Abstract
Chagas cardiomyopathy (CCC) is associated with coagulation disorders that frequently culminate in thrombotic events, contributing to increased mortality rates in this clinical condition. Considering the demonstrated effect that extracellular vesicles (EVs) have on regulating inflammatory processes, coagulation, and angiogenesis, the present study aims to characterize plasma EVs and their relationship with coagulation disorders in patients with CCC. A total of 78 patients were assessed with 46.1% (36/78) representing the CCC group, 8.9% (7/78) with cardiomyopathy unrelated to Chagas disease (CM group), and 44.8% (35/78) comprising the control group, which included individuals without cardiomyopathy and negative for T. cruzi infection. Plasma EVs concentration (EVs/mL) for each individual was evaluated by flow cytometry, along with the proportion of EVs expressing PSGL-1 (PSGL-1+ EVs), Tissue Factor (TF + EVs), and CD41a (CD41a + EVs). The ability of EVs to induce platelet aggregation was evaluated by spectrophotometry. We also evaluated other prothrombotic biomarkers, including platelet count, activated partial thromboplastin time (PTT), prothrombin time (PT), and D-dimer levels. The results revealed elevated D-dimer levels in the CCC group, accompanied by a decrease in the count of EVs per mL of plasma and a significant increase in the proportion of PSGL-1+ EVs (P < .05) compared to the control group. Other parameters did not exhibit significant differences between groups. The elevated levels of PSGL-1+ EVs in the CCC group may be attributed to myocardial inflammatory processes, which, upon interaction with platelet-derived P-selectin, could promote thrombus formation, as indicated by the increased D-dimer levels in this group.
Introduction
American trypanosomiasis, also known as Chagas disease (CD), is a neglected disease caused by the hemoflagellate parasite Trypanosoma cruzi (Trypanosomatidae). 1 It is primarily transmitted through hematophagous vectors known as triatomines, accounting for 70% of cases, and vertical transmission, including approximately 26% of cases. 2 Additional routes include oral transmission, blood products, solid organ transplantation, and laboratory accidents. 3 Chagas disease is endemic in 21 Latin American countries, where approximately eight to ten million people are infected, and about 30 thousand new cases are reported annually. 4 Due to population mobility, the disease is not restricted to endemic areas. In the USA, 300 167 individuals live with T. cruzi infection, and nearly 63 to 315 congenital infections occur yearly. 5 In Europe, it is estimated that 90 000 to 140 000 American migrants are infected with T. cruzi, of which 0.1% are children. 6
The clinical manifestations are divided into two phases. The acute phase is often asymptomatic or presents as an acute febrile undifferentiated syndrome, with rare cases exhibiting localized inflammation, unilateral eyelid edema, and occasional deaths associated with oral infection. The chronic phase consists of an indeterminate form where patients are infected and asymptomatic, accounting for 70% of cases, and a determinate form where the patient may present Chagas cardiomyopathy (CCC) (20-30% of cases) or gastrointestinal megasyndrome (10% of cases). 7 Individuals with CCC have a twofold higher risk of mortality compared to other cardiomyopathies, including idiopathic dilated, ischemic, and hypertensive cardiomyopathies. This is due to the unique pathophysiology of the disease, wherein chronic myocarditis leads to the loss of cardiomyocytes and replacement of fibrotic tissue, resulting in cardiomyopathy, ventricular arrhythmias, aneurysms, and an increased risk of cardioembolic events that can produce ischemic stroke. 8
A high incidence of ischemic stroke is frequently noted in CCC patients compared to the general population. Cross-sectional studies in the Americas reported a higher frequency of stroke in CCC than in non-CD individuals (14%–15% vs 2%–6%, P < .01 in Brazil; 25% vs 2%, P < .05 in Colombia).9–11 The potential hypercoagulable state in patients with CCC remains unclear. However, some studies propose that chronic inflammation, oxidative stress, and the release of pathogen-associated molecular patterns (PAMPs) by T. cruzi or damage-associated molecular patterns (DAMPs) released by infected cells could contribute to aggregation and activation of platelets. 12 Similarly, a recent systematic review and meta-analysis revealed that serological levels of prothrombin fragment 1 + 2 (F 1 + 2), plasminogen activator inhibitor-1 (PAI-1), and P-selectin were significantly increased in CCC patients compared to healthy controls. The elevated serological levels of these molecules, coupled with concurrent platelet activation, enhance the release of procoagulant factors such as extracellular vesicles (EVs), adhesion molecules, and coagulation factors, among others, promoting the coagulation cascade and thrombin production in CCC. 13
Extracellular vesicles are small lipid bilayer-delimited particles (150-1000 nm) produced by all cells during activation or cell death. 14 Due to the variety of molecules inside and on their surface, with agonist and antagonist activities, they play a pivotal role in various physiological and pathological processes, including the proliferation of endothelial cells, coagulation, thrombosis, inflammation, and other events related to innate and adaptive immunity.15,16 Platelet-derived extracellular vesicles (PDEVs) are the most abundant among all types of EVs in circulation. However, the mechanisms leading to PDEV release, their role in coagulation, and phenotypic composition are poorly understood. 17 Total EVs and specific populations expressing CD41a, PSGL-1, and Tissue Factor are increased in the blood of patients with cancer, lupus, and cardiovascular diseases, including atherosclerosis, coronary artery disease, ischemia, myocardial infarction, graft occlusion, and heart failure. These EVs are associated with the modulation of coagulation, inflammation, and disease progression.18–22 Considering the importance of EVs in the pathophysiology of cardiovascular disease, this study aims to assess the count of EVs and characterize the populations of EVs expressing CD41a, PSGL-1, and Tissue Factor in patients with CD and their relationship with the hypercoagulable state.
Material and Methods
Ethics Statement
This research was conducted in accordance with Resolution No. 8430 of 1993 from the Colombian Ministry of Health. The study is considered “Research with Minimal Risk”. Patients were fully informed about the study and voluntarily agreed to participate by completing an informed consent form in accordance with international ethical regulations. It was endorsed by the Scientific Technical Committee and the Research Ethics Committee of the Cardiovascular Foundation of Colombia (FCV), documented in Minutes Number 214 of 2022.
Study Area and Subject Sampling
This research was an analytical cross-sectional study, with the population consisting of a group of patients who attended the Heart Failure Clinic of the FCV between June 2022 and June 2023. This center is in Floridablanca, Santander, a hyperendemic area for CD in the northeast of Colombia. Three groups of patients over 18 years of age were included; the first group included patients with CCC exhibiting changes in electrocardiographic and echocardiographic parameters (Group: CCC); the second comprised patients with cardiomyopathy and a reduced ejection fraction (EF ≤ 40%), and negative for T. cruzi infection (Group: CM). The third group represented a control group, consisting of individuals that attended routine check-ups, in which cardiological evaluations showed no evidence of heart failure (EF ≥ 55%) and were negative for T. cruzi infection (Group: Control). T. cruzi infection was evaluated using indirect immunofluorescence (IFAT) and enzyme-linked immunosorbent assay (ELISA) following the protocols described by Instituto Nacional de Salud (INS).23,24 Patients with CCC were determined according to the guidelines for the diagnosis and treatment of CD. 25 Exclusion criteria for this study encompassed pregnancy or the postpartum period, those with acquired or hereditary thrombophilias, liver diseases, chronic kidney failure, and individuals undergoing treatment with any anticoagulant within the last six months.
Blood Sample and Coagulation Function Measurements
After signing the informed consent form, participants were asked for clinical and epidemiological data, including area of origin and risk factors for CD transmission. The information recorded also included cardiovascular risk factors and use of cardiac therapy medications during the last six months. Heart failure patients were classified by the New York Heart Association (NYHA) functional class: I: No limitations in daily activity, II: Slight limitations in daily activity, III: Marked limitations in daily activity, IV: Unable to engage in any activity without discomfort. Three blood samples were collected for each patient, two collected in citrate tubes (BD-Vacutainer® 3.8% sodium citrate tubes; Becton Dickinson, San Jose, CA) and the last one in EDTA tubes (BD-Vacutainer® EDTA K2; Becton Dickinson, San Jose, CA). The samples were obtained through a clean phlebotomy of an antecubital vein using 20-gauge needles (BD Vacutainer needles). Coagulation function was evaluated in all patients by measuring platelet count, activated partial thromboplastin time (PTT), prothrombin time (PT), and D-dimer. Platelet count was determined from EDTA blood in an automated hematology analyzer (CELL-DYN Ruby; Abbott, Abbott Park, Illinois, USA). Coagulation times and D-dimer in plasma were evaluated using an automatic coagulation analyzer according to the manufacturer's recommendations (Sysmex® CA-1500; Siemens Healthcare GmbH, Germany). These measurements were conducted within six hours after the sampling procedure.
Isolation and Validation of EVs
EVs were isolated from 4 mL of citrated venous blood. Briefly, the blood sample was centrifuged at 1800×g for 10 min at 21 °C to obtain total plasma. This plasma was centrifuged twice at 3000×g for 20 min at 21 °C to remove the platelets and obtain platelet-poor plasma (PPP). Finally, 1 mL of PPP was centrifuged at 17 000×g for one h to allow the sedimentation of EVs. These vesicles were suspended in 100 μL of filtered Dulbecco's phosphate-buffered saline (1X DPBS; Gibco, Grand Island, NY), taking care not to disturb them, and kept at −70 °C until use.
To corroborate the size of EVs, their diameter was evaluated in a representative sample using dynamic light scattering (DLS) and field emission scanning electron microscopy (FESEM). For DLS, measurements were conducted in an aqueous medium at neutral pH and room temperature using a Zetasizer Pro instrument (Malvern Instruments Ltd, Malvern, UK). For FESEM, vesicles were fixed with 2.5% glutaraldehyde, deposited on copper-coated carbon scanning transmission electron microscope (STEM) grids, dried on the grid at 40 °C, and contrasted with uranyl acetate and lead citrate. The samples were then dried at room temperature and evaluated using a field emission scanning electron microscope, Scios 2 LoVac field emission electron microscope (Thermo Fisher Scientific, USA). Finally, to ensure the lipidic nature of EVs, they were stained with anti-human CD41a monoclonal antibody (Figure 1C) and exposed to 0.1% Triton X-100 (Sigma-Aldrich, MO).
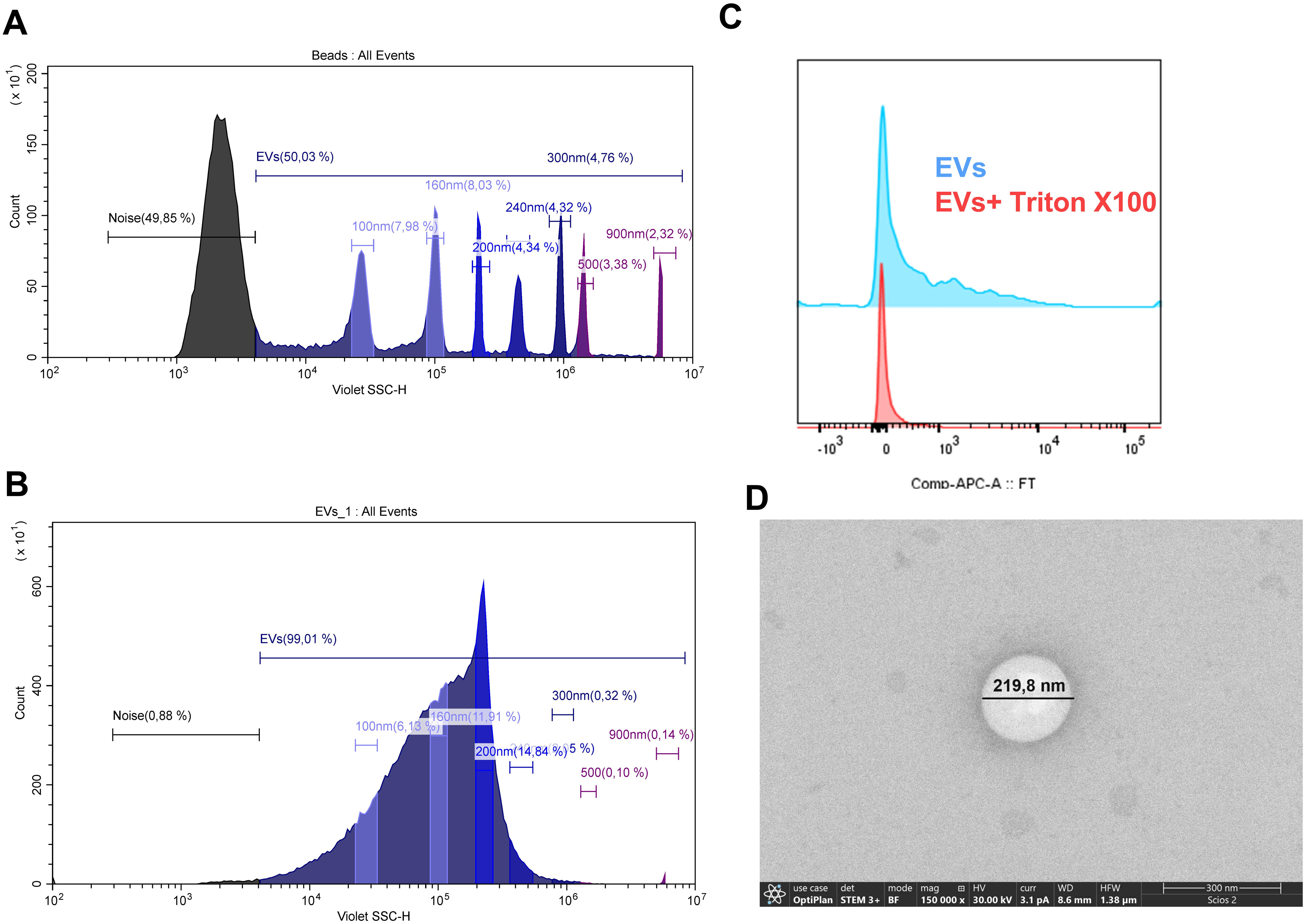
Counting and caracterization of extracelular vesicles. A) The EV-gate was determined using MegamixPlus FSC beads (BioCytex a Stago group company, Marseille, France) which have beads with different sizes (100, 160, 200,240, 300, 500, 900 nm). B) Representative sample of EV size according to the parameters defined by using MegamixPlus FSC beads. C) Representative CD41a histograms of EVs untreated and treated with 0.1% Triton X-100. CD41a positivity was rapidly altered after Triton X-100 addition. D) Representative microphotographs of field emission scanning electron microscopy dual beam (FIB-FESEM) of EVs. These images demonstrate the vesicular shape and size expected for EVs.
Counting and Characterization of EVs
Frozen suspensions of EVs (200 μL) were brought to a volume of 500 μL with filtered DPBS. A 50 μL aliquot of the suspension of EVs was diluted with 50 μL of filtered DPBS, and used for direct counting of EVs on the CytoFLEX flow cytometer (Beckman Coulter, Brea, CA, USA). The EV-gate was determined using MegamixPlus FSC beads (BioCytex, a Stago group company, Marseille, France) with beads of different sizes (100, 160, 200, 240, 300, 500, and 900 nm). EVs were defined as vesicles <1.0 µm in size (Figure 1A-B). EV counts were used to calculate each individual's plasma EVs concentration (EVs/mL).
For characterization, EVs were stained with monoclonal antibodies (mAbs) specific for markers of platelets (anti-CD41a-PE; clone HIP8; eBiosciences), Tissue Factor (anti-TF-APC; clone FLEG; eBiosciences) and P-selectin glycoprotein ligand-1 (anti-PSGL-1-APC; clone FLEG; eBiosciences). The samples were incubated in the dark at room temperature for 30 min and acquired on the BD LSRFortessa™ flow cytometer (BD Biosciences). The frequencies of EVs positive for each marker were analyzed using FlowJo vX software.
Aggregometry Evaluation of EVs
Platelet aggregometry was evaluated in a platelet-rich plasma (PRP) obtained from a healthy donor at a concentration of 500 000 platelets/μL as described elsewhere. 26 A total of 500 µL of PRP from the donor was incubated with 50 µL of suspensions of EVs from each patient in 1.5 mL plastic cuvettes. Cuvettes were shaken (900 rpm, orbital shaking) for 8 min at 37 °C, and after that, the absorbance was measured using a spectrophotometer set at 595 nm, according to Vinholt et al, 2017. Platelet aggregation was determined by calculating the optical density (OD) in paired measurements with controls, using platelet-rich plasma (PRP) as the reference for 0% aggregation and platelet-poor plasma (PPP) as the reference for 100% aggregation, as previously described. 27
Data Analyses
Quantitative variables were presented as medians and interquartile ranges (IQR) and were compared between groups using the Kruskal-Wallis or Mann-Whitney U test. A Dunn's Multiple Comparisons test was conducted to identify specific group differences. Qualitative variables were reported using absolute frequencies and percentages, and between-group comparisons were made using the chi-squared test (χ2). A Principal Component Analysis (PCA) was conducted to explore relationships between observations and variables. IBM SPSS Statistics software, version Armonk, NY, USA, 29.0.10 (APA, MLA, Chicago), was employed for all statistical analyses. Graphs were generated using GraphPad Software LLC, San Diego, CA, USA, version 9.0. Statistical significance was determined at P < .05.
Results
Characterization of Sociodemographic and Clinical Information
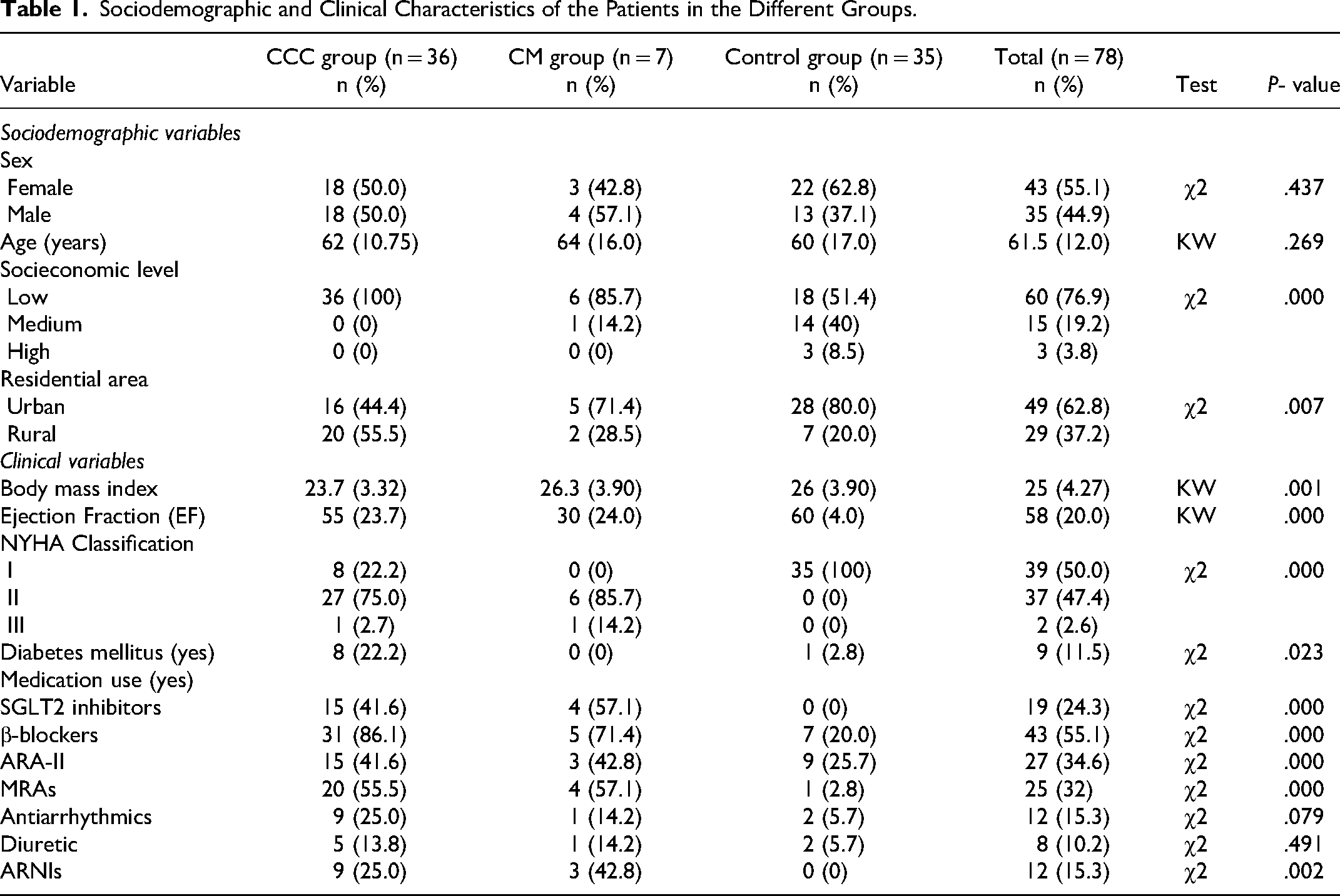
A total of 78 patients were evaluated, with 46.1% (36/78), 8.9% (7/78), and 44.8% (35/78), corresponding to CCC, CM, and control groups, respectively. All groups were similar in age and sex. Regarding socioeconomic level and residential areas, the CCC group had a higher proportion of patients with low socioeconomic status residing in rural areas. Regarding clinical variables, the CM group exhibited a higher body mass index and lower ejection fraction (EF). For NYHA classification, 22.2%, 75%, and 2.7% of CCC group patients belonged to classes I, II, and III, respectively. In contrast, 0%, 85%, and 14.2% of CM group patients belonged to these classes. All control group patients belonged to class I. Additionally, the CCC group showed a higher proportion of patients with diabetes mellitus. Finally, the CCC and CM groups demonstrated more medication use (Table 1). Additional clinical characteristics of each patient are provided in Table S1.
Sociodemographic and Clinical Characteristics of the Patients in the Different Groups.
Coagulation Function and Prothrombotic Biomarkers
The measurement of D-dimer revealed elevated values in CM group, followed by the CCC and control groups (KW, P < .015). However, after multiple comparisons, a significant difference was found between the CCC and control group (Dunn's test, P < .05), but not between the CM and control group (Table 2). Additionally, 57.1%, 51.6%, and 28.1% of patients in the CCC, CM, and control groups had abnormal D-dimer values, respectively. Platelet count, PTT, PT, and platelet aggregation did not show significant differences between groups (Table 2).
Results of the Prothrombotic Biomarkers and Platelet Aggregation in the Different Groups.
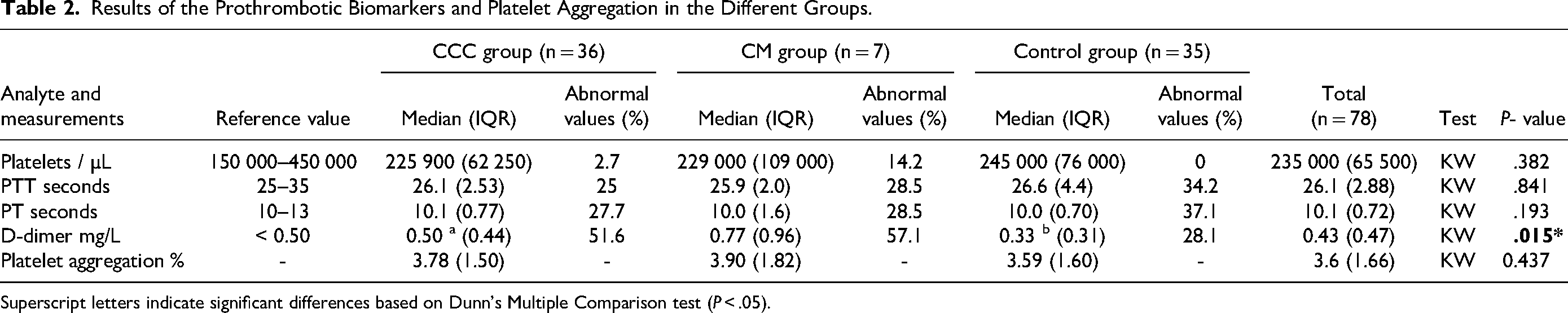
Superscript letters indicate significant differences based on Dunn's Multiple Comparison test (P < .05).
Validation, Counting and Characterization of EVs
EVs showed a lipidic nature because of the absence of CD41a expression in the presence of Triton X-100 (Figure 1C). The vesicular structure was verified through FESEM (Figure 1D) and DLS, showing that EVs vesicles isolates displayed vesicular shape and expected sizes of < 1000 nm; according to Minimal Information for Studies of Extracellular Vesicles 2018 (MISEV2018) guidelines (Théry et al, 2018).
The quantification of EVs per mL of plasma revealed higher values in the control group, followed by the CM and CCC groups, respectively (KW, P = .005), indicating a significant difference between the CCC group and the control group (Dunn's test, P < .05) (Figure 2). Regarding the characterization of EVs, the percentage of EVs expressing PSGL-1 (PSGL-1+ EVs) was higher in the CM group, followed by the CCC and control groups, respectively (KW, P < .0001), showing a significant difference between CM and CCC in comparison to the control group (Dunn's test, P < .05) (Figure 2). No significant differences were observed in the proportion of TF + EVs and CD41a + EVs between groups. Associative analyses of EV characterization and clinical features of patients, independent of clinical group, revealed notable differences in the proportion of PSGL-1+ EVs concerning NYHA classification, SGLT2 inhibitors, Beta-blocker, MRAs, and ARNIs usage (KW, P < .05). Similarly, significant differences in the proportion of TF + EVs regarding NYHA Classification, SGLT2 inhibitors, and ARA-II usage were noted. There were no significant associations between the quantification of EVs and CD41a + EVs (Table S2).
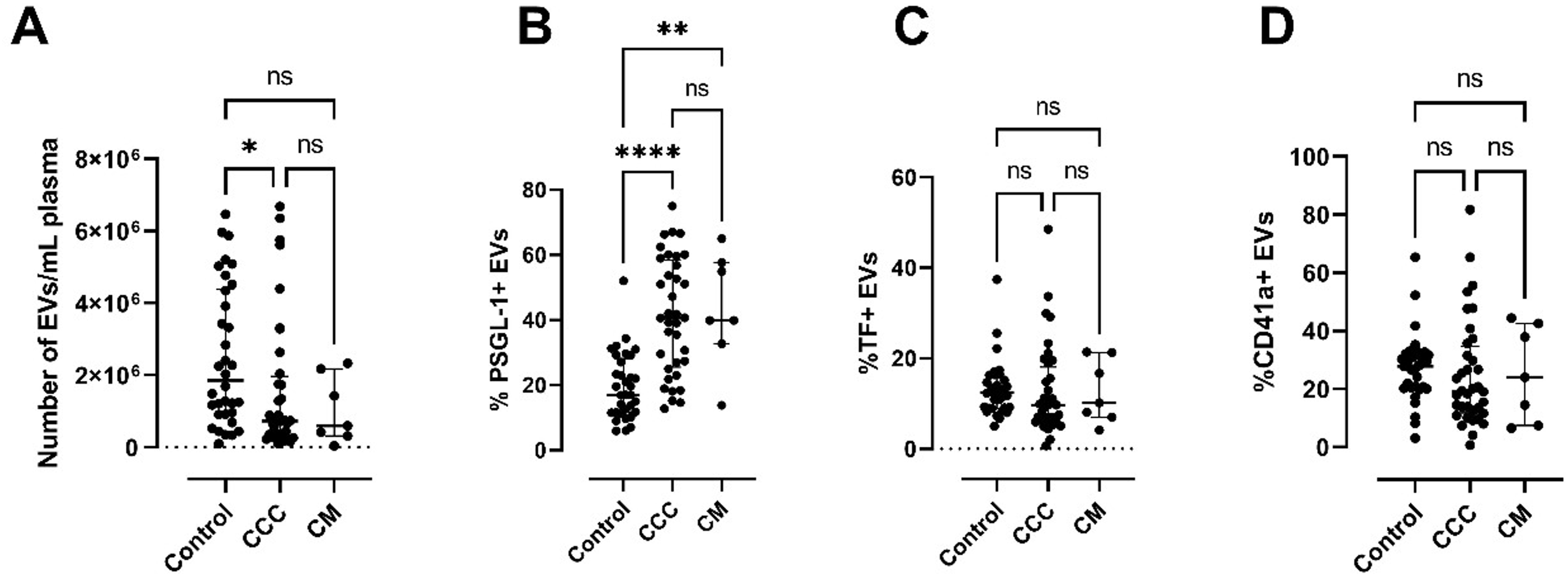
Results of EVs in the different groups as determined by flow cytometry. A. Count of EVs per mL of plasma. B. Frequency of EVs expressing PSGL-1 (PSGL-1+ EVs). C. Frequency of EVs expressing Tissue Factor (TF + EVs). D. Frequency of EVs expressing and CD41a (CD41a + EVs).
Exploratory Cluster Analysis
PCA identified three principal components explaining 50.2% of the variability. The dimensional reduction suggests two clusters of variables. The first cluster is represented by PTT, PT, platelet aggregation, D-dimer, and PSGL-1+ EVs, along with the CCC and CM control groups. The second cluster consists of platelet count, quantification of EVs per mL of plasma, and CD41a + EVs, along with the control group (Figure 3). Both clusters suggest a possible correlation between these groups of variables.
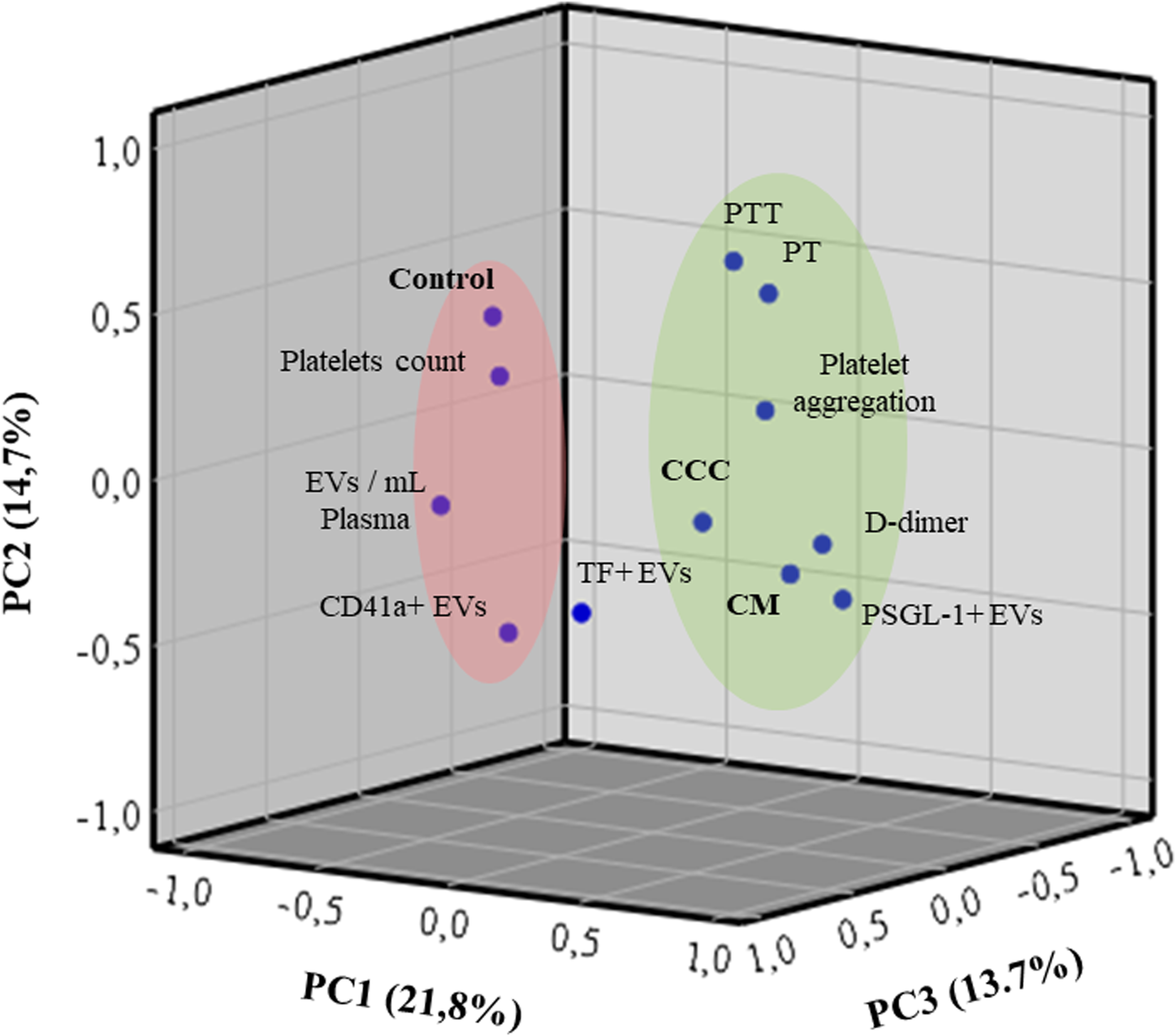
Principal component analysis (PCA) between EVs, prothrombotic biomarkers and clinical groups.
Discussion
Multiple studies have found a high incidence of embolic events in patients with CCC, highlighting the significant risk of thrombotic events associated with this condition.13,28 In terms of pathophysiology, EVs play a crucial role in thrombotic events due to their procoagulant surface and the expression of highly procoagulant proteins like PSGL-1 and TF. 29 This study examined the EVs profile with other prothrombotic biomarkers in patients with CCC and their respective controls. The results reveal a prothrombotic state in the CCC group, as indicated by an increase in D-dimer levels, a reduction in EVs per mL of plasma, and a significant increase in the proportion of PSGL-1+ EVs. The clinical and epidemiological implications of these findings are discussed herein.
Elevated levels of EVs have been observed in various health conditions, including systemic lupus erythematosus,30,31 rheumatoid arthritis,32,33 cancer,34,35 acute coronary syndromes.36,37 These EVs have emerged as promising biomarkers for tracking disease progression. In contrast, in our study, a low count of EVs was observed in the CCC group compared to the control group (P < .05; Figure 2A). The treatment received by patients could explain this observation. The association analysis suggested that patients receiving pharmacological treatment with β-blockers independent of the clinical group exhibited reduced EVs levels (P = .092; Table S2), indicating a negative impact of this specific treatment on EVs modulation. In a study involving patients with acute heart failure, stable heart failure, and stable coronary syndrome, no significant differences were found in platelet-derived EVs counts between groups. However, after pharmacological treatments, it became evident that a low count of these EVs was associated with the use of β-blockers (P < .016). 36 From a pharmacological standpoint, β-Blockers indirectly prevent calcium entry into myocardial cells; therefore, they slow conduction velocity and automaticity and prolong the atrial refractory period.38,39 Considering that an increase in intracellular calcium appears to be the initiating step in EVs biogenesis,40,41 the low count of EVs in the CCC group could be associated with the use of β-blockers. However, further research is needed to confirm the effects of these drugs on EV biogenesis.
Although a reduced EVs count was observed in the CCC patients, the increase in the proportion of PSGL-1+ EVs compared to the control group suggests a high risk of thrombosis in this group (P < .05; Figure 2B). P-selectin is stored in α-granules of platelets and Weibel-Palade bodies of endothelial cells, while one of its ligands, PSGL-1, is a molecule constitutively expressed on the surface of numerous hematopoietic and some nonhematopoietic cells, 42 therefore, EVs released from different cells could be PSGL-1 + . P-selectin and PSGL-1 are vascular adhesion molecules that play a crucial role in leukocyte-endothelial and leukocyte-platelet interactions during inflammatory responses. Previous studies have revealed that inappropriate expression of these molecules contributes to leukocyte-mediated tissue damage in various inflammatory and thrombotic disorders, as recently reviewed. 43 In mice models, PSGL-1+ EVs are captured by thrombus-associated platelets through the interaction of PSGL-1 and platelet P-selectin, leading to a concentration of Tissue Factor and the initiation of blood coagulation and fibrin formation. 44 A study evaluating hypercoagulability factors in CD patients showed an increase in P-selectin in patients with CCC compared to the healthy group (P = .02). 45 These findings suggest that PSGL-1+ EVs may contribute to thrombus formation and could explain the elevated D-dimer levels observed in 51.6% of CCC patients. Similar results regarding elevated D-dimer levels in patients with CCC have been consistently observed (P < .05), confirming the prothrombotic state in this clinic condition.45–47 Considering that intracavitary thrombi have been identified in up to 36% of CD patients who died from sudden death or heart failure, 48 early identification of patients with elevated D-dimer levels could allow for proactive interventions, such as increased monitoring for thrombotic complications or earlier initiation of antithrombotic therapy, particularly in those who have not yet developed significant heart failure symptoms.
P-selectin and PSGL-1 have essential roles in cardiovascular disease development. During the first stages of myocardial infarction, blood neutrophils (PMN) infiltrate the ischemic myocardium in large quantities. 49 They are attracted by cell debris and inflammatory mediators released by activated resident cells. Recruited neutrophils aim to phagocytose and clear dead cell debris caused by ischemia. However, they concomitantly cause collateral cardiac injury by releasing reactive oxygen species (ROS), proteolytic enzymes, and inflammatory mediators.50,51 In addition, neutrophils can form extracellular traps (NETs) and release EVs that contain a multitude of inflammatory mediators, including PSGL-1. 49 The progression of PMN–endothelium interactions is largely mediated by the P-selectin, which is translocated from Weibel-Palade bodies to the endothelial cell surface. 52 The inhibition of P-selectin with an anti-P-selectin monoclonal antibody reduced myocardial damage during PCI in patients with non-ST-segment elevation myocardial infarction. 53 Whereas the expression levels of PSGL-1 on CD14 + CD16 + monocytes are significantly higher in patients with plaque rupture or intracoronary thrombi, as assessed by frequency-domain optical coherence tomography. 54 Since the analysis of other coagulation biomarkers (platelet count, PT, PTT, TF + EVs, and CD41a + EVs) did not reveal significant differences between the groups, the prothrombotic state observed in the CCC and CM groups, as indicated by elevated D-dimer levels, could be explained by PMN infiltration during myocardial infarction characteristic from these clinical conditions. This infiltration could trigger the release of PSGL-1+ EVs, which may promote thrombus formation through interactions with platelet-derived P-selectin. Evaluating PSGL-1+ EVs could potentially lead to the development of a non-invasive blood test to monitor disease severity, particularly in relation to the inflammatory processes and their complications, such as prothrombotic states. However, further research is needed to explore these possibilities.
Finally, although this is the first study to evaluate EVs related to coagulation disorders in CCC patients, our study has some limitations. The small sample size in the CM group, primarily due to the higher use of anticoagulants in this population, and the absence of a healthy control group are notable constraints. These factors reduce the study's statistical power and affect the robustness of the results. Despite these limitations, the elevated D-dimer levels observed in the CCC group highlight the high thromboembolic risk in these patients, as reported in previous studies.55,56 On the other hand, this study was conducted in a hyperendemic area for CD, where the specific demographic, socioeconomic, and environmental characteristics of the patient population may differ from those in other regions. This variability makes it difficult to generalize the findings to CCC patients in non-endemic areas or under different healthcare conditions. Finally, the limited understanding of EVs in heart failure poses challenges in identifying potential biomarkers that could be implicated in thrombus processing.
In conclusion, EVs levels were notably lower in patients with CCC compared to the control group, possibly influenced by the use of β-blockers. Conversely, the proportion of PSGL-1+ EVs and D-dimer levels were significantly higher in this group. The elevated levels of PSGL-1+ EVs in CCC patients may be attributed to myocardial inflammatory processes, which, upon interaction with platelet-derived P-selectin, could promote prothrombotic stages, as indicated by the increased D-dimer levels. To our knowledge, this is the first characterization of EVs in patients with CCC and their association with thrombotic events, providing valuable insights into the pathophysiology of thrombotic events in these patients.
Supplemental Material
sj-docx-1-cat-10.1177_10760296241295742 - Supplemental material for Assessment of Plasma Exovesicles and Prothrombotic Biomarkers Suggest Prethrombotic Conditions in Chagas Cardiomyopathy in Colombia
Supplemental material, sj-docx-1-cat-10.1177_10760296241295742 for Assessment of Plasma Exovesicles and Prothrombotic Biomarkers Suggest Prethrombotic Conditions in Chagas Cardiomyopathy in Colombia by Jeiczon Elim Jaimes-Dueñez, Karen Álvarez, Luis Eduardo-Echeverria, Diana Isabel Cáceres-Rivera, Lyda Z. Rojas, Sergio Alejandro Gómez-Ochoa, Laura Daniela-Muñoz, María Cantillo-Reines, Marisol Tique-Oviedo, Yurany Eresbey-Granada and Omar Triana-Chávez Biol in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Acknowledgments
We extend our heartfelt gratitude, foremost, to the patients whose generosity in granting access to samples and information made this study possible. Our deepest appreciation also goes to the dedicated professionals at the FCV Heart Failure Clinic for their invaluable assistance throughout. Special thanks to Dr Yamile Higuera Blanco of the FCV Laboratory for her unwavering support in sample processing, and to Dr Mauricio Rojas of the Cytometry Unit at the University of Antioquia for her expert guidance in the evaluation and analysis of EVs. Finally, we would like to thank PhD student Tulio José Lopera Restrepo for his support in vesicle analysis, and BECKMAN COULTER for their guidance in establishing the parameters for exovesicle acquisition and characterization using the CytoFLEX cytometer.
CRediT Authorship Contribution Statement
JEJD: Investigation, Methodology, Writing –original draft, Formal analysis, Writing – review & editing, Visualization, Resources. KA: Conceptualization, Methodology, Investigation, Writing – original draft. LEE: Formal analysis, Investigation, Writing – original draft, Resources. LZR: Conceptualization, Methodology, Investigation, Writing – original draft, Resources. SAGO: Conceptualization, Methodology, Investigation, Writing – original draft. LDM: Methodology, Investigation, Writing – original draft. MCR: Methodology, Investigation, Writing – original draft. DICR: Methodology, Investigation, Writing – original draft. YEG: Methodology, Investigation, Writing – original draft. OTC: Investigation, Methodology, Writing –original draft, Visualization, Resources.
Declaration of Generative AI and AI-Assisted Technologies in the Writing Process
During the preparation of this work the authors used the assistance of ChatGPT 4.0 by OpenAI, to providing helpful insights and corrections during the process of improving the drafting and readability. After using this tool, the authors reviewed and edited the content as needed and take full responsibility for the content of the publication.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
Data will be made available on request at
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Universidad Cooperativa de Colombia,
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
