Abstract
Background
Rivaroxaban, a direct Factor Xa inhibitor, is commonly used for cerebral venous thrombosis (CVT) correction. However, pharmacokinetic differences in Chinese may vary in sensitivity and tolerance, resulting in either insufficient or excessive anticoagulation. Herein, the optimizing dosages of rivaroxaban in Chinese patients with CVT were analyzed based on monitoring anti-Xa activity dynamically, to maintain therapeutic efficacy and reduce rivaroxaban-related bleeding.
Methods
A real-world cohort study was conducted involving 112 CVT patients in Xuanwu Hospital, from August 2021 through January 2024. Patients were grouped according to their doses of rivaroxaban use (5, 10, 15, and 20 mg daily) based on dynamic plasma anti-Xa activity monitored using the chromogenic anti-Xa assay. Plasma levels of anti-Xa activity reached the therapeutic range, bleeding events and the dosage of rivaroxaban among these groups were analyzed.
Results
The ratios of the patients whose plasma anti-Xa levels reached the standard therapeutic level (0.3–0.7 IU/mL) between the cohorts less than 20 mg/d and 20 mg/d showed no statistical difference, and no significant disparities were observed among 5, 10, 15, and 20 mg/d dose groups. There was a discernible increase in the proportion of patients with bleeding events in the 20 mg/d group, even though the results did not reach a statistical difference. Meanwhile, in patients with bleeding events, their plasma anti-Xa levels could exceed 0.7 IU/mL.
Conclusion
Sensitivity and tolerance to rivaroxaban in Chinese may vary. Individualized therapy dosage under the guidance of anti-Xa activity monitoring may not only guarantee anticoagulation effect, but also reduce rivaroxaban-related bleeding events.
Introduction
Cerebral venous thrombosis (CVT), is a special subtype of stroke. 1 The standard medical strategy of CVT at present is still anticoagulation, in which, direct oral anticoagulants (DOACs) such as rivaroxaban, a direct Factor Xa inhibition, is commonly used due to its convenience during treatment, which need not require frequent blood monitoring like warfarin use, 2 and it was demonstrated non-inferiority to warfarin in CVT correction. 3 Based on European and American guidelines, Chinese guidelines still recommend the standard dose of rivaroxaban is 20 mg per day, with a reduced dose of 15 mg daily for patients whose creatinine clearance rate is less than 50 ml/min. 4 However, with the recommended dosage, we often encounter rivaroxaban-related bleeding events in clinical settings. A previous study revealed that over 30% of patients in Asian countries, excluding Singapore, received inappropriate doses of DOACs, including rivaroxaban, and this discrepancy might be attributed to differences in pharmacokinetics between Chinese and Western populations.5,6
Also, research on Asian populations revealed that the exposure concentration of rivaroxaban was higher in Asians than in Caucasians at equivalent therapeutic doses. 7 Furthermore, a subgroup analysis of the ROCKET AF trial indicated that East Asian patients taking the standard 20 mg dose of rivaroxaban had a significantly higher incidence of major bleeding events compared to non-East Asians. 8 The absence of a universally accepted dosing standard for rivaroxaban in Chinese CVT patients, along with the potential risk of bleeding and the lack of a specific reversal agent, underscores the urgency for optimized dosing strategies.
Previous research has suggested that liquid chromatography-tandem mass spectrometry (LC-MS/MS) is the preferred method for quantitatively evaluating the blood concentration of rivaroxaban due to its high specificity and sensitivity. 9 However, this approach necessitates sophisticated and costly equipment as well as skilled professionals, thereby limiting its widespread application. In this context, anti-Xa activity monitoring has surfaced as a promising tool to assess rivaroxaban levels and effectiveness as well as fine-tune dosages accordingly. The chromogenic anti-Xa assay, which measures the inhibition of plasma Factor Xa by rivaroxaban, offers a direct link between drug concentration and anticoagulant effect. Previous studies established a good linear relationship between anti-Xa activity and rivaroxaban plasma concentrations.10,11 It is crucial to emphasize that the low molecular weight heparin anti-FXa assay employed in this study, while previously suggested as feasible for rivaroxaban monitoring, has not undergone specific validation for quantifying rivaroxaban levels. In the absence of an international standard, we preliminarily explore that the assay qualitatively measures the inhibition of Factor Xa by rivaroxaban, allowing for the classification of patients’ anti-Xa levels as therapeutic, sub-therapeutic, or supra-therapeutic. We emphasized that the results presented herein must be interpreted with caution, recognizing the qualitative rather than quantitative nature of the assay outcomes.
Methods
Study Design and Population
A total of 112 Chinese patients with imaging confirmed CVT enrolled in this real-world cohort study consecutively after signing the informed consent, from August 2021 through January 2024 in Xuanwu Hospital, Capital Medical University, China. The diagnosis imaging tools included magnetic resonance venography, computed tomographic venography, magnetic resonance black-blood thrombus imaging, and digital subtraction angiography.
The inclusion criteria were as follows:
Patients match the diagnostic criteria of CVT; Age ranging from 18 to 80 years; Anticoagulation with rivaroxaban for at least 7 days; Signed informed consent form. Use of anticoagulants other than rivaroxaban for long-term anticoagulation therapy such as dabigatran, warfarin, unfractionated heparin, or low molecular heparin; Inability to complete cerebral venography and stenting or allergy to contrast agent; Those with severe bleeding events; Patients with life-threatening conditions including severe cardiovascular or respiratory disorders or malignancy; Subjects lacking complete data.
The exclusion criteria were as follows:
This study complied with the Declaration of Helsinki and was approved by the ethics committee of Xuanwu Hospital, Capital Medical University. All patients signed the informed consent.
Intervention and Grouping
After completing all relevant examinations, patients underwent rivaroxaban (manufactured by Suzhou Third Pharmaceutical Factory Co., Ltd, Suzhou, China) with the approval number H20213398. The initial dosage was determined based on each patient's baseline conditions, including clinical symptoms, neuroimaging findings, and creatinine clearance. Patients were grouped according to different dosages of the rivaroxaban (5/10/15/20 mg) daily for equal or more than 7 days. Meanwhile, plasma anti-Xa activities were assayed dynamically during rivaroxaban use.
Data Collection
Data, including demographic information (age, sex, etc), clinical symptoms, and imaging findings, baseline anti-Xa activity were comprehensively recorded. Additionally, both the initiation time of oral rivaroxaban and the modified Rankin Scale (mRS) scores were also documented. Plasma samples were collected at baseline prior to rivaroxaban use and a minimum of seven consecutive days post-rivaroxaban, to ensure optimal assessment conditions. On the day of blood collection post-rivaroxaban, blood samples were drawn through aseptic venipuncture from the antecubital vein at 4–6 h post-rivaroxaban orally. Immediately after collection, the blood was transferred to 3.8% sodium citrate test tubes to preserve its integrity. Exercise great care in the centrifugation of the samples in order to minimize platelet alteration, as well as to achieve maximum removal of the platelets. Perform centrifugation for 15 min at a speed of 2000–2500 g, and at a temperature of approximately 18 °C. Blood samples should be tested without dilution and loaded into the instrument to measure the anti-Xa activity, which was carried out automatically in the analyzer device at 405 nm as soon as the samples were loaded.
The anti-Xa assay in this study is a competitive assay using a synthetic chromogenic substrate on STA Compact, which is an automatic coagulation analyzer from Diagnostica Stago. The test principle is: upon the addition of factor Xa to the plasma-substrate mixture, two simultaneous reactions occur - hydrolysis of the substrate by factor Xa and inhibition of factor Xa by DOACs. After the necessary period of time for the competitive reaction to reach equilibrium, the quantity of paranitroaniline that is released is inversely proportional to the concentration of anticoagulant present in the test medium. The low molecular weight heparin level is provided in IU/mL, with a detection threshold of 0.1 IU/mL and a linear range extending up to 2.0 IU/mL. Reproducibility studies using samples containing rivaroxaban yielded consistent results. It is important to clarify that this assay, along with its calibration and validation, was specifically designed and optimized for low molecular weight heparin and has not been officially developed or validated for DOACs such as rivaroxaban, despite the above detection threshold and linearity range. In the absence of an international standard, we preliminarily explore that the assay qualitatively measures the inhibition of Factor Xa by rivaroxaban, allowing for the classification of patients’ anti-Xa levels as standard therapeutic (0.3–0.7 IU/mL), supratherapeutic (over 0.7 IU/mL), or subtherapeutic (below 0.3 IU/mL). We emphasized that the range is adapted from low molecular weight heparin therapy and should not be interpreted as quantitatively accurate for rivaroxaban monitoring, but this range serves as a rough indicator of potential anticoagulant effect.
Statistical Analysis
All statistical analyses in this study were conducted by the SPSS version 26.0 program (IBM). Continuous variables following normal distribution were calculated as mean ± standard deviation and compared using t-test or two-way analysis of variance (ANOVA). Alternatively, non-normally distributed data were presented as median (interquartile range) and assessed using Mann–Whitney U test or non-parametric Wilcoxon signed-rank test. Categorical variables were expressed as counts and percentages and Pearson's chi-square test or Fisher’s exact test was applied to evaluate differences. Two-tailed p-value <0.05 was considered statistically significant.
Results
Baseline Characteristics
A total of 112 patients who matched the inclusion and exclusion criteria entered into final analysis. They were divided into four groups based on the dosage of their rivaroxaban use: including 15 cases in 5 mg/d group, 54 cases in the 10 mg/d group, 18 cases in the 15 mg/d group, and 25 cases in the 20 mg/d group. Notably, the patients enrolled hailed from 20 diverse provinces across China, accounting for 68% of the country's geographical regions (refer to Figure 1). The average age was 41.2 ± 15.6 years, with females accounting for 60.71%. Importantly, there were no significant disparities in the prevalence of comorbidities, including hypertension, diabetes, and hyperlipidemia, among the groups. Furthermore, symptoms such as headache, tinnitus, neurological deficits, visual impairment, epilepsy as well as veins involvement except for superior sagittal sinus involvement, did not vary significantly among the groups. There were also no notable differences about the baseline mRS scores among the groups, and the baseline plasma anti-Xa activity in all the patients enrolled was 0.0 IU/mL, more details were shown in the Supplemental Material.
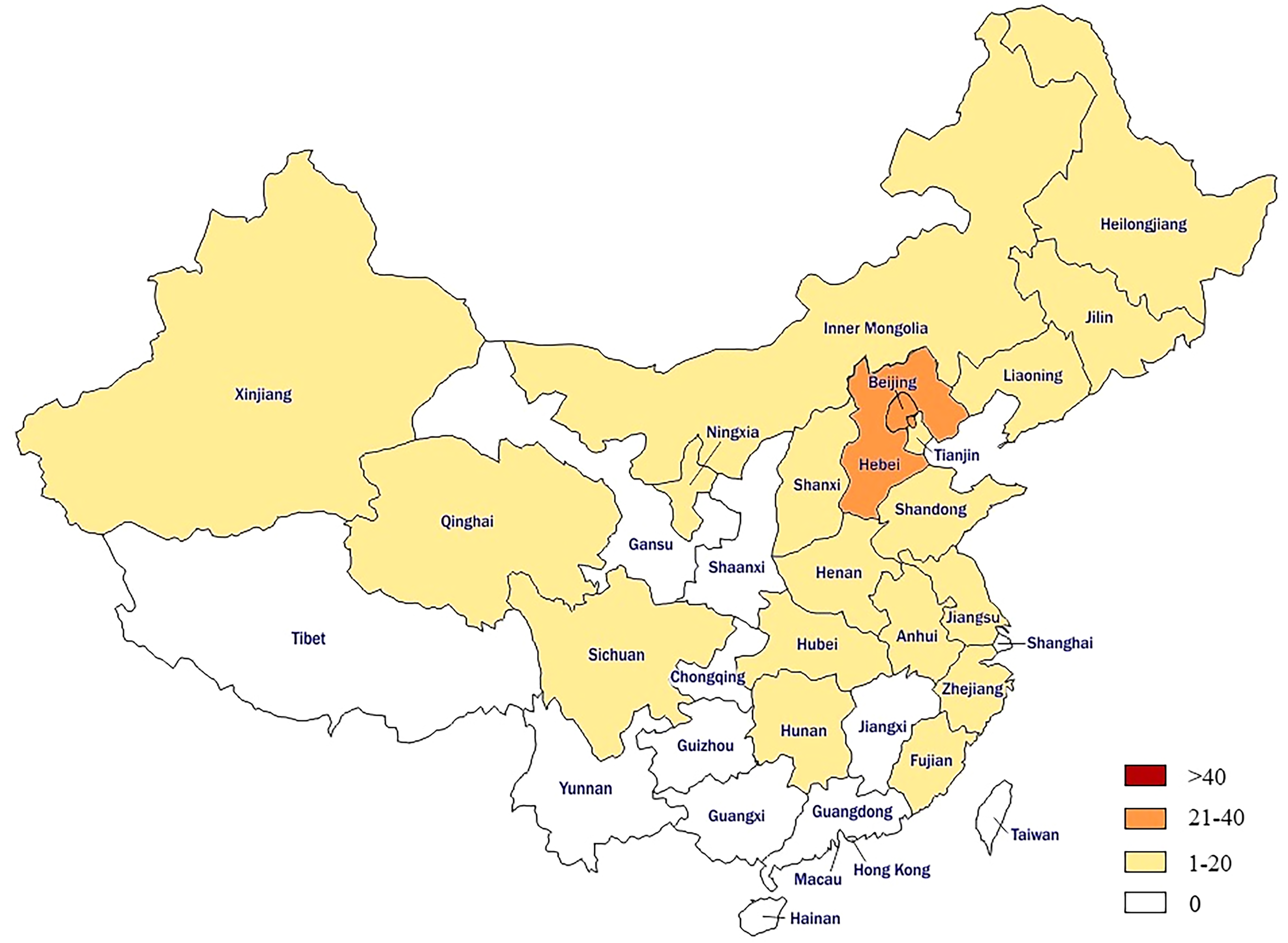
The participants came from 20 diverse provinces across China, representing 68% of the country's geographical regions.
Achievement of Therapeutic Anti-Xa Levels
Firstly, patients were categorized based on the rivaroxaban doses they underwent: 20 mg/d cohort and <20 mg/d cohort. The anti-Xa level at 0.3 to 0.7 IU /mL was considered as ideal therapeutic anticoagulation effect, supratherapeutic (over 0.7 IU/mL) was with a high risk of bleeding and subtherapeutic (below 0.3 IU/mL) was weak anticoagulation effect. Our data revealed that no statistical difference in achieving the standard therapeutic anti-Xa levels between these two cohorts (<20 mg/d cohort vs 20 mg/d cohort was 27.59% vs 40.00%, p = 0.234, details were provided in Table 1), although there was a trend of low achievement rates with low dosages (refer to Figure 2), no significant disparities were observed among the 5, 10, 15, and 20 mg/d groups (20.00% vs 25.93% vs 41.18% vs 40.00%, p = 0.394).
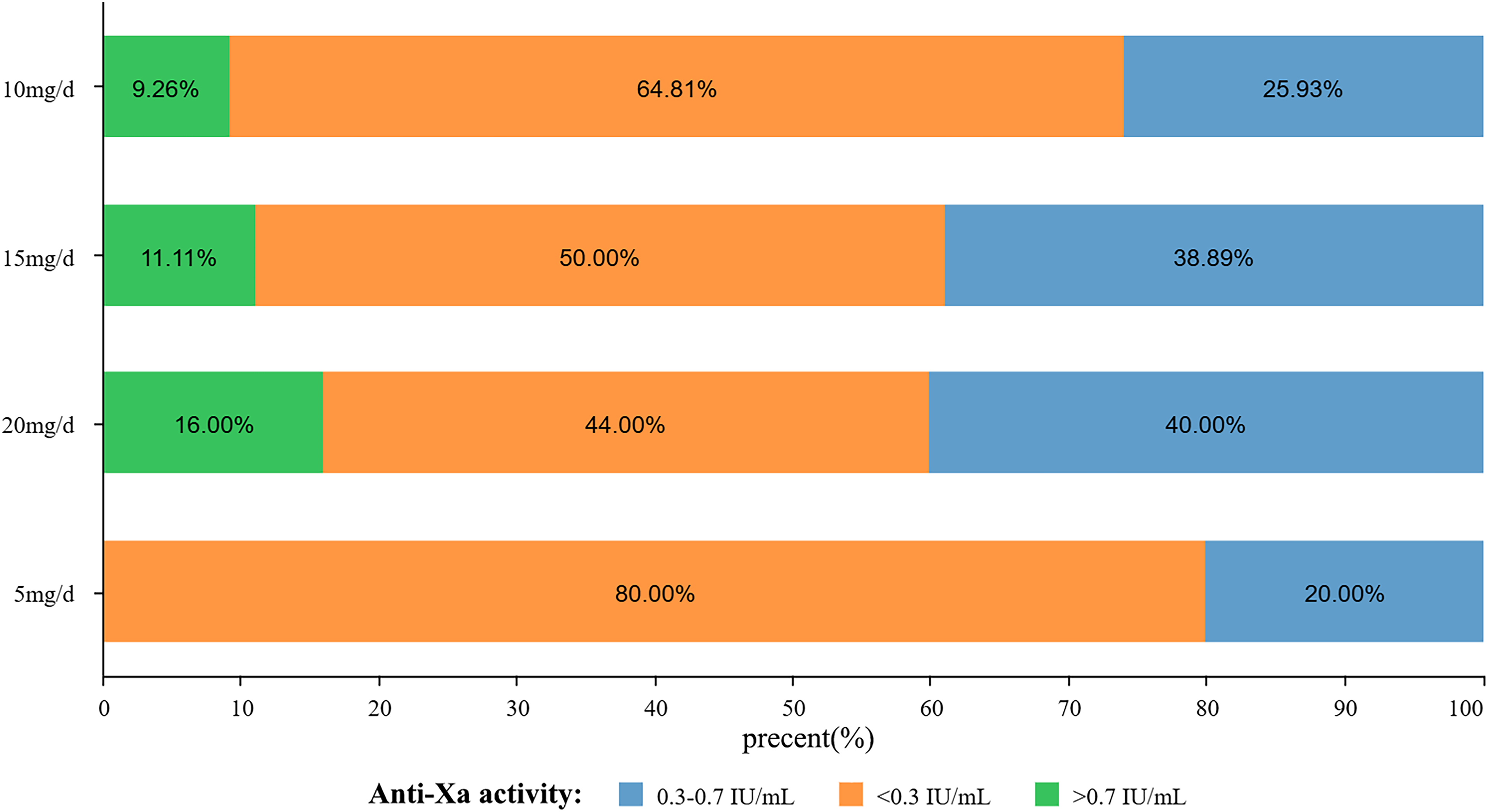
Achievement of therapeutic anti-Xa levels: A trend of low achievement rates with low dosages.
Achievement of Therapeutic Anti-Xa Levels.
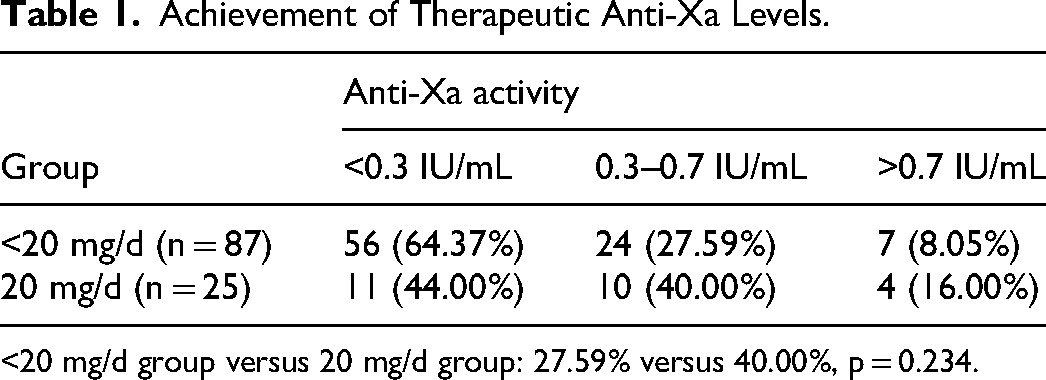
<20 mg/d group versus 20 mg/d group: 27.59% versus 40.00%, p = 0.234.
Bleeding Risk
Chi-square tests were employed to compare bleeding risk among the 5, 10, 15, and 20 mg/d groups, there was a discernible increase in the proportion of patients with bleeding risk in the 20 mg/d group (refer to Figure 3) even though the results did not reach statistical difference (0 vs 9.26% vs 11.11% vs 16.00%, p = 0.429). Meanwhile, in the patients with bleeding events, their plasma anti-Xa levels could exceed 0.7 IU/mL, details were shown in Table 2.
Bleeding risk: A discernible increase in the proportion of patients with high bleeding risk with higher dosages.
Bleeding Risk.
The Rate to Reach Therapeutic Anti-Xa
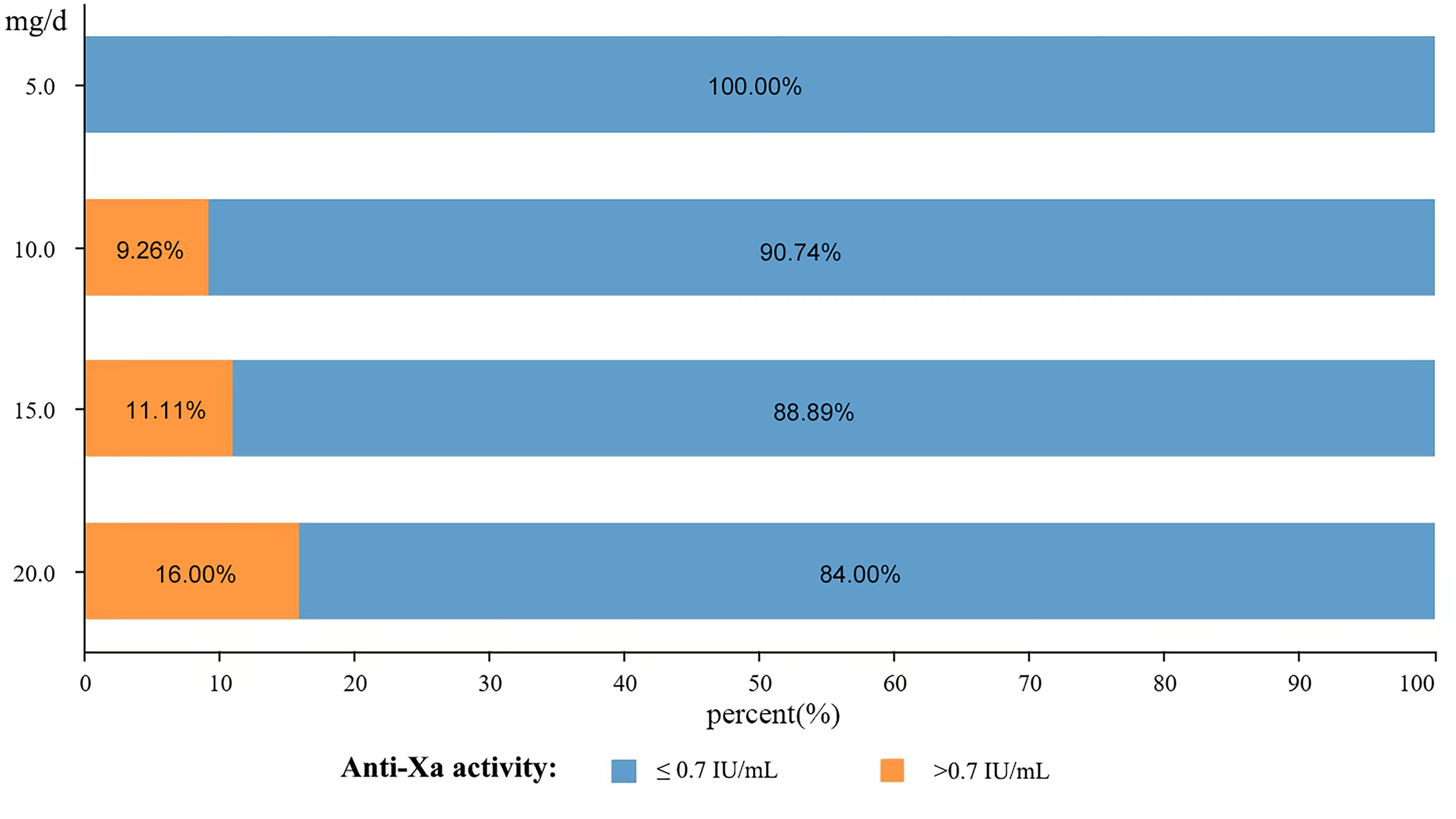
Finally, the sensitivity of rivaroxaban was evaluated by the rate to reach therapeutic plasma anti-Xa level at Day7 post-rivaroxaban continuous use: below 0.3 IU/mL was considered as rivaroxaban -insensitive, equal to or above 0.3 IU/mL was rivaroxaban -sensitive. We found that plasma anti-Xa in the majority of patients in <20 mg/d of rivaroxaban group could obtain equal or above 0.3 IU/mL, although no significant difference was found between <20 mg/d cohort and 20 mg/d cohort (35.63% vs 56.00%, p = 0.067). Nevertheless, a downward trend of the rate to reach therapeutic anti-Xa level was evident with decreasing dosages (illustrated in Figure 4). Specifically, a notably higher proportion of patients in the 5 mg/d group exhibited anti-Xa levels below 0.3 IU/mL compared to the 20 mg/d cohort (20.00% vs 56.00%, p = 0.026; detailed data were in Table 3).
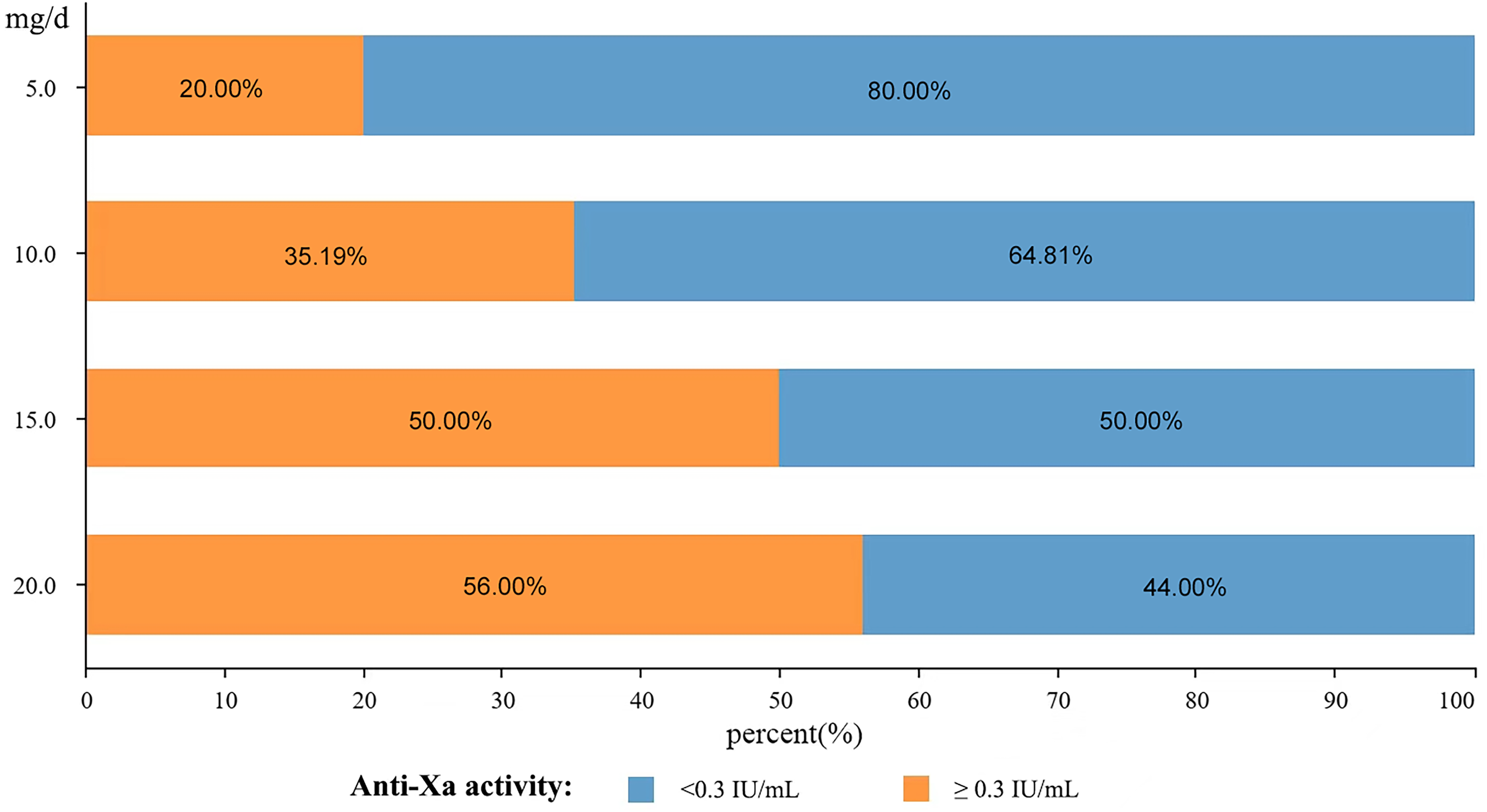
Drug Sensitivity: A downward trend of the rate to reach therapeutic anti-Xa level was evident with decreasing dosages.
Drug Sensitivity.
Discussion
In the current study, we aimed to refine the dosing regimen of rivaroxaban for Chinese patients with cerebral venous thrombosis by utilizing anti-Xa activity monitoring. Our primary goal was to establish an effective dosing strategy that balances therapeutic efficacy with minimal bleeding risk. Our results demonstrated that reduced doses of rivaroxaban could achieve comparable therapeutic anti-Xa levels while maintaining a low bleeding risk compared to traditional doses(The dosage recommended in the drug instructions). This provides valuable insights into the optimization of anticoagulation therapy for Chinese patients with CVT.
The standard dosage recommendation for rivaroxaban in China, which aligns with European and American guidelines, is 20 mg per day. For patients with creatinine clearance rates below 50 ml/min, a reduced dose of 15 mg daily is suggested. 4 However, a significant discrepancy exists between these recommendations and actual dosing practices in Asian countries. Over 30% of patients in Asian regions, excluding Singapore, receive inappropriate doses of DOACs, including rivaroxaban. This divergence may be attributed to pharmacokinetic differences between Chinese and Western populations.5,6 Notably, a subgroup analysis from the ROCKET AF trial indicated that East Asian patients receiving the standard 20 mg dose of rivaroxaban had a significantly higher incidence of major bleeding events compared to non-East Asian patients. 8 This underscores the necessity for tailored dosing strategies in this specific patient segment.
Our study bridges this knowledge gap by using anti-Xa activity monitoring as a precise tool to assess rivaroxaban levels and adjust dosages accordingly. The use of the low molecular weight heparin anti-Xa assay in our study represents an interim approach, necessitated by the current lack of a standardized, commercially available assay for rivaroxaban. This interim solution is grounded in published reports demonstrating the feasibility of this method for qualitative assessment of rivaroxaban levels.10,11 The absence of statistically significant differences in therapeutic achievement and bleeding risk among different dosage groups highlights the potential of reduced rivaroxaban doses in Chinese CVT patients. This finding is consistent with previous studies reporting similar efficacy and safety profiles between reduced and standard rivaroxaban doses in various patient populations.12,13 Furthermore, our study contributes to the existing evidence by identifying a trend toward reduced sensitivity at lower rivaroxaban doses, specifically in the 5 mg group. This observation aligns with the understanding that anti-Xa activity directly reflects the drug's anticoagulant effect, and lower levels may indicate inadequate Factor Xa inhibition. Consequently, suboptimal anti-Xa levels could compromise the anticoagulant effect of rivaroxaban, potentially elevating the risk of thromboembolic events.
In comparison to previous research, our study's findings provide valuable insights into the management of Chinese patients with CVT undergoing rivaroxaban therapy. Specifically, our results may support the utilization of anti-Xa activity monitoring to adjust rivaroxaban dosing in this patient population. This tailored approach may ensure optimal therapeutic effectiveness while significantly reducing the risk of bleeding complications. Based on the comprehensive analysis of our data, we recommend initiating anticoagulation therapy with a reduced rivaroxaban dose of 10–15 mg daily for Chinese CVT patients. This initial dosing regimen should be followed by regular anti-Xa activity checks to monitor the drug's effectiveness. In cases where anti-Xa levels drop below the therapeutic range, a gradual increase in dosage may be necessary to maintain optimal anticoagulation. Conversely, if anti-Xa levels exceed the upper limit of the therapeutic range, a reduction in dose would be advisable to mitigate the risk of bleeding. This individualized dosing strategy, tailored to each patient's unique pharmacokinetic profile, offers the potential to significantly enhance therapeutic outcomes in Chinese CVT patients undergoing rivaroxaban therapy.
Limitations
Our study has several strengths, including its prospective design, use of validated anti-Xa assays, and inclusion of a diverse range of rivaroxaban doses. However, some limitations should be acknowledged. Firstly, it is important to note that the heparin anti-FXa assay used in this study has not been specifically validated for monitoring rivaroxaban levels. While previous studies have suggested its feasibility, the use of this assay for rivaroxaban monitoring remains non-standardized and potentially suboptimal. Therefore, the application of this assay in a clinical setting without proper validation poses potential risks to patients. Until such validation is complete, caution should be exercised when interpreting our results and applying them to clinical practice. Second, the sample size was relatively small Chinese patients, limiting the power to detect small differences between groups and generalize to other ethnicities. Third, potential confounding factors such as renal function and concomitant medications were not fully explored.
Future investigations should strive to overcome the limitations of our current study. Specifically, developing and validating accurate and standardized assays for rivaroxaban, as well as other DOACs. Such assays will enable more precise dosing and monitoring, ultimately leading to optimizing the outcomes of the patients. Besides, expanding sample sizes and including multi-ethnic groups can enhance the study's power and generalizability. A comprehensive examination of confounding factors, such as renal function, concomitant medications, and genetic variations, could offer deeper insights into rivaroxaban's pharmacokinetics and pharmacodynamics in CVT patients.
Conclusion
Our pilot study revealed that the sensitivity of rivaroxaban in Chinese patients might be variable, the dosage lower than that recommended in the instructions might achieve therapeutic plasma anti-Xa levels and decrease bleeding risk. Dynamic plasma anti-Xa monitoring during rivaroxaban use may help to adjust the optimal dose for the patient individually, to achieve safe and effective outcomes.
Supplemental Material
sj-docx-1-cat-10.1177_10760296241286507 - Supplemental material for Anti-Xa Activity Monitoring for Optimizing Rivaroxaban Dosage in Chinese Patients with Cerebral Venous Thrombosis
Supplemental material, sj-docx-1-cat-10.1177_10760296241286507 for Anti-Xa Activity Monitoring for Optimizing Rivaroxaban Dosage in Chinese Patients with Cerebral Venous Thrombosis by Xiaoming Zhang, Meini Gao, Duo Lan, Zhongao Wang, Kun Fang, Xiangyu Ren, Naiqi Liu, Da Zhou, Xunming Ji and Ran Meng in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Abbreviations:
Acknowledgments
We want to thank all colleagues in this study for their cooperation.
Author Contributions
XZ, MG, and DL contributed equally to this work and should be considered co-first authors. XZ, MG, and DL wrote the first draft of the manuscript; KF, XR, NL, and ZW performed the material preparation, data collection, and statistical analysis; DZ, XJ, and RM wrote sections of the manuscript and contributed to manuscript revision; RM takes full responsibility for the data, the analyses and interpretation, and the conduct of the research. All authors read and approved the submitted version.
Data Availability Statement
The data supporting this study's findings are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval and Patient Consent Statement
The study was approved by the Institutional Ethic Committee of Xuanwu Hospital, Capital Medical University, and was conducted according to the guidelines laid down in the Declaration of Helsinki. Prior to any study procedure, written informed consent was obtained from the participants.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China, (grant number 82171297, 82101390).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
