Abstract
Introduction:
Although the role of inflammation in DVT has been investigated in different studies, there is no definite answer as to whether increased systemic inflammation is the cause or the consequence of DVT.
Aim:
To follow inflammatory parameters in a cohort of patients with idiopathic DVT.
Methods:
Out of 49 patients with an acute idiopathic DVT, which were investigated four months after an acute episode (DEVTA 1), 43 patients were included in the follow-up study investigating inflammatory markers and hemostatic markers of endothelial damage five years after an acute DVT (DEVTA 2). A control group consisted of 43 sex and age matched healthy subjects (CONTROLS).
Results:
The levels of inflammatory markers were significantly higher in DEVTA 2 in comparison to CONTROLS: tumor necrosis factor alpha 2.0 pg/mL (1.1-2.3) vs 1.3 pg/mL (0.8-1.9), p < .001, high sensitivity C-reactive protein 3.2 mg/L (1.5-5.2) vs 1.7 mg/L (0.9-3.0), p = .008, interleukin-6 (IL-6) 2.7 pg/mL (2.0-3.5) vs 2.1 pg/mL (1.5-2.6), p = .025, IL-8 5.0 pg/mL (3.6-7.3) vs 2.4 pg/mL (1.8-2.8), p < .001. IL-10 was significantly decreased (0.9 pg/mL (0.7-1.8) vs 1.8 (1.5-2.2), p < .001. Most of the proinflammatory markers remained elevated in the DEVTA 2 in comparison to DEVTA 1. Markers of endothelial damage were higher in DEVTA 2 in comparison to CONTROLS and higher than in DEVTA 1.
Conclusion:
Patients with idiopathic DVT have long-term increased inflammatory markers and markers of endothelial damage. These findings favor the hypothesis that inflammation is a cause and not merely a consequence of acute DVT.
Introduction
Deep vein thrombosis (DVT) is a frequent disease that predominantly affects the deep veins of the lower limbs. The underlying pathogenic mechanisms are not completely understood in patients with known risk factors for DVT and even less in those patients without known risk factors. The most probable pathophysiological mechanisms that increase a prothrombotic tendency include hypercoagulability of the blood, endothelial damage, and blood stasis. 1 Vessel wall damage is closely related to an inflammatory response. The role of inflammation in the pathogenesis of arterial thrombosis has been elucidated. 2 However, little is known about the relationship between inflammation and DVT. A few studies have confirmed an increased systemic inflammatory response in patients with DVT. 3,4 Data from investigations of physiological processes for the development of DVT also indicate that inflammation could be the primary process that damages the vessel wall and initiates thrombus formation at the venous wall and that the coagulation system in both arterial and venous systems are coupled by a common pathway. 3 In our previous studies, we showed that some inflammatory markers and markers of endothelial damage in patients with idiopathic DVT were still increased 4 months after an acute phase of the disease in comparison to healthy controls. 4,5 Proinflammatory cytokines such as interleukin 6 (IL-6), IL-8, and high-sensitivity C-reactive protein (hsCRP) were increased, while the anti-inflammatory interleukin 10 (IL-10) was decreased.
There is no definite answer as to whether increased inflammatory markers are the cause or the consequence of DVT. In the Leiden thrombophilia study (LETS), both cytokines (IL-6 and IL-8) and tumor necrosis factor α (TNF-α) were associated with a 2- to 3-fold increased risk of first DVT. 6 However, in most of these studies, cytokine levels were investigated only in the phase of an acute event.
Therefore, the aim of our study was to investigate whether persistent inflammatory activity and endothelial damage exist in patients with DVT 5 years after the initial event. The hypothesis is that if an increased inflammatory response and endothelial dysfunction still exist after 5 years, the deterioration must be long-lasting and is probably present before the appearance of the acute DVT and as such is involved in the pathogenesis of DVT.
Patients and Methods
Between June 2006 and November 2008, 49 consecutive patients admitted to our hospital or presented at the outpatient department (University Medical Centre) with a first episode of acute, symptomatic idiopathic DVT of the lower limbs and who fulfilled the strict inclusion criteria were included in the case–control study, that is, dysfunction of endothelium in venous thrombosis analysis (DEVTA 1) as described elsewhere. 4 In brief, patients with secondary DVT or symptomatic atherosclerosis (known coronary artery disease, ischemic stroke, transient ischemic attack, or intermittent claudication) and patients with renal and liver disease or dysfunction were excluded. All patients in the acute phase of the disease were treated with single daily subcutaneous injections of low-molecular-weight heparin at therapeutic dose (dalteparin 200 IU/kg) along with vitamin K antagonist (warfarin), starting from the first treatment day. Dalteparin and warfarin were simultaneously administered until an international normalized ratio (INR) of 2.0 was maintained for at least 2 consecutive days. All patients received anticoagulant therapy with warfarin for 6 months with regular INR monitoring. Thereafter, patients were followed every year at our outpatient clinic. Between September 2013 and December 2014, we invited the same cohort of patients to participate in the DEVTA 2 study, which aimed to reevaluate markers of inflammation and endothelial dysfunction in the chronic phase of the disease, that is, 5 years after an initial diagnosis. Of the 49 patients, 43 were included in this follow-up study. Of the 6 patients not included in the study, 3 patients died. One of the male patients died at the age of 85 due to comorbidities associated with Alzheimer dementia, the other at the age of 82 due to Parkinson disease, while the female patient died of ischemic stroke at the age of 74. According to our records and additional data obtained from their family physicians, none of them suffered recurrent DVT or pulmonary embolism (PE). Of the remaining 3 patients, 1 of the patients could not be located (moved out of the country) and 1 patient refused to sign the informed consent form for the reevaluation study. One of the male patients was treated for 2 cancers, that is, for colon carcinoma in 2009 and for epidermoid carcinoma of the lungs in 2011, and therefore he was not included in the second cohort (Figure 1). A control group was composed of 43 age- and sex-matched healthy participants who were randomly sampled from an institutional research database (controls). The demographic characteristics of all participants are depicted in Table 1.
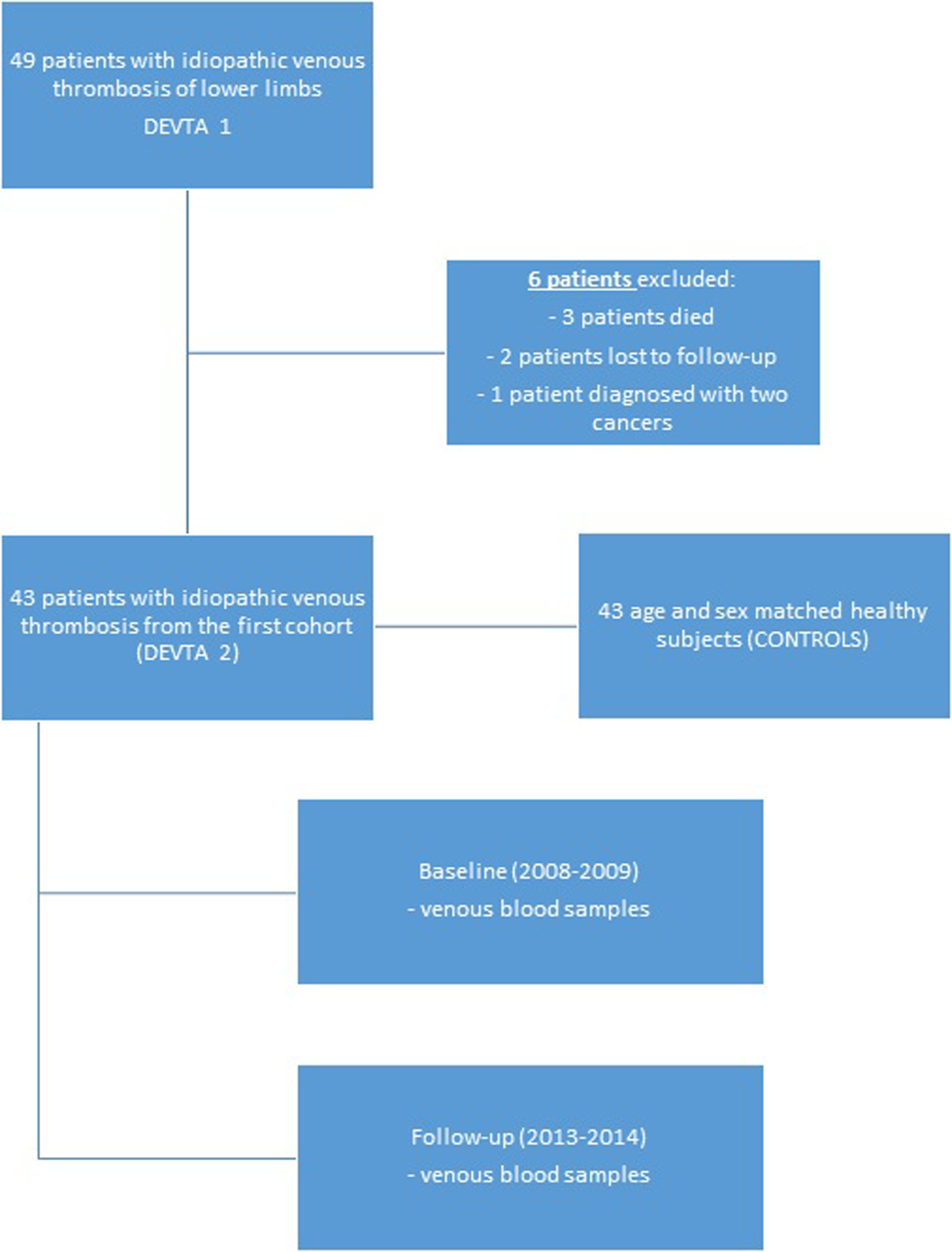
Flowchart of the study protocol for DEVTA study. DEVTA indicates dysfunction of endothelium in venous thrombosis analysis.
Demographic and Clinical Characteristics of the Study Population.a
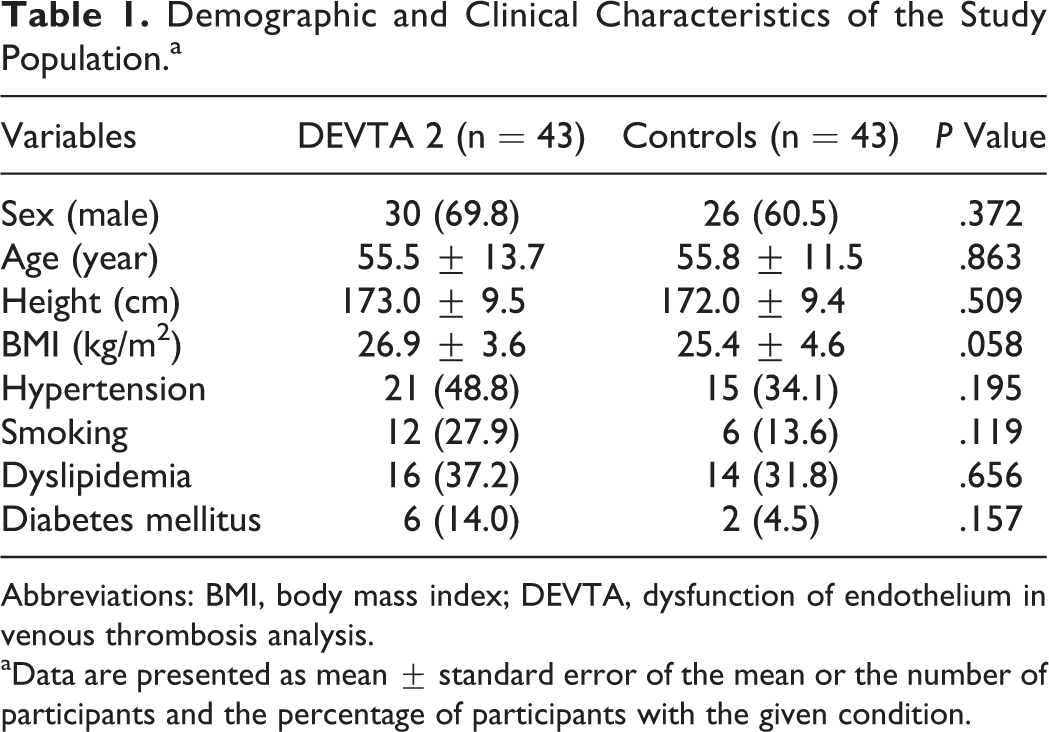
Abbreviations: BMI, body mass index; DEVTA, dysfunction of endothelium in venous thrombosis analysis.
aData are presented as mean ± standard error of the mean or the number of participants and the percentage of participants with the given condition.
Laboratory Analysis
Blood for laboratory analysis was collected in the morning after a 12-hour overnight fast. Samples were drawn from the antecubital vein using a 19-gauge needle with minimal venous stasis. For the analysis of white blood cell count, hemoglobin concentration, and platelets, peripheral venous blood specimens were sampled in Vacutainer tubes containing K-ethylenediaminetetraacetic acid and placed immediately to an automated hematologic analyser, K-4500 (TOA Medical Electronics, Kobe, Japan). Blood for the analysis of glucose and lipids was collected into Vacutainers without additives. Plasma glucose level was determined using an autoanalyzer (Express Plus; Ciba Corning Diagnostics, Palo Alto, California). Concentrations of total cholesterol, high-density lipoprotein cholesterol (HDL-C), and triglycerides (TG) were determined by standard colorimetric assays (Ektachem 250 Analyser; Eastman Kodak Company, Rochester, Minnesota). Low-density lipoprotein cholesterol (LDL-C) was calculated from the Friedewald’s formula by dividing the TG by 5 and subtracting the HDL-C. 7
For detection of the inflammatory markers and endothelial damage markers, blood was anticoagulated with the addition of 4.5 mL of whole blood to 0.5 mL of either 500 mmol/L acidified citrate (tPA, tissue plasminogen activator [t-PA] and plasminogen activator inhibitor 1 [PAI-1] assays) or 105 mmol/L sodium citrate. Platelet poor plasma was prepared with centrifugation at 3000g for 20 minutes at 22°C; the plasma was removed, transferred into sterile cryovials in aliquots of 1 mL, and immediately frozen and stored at −70°C for later assay analysis. At the time of the analysis, the samples were thawed in a water bath at 37°C for 10 minutes and after mixing and were allowed to stand at room temperature for at least 15 minutes before use.
The concentrations of TNF-α (Quantikine HS Human TNF-α Immunoassay; R&D Systems, Minneapolis, MN, USA), IL-6 (Quantikine Human IL-6 Immunoassay; R&D Systems), IL-8 (Quantikine HS Human CXCL-8, IL-8 Immunoassay; R&D Systems), IL-10 (Quantikine HS Human IL-10 Immunoassay; R&D Systems), von Willebrand factor (vWF) antigen (Axis Shield, Scotland, UK), and P-selectin (Human sP-Selectin/CD62P; R&D Systems) were measured by an enzyme-linked immunosorbent assay (ELISA) using commercially available kits according to the manufacturers’ instructions. The minimum detectable amounts of vWF, TNF-α, and P-selectin were 1.0%, 1.2 pg/mL, and 0.59 ng/mL, respectively. All the experiments were run in duplicate and the intra- and interassay coefficients of variation were 2.4% and 2.8% for vWF, 5.6% and 6.9% for P-selectin, and 12.3% and 14.3% for TNF-α. The hsCRP levels in the serum were assayed by a fully automated latex-enhanced nephelometric immunoassay (Dade Behring, Marburg, Germany).
Determination of plasminogen activators and inhibitors were done by using commercially available assay kits. The levels of tPA and PAI-1 antigen were assessed by using IMULYSE tPA and PAI-1 ELISA kits, both from Biopool (Umeå, Sweden). The tPA activity was measured by using a chromogenic assay (Chromolyse tPA) and PAI-1 activity was measured using a amidolytic assay (Spectrolyse/pL), both from Biopool (Umeå, Sweden). The inter- and intraassay variations were3.5% to 5.4% and 4.9% to 5.5% for tPA antigen;2.4% to 3.3% and 1.9% to 2.9% for PAI-1, and5.2% to 5.3% and 3.9% to 7.0% for tPA activity.
t-PA activity, t-PA antigen, and PAI-1 activity levels follow a circadian rhythm with peak marker activity levels occurring in the morning. Therefore, all blood samples were drawn in the morning between 7 AM and 9 AM. 8
Statistical Analysis
The normality of data was tested using the Shapiro-Wilk test. Normally distributed data were expressed as mean ± standard deviation for continuous variables and number with percentages for categorical variables. Nonparametric variables were expressed as median and interquartile range (25th-75th percentile). The variables age, height, and body mass index (BMI) were normally distributed. Most of the laboratory variables were not uniformly distributed, therefore, laboratory data are reported as the median and interquartile range. The significance of differences in concentrations between the patients and the controls was tested with independent Student t test for unpaired samples, or the Mann-Whitney U test for nonparametric data. Due to violations of parametric test assumptions (nonnormal distribution), comparison of changes in measured values over time (DEVTA 1 vs DEVTA 2) was carried out using the Friedman test. The Wilcoxon signed rank test was then performed to show the significance of pairwise differences. All tests were 2 sided, and a P value of less than .05 was considered significant.
The correlation coefficient was used for the assessment of the relation between inflammatory parameters and levels of markers of endothelial damage. The correlation was defined as very good if it was more than 0.75, as good if it was between 0.50 and 0.75, and as moderate if it was less than 0.50. With the Spearman ρ correlation test, the significance of the correlation coefficient was calculated and defined as significant at the .01 level (2 tailed).
Statistical analysis was performed with Statistical Package for the Social Sciences software for Windows, version 23 (SPSS Inc, Chicago, Illinois).
Ethics
The study was conducted according to the principles of the Declaration of Helsinki and approved by the National Medical Ethics Committee. Written informed consent was obtained from all included participants.
Results
Patient Cohort and Demographics
The study included a cohort of patients with idiopathic DVT followed up prospectively, since 2008. Data on the first cohort have already been published on venous 4 and arterial 1,9 studies.
The mean age of the patients included in the second cohort was 55.5 ± 13.7 years. There were 70% males. After 5 years, 33 patients were free of recurrent thromboembolic events (76.7%), 8 (18.6%) suffered recurrent proximal DVT (2 with concurrent PE), and 2 (4.7%) distal DVT accompanied by superficial vein thrombosis.
In comparison to controls, the patients didn’t differ in their clinical characteristics with the exception of BMI, which was slightly higher in patients than in the controls; however, this did not reach statistical significance (Table 1). The biochemical and hematological parameters did not differ between DEVTA 1 (4 months after an acute DVT episode) and DEVTA 2 (5 years after an acute DVT). However, the levels of total cholesterol and LDL-C were higher in the cohort of patients (DEVTA 2) in comparison to controls (Table 2).
Biochemical and Hematological Parameters in Patients Investigated 4 Months (DEVTA 1) and 5 Years (DEVTA 2) After an Acute Episode of DVT in Comparison to Healthy Participants (Controls).a
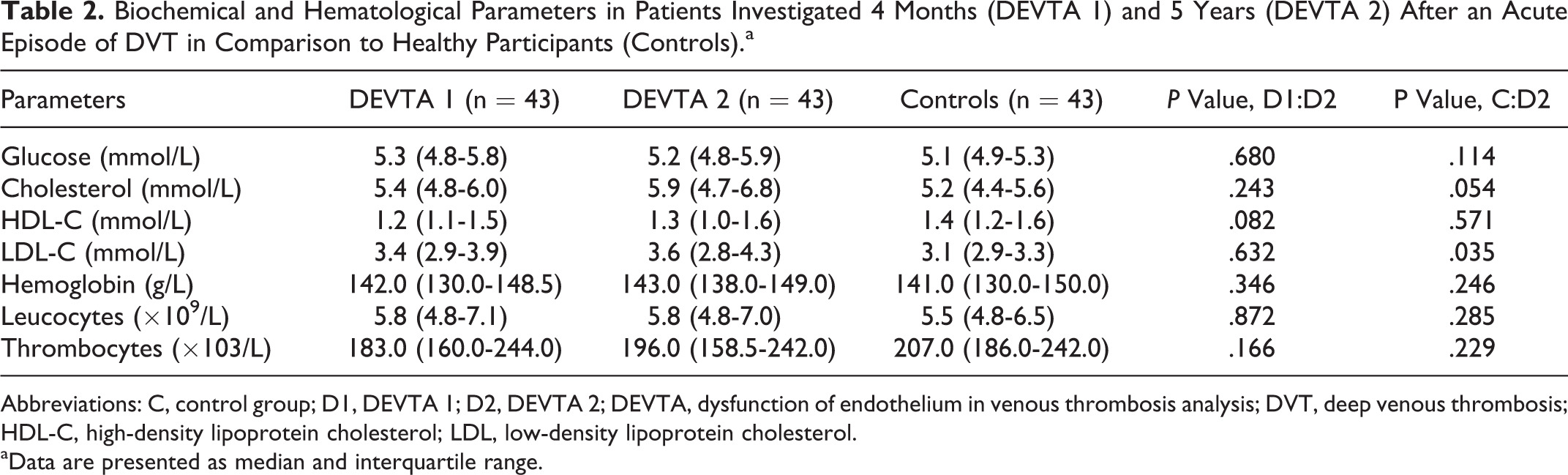
Abbreviations: C, control group; D1, DEVTA 1; D2, DEVTA 2; DEVTA, dysfunction of endothelium in venous thrombosis analysis; DVT, deep venous thrombosis; HDL-C, high-density lipoprotein cholesterol; LDL, low-density lipoprotein cholesterol.
aData are presented as median and interquartile range.
Inflammatory Markers
Table 3 shows the levels of inflammatory markers in both cohorts of patients and the controls. Some inflammatory markers (TNF-α, hsCRP, and IL-6) did not significantly differ between the cohorts. They were significantly elevated in comparison to the levels in the healthy controls. However, the circulating levels of IL-8 were higher after 5 years of observation compared to the controls and patients after 4 months of follow-up. In contrast, the levels of the anti-inflammatory IL-10 were significantly lower in the DEVTA 2 than in the DEVTA 1 cohort and lower than that in the healthy participants.
Inflammatory Markers in the Patients Who Participated in DEVTA 1 and DEVTA 2 in Comparison to the Healthy Participants (Controls).a
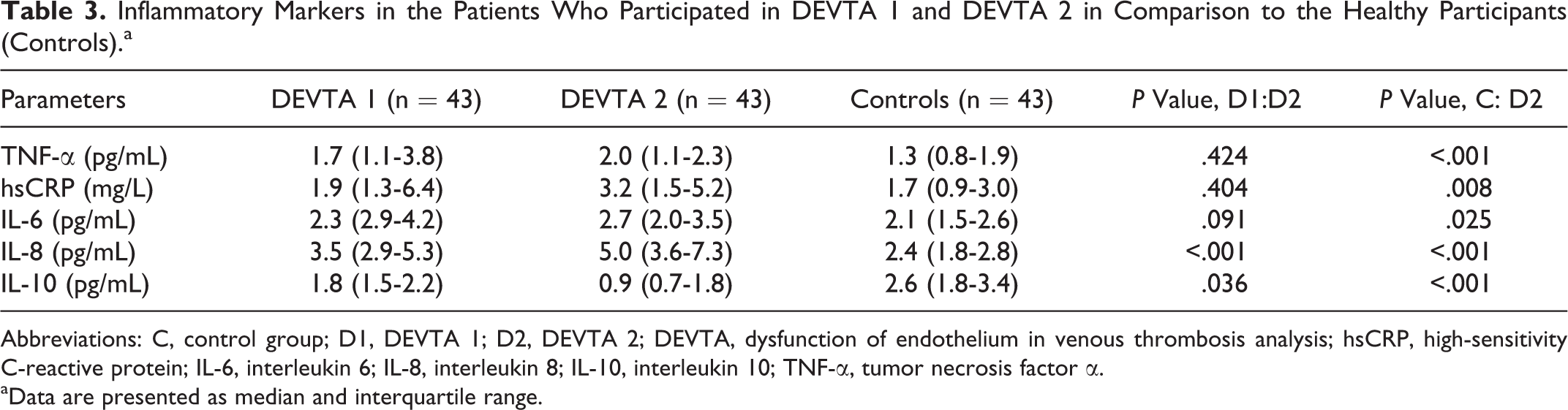
Abbreviations: C, control group; D1, DEVTA 1; D2, DEVTA 2; DEVTA, dysfunction of endothelium in venous thrombosis analysis; hsCRP, high-sensitivity C-reactive protein; IL-6, interleukin 6; IL-8, interleukin 8; IL-10, interleukin 10; TNF-α, tumor necrosis factor α.
aData are presented as median and interquartile range.
Hemostatic Markers of Endothelial Damage
The von Willebrand factor antigen, one of the circulating factors of endothelial damage, remained significantly elevated at 5 years and was higher than that at 4 months after the acute episode of DVT: in healthy participants: 92.0% (77.0-105.0), in DEVTA 1 participants: 146% (119-173), and in DEVTA 2 participants: 155.1% (128.0-223.4), P < .001 and P = .006, respectively. The tPA activity and the levels of P-selectin remained stable over time and did not differ from the controls. The levels of tPA antigen and PAI-1 activity, known markers of impaired fibrinolytic activity, were similar in DEVTA 1 and DEVTA 2 patients. However, significantly higher levels of tPA antigen and PAI-1 activity were observed in the patients after 5 years (DEVTA 2) in comparison to controls (Table 4).
Levels of Hemostatic Markers of Endothelial Damage in the Patients Who Participated in DEVTA 1 and DEVTA 2 in Comparison to Healthy Participants (controls).a
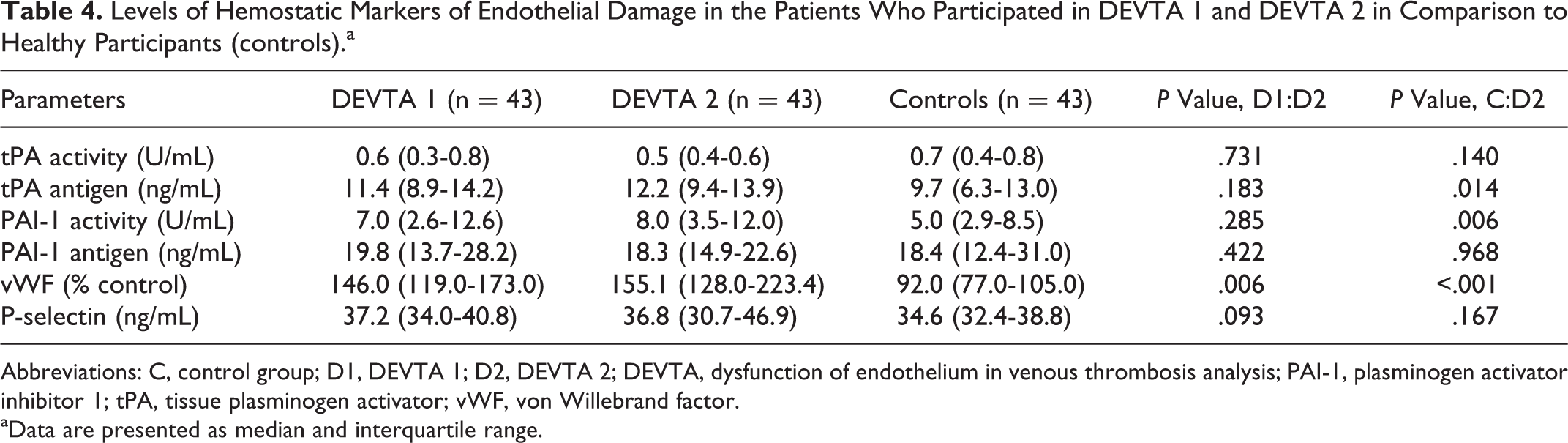
Abbreviations: C, control group; D1, DEVTA 1; D2, DEVTA 2; DEVTA, dysfunction of endothelium in venous thrombosis analysis; PAI-1, plasminogen activator inhibitor 1; tPA, tissue plasminogen activator; vWF, von Willebrand factor.
aData are presented as median and interquartile range.
Correlations Between the Inflammatory Markers and Markers of Endothelial Damage
In the DEVTA 2 cohort, significant correlations were found between the inflammatory markers (hsCRP, TNF-α, IL-6, and IL-8) and hemostatic markers of endothelial damage (vWF, tPA antigen, and PAI-1 activity; Table 5). Moderate to good correlations were found between hsCRP, IL-8, IL-6, and P-selectin or vWF, respectively. Further, moderate correlations between the inflammatory markers and the levels of some lipids were detected. Additionally, the relationship between endogenous fibrinolysis and some biochemical markers was established (Table 6).
Correlation Between Different Laboratory Parameters in the Follow-Up Cohort of Patients With Idiopathic Venous Thrombosis (DEVTA 2).
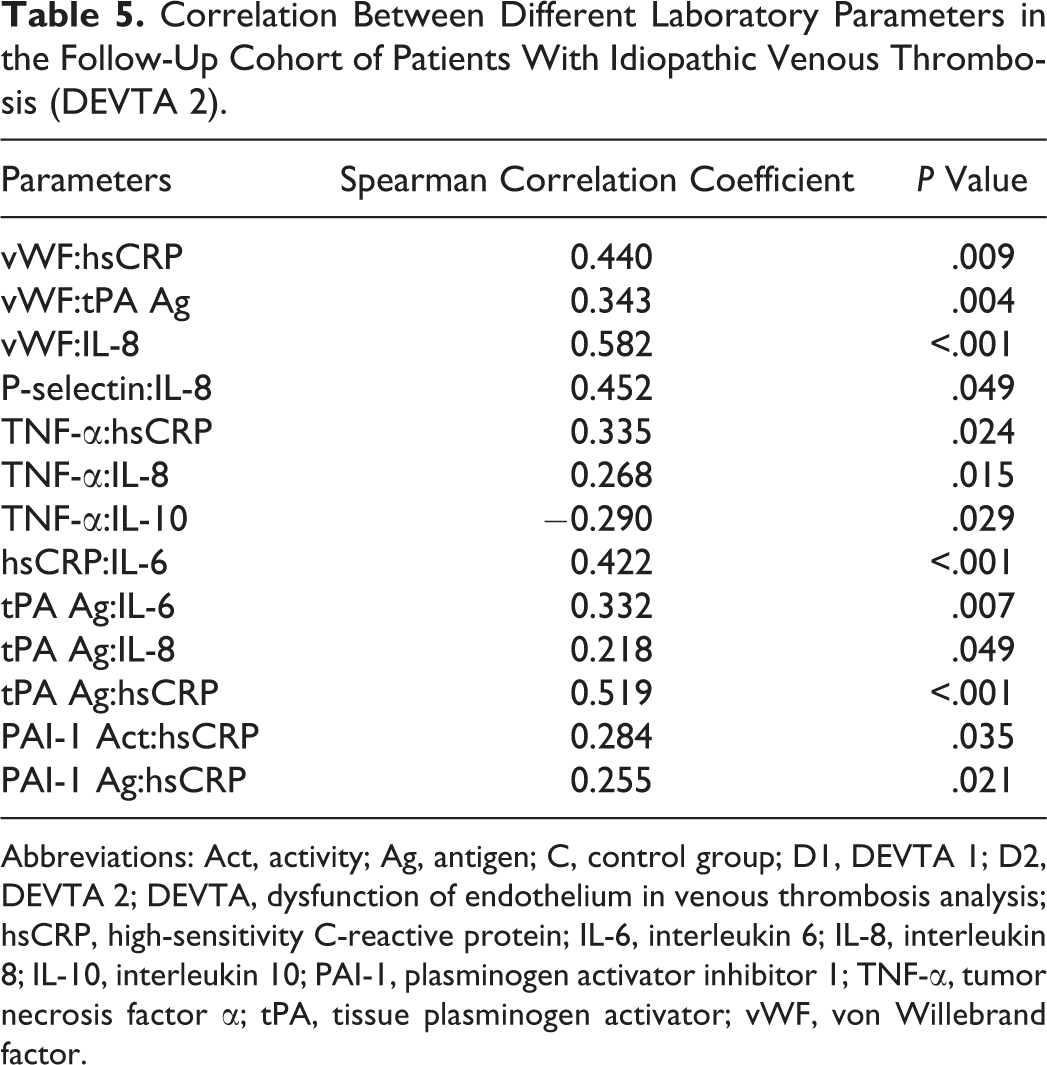
Abbreviations: Act, activity; Ag, antigen; C, control group; D1, DEVTA 1; D2, DEVTA 2; DEVTA, dysfunction of endothelium in venous thrombosis analysis; hsCRP, high-sensitivity C-reactive protein; IL-6, interleukin 6; IL-8, interleukin 8; IL-10, interleukin 10; PAI-1, plasminogen activator inhibitor 1; TNF-α, tumor necrosis factor α; tPA, tissue plasminogen activator; vWF, von Willebrand factor.
Correlation Between Different Laboratory Parameters of Endothelial Damage, Platelets, and Lipid Parameters in the Follow-Up Cohort of Patients With Idiopathic DVT (DEVTA 2).
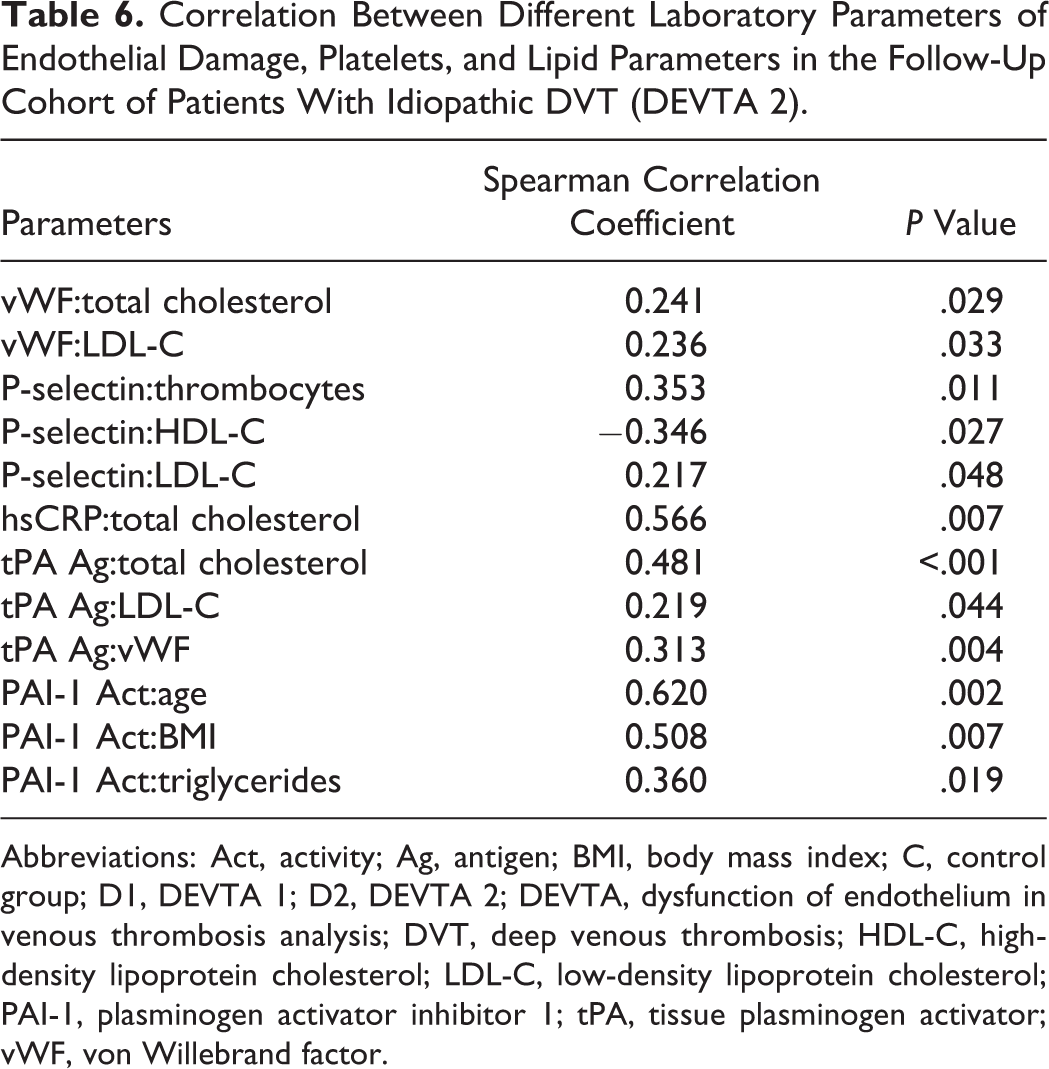
Abbreviations: Act, activity; Ag, antigen; BMI, body mass index; C, control group; D1, DEVTA 1; D2, DEVTA 2; DEVTA, dysfunction of endothelium in venous thrombosis analysis; DVT, deep venous thrombosis; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; PAI-1, plasminogen activator inhibitor 1; tPA, tissue plasminogen activator; vWF, von Willebrand factor.
Discussion
To the best of our knowledge, this is the first study investigating inflammatory markers and hemostatic markers of endothelial damage 5 years after the diagnosis of DVT of the lower limbs. Most of the inflammatory markers remained elevated over a period of 5 years, the levels of fibrinolytic parameters were decreased and the levels of anti-inflammatory cytokine showed a further decline.
A DVT is a chronic disease with some well-known risk factors; however, its underlying pathogenic mechanisms have not been completely elucidated. On the basis of the results of some studies, we have proposed that besides an inherited or acquired thrombophilic state, inflammation represents one of the most important underlying stimulating factor of thrombogenesis. In the previous study (DEVTA 1), we confirmed the presumption that patients would still have elevated inflammatory markers 4 months after the acute phase of the disease. 4 During a much longer follow-up period, up to 5 years (DEVTA 2), the majority of the levels of the inflammatory markers were increased and were comparable between DEVTA 1 and DEVTA 2 and remained significantly elevated in comparison to healthy participants. These results indicate that the increased systemic inflammatory response is a long-lasting deterioration, which was most likely present before the acute DVT and as such is involved in the pathogenesis of DVT and is thus not merely the consequence of DVT. Therefore, it could be one of the mechanisms of pathogenesis of DVT, particularly idiopathic DVT. The reasons for permanently increased proinflammatory status are probably due to different mechanisms, which could be the result of known and unknown risk factors for DVT. This indicates that the pathogenesis of DVT is similar to the etiopathogenesis of atherosclerosis.
The inflammatory process most likely promotes thrombus formation through different mechanisms: the procoagulant and antifibrinolytic activity of blood and the morphological and functional deterioration of the venous wall.
The involvement of the inflammatory process in thrombogenesis was also confirmed by the results of some basic science studies. One of the histochemical studies on thrombotic occluded inferior vena cava showed that there is a significant increase in neutrophils and monocytes and increase in P-selectin messenger RNA (mRNA), E-selectin mRNA, and IL-10 mRNA in the venous wall in the acute phase of thrombosis. 10 This demonstrates that venous thrombosis is associated with an inflammatory response in the venous wall.
The role of individual inflammatory markers in the etiopathogenesis of venous thromboembolism (VTE) is most likely different. The hsCRP promotes a prothrombotic state through the induction of tissue factor synthesis, leading to the activation of the extrinsic coagulation pathway. Furthermore, hsCRP appears to play an important role in regulating the function of blood platelets, the extrinsic blood coagulation cascade, and the fibrinolytic system. 11 However, its involvement in the etiopathogenesis of DVT has not been definitely elucidated. 12 The predictive value of plasma hsCRP levels in the development of DVT has been investigated in 2 large prospective studies. In the Physician Health Study, which included more than 1000 male physicians, the mean hsCRP plasma levels in patients who developed DVT during 8 years follow-up were not statistically different from patients who did not develop DVT. 13 Similar analyses of participants of the Cardiovascular Health Study and in the Atherosclerosis Risk Community Study (n = 19 293) during 8 years’ follow-up also demonstrated that there was no association between the baseline hsCRP levels and the subsequent development of DVT. 14 Although these results contradict our findings, patients in our study already survived VTE in contrast to the above-mentioned studies, where the prognostic value of hsCRP level was studied in healthy participants and where only a small number of participants developed DVT despite the large number of participants.
The finding of increased levels of cytokines and TNF-α in DEVTA 1 and DEVTA 2 in comparison to the healthy participants is in line with the results of the LETS conducted by van Aken et al. 15 This study showed increased levels of IL-8 in patients with recurrent DVT and also in patients with a first event of DVT compared to healthy controls. The levels of IL-6, IL-8, and TNF-α were associated with 2- to 3-fold increased risk of first DVT. It was also shown that the risk for DVT increased with the actual level of observed IL-8. 6 However, in all these studies, cytokine levels were investigated after the thrombotic events. Our findings again favor the presumption that patients with DVT have some inflammatory markers that increased chronically.
An argument against the role of inflammation in the development of DVT is the fact that the levels of inflammatory markers are high in the acute phase of DVT and decrease afterward, as shown in the study by Roumen-Klappe et al. 16 The levels of IL-6, IL-8, and hsCRP were determined at the time of the diagnosis of acute DVT and 5 days later. The IL-8 levels were elevated at both points in time. However, plasma IL-6 and hsCRP levels were decreased at day 5. This may be an indication that some inflammatory markers are a consequence rather than a cause of DVT. The observed decrease in some plasma inflammatory markers may be particularly attributable to the treatment. In most studies, patients were treated with heparin, which has an anti-inflammatory effect. 17 In contrast, the findings of van Aken et al favor the hypothesis that the inflammatory state preceded the appearance of DVT because in their study, the levels of inflammatory markers did not differ in patients with recent or demote DVT. 15
In our DEVTA 1 and DEVTA 2 study, circulating markers of endothelial dysfunction (vWF, P-selectin) were also studied and it was shown that patients with idiopathic DVT have a long-term increase of these markers in comparison to healthy participants. Furthermore, studies in participants with risk factors for atherosclerosis and patients with atherosclerosis showed that the vWF is elevated and is therefore an indicator of endothelial damage. 18 In addition, the vWF participates in the formation of platelet plugs at the site of endothelial damage. 19
An important finding of our study was the interrelationship between individual markers of systemic inflammation and lipid levels (total cholesterol, TG, and HDL-C). This indicates that lipid metabolic disorders are most likely in some patients and responsible for an increased systemic inflammatory response. This finding is in line with the findings in other studies. 20,21 Furthermore, a relationship was shown to exist between vWF, which is an indicator of endothelial damage and is involved in thrombogenesis, and some inflammatory markers. These results again confirm the interrelationship between inflammation and coagulation as well as with endothelial damage. Various studies have confirmed that vWF is an index of endothelial damage in vascular disease and that its levels rise in proportion with the extent of vascular involvement. 22 Significant interrelationships were also found between individual circulating inflammatory markers, indicating the complexity of the inflammatory process in patients with DVT. Correlations were also shown to exist between PAI-1 activity and some clinical characteristics of patients such as age and BMI. This indicates that the prothrombotic potential increases with the age of patients and BMI. With increase in body weight, the level of PAI-1 increases, and weight loss can improve the abnormality in the hemostatic and fibrinolytic factors associated with obesity. 23
Conclusion
The results of our study confirm the hypothesis that DVT is a chronic disease and that increased inflammatory response not only is present in the acute and subacute phase of the disease but persists later on. These findings favor the hypothesis that inflammation is a cause and not merely a consequence of VTE and as such could be involved in thrombus formation and the pathogenesis of acute DVT.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
