10 mg daily across the dosing interval.
Methods
This single center, prospective cohort study was conducted at the UCHealth University of Colorado Hospital (UCH) from June 1, 2023, to April 1, 2024. Enrolled patients were between the ages of 18-89 years and received at least one dose of rivaroxaban 10 mg daily for VTE prophylaxis during their hospitalization. Patients were required to have active laboratory orders to add-on anti-Xa levels to previous blood draws. Patients were excluded from the study if they received any other anticoagulant while on rivaroxaban. Of note, UCH institutional guidance supports the use of VTE prophylaxis in patients considered critically ill or with a PADUA score of ≥4. Approved institutional VTE prophylaxis guidance for using rivaroxaban 10 mg daily is preferred in patients with low bleeding risk, those unlikely to have procedures, and those with active or history of heparin induced thrombocytopenia (HIT). It is recommended that rivaroxaban be avoided in those with a CrCl < 15 ml/min, end-stage renal disease (ESRD), or concomitant use with strong CYP3A4 and/or PGP inhibitors/inducers.
Eligible patients were identified via the electronic medical record. Providers consented for the ordering of LMWH calibrated (dalteparin, enoxaparin) anti-Xa levels as an add-on to the patient's routine labs. UCH clinical laboratory performed anti-Xa testing and reporting per usual procedures using the STA-R MAX laboratory instrument. Both heparin and LMWH anti-Xa assays utilize the same reagents and laboratory instrument at the UCH anticoagulation laboratory and only differ by the reported reference ranges. LMWH calibrated anti-Xa assays are reported within reference ranges of 0.1-1.7 IU/ml. Results outside the assay's reference ranges are reported as <0.1 IU/ml or >1.7 IU/ml by the UCH clinical laboratory. Samples of unused blood for anti-Xa level testing were collected by the anticoagulation laboratory from the patient's routine morning labs. Investigators notified the UCH anticoagulation laboratory for collection and storage of identified samples. Timing and volume of blood sample collection occurred as a result of normal practice procedures and were not research guided. Collected samples were obtained in 3.2% sodium citrate tubes (BD Vacutainer 1.8 ml) and plasma samples were routinely stored at −70 °C. The laboratory stored samples until assessment of rivaroxaban drug concentrations was completed. Drug concentration testing was conducted via the same STA-R MAX laboratory instrument as the LMWH anti-Xa assay. However, the calibration, controls, and protocols were supplied by Diagnostica Stago to perform drug concentration testing with the device. 21
The primary outcome of the trial was to assess the correlation between rivaroxaban drug concentrations and LMWH calibrated anti-Xa levels at various time points following administration. Secondary outcomes included evaluating rivaroxaban drug concentrations at various time points after administration and LMWH calibrated anti-Xa levels from time of rivaroxaban drug administration.
A linear regression model was created by plotting the relationship between rivaroxaban drug concentrations and LMWH calibrated anti-Xa levels. A coefficient of determination was used to analyze the data to determine the relationship between the dependent variable of drug concentrations to the independent variable of LMWH calibrated anti-Xa levels. A correlation coefficient of 0.80 or greater was considered a strong correlation between variables. 22 Descriptive and inferential statistical testing was conducted for data analysis via a Pearson's correlation coefficient between variables plotted on the predictive linear regression model. Statistical analyses of the results were performed using SAS software version 9.2 (SAS Institute, Cary, NC),GraphPad Prism 4.0 (GraphPad Software, San Diego, CA), and InStat3.06 (GraphPad Software, San Diego, CA). An alpha level of less than 0.05 was considered to be statistically significant. Scatter plots analysis was performed via Microsoft Excel 2024. This research was approved by the University of Colorado Institutional Review Board under the protocol number 23-1911.
Results
From June 2023 through April 2024, a total of 73 patients were enrolled from the UCHealth University of Colorado Hospital. A total of 151 samples were collected for assessment of rivaroxaban drug concentrations and LMWH calibrated anti-Xa assays for rivaroxaban 10 mg daily. Roughly 77% of the 151 samples collected were at steady-state concentrations of rivaroxaban. Additional baseline characteristics and clinical information of patient population included are represented in Table 1.
Baseline Characteristics.

The primary outcome of determining the relationship of rivaroxaban drug concentrations and LMWH calibrated anti-Xa levels was analyzed via a linear regression model (Figure 1). Rivaroxaban concentrations and LMWH calibrated anti-Xa activity were strongly correlated, r2 = 0.99, P < .00001. A broad range of rivaroxaban concentrations (1-59 ng/ml) and anti-Xa levels (0.1-1.7 IU/ml) were noted for collected samples.

Correlation between rivaroxaban drug concentrations versus LMWH calibrated anti-Xa activity.
The secondary outcome assessing the relationship between rivaroxaban drug concentrations and time since administration were weakly correlated, r2 = 0.16, P = .049 (Figure 2). Similarly, the time since administration (median 17 h, interquartile range, 12 to 20) of rivaroxaban and anti-Xa activity were also weakly correlated r2 = 0.15, P = .066 (Figure 3). The time of sample collection in relation to the time of rivaroxaban administered was variable and ranged from 1-48 h post-administration.

Correlation between time since administration versus rivaroxaban concentrations.
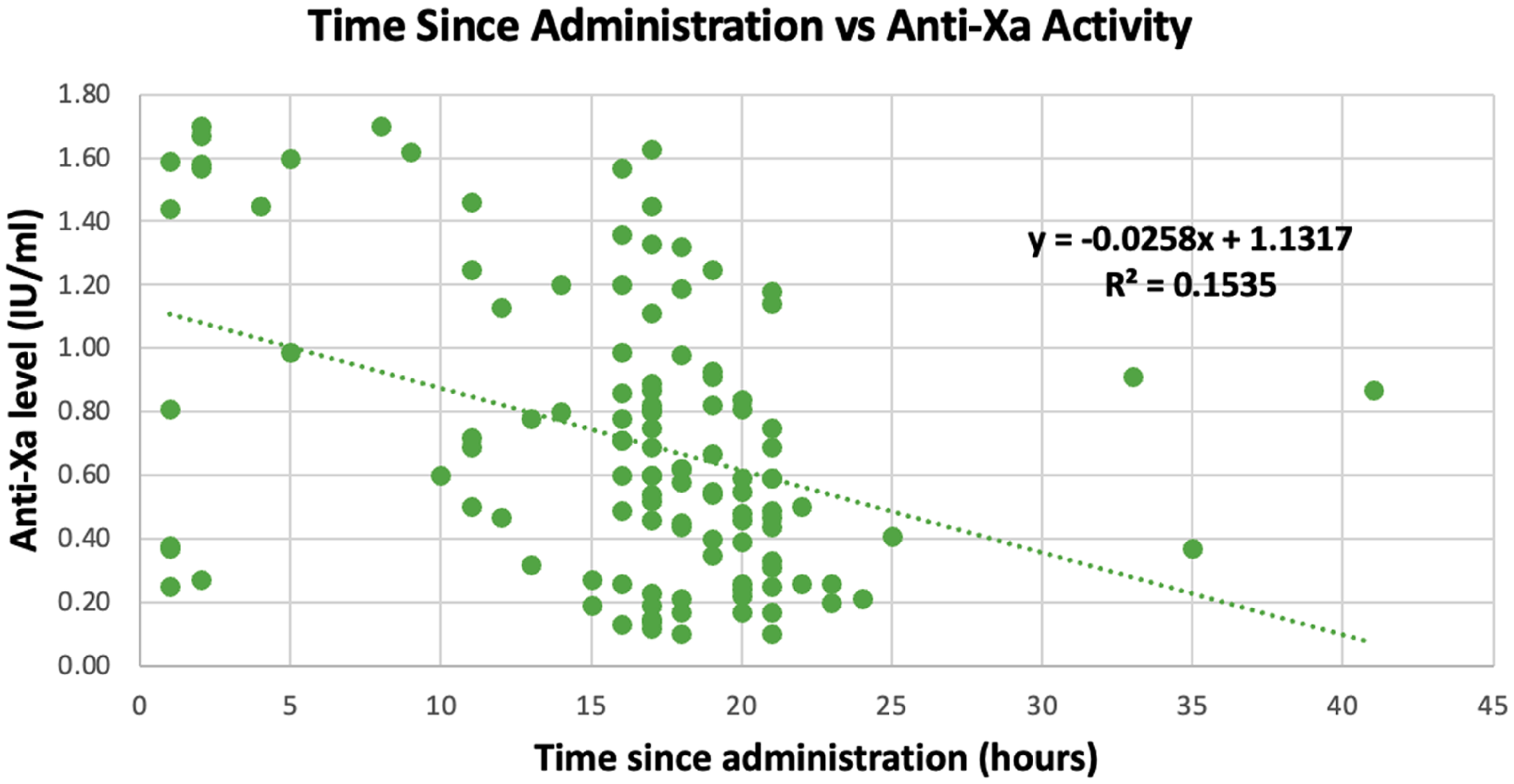
Correlation between time since administration versus LMWH calibrated anti-Xa activity.
Expected trough concentrations of rivaroxaban 10 mg daily range from 1-38 ng/ml (yellow) and drug concentrations <50 ng/ml (blue) are considered to have minimal to no residual anticoagulation effect.
Discussion
The strong and linear correlation seen in the primary outcome is consistent with previously published data on treatment doses of rivaroxaban. 21 Correlation was consistently seen over a broad rivaroxaban concentration (1-59 ng/ml). These findings provide additional evidence for the potential use of LMWH calibrated anti-Xa assays to assess anticoagulant effects for lower doses of rivaroxaban. Considering there are no commercially available DOAC calibrated anti-Xa assays, such data may be beneficial for decision making in clinical practice. Currently, no guidance exists on how to clinically interpret LMWH calibrated anti-Xa levels for factor Xa inhibitors. Our institution has observed provider hesitation in using rivaroxaban for VTE prophylaxis due to anticipation of procedures during admission. Providing such guidance on clinical interpretation of LMWH calibrated anti-Xa levels for factor Xa inhibitor monitoring can be beneficial in improving provider comfortability. Other clinical situations where monitoring may be beneficial include: acute bleeding events, renal insufficiency, recurrent thrombosis, overdose, concern for malabsorption, or in combination with CYP3A4 or P-glycoprotein inhibitors or inducers.
Findings from this research may help provide preliminary guidance, in addition to previously published data. 23 Current literature demonstrates rivaroxaban plasma concentrations range from 20-660 ng/ml at therapeutic doses and 1-196 ng/ml for prophylactic doses.13,14 Furthermore, treatment rivaroxaban dosing has been reported to have peak anti-Xa levels from 3.80-6.20 IU/ml and trough anti-Xa levels around 0.60-1.00 IU/ml. 21 Our results show LMWH calibrated anti-Xa assay values of 0.8 −1.40 IU/ml are produced when drug concentrations are around 30-50 ng/ml. Suggesting that LMWH calibrated anti-Xa levels less than 1.40 IU/ml may indicate minimal anticoagulation effects for rivaroxaban VTE prophylaxis.
Current guidelines in the pre-procedural setting recommend holding rivaroxaban for 5-half-life intervals or 2-3 days prior to a medium-high risk procedure and at least 2-half-lives or 1 day prior to a low-risk procedure.16,17 A majority of data used to base such a recommendation is largely from patients receiving treatment doses of rivaroxaban and a broad generalization of the patient population included. Obtaining LMWH-calibrated anti-Xa assays as a means of assessing anticoagulation effect has the potential for shortening the time to procedures if they correlate to drug concentrations of less than 50 ng/ml; which were not associated with increased bleeding events in perioperative setting. 16 As well as, increasing the time to procedure for patients whose LMWH calibrated anti-Xa assays correlate to high anticoagulation effects and potentially an increased risk of perioperative bleeding.
One limitation of this study was that not all patients obtained steady-state concentrations for rivaroxaban 10 mg daily dosing at the time the anti-Xa levels were drawn. This is likely due to patients being placed on rivaroxaban for VTE prophylaxis once clinically stable and close to discharge. Plasma samples in which anti-Xa assays were obtained were collected and stored for a period of time at −70 °C until materials to run drug concentrations were available.
Additionally, LMWH calibrated anti-Xa assay parameters are reported between 0.1 to 1.7 IU/ml. Exact anti-Xa levels outside of these parameters are not reported by the laboratory and therefore, were not included in the data analysis. In the research setting, we are able to dilute samples with blank plasma to determine actual anti-Xa levels above the reference of quantification. 21 Due to the high correlation between primary outcome variables, it is possible to extrapolate the anti-Xa assays from the line of best fit using drug concentrations data. However, the decision was made to not include data points falling outside the reference ranges to avoid skewing research results. Values outside the limits of quantification are not available in routine clinical practice and would not follow standards set by the Clinical and Laboratory Standards Institute (CLSI).
Knowledge gaps remain for how different variables may affect the correlation between rivaroxaban drug concentrations and LMWH calibrated anti-Xa assays. In particular, the effect of renal function, extremes in body weight, and route of administration via feed tube is not well defined in this study.
Conclusion
Our study demonstrated that LMWH calibrated (dalteparin, enoxaparin) anti-Xa assays strongly correlate to rivaroxaban concentrations ranging from (1-59 ng/ml) at VTE prophylactic doses. Only a weak correlation was seen when comparing time of administration to LMWH calibrated anti-Xa assays and rivaroxaban drug concentrations. The next step at our institution is to translate clinical interpretation of LMWH calibrated anti-Xa assays in determining anticoagulation effect when using rivaroxaban for VTE prophylaxis. Further research is needed to assess the effects of renal function, extreme body weights, and route of administration on LMWH calibrated anti-Xa levels.
Footnotes
Acknowledgments
The calibration, reagent, and control materials used to determine rivaroxaban concentration within this article were provided kindly by Diagnostica Stago.
Consent for Publication
Not applicable.
Consent to Participate
Informed consent to participate was waived by the University of Colorado Institutional Review Board (IRB). Informed consent was not required due to research being conducted under routine clinical practice allowing for exemption from IRB review
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Standards
This research was approved by the University of Colorado Institutional Review Board under the protocol number 23-1911.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
