Abstract
Previous clinical and epidemiological studies have shown that patients with venous thromboembolism (VTE) are comorbid with symptoms of ischaemic stroke (IS). Current understanding about increased risk of IS after VTE remain inconclusive. This study use a bidirectional two-sample Mendelian randomization (MR) study to explore the causality of VTE, pulmonary embolism (PE), deep vein thrombosis (DVT), and IS. This study used pooled data from published genome-wide association studies (GWAS). GWAS statisics of IS (from EBI database, n = 484 121), VTE (from Finngen database, n = 218 792), PE (from Finngen database, n = 218 413), and DVT (from UK biobank database, n = 337 159) were assessed. Forward and reverse MR analysis were conducted to explore the causal relationship between three type of the exposure (VTE, PE, and DVT) and the outcome (IS). Our primary causal inference method was Inverse Variance Weighted (IVW). Secondary inference methods were Weighted Median and MR-Egger. For the sensitive analysis, MR-PRESSO, MR-Egger intercept, Cochran's Q, leave-one method were used to consolidate our findings. In the foward MR analysis, VTE increased the risk of IS (ORIVW = 1.034, PIVW = 0.021) and PE was also a risk factor for IS (OR = 1.055, PIVW = 0.009). There was no causality that DVT influenced on IS (PIVW > 0.05). In the reverse MR analysis, IS came to be a risk factor for DVT (OR = 1.003, PIVW = 0.046). Meanwhile, IS took not any causal effect on VTE and PE. All the results passed the reasonable sensitive analysis. Our findings provided genetic evidence that PE and VTE can lead to an increased risk of IS, whereas increased IS promoted the risk of DVT further. Our findings provided novel insights about the risk factors and management for IS.
Keywords
Introduction
Strokes, which are important contributors to global mortality and disability, occur when blood vessels in the brain either burst or become blocked, cutting off the flow of blood to the brain.1,2 Strokes are classified as either hemorrhagic or ischemic. Accounting for 62.4% of all stroke events, ischemic stroke (IS) is the most common type of stroke. 3 The pathophysiological mechanism of IS currently include atherosclerosis, injury of vascular endothelial cells, metabolic disorders, immune microenvironment, and hereditary factors.4–8 Meanwhile, the management of various modifiable risk factors related to IS, including smoking, hypertension, diabetes, hyperlipidemia, atrial fibrillation, and coronary heart disease, are considered to be critical in reducing the occurrence of IS.9,10 Current studies have found that more risk factors come to be the main culprits of different subtypes of IS. 11 Therefore, it is important to find potential unknown risk factors for current IS risk management.
Venous thromboembolism (VTE) is a complete or incomplete obstruction of a blood vessel resulting from an abnormality in the clotting of blood within a vein, including both deep vein thrombosis (DVT) and pulmonary embolism (PE). 12 The formation of VTE is dependent on a number of factors, including venous stasis, a hypercoagulable state of blood, and endothelial damage. 13 DVT is defined as a blood clot forming in the deep veins of the legs, where as PE is a clinical syndrome that result from the obstruction of the pulmonary arterial system by various embolism, including fat embolism, amniotic fluid embolism, or air embolism. 14 The relationship between VTE and IS is relatively close in pathogenesis, clinical research, and hereditary factors. From the perspective of pathogenesis, the hypercoagulable state after IS onset is the risk of VTE, whereas severe VTE would increase the risk of thrombus entering the cerebrovascular, aggravating the occurrence of IS. 15 European Stroke Organisation highlighted the high incidence of VTE (DTV and PE) and related preventive measures for IS patients. 16 Meanwhile, considering the shared biological pathways and aetiological factors between IS and VTE, it is plausible that the severity of IS may also be influenced by genetic variants associated with VTE. 17 The single nucleotide polymorphisms (SNP) rs4253417, derived from a VTE associated genetic variant, coagulation factor XI (FXI) levels, has been considered as the most important IS risk factor and biomarker. 18 Therefore, the association between IS and TVE is evident, but the causal relationship remains unclear. Recent observational studies of correlation between VTE and IS are often limited by bidirectional causality and confounding factors, which cannot reach a more convincing conclusion. 19
Mendelian randomization (MR) is a new type of epidemiological methodology that can reveal causal relationship. It uses SNPs as the instrumental variable, assuming that the alleles are independent and randomly assigned. 20 Therefore, MR can reduce the impact of confounding variables and bias due to reverse causation, and is employed to ascertain causal relationships between exposures and outcomes, as well as to differentiate the temporal sequence in which exposures and outcomes occur in a sequential manner. 21 This enables a more comprehensive investigation of the causal relationship between VTE, DVT, PE, and IS in larger sample sizes and across a broader population. To date, no studies have utilized genome-wide association study (GWAS) data to investigate the potential association between IS, DVT, PE, and VTE using a two-sample MR approach. This research serves two main purposes. Firstly, it aims to provide an evidence-based supplement on modifiable risk factors and offer insights into preventive strategies for IS. Secondly, it explores the bidirectional causal relationship between IS and VTE within the context of their complex interaction dynamics, enhancing clinical understanding of IS and VTE.
Method
Study Design
To explore the causality between VTE, DVT, PE, and IS, we conducted a bidirectional MR analysis and mapped the flowchart in Figure 1. The exposure of forward MR were VTE, DVT, PE, and IS was the outcome, where the reverse MR referred to an exchange between the exposure and the outcome. Forward and reverse MR analysis were implemented by using the instrumental variable (IV) approach to replace the exposure and outcome as far as possible. There are some major limitations about IV, including weak instrument bias, confoundings, and pleiotropy. 22 Therefore, in order to obtain reliable results, the IV must satisfy three key principles of MR. 3 Firstly, the IV must be strongly associated with exposure. Furthermore, the IV is not associated with all potential confounding factors that may influence exposure and outcome. Ultimately, the IV can only influence the outcome by associating it with exposure, not by other means. To adhere to the established norms of MR, this study was conducted in accordance with the most recent guidelines on the utilization of MR for epidemiological research (STROBE-MR). 24 The study comprised the following core steps: (1) the selection of genetic IVs associated with exposure and outcome; (2) the utilization of multiple MR methods to assess multiplicity of effects; (3) the incorporation of heterogeneity and sensitivity analyses.
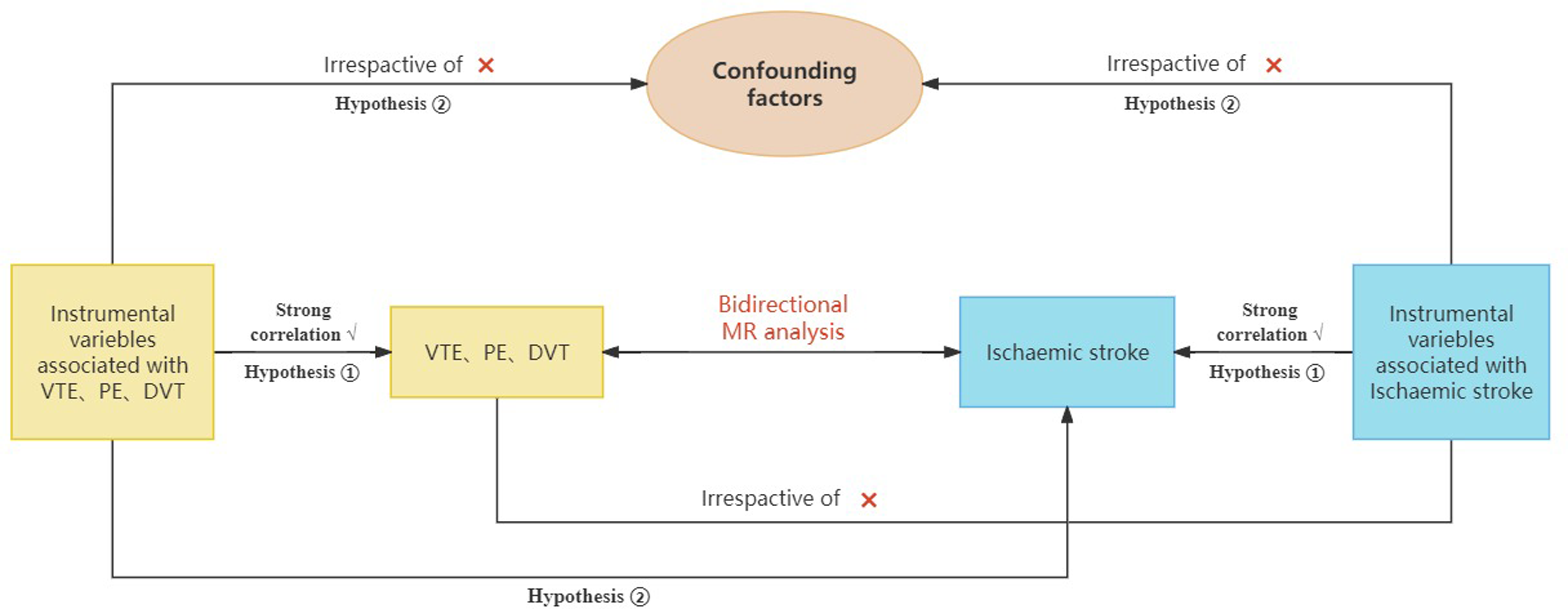
The fowchart of the MR research.
Data Source
To minimize bias due to population stratification and ethnic differences, only samples from European populations were selected. 25 The data source for the genetic associations related to IS came from the IEU Open GWAS database (https://gwas.mrcieu.ac.uk/), which included 484 121 individuals, comprising 11 929 CI cases and 472 192 control samples. Meanwhile, GWAS data for VTE and PE were procured from Finngen database (https://www.finngen.fi/en). GWAS data for DVT were assessed from UK biobank database (https://www.ukbiobank.ac.uk/). The data between exposures and outcomes were from different countries, environmental factors and population structure, thereby ensuring the scientific validity and avoiding the homogeneity of the study. The total sample size for venous thrombosis was 218 792, comprising 9176 patients with VTE and 209 616 controls. There were 4185 cases and 214 228 controls in the GWAS data for PE, with a sample size of 218 413. The GWAS data sample size for DVT was 337 159, including 6767 cases and 330 392 controls.
IVs Selection
IVs Screening Threshold
In selecting the instrumental variables, we selected SNPs that were found to be associated significantly with exposure based on stringent criteria (P < 5 × 10−6) and independence (r² < 0.001, kb = 10 000). It was confirmed that these selected SNPs were strongly associated with VTE, DVT, and PE. Similarly, for the reverse MR analysis, the threshold was set at P < 5 × 10−6 and independence (r² < 0.001, kb = 10 000). These thresholds were used in another valid research and approved. 26
IVs Harmonization
Palindromic SNPS referred to the identical and complementary part of the duplex DNA, and duplicate SNPS refer to the detection of rs sites with exactly the same name, both of which should be deleted at screening IVs. 21 After excluding all SNPs with palindromes and duplicates, we harmonised the effect alleles among the exposure and outcome datasets. An F-value higher than 10 is an indication of a low risk of weak IV bias and avoids weak instrumental bias. 27 The F statistic, calculated as F = (N − 2) × R^2/(1 − R^2), where R^2 is the variance of exposure explained by the genetically determined effect allele frequency (EAF) and genetic effect of exposure, and N is the sample size, was used to assess the strength of the instrumental variable (IV).28,29 A comprehensive account of all selected SNPs can be found in the Supplementary Table.
Statistical Analyses
MR Analysis
Three methods - inverse variance weighted (IVW), MR-Egger and weighted median (WM) - were used to determine the causal relationship. 30 Of these, when all IVs are validated tools, the IVW method is the most effective and produces the greatest statistic validity. 31 In order to establish causality, the following criteria were employed: a P-value of less than 0.05 in IVW analyses and a consistency of results between the WM and MR-Egger analyses and those of IVW. The same methods were applied in the reverse MR analysis as above in the forward analysis. The set of SNPs associated with IS was used to examine its causal effect with VTE, DVT, and PE. The TwoSampleMR software package was used for all analyses in RStudio (version 4.3.2).
Sensitive Analysis
Sensitivity analysis were performed using the MR-Egger intercept and MR-PRESSO for determining the presence of pleiotropy. 32 Pleiotropy referred to the phenomenon that a single DNA variant or gene affects multiple traits simultaneously, which must be avoided and verified in MR studies. 33 The absence of horizontal pleiotropy was indicated by an intercept close to 0 and a p-value greater than 0.05. Subsequently, Cochran's Q-test was employed to quantify heterogeneity in the IVW estimates, with a P-value > 0.05 indicating the absence of heterogeneity. 34 In the case of heterogeneity, the IVW MR analyses were carried out with random effects. Leave-one-out analyses were performed to determine the influence of individual SNPs on the overall causal effect to ensure the reliability of the results. Symmetries of selected SNPs were assessed using funnelplotting. Scatter plots were used to visualise the effect association between exposure and outcome.
Result
Selection of IVs
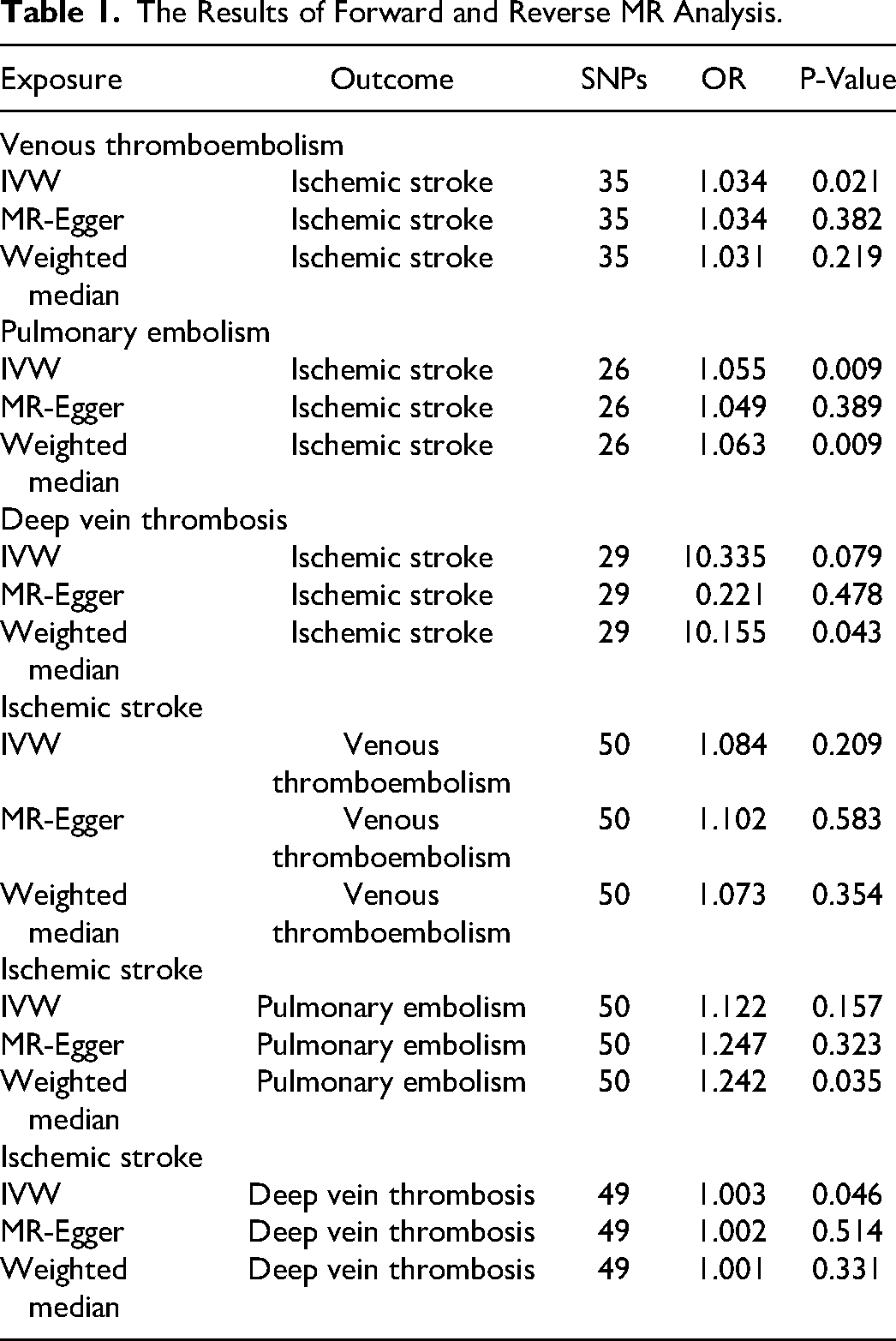
In the forward MR analysis, a selection of IVs was made. Respectively, VTE, PE, and DVT were ultimately selected as 35, 26, and 29 SNPs (Supplementary Tables 1-3). When IS was considered the exposure, 50,50,49 SNPs were included in the reverse analyses for venous VTE, PE, and DVT, respectively (Supplementary Tables 4-6). Finally, the F-value for the IVs was greater than 10, which indicated that it is unlikely that a weak IV bias was actually present (Supplement Table 7).
Mendelian Randomization and Sensitive Analysis
IVW analysis showed that VTE significantly increased the risk of IS (ORIVW = 1.034, PIVW = 0.021, ORWM = 1.031, ORMR-Egger = 1.034). Similarly, PE was also a risk factor for IS (OR = 1.055, PIVW = 0.009, ORWM = 1.063, ORMR-Egger = 1.049). However, the IVW method also demonstrated an absence of statistical evidence to support a causal relationship between DVT (PIVW > 0.05) and IS. The results of all forward MR analysis were presented in Table 1. The scatter plots of the effect sizes of the SNPs for the individual phenotypes in the forward analyses were shown in Figure 2. In addition, when the line of the other two methods in the scatter plot is in the same direction as the line of the IVW, the validity of the IVW can be testified. For the reverse MR analysis, the IVW results showed a reverse causality between IS and DVT, meaning that IS is a risk factor for DVT (OR = 1.003, PIVW = 0.046, ORWM = 1.001, ORMR-Egger = 1.002). In addition, the inverse analysis between IS with VTE and PE was not statistically significant (PIVW < 0.05). The specific P-values were presented in Table 1. Figure 2 also showed the scatter plots for individual phenotypes in the reverse analysis.
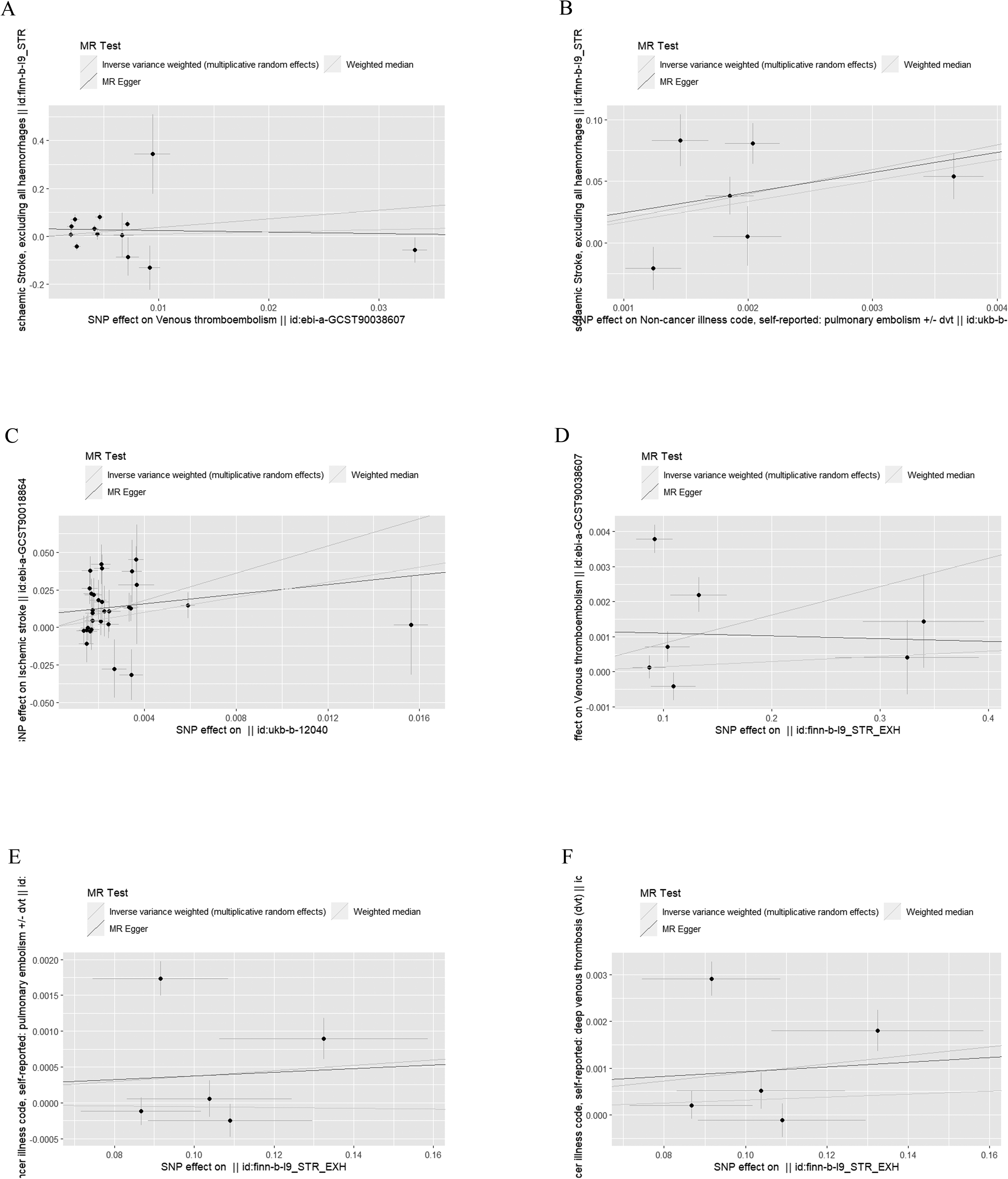
The scatter plots about the forward and reverse MR analysis. (A) VTE on IS; (B) PE on IS; (C)DVT on IS; (D) IS on VTE; (E) IS on PE; (F) IS on DVT. Venous thromboembolism:VTE; Pulmonary embolism:PE; Deep vein thrombosis:DVT; Ischaemic stroke:IS.
The Results of Forward and Reverse MR Analysis.
For sensitive analysis, there was no evidence of horizontal pleiotropy in the results of the MR-Egger intercept test and MR-PRESSO (P > 0.05)(Table 2). In this study, a random effects model was employed for all analyses, given that the P value for Cochran Q was less than 0.05 (Table 2). The results of the ‘leave one out’ analysis and visualisation demonstrated the robustness of the MR results. Supplement Figures 1-3 exhibited the forest plots, funnel plots and ‘leave one out’ visualisations of this study.
The Results of MR Analysis Results.
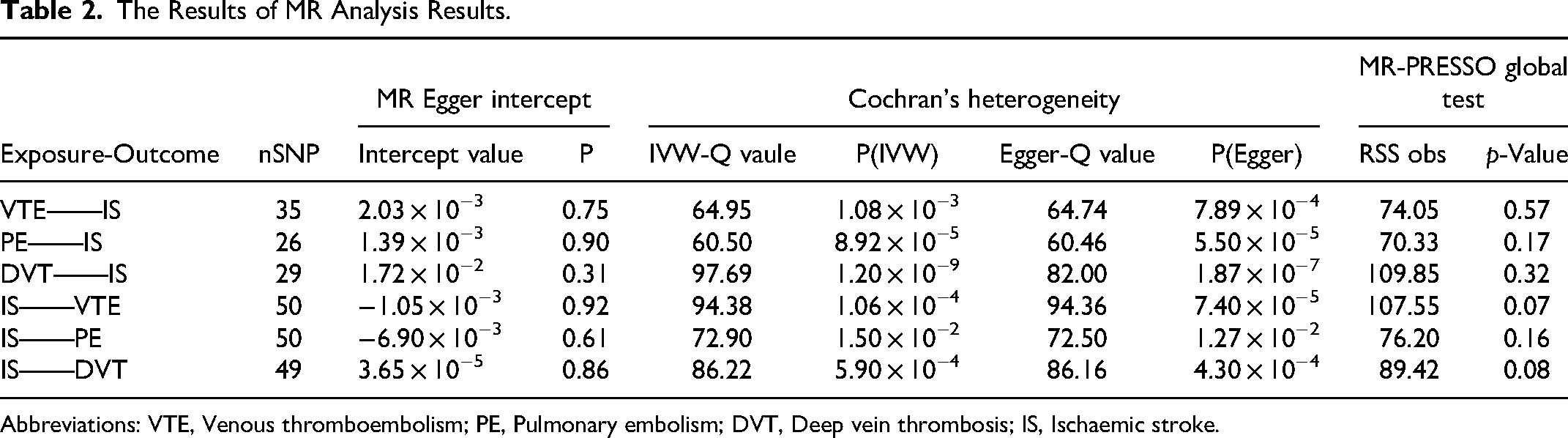
Abbreviations: VTE, Venous thromboembolism; PE, Pulmonary embolism; DVT, Deep vein thrombosis; IS, Ischaemic stroke.
Discussion
The results of this study suggested that VTE and PE were associated with an increased risk of IS. It exhibited a positive correlation between the two conditions, rather than inverse causation. These findings have considerable ramifications for the prevention and early detection of IS. In addition to this, we also found that IS may increase the risk of DVT. It also reminds us of the need to focus on the care of patients with IS and the prevention of complications.
The primary pathological process underlying an IS is cerebral infarction. A lack of sufficient blood supply to brain tissue results in an initial reversible loss of function, which ultimately culminates in cerebral infarction. This is characterised by the death of neurons and the destruction of supportive structures. Ischemia initiates a series of events that commences with a loss of electrical function and subsequently progresses to membrane dysfunction, accompanied by calcium influx.This results in the formation of calcium-dependent ROS, which ultimately leads to the disruption of cell membranes and the subsequent cytolysis and necrosis of brain tissue. IS represents one of the most significant neurovascular causes of mortalities and morbidities.Stroke is one of the most prevalent incapacitating diseases, the second most common cause of mortality, and the third most common cause of disability-adjusted life years. This highlights the significant global burden of IS and underscores the importance of prevention strategies.35,36
It is of paramount importance to identify patients who have suffered an IS and who are at an enhanced likelihood of experiencing a VTE in order to guarantee that they receive suitable thromboprophylaxis. A number of potential risk factors and indicators of VTE in patients with ischaemic stroke have been identified in previous observational studies. However, the potential for bias in these studies was not always adequately assessed using a structured framework. However, the paucity of studies of sufficient quality on each factor, coupled with the limitations of the research methodology and modelling framework, has resulted in a dearth of evidence-based conclusions. The meta-analyses provided by the studies indicated an association, but lacked validation of exposures and outcomes, and could not adjust for obvious confounders such as body mass index (BMI). Furthermore, the existing studies do not provide convincing results, do not confirm strong associations, and are contradictory and controversial. Consequently, further confirmation is imperative.37–39
VTE is responsible for a quarter of global deaths, with thrombosis representing the most common form of VTE. 37 Despite a decline in age-standardised mortality rates for heart attack and stroke in developed countries over the past two decades, the mortality rate from VTE has remained stable. VTE is primarily constituted of two discrete conditions: DVT and PE.40,41 DVT is defined as the formation of an abnormal blood clot within the deep veins of the body. DVT has the capacity to manifest in any deep vein throughout the body, though it is most frequently observed in the deep veins of the lower extremities, particularly the popliteal and femoral veins. The clinical manifestations of DVT include oedema of the affected limb, pain, increased skin temperature and dilation of superficial veins. A PE represents a significant complication of VTE, typically resulting from the dislodgement of a thrombus from the deep veins of the lower extremities. The dislodged thrombus enters the Pulmonary artery along with the blood flow, causing an obstruction of the Pulmonary artery and its branches, which results in an obstruction of the Pulmonary circulation. The clinical manifestations of PE are diverse, ranging from asymptomatic in mild cases to severe symptoms such as respiratory distress, chest pain, hemoptysis and even shock.42,43
It is essential to acknowledge that the conclusions of this MR study are not without inherent limitations. As MR is a causal inference method based on genetic variation, there may be discrepancies in the genetic background between different population samples, which will consequently impact the precision of the MR results. Additionally, there may be instances of horizontal pleiotropy. Despite utilising rigorous criteria for the identification of SNPs and employing a range of testing and correction methods, including MR-Egger regression and MR-PRESS0 global test, it is still plausible that the genetic instrumental variables may not be associated with IS through PE, and that alternative pathways may contribute to this risk. Furthermore, it is not always possible to fully identify all potential biases through sensitivity analysis,the presence of linkage disequilibrium (LD), defined as the correlation between genetic variants within a population, must be considered. If the genetic variant employed to represent the exposure factor exhibits LD with another genetic variant associated with the outcome, the potential for confounding effects to arise exists, thereby affecting the accuracy of the MR results. 44
In conclusion, our MR study sought to elucidate the causal relationship between VTE, PE, DVT and IS. The results of this study indicate that PE and VTE, rather than DVT, may act as a risk factor for IS. On the contrary, IS may further aggravate the occurrence DVT. In the future, it would be possible to increase the sample size, include more racial groups, and perform population stratification in order to reduce the effect of genetic heterogeneity. It is necessary to conduct further research in order to ascertain the biological mechanisms involved and to identify any possible trends between the two.
Conclusion
The results of this MR study lend support to the hypothesis that VTE and PE are risk factors for IS. On the contrary, IS may further aggravate the occurrence DVT. This could provide a novel approach to clinical practice for the treatment of patients with VTE by implementing care and prevention strategies for IS. In order to mitigate the likelihood of complications, it is recommended that patients engage in lifestyle modifications, including dietary adjustments, weight loss, smoking cessation, and regular physical activity. Additionally, it is advised that patients maintain optimal blood pressure and adhere to oral anticoagulant therapy, with the objective of reducing the risk of atrial fibrillation and thus preventing IS.
Supplemental Material
sj-xlsx-1-cat-10.1177_10760296241293333 - Supplemental material for A Bidirectional two-Sample Mendelian Randomization Study of the Association Between Venous Thromboembolism and Ischaemic Stroke
Supplemental material, sj-xlsx-1-cat-10.1177_10760296241293333 for A Bidirectional two-Sample Mendelian Randomization Study of the Association Between Venous Thromboembolism and Ischaemic Stroke by Haibing Xiong, Xinhong Tian, Aiwei He, Tingting Chen, Yanlin Li, Jiajie Leng and Letai Li in Clinical and Applied Thrombosis/Hemostasis
Supplemental Material
sj-docx-2-cat-10.1177_10760296241293333 - Supplemental material for A Bidirectional two-Sample Mendelian Randomization Study of the Association Between Venous Thromboembolism and Ischaemic Stroke
Supplemental material, sj-docx-2-cat-10.1177_10760296241293333 for A Bidirectional two-Sample Mendelian Randomization Study of the Association Between Venous Thromboembolism and Ischaemic Stroke by Haibing Xiong, Xinhong Tian, Aiwei He, Tingting Chen, Yanlin Li, Jiajie Leng and Letai Li in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Acknowledgements
We would like to express our sincere gratitude to the researchers and participants in the GWAS for their invaluable contributions to the collection and management of the extensive data resources.
Data availability statement
All datasets used for MR analysis in this study are publicly available and available for use on reasonable demand. The authors are grateful to all researchers who have made their data available for use in this study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Statement
No further institutional review board approval was required as the data used were derived from published studies and public databases.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funded project: Chongqing Science and Health Joint Medical Research Project (2021MSXM261).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
