Abstract
Background
Recently, the effect of Lipoprotein(a) [Lp(a)] on thrombogenesis has aroused great interest, while inflammation has been reported to modify the Lp(a)-associated risks through an unidentified mechanism.
Purpose
This study aimed to evaluate the association between platelet reactivity with Lp(a) and high-sensitivity C-reactive protein (hs-CRP) levels in percutaneous intervention (PCI) patients treated with clopidogrel.
Methods
Data were collected from 10,724 consecutive PCI patients throughout the year 2013 in Fuwai Hospital. High on-treatment platelet reactivity (HTPR) and low on-treatment platelet reactivity (LTPR) were defined as thrombelastography (TEG) maximum amplitude of adenosine diphosphate-induced platelet (MAADP) > 47 mm and < 31 mm, respectively.
Results
6615 patients with TEG results were finally enrolled. The mean age was 58.24 ± 10.28 years and 5131 (77.6%) were male. Multivariable logistic regression showed that taking Lp(a) < 30 mg/dL and hs-CRP < 2 mg/L as the reference, isolated Lp(a) elevation [Lp(a) ≥ 30 mg/dL and hs-CRP < 2 mg/L] was not significantly associated with HTPR (P = 0.153) or LTPR (P = 0.312). However, the joint elevation of Lp(a) and hs-CRP [Lp(a) ≥ 30 mg/dL and hs-CRP ≥ 2 mg/L] exhibited enhanced association with both HTPR (OR:1.976, 95% CI 1.677–2.329) and LTPR (OR:0.533, 95% CI 0.454–0.627).
Conclusions
The isolated elevation of Lp(a) level was not an independent indicator for platelet reactivity, yet the concomitant elevation of Lp(a) and hs-CRP levels was significantly associated with increased platelet reactivity. Whether intensified antiplatelet therapy or anti-inflammatory strategies could mitigate the risks in patients presenting combined Lp(a) and hs-CRP elevation requires future investigation.
Keywords
Introduction
Patients with coronary heart disease (CHD) suffer from residual cardiovascular risks, despite revascularization with percutaneous intervention (PCI) and accepting the guideline-recommended statin and antiplatelet therapy. In the past few years, lipoprotein(a) [Lp(a)], a well-recognized causal indicator for atherosclerosis, has generated heated discussion in the field of coronary heart disease (CHD). 1 Interestingly, recent research has reported that elevated hs-CRP level had an amplifying effect on Lp(a)-associated cardiovascular risks,2,3 though the mechanism was not elucidated.
In CHD patients undergoing PCI treated with clopidogrel, platelet reactivity varies between patients, as the response to antiplatelet medication is affected by multiple clinical factors (including patient characteristics, comorbidities, inflammation, etc), drug-drug interactions, and genetic polymorphisms.4,5 High on-treatment platelet reactivity (HTPR) and low on-treatment platelet reactivity (LTPR) were regarded as effective indicators of haemorrhagic and ischemic risks.6,7 Furthermore, several studies have shown that Lp(a) and high-sensitivity C-reactive protein (hs-CRP) were related to platelet reactivity and thrombogenesis.8,9 Thus, we postulate that there may be a synergic interaction between hs-CRP and Lp(a) in the association with platelet reactivity, further influencing the residual risks in CHD patients.
The primary objective of our study was to evaluate the association of Lp(a) and hs-CRP levels with platelet reactivity in a large-sample cohort of PCI patients treated with clopidogrel. The findings may not only offer insights into the mechanism underlying the adverse prognosis of CHD patients presenting high levels of Lp(a) and hs-CRP, but also provide valuable information for the tailored antithrombotic therapy of high-risk patients.
Methods
Study Population
A total of 10,724 consecutive patients who underwent PCI in Fuwai Hospital from January 2013 to December 2013 were enrolled in this single-centered, large-scale observational study. After excluding 16 patients administrated with other P2Y12 inhibitors instead of clopidogrel, 3940 patients not been examined for thrombelastography (TEG), and 153 patients missing Lp(a) or hs-CRP test results, a dataset of 6615 patients was eventually established (Supplementary Figure 1).
All patients received guideline-recommended antiplatelet therapy under the guidance of their clinicians. For patients without contraindications, aspirin and clopidogrel were administered for ≥ 6 days preoperatively. For those undergoing emergency PCI, a loading dose of aspirin 300 mg and clopidogrel 300–600 mg were given as soon as possible before the procedure. After the procedure, all patients continued dual antiplatelet therapy with aspirin 100 mg/day and clopidogrel 75 mg/day for at least one year.This study was approved by the Institutional Ethics Committee of Fuwai Hospital (approval number: 2013–449), and informed consent was obtained from all participants.
Platelet Function Measurement and Definition
Platelet function was evaluated through adenosine diphosphate (ADP) induced platelet maximum amplitude (MAADP) of TEG platelet mapping assay, which reflected fibrin clot strength under the stimulation of ADP. All patients underwent TEG the morning after PCI, within 12–24 h post-procedure. 2 ml of fasting venous blood samples were collected from patients in a supine position. Platelet function was assessed using the TEG 5000 Thrombelastograph Hemostasis Analyzer system (Haemonetics Corporation, MA, USA). Whole blood specimens were anticoagulated with 3.2% sodium citrate, and all procedures from blood collection to testing were completed within 2 h. Definitions of HTPR and LTPR were in accordance with the widely recognized cut-off points, which were as follows: MAADP > 47 mm as HTPR and MAADP < 31 mm as LTPR.10,11
Laboratory Measurement and Grouping
Venous blood samples were collected within 24 h of admission after overnight fasting. Biomarkers, including glucose, triglyceride (TG), total cholesterol (TC), high-density lipoprotein cholesterol (HDL-C), high-density lipoprotein cholesterol (LDL-C), Lp(a) and hs-CRP were measured using the automatic biochemical analyzer LABOSPECT 008 (Hitachi High-Tech Corporation, Tokyo, Japan). White blood cell count (WBC) and platelet count were measured using the Sysmex XN 2000 automatic blood cell counter (Sysmex Corporation, Kobe, Japan).
In this study, Lp(a) and hs-CRP were examined as categorical variables. The cut-off point of Lp(a) was set to 30 mg/dL, which was more applicable to the Chinese population. 12 Hs-CRP level was classified by 2 mg/L, according to the recommendations of the ACC/AHA guidelines.13,14 Based on the elevation of Lp(a) and hs-CRP levels, participants were categorized into four groups, as follows: Group1, no elevation of Lp(a) or hs-CRP [Lp(a) < 30 mg/dL and hs-CRP < 2 mg/L]; Group 2, isolated elevation of Lp(a) [Lp(a) ≥ 30 mg/dL and hs-CRP < 2 mg/L]; Group 3, isolated isolation of hs-CRP [Lp(a) < 30 mg/dL and hs-CRP ≥ 2 mg/L]; Group 4, elevation of both Lp(a) and hs-CRP [Lp(a) ≥ 30 mg/dL and hs-CRP ≥ 2 mg/L].
Statistical Analysis
Statistical analyses were performed using the SPSS Statistics 28.0 (IBM Corporation, Armonk, NY, USA). Continuous variables were presented as means with standard deviations (SDs) for normally distributed variables and as medians and interquartile ranges (IQRs) for non-normally distributed variables. Categorical variables were expressed as frequencies and percentages. For significance test, ANOVA or Kruskal-Wallis H test was performed for continuous variables, as appropriate, and the Pearson chi-square test or Fisher's exact test was performed for categorical variables. To assess the effect size of differences between mean values, Cohen's d (dCohen) was calculated for normally distributed continuous variables, with the following formula: dCohen = mean difference / pooled standard deviation. The computation was performed using a specialized effect size calculator provided by Lenhard et al 15 Potentially relevant factors of HTPR and LTPR were screened by univariable logistic regression and later included in the multivariable analysis. Multivariable logistic regression was conducted to analyze the association of Lp(a) and hs-CRP with platelet reactivity. Odds Ratios (OR) with 95% Confidence Intervals (CI) were calculated as measure of effect. Subgroup analyses were conducted as a robustness check and the P-values for interaction were assessed. Subgroup populations were stratified according to age (whether ≥ 65 years), diagnosed acute coronary syndrome (ACS) on admission, comorbid diabetes, and prescription of statin. Sensitivity analyses were applied to further assess the impact of systemic inflammation, including ACS and suspected infection (WBC > 10 × 109/L) on the result. For all analyses, P < 0.05 (two-sided) was considered statistically significant.
Results
Baseline Characteristics
Among the 6615 patients finally included in the study, the mean age was 58.24 ± 10.28 years and 5131(77.6%) were male. Based on the value of MAADP, 2023 (30.6%), 2078 (31.4%), and 2514 (38.0%) patients were recognized as presenting HTPR, NTPR, and LTPR, respectively. Baseline characteristics of the patients in the 3 groups, stratified according to platelet reactivity, are listed in Table 1. Higher Lp(a) and hs-CRP levels were observed in the HTPR group, along with higher mean age, a greater proportion of females, higher prevalences of diabetes and ACS, a higher platelet count, and higher levels of glucose, triglycerides, total cholesterol, low-density lipoprotein cholesterol (LDL-C), while the LTPR group exhibited a higher proportion of current smoker, as well as a higher leukocyte count (P < 0.05 for all comparisons). However, it should be noted that the differences in glucose and leukocyte count between the two groups have very small effect sizes, indicating that these differences may not be practically significant (Cohen's d < 0.2).
Baseline Characteristics, Patients Stratified by Platelet Reactivities.
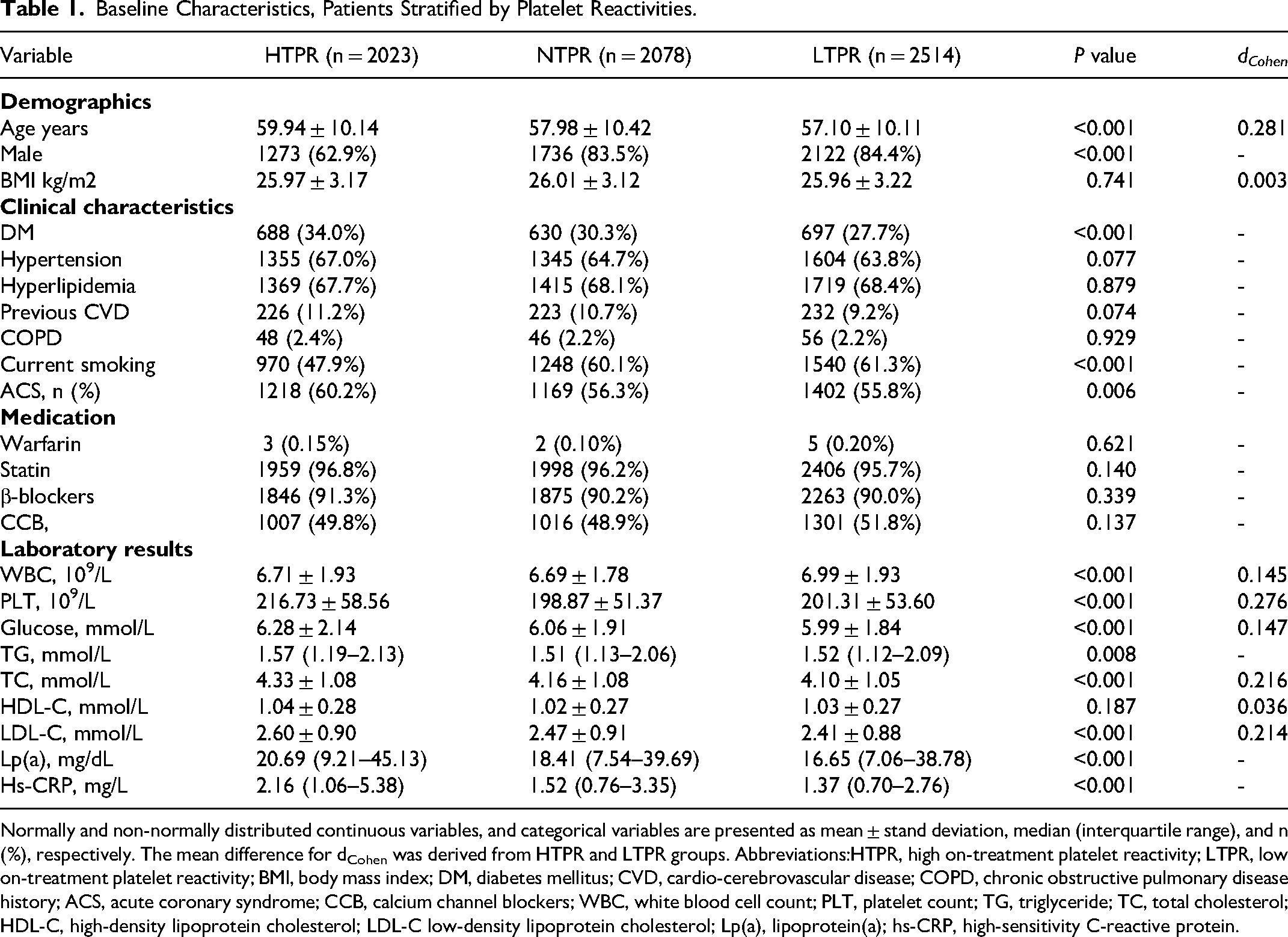
Normally and non-normally distributed continuous variables, and categorical variables are presented as mean ± stand deviation, median (interquartile range), and n (%), respectively. The mean difference for dCohen was derived from HTPR and LTPR groups. Abbreviations:HTPR, high on-treatment platelet reactivity; LTPR, low on-treatment platelet reactivity; BMI, body mass index; DM, diabetes mellitus; CVD, cardio-cerebrovascular disease; COPD, chronic obstructive pulmonary disease history; ACS, acute coronary syndrome; CCB, calcium channel blockers; WBC, white blood cell count; PLT, platelet count; TG, triglyceride; TC, total cholesterol; HDL-C, high-density lipoprotein cholesterol; LDL-C low-density lipoprotein cholesterol; Lp(a), lipoprotein(a); hs-CRP, high-sensitivity C-reactive protein.
Lp(a) and Hs-CRP Levels in Different Groups
In the total study population, the levels of Lp(a) and hs-CRP were skewed distributed, with median values of 18.32 (IQR 7.81–41.21) mg/dL and 1.61 (IQR 0.80–3.61) mg/L, respectively. Notably, no patients had an hs-CRP level exceeding 20 mg/L. When comparing patients in the 3 groups of varied platelet reactivity, those presenting HTPR demonstrated the highest median levels of Lp(a) (20.69 mg/dL) and hs-CRP (2.16 mg/L). On the contrary, patients with LTPR exhibited the lowest levels of Lp(a) (16.65 mg/dL) and hs-CRP (1.37 mg/L) (P < 0.001 for all comparisons). Based on the levels of Lp(a) and hs-CRP, the study population was further categorized into four groups. The baseline characteristics of patients in different groups were described in Supplementary Table 1.
The Association Between Lp(a) and hs-CRP Levels with HTPR and LTPR
When HTPR was the dependent variable (Table 2), multivariable logistic regression analysis was adjusted with age, sex, diabetes, hypertension, current smoking, leukocyte count, platelet count, glucose, total cholesterol, and LDL-C (all P < 0.05 in univariable analysis) revealed that, compared with the reference (Group 1), the isolated elevation of Lp(a) (Group 2) exhibited no significant association with HTPR (P = 0.166). Conversely, isolated hs-CRP elevation (Group 3) was significantly associated with an increased risk of HTPR (OR: 1.881, 95% CI 1.630–2.171, P < 0.001). In addition, the combined elevation of hs-CRP and Lp(a) (Group 4) presented an enhanced association with HTPR, with the odds increased by 1.985 folds (OR: 1.985, 95% CI 1.684–2.340, P < 0.001).
Results of Logistic Regression Analysis for HTPR.
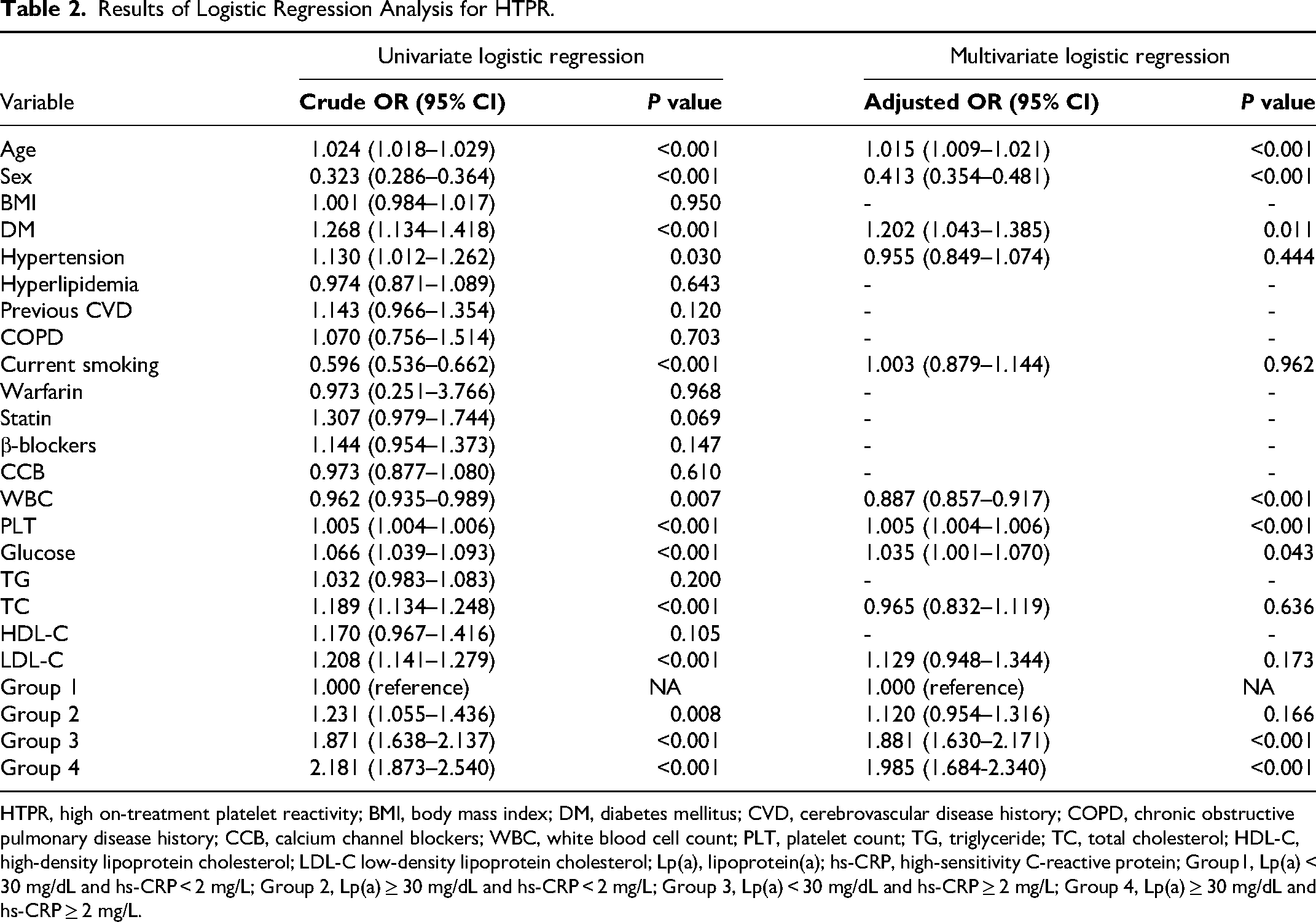
HTPR, high on-treatment platelet reactivity; BMI, body mass index; DM, diabetes mellitus; CVD, cerebrovascular disease history; COPD, chronic obstructive pulmonary disease history; CCB, calcium channel blockers; WBC, white blood cell count; PLT, platelet count; TG, triglyceride; TC, total cholesterol; HDL-C, high-density lipoprotein cholesterol; LDL-C low-density lipoprotein cholesterol; Lp(a), lipoprotein(a); hs-CRP, high-sensitivity C-reactive protein; Group1, Lp(a) < 30 mg/dL and hs-CRP < 2 mg/L; Group 2, Lp(a) ≥ 30 mg/dL and hs-CRP < 2 mg/L; Group 3, Lp(a) < 30 mg/dL and hs-CRP ≥ 2 mg/L; Group 4, Lp(a) ≥ 30 mg/dL and hs-CRP ≥ 2 mg/L.
Results were comparable when taking LTPR as the dependent variable (Table 3). Multivariable logistic regression was adjusted with age, sex, diabetes, history of cardiovascular diseases (CVDs), current smoking, leukocyte count, platelet count, glucose, total cholesterol, and LDL-C (P all < 0.05 in univariable analysis). Without the elevation of hs-CRP level, elevated Lp(a) (Group 2) was not an independent predictor for LTPR (P = 0.312) either. Nevertheless, isolated hs-CRP elevation (Group 3) was significantly associated with a decreased risk of LTPR (OR: 0.583, 95% CI 0.509–0.667, P < 0.001). Moreover, when there was a concomitant elevation of both markers (Group 4), the risk further decreased to 0.533 folds (OR: 0.533, 95% CI 0.454–0.627, P < 0.001).
Results of Logistic Regression Analysis for LTPR.
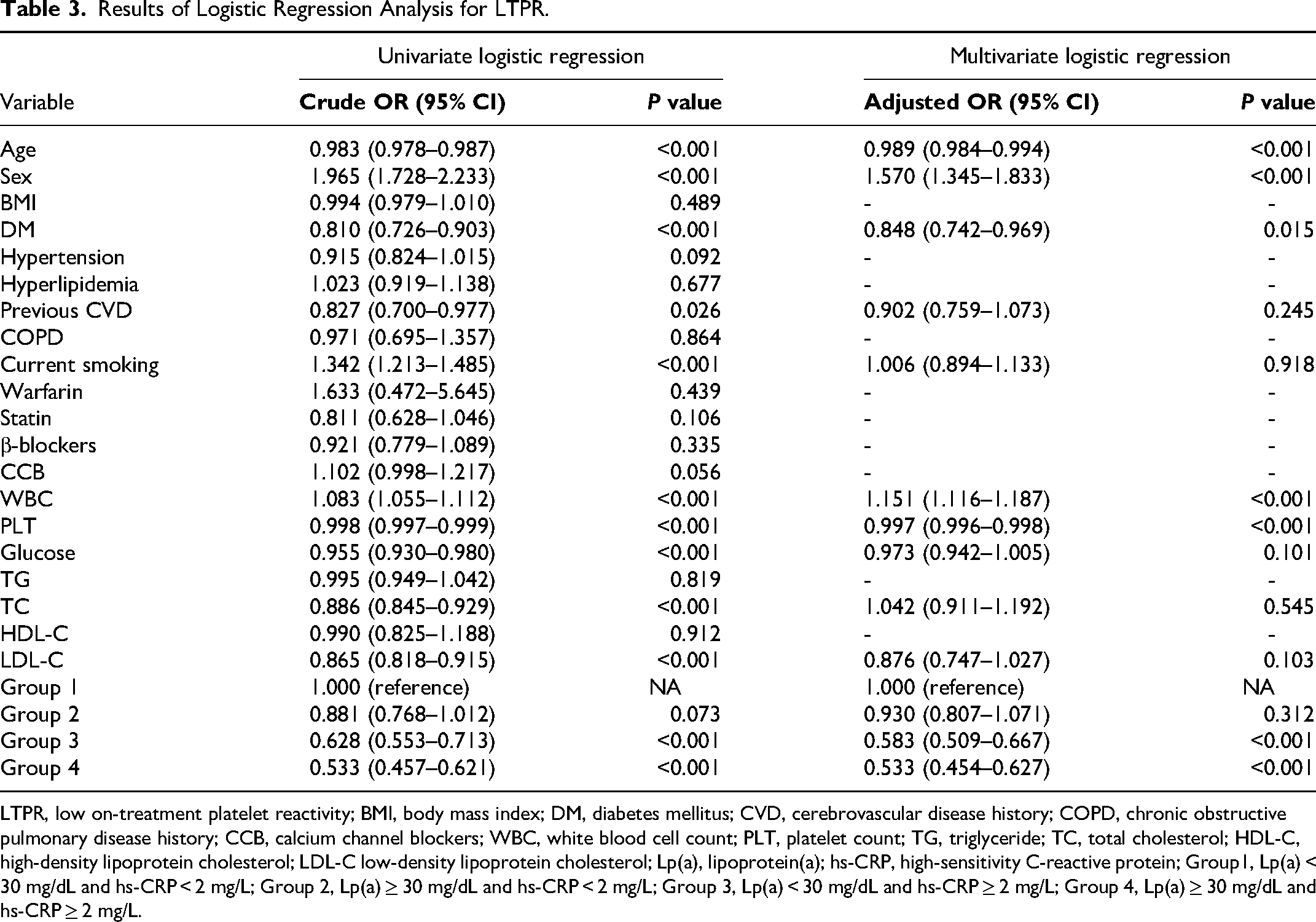
LTPR, low on-treatment platelet reactivity; BMI, body mass index; DM, diabetes mellitus; CVD, cerebrovascular disease history; COPD, chronic obstructive pulmonary disease history; CCB, calcium channel blockers; WBC, white blood cell count; PLT, platelet count; TG, triglyceride; TC, total cholesterol; HDL-C, high-density lipoprotein cholesterol; LDL-C low-density lipoprotein cholesterol; Lp(a), lipoprotein(a); hs-CRP, high-sensitivity C-reactive protein; Group1, Lp(a) < 30 mg/dL and hs-CRP < 2 mg/L; Group 2, Lp(a) ≥ 30 mg/dL and hs-CRP < 2 mg/L; Group 3, Lp(a) < 30 mg/dL and hs-CRP ≥ 2 mg/L; Group 4, Lp(a) ≥ 30 mg/dL and hs-CRP ≥ 2 mg/L.
Sensitivity Analysis
When examining the associations between the levels of Lp(a) and hs-CRP with platelet reactivity across subgroup, robust results were observed (Figure 1 and Supplementary Figure 2), as there were no significant interactions with age, ACS at admission, history of diabetes, and lipid management through statin prescription (all P-value for interaction > 0.05). In addition, the results remained consitent in further sensitivity analyses when adjusting ACS as a confounder and excluding patients with WBC > 10 × 109/L (Supplementary Table 2).
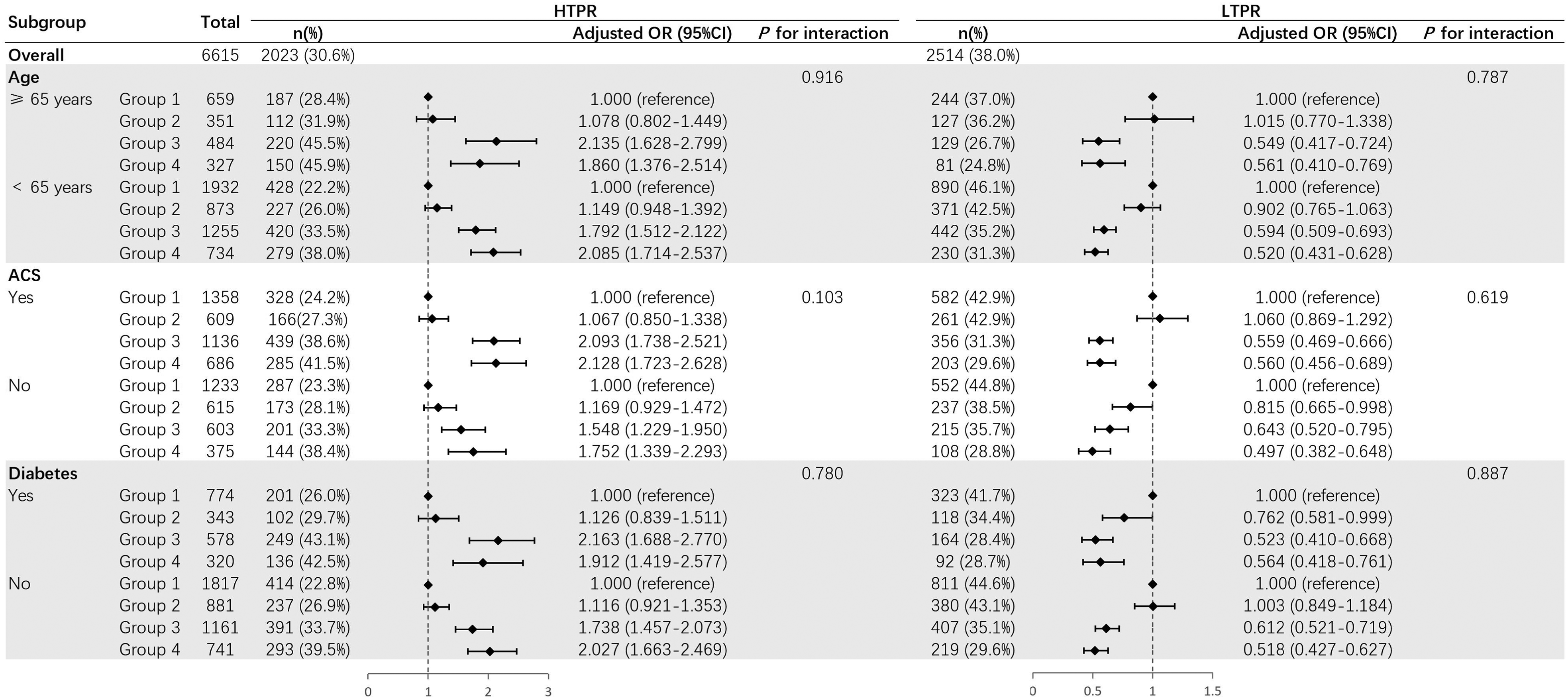
Subgroup analysis. HTPR, high on-treatment platelet reactivity; LTPR, low on-treatment platelet reactivity; ACS, acute coronary syndrome; DM, diabetes mellitus; Group 1, Lp(a) < 30 mg/dL and hs-CRP < 2 mg/L; Group 2, Lp(a) ≥ 30 mg/dL and hs-CRP < 2 mg/L; Group 3, Lp(a) < 30 mg/dL and hs-CRP ≥ 2 mg/L; Group 4, Lp(a) ≥ 30 mg/dL and hs-CRP ≥ 2 mg/L.
Discussion
This prospective, large-scale real-world study was the first to report that, among PCI patients treated with clopidogrel, Lp(a) influences platelet reactivity only in the presence of elevated hs-CRP levels (≥ 2 mg/L), and there is a significant enhancement in platelet reactivity when both Lp(a) and hs-CRP levels are elevated: (1) Elevated hs-CRP level (hs-CRP ≥ 2 mg/L) is an independent risk factor for HTPR, as well as an independent protect factor for LTPR; (2) Without the simultaneous elevation of hs-CRP, isolated elevation of Lp(a) level [Lp(a) ≥ 30 mg/dL] is not a significant marker for HTPR or LTPR; (3) Concomitant elevation of Lp(a) and hs-CRP levels is significantly associated with an increased risk of HTPR and a decreased risk of LTPR, videlicet heightened platelet reactivity.
It is well-established that Lp(a) is a causal risk factor for atherosclerotic cardiovascular diseases, independent of LDL-C. 16 Multiple studies including a Mendelian randomization analysis have indicated that elevated Lp(a) level is associated with cardiovascular risks regardless the level of LDL-C.17,18 The Lp(a)-related risks increase in a linear manner as plasma Lp(a) level elevates to >30 mg/dl. 19 In recent days, the interaction between inflammation and Lp(a)-associated risks has aroused widespread attention. Lp(a) may enhance inflammation via the bounded oxidized phospholipids (OxPL), 20 while inflammatory conditions could refine the Lp(a)-associated cardiovascular risks. In the prespecified secondary analysis of the ACCELERATE trial, 21 increased Lp(a) level was significantly associated with major adverse cardiovascular events only when hs-CRP level was 2 mg/L or higher. 2 Besides, the study of Zhang et al included 4600 participants from the Multi-Ethnic Study of Atherosclerosis and discovered a strong interaction between Lp(a) and hs-CRP in residual cardiovascular risk 3 : The Lp(a)-related CVD events were only observed with the joint elevation of hs-CRP; Higher incidence of CVD events and all-cause death were observed in patients with combined elevation of both markers, despite ethnicity. In a previous study conducted by our team on the same study population, we also identified a similar interaction between Lp(a) and hs-CRP regarding the risk of major adverse cardiovascular and cerebrovascular events (MACCE). 22 Specifically, when the hs-CRP level was ≥ 2 mg/L, Lp(a) levels ≥ 30 mg/dL were significantly associated with MACCE (adjusted HR: 1.20; 95% CI: 1.04–1.39; p < 0.05). In contrast, no such association was observed when the hs-CRP level was < 2 mg/L. The results of our current study are in line with these previous findings, indicating that Lp(a) does not significantly contribute to on-treatment platelet reactivity or the related thrombotic risk unless hs-CRP level is concomitantly elevated. This effect between hs-CRP and Lp(a) on platelet reactivity may be a potential mechanism underlying the phenomenon of the greatest residual risk in patients with dual elevation of Lp (a) and hs-CRP.
To the best of our knowledge, variability of platelet response to antiplatelet therapy in CHD patients is correlated to adverse cardiac and cerebrovascular events. HTPR is a strong predictor for thrombotic events and all-cause mortality, whereas LTPR is associated with an increased risk of bleeding.7,23 A study enrolling 20,839 patients (97% treated with clopidogrel) reported that compared with patients displaying optimal platelet reactivity, patients with HTPR had an approximately 2.7-fold higher risk of stent thrombosis, while patients with LTPR had an approximately 1.7-fold higher risk of major bleeding. 24 According to the expert consensus on platelet function test, MAADP by TEG platelet mapping assay is generally advised for the definition of HTPR and LTPR, 11 as MAADP demonstrated strong prognostic value of in predicting long-term thrombotic and bleeding events.25,26 It is acknowledged that various factors, including genetic polymorphisms, comorbidities, and levels of biomarkers may cast influence on platelet reactivity. Our research identified older age, female sex, comorbid diabetes, and higher platelet count as risk factors associated with HTPR, while leukocyte count was associated with LTPR. Age, sex, and diabetes are well-established contributors to platelet reactivity. 10 Previous studies have also noetd that lower leukocyte count, 9 and higher platelet count 27 correlated with increased platelet reactivity. Our findings align with the exsiting research; thus, we will not delve into those aspects further. Instead, we focused on exploring the role of Lp(a) and hs-CRP in this context. Recent evidence has suggested that Lp(a) enhances platelet activation and aggregation, which consequently promotes thrombosis. A prospective study by Zhu et al reported that patients with elevated Lp(a) levels showed more active platelet response to ADP. 8 Another study by Liu et al also. found that Lp(a) was positively correlated to arachidonic acid (AA)-induced platelet aggregation. 28 On the other hand, hs-CRP was elucidated to be independently associated with platelet reactivity. In our previous research, patients with higher hs-CRP displayed an approximately 1.1-fold increased risk of HTPR. 9 Notably, prior studies focused on the individual impact of Lp(a) and hs-CRP on platelet reactivity, while our current study first investigated the interaction of these biomarkers and revealed the heightened platelet reactivity towards clopidogrel treatment among patients exhibiting conjoint elevation of Lp(a) and hs-CRP levels, which indicates increased susceptibility to thrombosis events. These findings may shed light on the mechanism of interaction between inflammation, measured by hs-CRP, and Lp(a) in platelet aggregation, subsequent thrombotic processes, and adverse cardiovascular events.
Tailored treatment that optimizes the delicate balance between thrombotic and hemorrhagic risks has always been a research hotspot in the field of antithrombosis. Our study may offer new insights into the identification of patients at higher risk of low response to clopidogrel and the subsequent adverse cardiovascular event. Considering the concomitant elevation of Lp(a) and hs-CRP as a potential indicator for the risk of thrombotic events, it is reasonable for future research to delve deeper into the effectiveness and safety of intensified antithrombotic therapy in these patients, such as switching clopidogrel to a more potent P2Y12 inhibitor, or prolonging DAPT treatment, etc Besides, treatments targeting inflammation and Lp(a) may confer additional benefits. There are several Lp(a)-lowering medications, although their efficacy in the improvement of cardiovascular outcomes is yet lack of evidence, for instance, lepodisiran, niacin, 29 pelacarsen,30,31 mipomersen, 32 anacetrapib, 33 PCSK9 inhibitor, 34 etc In terms of anti-inflammatory therapies, agents targeting NLRP3 inflammasome such as colchicine has been validated to be effective in reducing cardiovascular events.35–37 In conclusion, for patients presenting conjoint elevation of Lp(a) and hs-CRP levels, the reasonableness of intensive antiplatelet therapy, and the potential enhancement of clinical outcomes through therapeutic adjustments of anti-inflammatory and lp-(a)-lowering strategies, need to be determined in future investigations.
Limitations
There are several limitations in our study. First, it was a single-center study. Subjects were mostly of the same race and from neighboring regions. Second, as an observational study, there were potentially uncontrolled confounders. Third, this study exclusively focused on the on-treatment platelet reactivity of clopidogrel. Whether the observed effects would alter when treated with other P2Y12 inhibitors was beyond the scope of this study. Fourth, the platelet function determination was according to the MAADP value of TEG. Although this is an evidence-based and acknowledged method, variations in the definition of platelet reactivity across different assays may exert a certain influence on the results. Moreover, baseline data on clopidogrel administration prior to admission were not recorded. Therefore, this variable was not assessed as a confounding factor in the analysis. These limitations should be further improved in future studies.
Conclusions
This prospective, large-scale real-world study revealed that, in patients undergoing PCI treated with clopidogrel, elevation of Lp(a) level was not independently correlated to platelet reactivity when hs-CRP < 2 mg/L, yet concomitant elevation of Lp(a) and hs-CRP levels was significantly associated with higher platelet reactivity. Therefore, clinicians should pay more attention to patients exhibiting elevated levels of both Lp(a) and hs-CRP. Further research is needed to determine the potential efficacy of intensive antiplatelet therapy in mitigating thrombotic events among these patients.
Supplemental Material
sj-docx-1-cat-10.1177_10760296241280711 - Supplemental material for Elevated High-Sensitivity C-Reactive Protein Level Enhances the Impact of Lipoprotein(a) on Platelet Reactivity in PCI Patients Treated with Clopidogrel
Supplemental material, sj-docx-1-cat-10.1177_10760296241280711 for Elevated High-Sensitivity C-Reactive Protein Level Enhances the Impact of Lipoprotein(a) on Platelet Reactivity in PCI Patients Treated with Clopidogrel by Kexin Zhang, Jiawen Li, Yulong Li, Kailun Yan, Pei Zhu, Xiaofang Tang, Deshan Yuan, Yuejin Yang, Runlin Gao, Jinqing Yuan and Xueyan Zhao in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Author Contributions
Kexin Zhang conceived the concept of this study, analyzed data, and drafted the manuscript. Jiawen Li, Yulong Li, Kailun Yan, Pei Zhu, Xiaofang Tang, and Deshan Yuan contributed to the acquisition, analysis, or interpretation of data. Xueyan Zhao, Jinqing Yuan, Yuejin Yang, and Runlin Gao developed the idea and revised the manuscript. All authors have approved the manuscript for submission.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The research was funded by the National Clinical Research Center for Cardiovascular Diseases, Fuwai Hospital, Chinese Academy of Medical Sciences [grant number NCRC2020013]; the National High Level Hospital Clinical Research Funding [grant number 2023-GSP-GG-40]; the CS Optimizing Antithrombotic Research Fund [grant number BJUHFCSOARF201801–06]; and the CAMS Innovation Fund for Medical Sciences (CIFMS) [grant number 2023-I2M-1–002].
the National Clinical Research Center for Cardiovascular Diseases, Fuwai Hospital, Chinese Academy of Medical Sciences, the National High Level Hospital Clinical Research Funding, the CAMS Innovation Fund for Medical Sciences (CIFMS), the CS Optimizing Antithrombotic Research Fund, (grant number NCRC2020013, 2023-GSP-GG-40, 2023-I2M-1-002, BJUHFCSOARF201801-06).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
