Abstract
The data from 254 patients with severe trauma were retrospectively analyzed. The patients were subdivided into disseminated intravascular coagulation (DIC) and non-DIC. There was a difference in the incidence of the continuous progression from the early to late phase of DIC between the patients with and without DIC on day 0. While 2 of 9 patients who newly developed late-phase DIC were complicated with sepsis, none of the 32 patients who showed a continuous progression of DIC from the early to late phase of trauma developed sepsis. The DIC and Sequential Organ Failure Assessment scores on day 0 were independent factors that predicted the continuous progression of the DIC from the early to late phase of trauma. Trauma itself, but not sepsis, contributes to the continuous progression of DIC from the early to late phase of trauma. The severity of DIC and organ dysfunction are involved in the pathogenesis of this continuous progression.
Keywords
Recently, 2 contradictory theories have emerged regarding the mechanisms of coagulation and fibrinolysis disorders during the early to late phase of trauma. The members of the Educational Initiative on Critical Bleeding in Trauma (EICBT) announced the new concept of acute coagulopathy of trauma shock and coagulopathy of trauma for the coagulation disorders at an early phase of trauma.1,2 They proposed that acute coagulopathy of trauma shock and coagulopathy of trauma are distinct from disseminated intravascular coagulation (DIC) and that there is no evidence to suggest a process of DIC during the course after trauma. Furthermore, they stated that both acute coagulopathy of trauma shock and coagulopathy of trauma terminate at an early phase of trauma and then either sepsis-induced or sepsis-like coagulopathy, but not DIC, develops at the late phase of trauma.1,2
There have been legitimate rebuttals to these concepts.3,4 Ample evidence demonstrates that new term acute coagulopathy of trauma shock and coagulopathy of trauma equals the DIC with fibrinolytic phenotype associated with consumption coagulopathy and excessive fibrinolysis. This type of DIC changes into DIC with the thrombotic phenotype 24 to 48 hours after trauma, leading to multiple organ dysfunction syndrome (MODS).3,4 The trauma itself, but not the sepsis associated with trauma, gives rise to the thrombotic phenotype of DIC at the late phase of trauma. A recent study showed that DIC with fibrinolytic phenotype at an early phase of trauma dependently proceeds to DIC at a late phase of trauma. 5 Although prospective, the major limitation of this study was that only a relatively small number of patients with trauma were investigated.
Therefore, the aim of the present study was to test the hypothesis that DIC at an early phase of trauma continuously proceeds to DIC at a late phase of trauma in a large cohort of patients with trauma.
Materials and Methods
Patient Selection and Data Collection
This was a retrospective study with a prospective data collection. With the approval of the Institutional Review Board, all patients with severe trauma defined with an Injury Severity Score (ISS) > 9 (at least 1 abbreviated injury scale > 3), from June 2000 to December 2009, were enrolled in the study. 6 Trauma patients with cardiac arrest or those resuscitated from cardiac arrest, individuals receiving anticoagulant therapy, and having known clotting disorders such as hematopoietic malignancies and severe liver cirrhosis were excluded. Furthermore, any patients who succumbed within 24 hours after arrival at the emergency department and those who lacked continuous data of global coagulation and fibrinolysis also were excluded. A systematic review of the computer-based medical records of these patients was retrospectively conducted to provide the baseline characteristics and DIC-related variables. The data of these variables were obtained at 6 time points from day 0 to day 5 after the arrival.
Definitions
The severity of illness of the patients was evaluated according to the Acute Physiology and Chronic Health Evaluation (APACHE) II score at the time of enrollment. 7 Organ dysfunction was assessed by the Sequential Organ Failure Assessment (SOFA) score. 8 Multiple organ dysfunction syndrome 8 was defined as an SOFA score > 12. The systemic inflammatory response syndrome (SIRS) was defined according to the American College of Chest Physicians/Society of Critical Care Medicine consensus conference. 9 Sepsis was defined as SIRS induced by infection. 9 The outcome measure was intensive care unit (ICU) mortality.
The DIC diagnosis was performed based on the Japanese Association for Acute Medicine (JAAM) DIC diagnosis criteria. 10 The overt DIC scores by the International Society on Thrombosis and Haemostasis (ISTH) were also calculated. 11 Fibrin/fibrinogen degradation product (FDP) was used as the fibrin-related marker in the ISTH criteria. No increase, moderate increase, and strong increase were defined as FDP of <9, 10 to 24, and >25 mg/L, respectively. The JAAM and ISTH overt DIC was established when the total score was >4 and >5, respectively. While the patients continuously met the JAAM DIC criteria from day 0 to until at least day 3 were defined as early-phase DIC persistently proceeded to DIC at the late phase of trauma, those who did not meet the JAAM DIC criteria on day 0 but met the criteria and maintained it continuously for 3 consecutive days during the period from day 1 to day 5 were defined as newly developed late-phase DIC. Therefore, the early phase means day 0 and the late phase was defined as days 1 to 5. The definition is presented as Figure 1 .

The definition of the early and late phase of DIC. A, The patients continuously met the JAAM DIC criteria from day 0 to until at least day 3 were defined as having early-phase DIC that persistently proceeded to DIC during the late phase of trauma; B, C, D, those who did not meet the JAAM DIC criteria on day 0 but met the criteria and maintained it continuously for 3 consecutive days during the period from day 1 to day 5 were defined as having newly developed late-phase DIC. Therefore, the early phase refers to day 0, while the late phase was defined as days 1 to 5. DIC indicates disseminated intravascular coagulation; JAAM, Japanese Association for Acute Medicine.
According to the EICBT, the term coagulopathy in the present study was defined as coagulation disorder due to “hypocoagulable” or “hypercoagulable” changes in coagulation system caused by miscellaneous factors such as inflammation, dilution, hypothermia, shock, acidemia, increased activated protein C, and sepsis.1,2
Measurement Methods and Treatment
The platelet count, prothrombin time, and fibrinogen were measured using plasma by XE-5000 (Sysmex, Kobe, Japan), Thromborel (Dahede Behring, Tokyo, Japan), and Thrombocheck Fib (L) (Sysmex), respectively. The serum FDP was measured using latex photometric immunoassay FDP (Mitsubishi Chemical Medience, Tokyo, Japan). Platelet concentrates, fresh frozen plasma, and packed red blood cells were promptly transfused based on the repeatedly obtained blood cell counts, coagulation, and fibrinolysis data to maintain the hemodynamics and to treat hemostatic disorders. The standard surgical protocols and ICU managements were appropriately applied to those who required an operation and intensive care.
Statistical Analysis
All measurements are expressed as the mean ± standard deviation. IBM SPSS PASW 18.0 software package for MAC OSX (IBM SPSS Inc, Chicago, Illinois) was used for all statistical calculation analyses. Comparisons between the 2 groups were made using the Mann–Whitney U test and either the chi-square test or Fisher exact test, if necessary. Multiple sets of data were analyzed by the KruskalWallis H test. The relationships between the variables measured and the continuous progression of DIC at an early phase to a late phase of trauma were analyzed by a stepwise logistic regression analysis (backward elimination based on likelihood), and the results were reported as the odds ratios (ORs) and 95% confidence intervals. A multiple regression analysis with stepwise method was also applied to predict DIC at late phase of trauma and the regression line was established. A value of P < .05 was considered statistically significant.
Results
Patient Characteristics
During the study period, 1501 patients with trauma were transported to our department; 254 patients met the inclusion criteria and were subclassified into patients with DIC (129) and non-DIC (125) based on the day 0 data. The basic characteristics of the patients show that the ISS are identical; however, the patients with DIC had a higher prevalence of SIRS and MODS and higher mortality rate than the non-DIC patients (Table 1
). All patients were further subdivided into 4 groups (Table 2
). While 32 early-phase DIC persistently proceed to DIC at the late phase of trauma and 9 newly developed late-phase DIC, 97 early-phase DIC improved and 116 patients could not be diagnosed as having DIC during the study period. Table 3
shows the serial changes in the JAAM DIC scores and the markers of fibrinolysis from day 0 to day 3, which suggests that hyperfibrinolysis observed as markedly increased levels of FDP and
Basic Characteristics and Day 0 Data of the patients
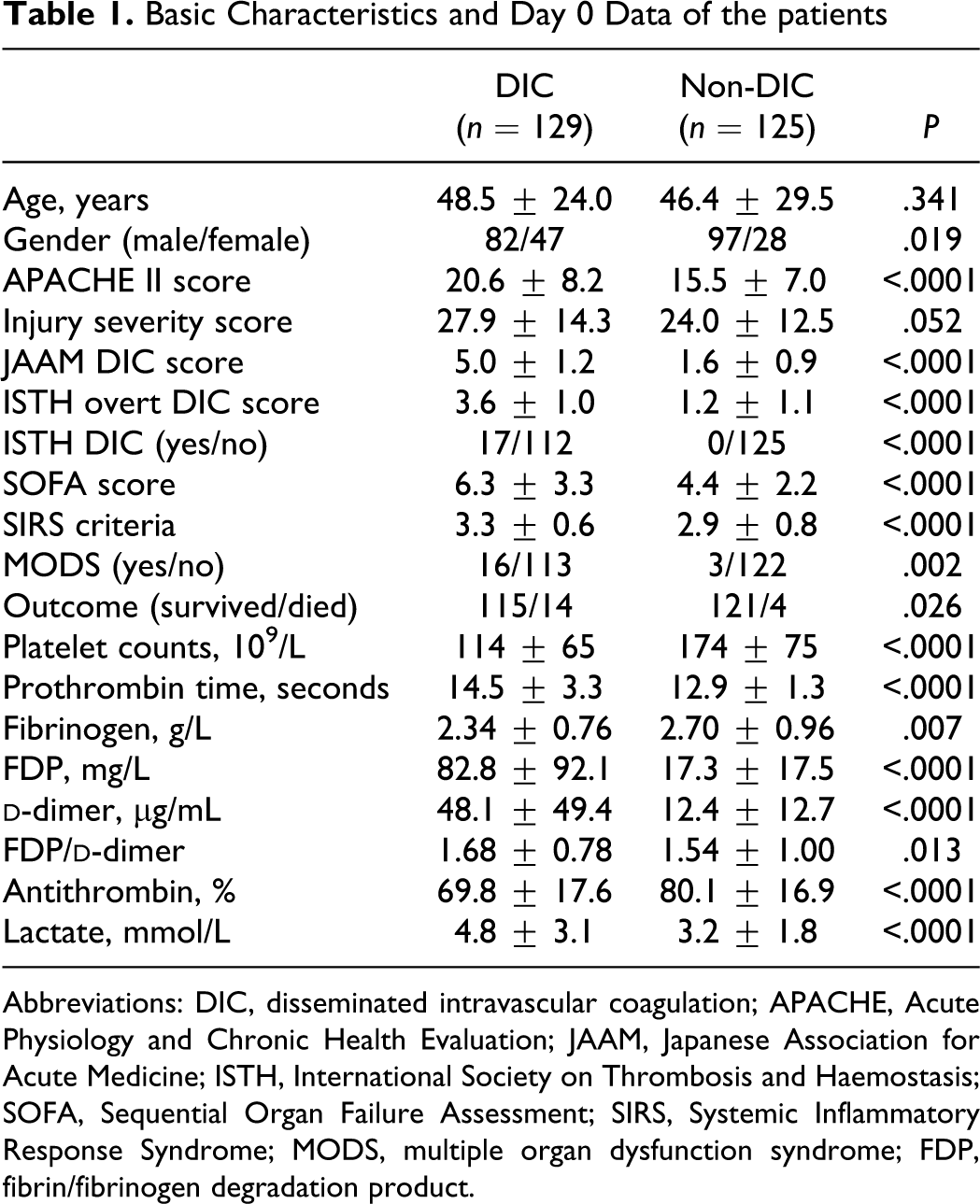
Abbreviations: DIC, disseminated intravascular coagulation; APACHE, Acute Physiology and Chronic Health Evaluation; JAAM, Japanese Association for Acute Medicine; ISTH, International Society on Thrombosis and Haemostasis; SOFA, Sequential Organ Failure Assessment; SIRS, Systemic Inflammatory Response Syndrome; MODS, multiple organ dysfunction syndrome; FDP, fibrin/fibrinogen degradation product.
Data Comparison Among the 4 Groups on Day 0
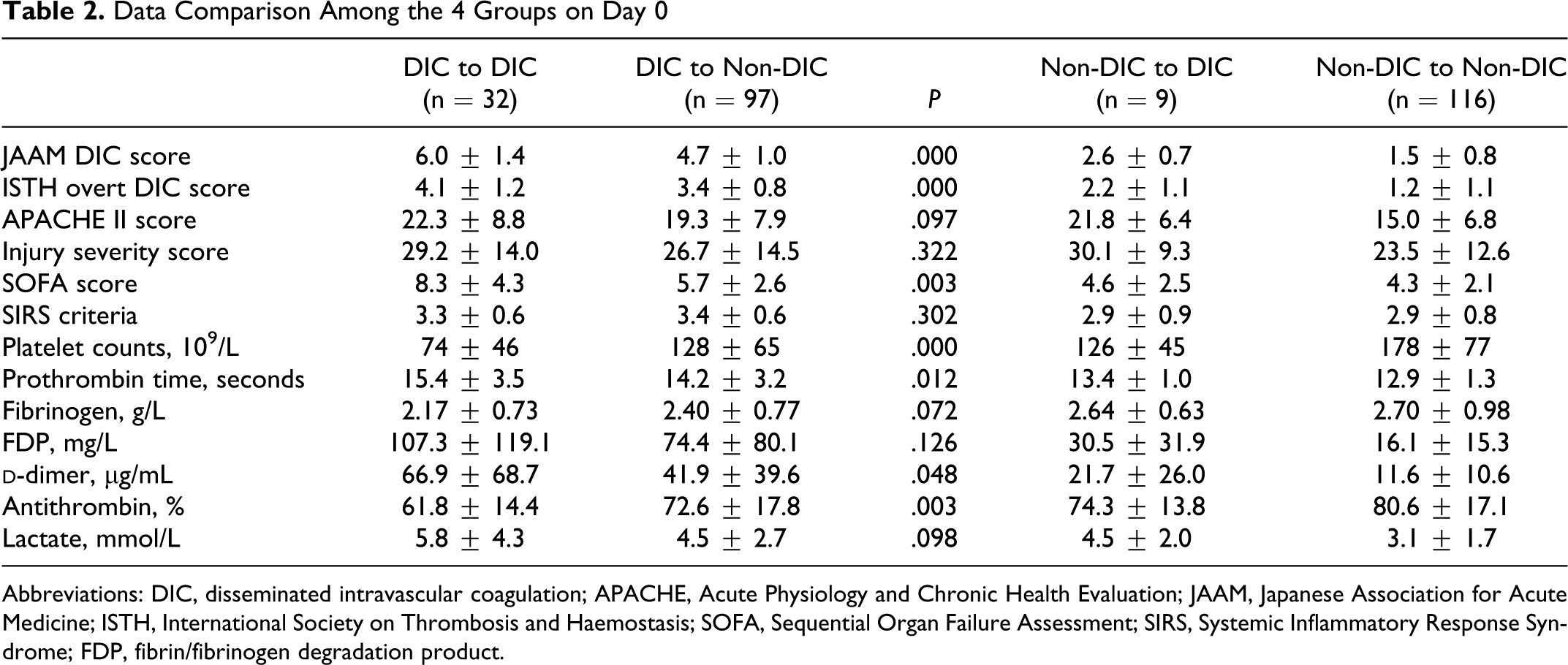
Abbreviations: DIC, disseminated intravascular coagulation; APACHE, Acute Physiology and Chronic Health Evaluation; JAAM, Japanese Association for Acute Medicine; ISTH, International Society on Thrombosis and Haemostasis; SOFA, Sequential Organ Failure Assessment; SIRS, Systemic Inflammatory Response Syndrome; FDP, fibrin/fibrinogen degradation product.
Serial Changes in the JAAM DIC Score, FDP, and d-Dimer in Patients With DIC at an Early Phase of Trauma, Who Thereafter Continuously Proceed to DIC at a Late Phase of Trauma
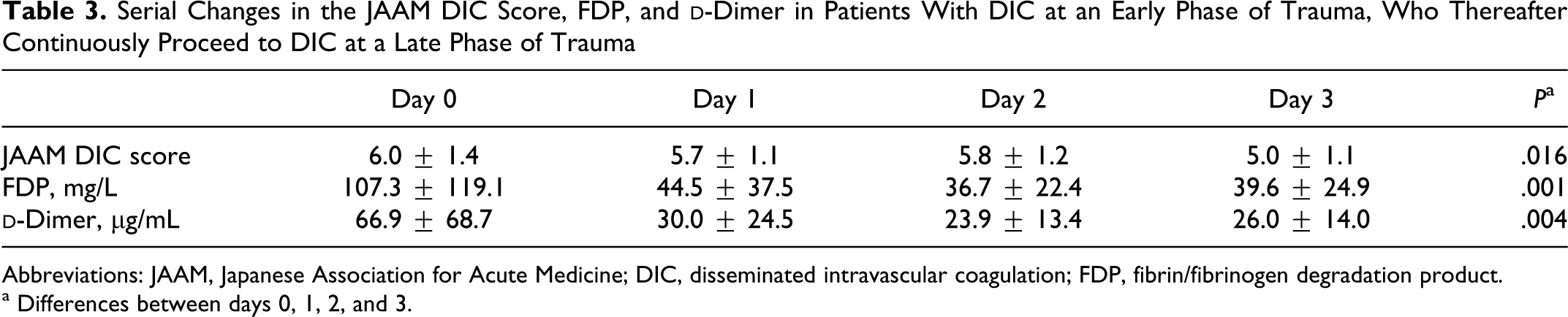
Abbreviations: JAAM, Japanese Association for Acute Medicine; DIC, disseminated intravascular coagulation; FDP, fibrin/fibrinogen degradation product.
a Differences between days 0, 1, 2, and 3.
Prediction of the Proceeding to DIC at a Late Phase of Trauma
Table 4
shows a significant difference in the incidence of late-phase DIC between the patients with (32 of 129, 24.8%) and without (9 of 125, 7.2%) JAAM DIC on day 0 (P < .0001). Almost half of the JAAM DIC patients who simultaneously met the ISTH overt DIC criteria continuously proceeded to DIC at the late phase of trauma (9 of 17, 52.9%). A stepwise logistic regression analysis demonstrated that the JAAM DIC score (P < .0001), SOFA score (P = .001), and
DIC At an Early Phase of Trauma Continues to DIC at Late Phase of Trauma
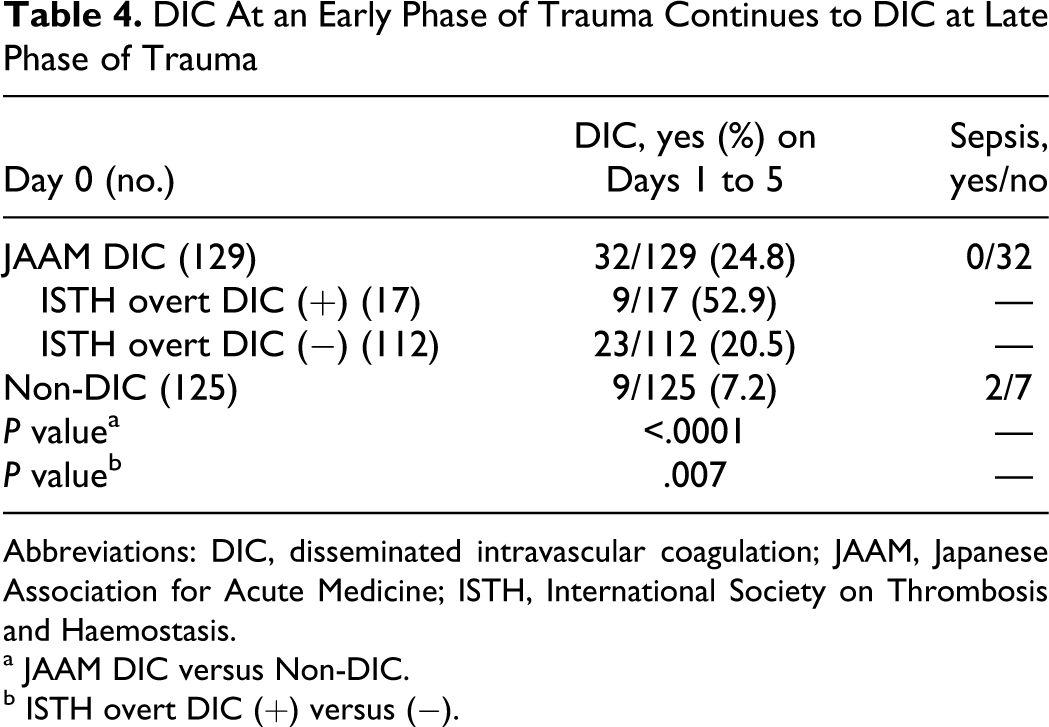
Abbreviations: DIC, disseminated intravascular coagulation; JAAM, Japanese Association for Acute Medicine; ISTH, International Society on Thrombosis and Haemostasis.
a JAAM DIC versus Non-DIC.
b ISTH overt DIC (+) versus (−).
Stepwise Logistic Regression Analysis With Backward Elimination on Likelihood Ratio for the Prediction of the Continuous Progression of DIC on Day 0 to DIC at the Late Phase of Trauma a
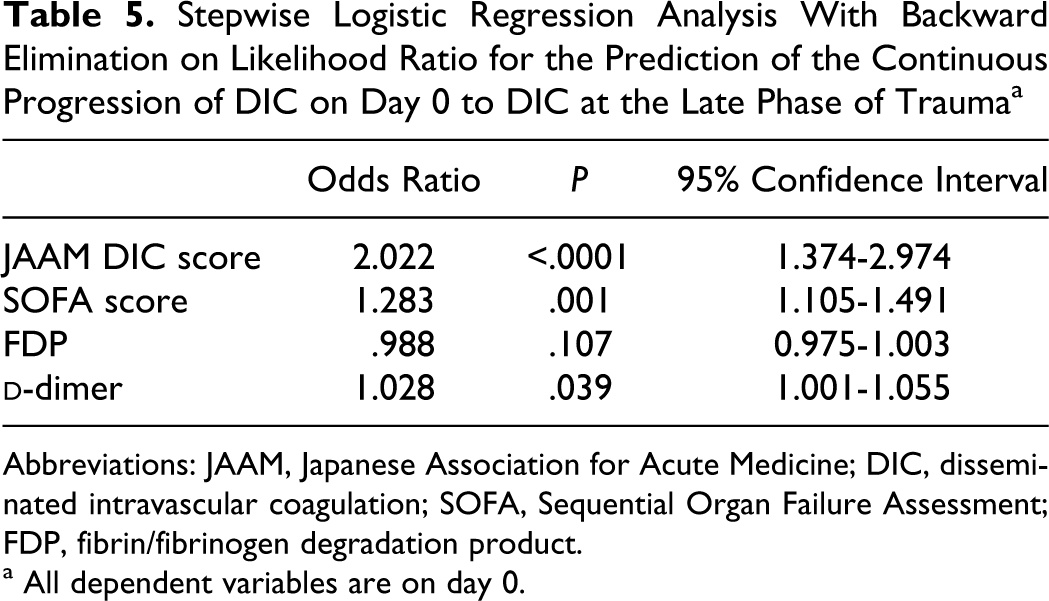
Abbreviations: JAAM, Japanese Association for Acute Medicine; DIC, disseminated intravascular coagulation; SOFA, Sequential Organ Failure Assessment; FDP, fibrin/fibrinogen degradation product.
a All dependent variables are on day 0.
Multiple Regression Analysis With the Stepwise Method for Predicting a Continuous Progression of DIC on Day 0 to DIC at a Late Phase of Trauma
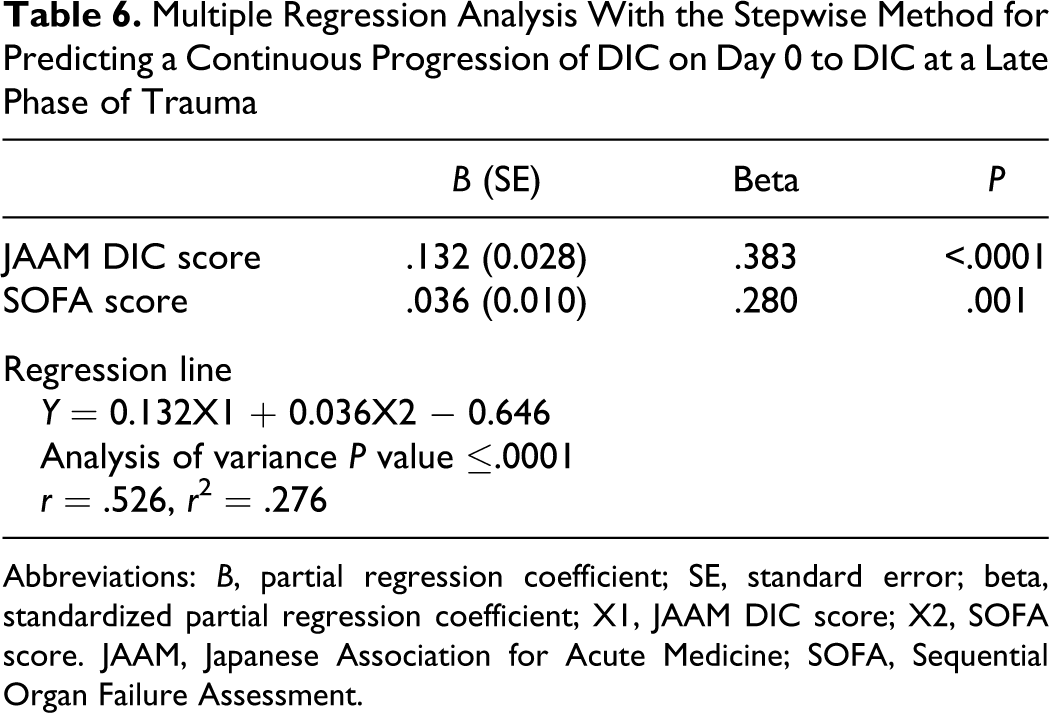
Abbreviations: B, partial regression coefficient; SE, standard error; beta, standardized partial regression coefficient; X1, JAAM DIC score; X2, SOFA score. JAAM, Japanese Association for Acute Medicine; SOFA, Sequential Organ Failure Assessment.
Two patients developed late-phase DIC complicated with sepsis; however, none of the patients that were continuously associated with the DIC from early to late phase of trauma experienced sepsis. Two patients complicated with sepsis died as a result of the sepsis- and DIC-related disorders. The mortality rate of patients continuously associated with DIC from the early to late phase was (5 of 32, 15.6%) and that of patients associated with newly developed DIC was (3 of 9, 33.9%), and no significant difference was observed between these 2 mortality rates (P = .342). The causes of death of the 5 patients with continuous DIC were pulmonary thrombosis in 2, severe head injury, massive bleeding-induced hypovolemic shock, and shock-induced severe ischemic hepatic dysfunction in 1 patient each.
Discussion
While the activation of the extrinsic coagulation pathway at the top of the cascade is the same, DIC is subdivided into fibrinolytic and thrombotic phenotype.12,13 Disseminated intravascular coagulation at an early phase of trauma from 24 to 48 hours after injury belongs to the fibrinolytic phenotype and DIC at a late phase of trauma is a thrombotic phenotype.3,4 The serial measurements of global and molecular markers of coagulation and fibrinolysis have repeatedly revealed that DIC at the early phase of trauma dependently continues to DIC at a late phase of trauma and those results are summarized in a review article of trauma and DIC.3,4 The EICBT correctly pointed out that patients with trauma are initially coagulopathic with increased bleeding but soon change to a hypercoagulable state, which puts them at an increased risk of a thrombotic event.1,2 However, they have never used the term DIC for these 2 coagulopathies. Coagulopathy with increased bleeding was referred to as acute coagulopathy of trauma shock and coagulopathy of trauma and a hypercoagulable state at late stage of trauma were called sepsis-induced or sepsis-like coagulopathy by the EICBT. 2 The present study as well as a recent prospective study leads to the speculation that the EICBT-proposed concepts may be incorrect. 5
The present study demonstrated that approximately 25% of the patients diagnosed by the JAAM DIC on day 0 continuously proceed to DIC at day 1 to day 3 after trauma. This prevalence increased to 52.9% when the patients simultaneously met the ISTH overt DIC criteria. The OR of the JAAM DIC and SOFA scores on day 0 for the prediction of this continuous proceeding of DIC from early to late phase was 2.02 and 1.28, respectively. These results indicate that the more severe the DIC and the organ dysfunction at an early phase of trauma, the greater the number of the JAAM patients with DIC that continuously proceed to the late-phase DIC. Furthermore, the results of the multiple regression analysis suggest that the JAAM DIC and SOFA scores on day 0 can explain about 30% of the continuous progression of the early-phase DIC to the late-phase DIC. The present study found sepsis had no role in the continuous proceeding of DIC at an early phase to the late phase of trauma. These results suggest that trauma itself but not sepsis induces the progression of DIC at an early phase to late phase of trauma.
On the contrary, 2 of 9 newly developed patients with DIC were complicated with sepsis and those 2 patients died. There was no difference in the mortality rate between the 2 types of DIC. These results suggest that sepsis may be one of the causes of newly developed DIC at the late phase, and this type of DIC is equally severe in comparison to trauma-induced DIC at an early phase. Sepsis-induced DIC is another typical type of DIC with a thrombotic phenotype. In this type of DIC, tissue factor-mediated massive and persistent thrombin generation, depression of anticoagulant pathways, and plasminogen activator inhibitor 1 (PAI-1)-mediated inhibition of fibrinolysis caused by inflammatory cytokines lead to disseminated microvascular fibrin thrombosis and organ dysfunction. 14 These findings suggest that irrespective of the causes of DIC, trauma, or sepsis, DIC during the late phase of trauma has an important role in the prognosis of patients with trauma due to the development of MODS.
High FDP and
Previous prospective study showed the incidences of the 2 types of DIC, continuous and newly developed, to be 60.0% and 22.2%, respectively. 5 Both incidences are higher than those of the present study, in which the former was 24.8% and the latter was 7.2%. The previous study intermittently measured parameters for coagulation and fibrinolysis during the study period. Therefore, the reason for the difference in the incidence of DIC between the 2 studies is based on the differences in the measurement method (intermittent or continuous) and the definition of both types of DIC (early and late phase). The data of the present study based on the continuous monitoring, strict definition, and larger patient cohort may represent the more exact incidence.
Limitations
Because this was a retrospective study, prospective validation of the natural history of the DIC after trauma is required. We speculate that the DIC with the fibrinolytic phenotype continuously proceeds to DIC with thrombotic phenotype; however, the present study did not demonstrate direct evidence for the 2 phenotypes.
In conclusion, the present study demonstrated that DIC at an early phase of trauma continuously progresses to DIC at a late phase of trauma without any complication of sepsis. The severity of DIC and the organ dysfunction are the independent predictors of this continuous progression of DIC. According to the findings observed in the present study, sepsis may be involved in the pathogenesis of newly developed DIC in the late phase of trauma.
Footnotes
Acknowledgment
The manuscript was presented at the 6th Congress of Asia Pacific Society on Thrombosis and Hemostasis, Bali, 2010.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
