Abstract
Background
Atrial fibrillation (AF) is prevalent among patients with end-stage kidney disease (ESKD) undergoing dialysis, and both conditions are associated with a heightened risk of cardiovascular diseases. Anticoagulation is essential for preventing thromboembolic complications in these patients. This study aimed to evaluate the effects of factor Xa inhibitors compared to vitamin K antagonists (VKAs) for AF patients on dialysis.
Methods
A comprehensive search of PubMed and Embase databases was conducted to identify relevant studies published up to June 2024. Eligible studies compared factor Xa inhibitors (rivaroxaban, apixaban, edoxaban) with VKAs in AF patients on dialysis, with primary outcomes of stroke or systemic embolism(SSE) and major bleeding.
Results
A total of 7 studies (3 randomized controlled trials and 4 observational cohorts) were included. For the RCTs, the use of factor Xa inhibitors was associated with a reduced risk of SSE compared to VKAs (odds ratio [OR] = 0.37, 95% confidence interval [CI]:0.15–0.93). There was no significant difference in the risk of major bleeding events between the two groups (OR = 0.65, 95%CI:0.32–1.33). Observational cohort studies yielded similar results with a decreased risk of SSE (hazard ratio [HR] = 0.74, 95%CI:0.57–0.96) and no significant difference in major bleeding (HR = 0.87, 95%CI:0.62–1.22). No differences in treatment effect between apixaban and rivaroxaban were observed for efficacy (p-interaction = 0.44) and safety (p-interaction = 0.21) outcomes.
Conclusion
Factor Xa inhibitors, particularly apixaban and rivaroxaban, were associated with a lower risk of SEE without an increase in major bleeding, which might be convenient alternatives to VKAs in managing AF in patients with ESKD on dialysis.
Introduction
Atrial fibrillation (AF) is a prevalent cardiac arrhythmia in patients with end-stage kidney disease (ESKD), particularly those undergoing dialysis. The coexistence of AF and ESKD amplifies the risk of cardiovascular diseases, including stroke. Anticoagulant therapy is crucial for reducing the risk of thromboembolic events in these patients. Vitamin K antagonists (VKAs), such as warfarin, have been the cornerstone of anticoagulation in AF patients with ESKD on dialysis. However, concerns about the increased bleeding risk and vascular calcification associated with VKAs have led to the exploration of alternative anticoagulants. In addition, warfarin has been linked not only to an elevated risk of bleeding and vascular calcification but also to an accelerated decline in renal function. In contrast, direct oral anticoagulants (DOACs) have demonstrated an association with a slower progression of kidney function deterioration, 1 suggesting a potentially more favorable impact on the long-term renal health of patients.
DOACs, including dabigatran, rivaroxaban, apixaban, and edoxaban, have emerged as potential alternatives to VKAs, offering advantages in terms of efficacy, safety, and convenience. 2 Despite the growing body of evidence supporting the use of DOACs in AF patients, the majority of phase III pivotal randomized controlled trials (RCTs)3–6 have excluded patients on dialysis, leading to uncertainty regarding their optimal use in this specific population. Given the impaired kidney function in dialysis patients with AF, which affects the clearance and metabolism of dabigatran, its use may not be suitable for this specific group of patients. Consequently, factor Xa inhibitors (rivaroxaban, apixaban, edoxaban), which have distinct pharmacokinetic profiles and less reliance on renal excretion, are being investigated as potentially more appropriate anticoagulants for AF patients undergoing dialysis.
Recent RCTs7–9 and observational studies10–13 have focused on the therapeutic efficacy and prognosis of factor Xa inhibitors in AF patients with ESKD on dialysis. Therefore, our current systematic review and meta-analysis aimed to synthesize the findings of recent RCTs and observational studies to provide a comprehensive overview of the therapeutic effects of factor Xa inhibitors compared with VKAs for AF patients with ESKD on dialysis.
Methods
Eligibility Criteria
This systematic review and meta-analysis was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. We included studies that met the following PICOS criteria:
Population: patients with ESKD undergoing dialysis (hemodialysis or peritoneal dialysis) with a diagnosis of AF. Intervention: treatment with factor Xa inhibitors, specifically rivaroxaban, apixaban, or edoxaban. Comparison: any form of VKAs, such as warfarin. Outcomes: the primary efficacy outcome was the incidence of stroke or systemic embolism, whereas the primary safety outcome was the occurrence of major bleeding events. Study design: RCTs and observational cohort studies were considered for this analysis. In the case of observational cohorts, the effect sizes were reported using either the propensity score method or the Cox model, which help in adjusting for potential confounding factors.
We excluded single-arm studies that solely focused on DOACs, as they did not offer a comparative analysis between DOACs and VKAs. Furthermore, studies lacking any data on the efficacy or safety outcomes pertinent to our research were also excluded. When encountering duplicated studies, we selected those with extended follow-up periods or larger sample sizes for inclusion in our analysis.
Data Sources and Search Strategy
A comprehensive literature search was performed in PubMed and Embase databases covering the period up to June 2024. The search strategy included the keywords related to AF, ESKD, dialysis, factor Xa inhibitors, and VKAs. Supplemental Table 1 shows the search strategy of this meta-analsyis. Additionally, we conducted a manual review of the reference lists from previous review articles to identify any potential studies that may have been overlooked in our database search.
Study Selection and Data Collection
Upon completion of the search, two independent reviewers examined the titles and abstracts of the identified studies. Applying our pre-established exclusion criteria, we eliminated studies that were classified as animal experiments, case reports, review articles, meta-analyses, editorial comments, conference abstracts, or any other documents deemed irrelevant to our research focus. Following this initial triage, we advanced to the full-text screening phase to confirm which studies fulfilled our eligibility criteria. A third reviewer was involved in case of any disagreements.
For each included study, the following data were extracted: first author, publication year, study design, country, sample size, demographics of the participants, duration of follow-up, type of factor Xa inhibitor and VKA used, and outcomes of interest. Any discrepancies were resolved by consensus.
Risk of Bias Assessment
We utilized the Risk of Bias 2 tool (RoB 2), 14 as endorsed by the Cochrane Collaboration, to evaluate the risk of bias in the included RCTs. Furthermore, for the observational studies, we conducted a risk of bias assessment using the ROBINS-I tool. 15 The outcomes of these assessments were visualized and presented through the robvis tool (https://mcguinlu.shinyapps.io/robvis/).
Quality of Evidence
We employed the Grading of Recommendations, Assessment, Development, and Evaluation (GRADE) system 16 to rate the quality of evidence for the studied outcomes. We adhered to the GRADE handbook for conducting the quality assessment. Utilizing the GRADEpro Guideline Development Tool (GRADEpro GDT: McMaster University and Evidence Prime, 2024. Available from gradepro.org), we generated the results. All evaluations were independently performed by two investigators, with consensus achieved through discussion.
Statistical Analysis and Data Synthesis
The I2 statistic was used to quantify the degree of heterogeneity, with an I2 value of 50% or more indicating substantial heterogeneity. Odds ratios (ORs) with 95% confidence intervals (CIs) were calculated for dichotomous data from RCTs, while adjusted hazard ratios (HRs) with 95% CIs were used for observational studies. The primary analysis was conducted using a random-effects model to account for the expected heterogeneity between studies. Subgroup analyses were conducted based on the type of DOACs (apixaban vs rivaroxaban) to explore potential differences in efficacy and safety. To explore potential differences in efficacy and safety using the p-interaction test, which helps to determine if there is a difference in treatment effects between apixaban versus rivaroxaban. According to the Cochrane book, the publication bias analysis was not assessed due to the limited number of included studies. Data analysis was performed using Review Manager (RevMan) version 5.4 software provided by the Cochrane Collaboration.
Results
Study Selection and Characteristics
The PRISMA flow diagram shown in
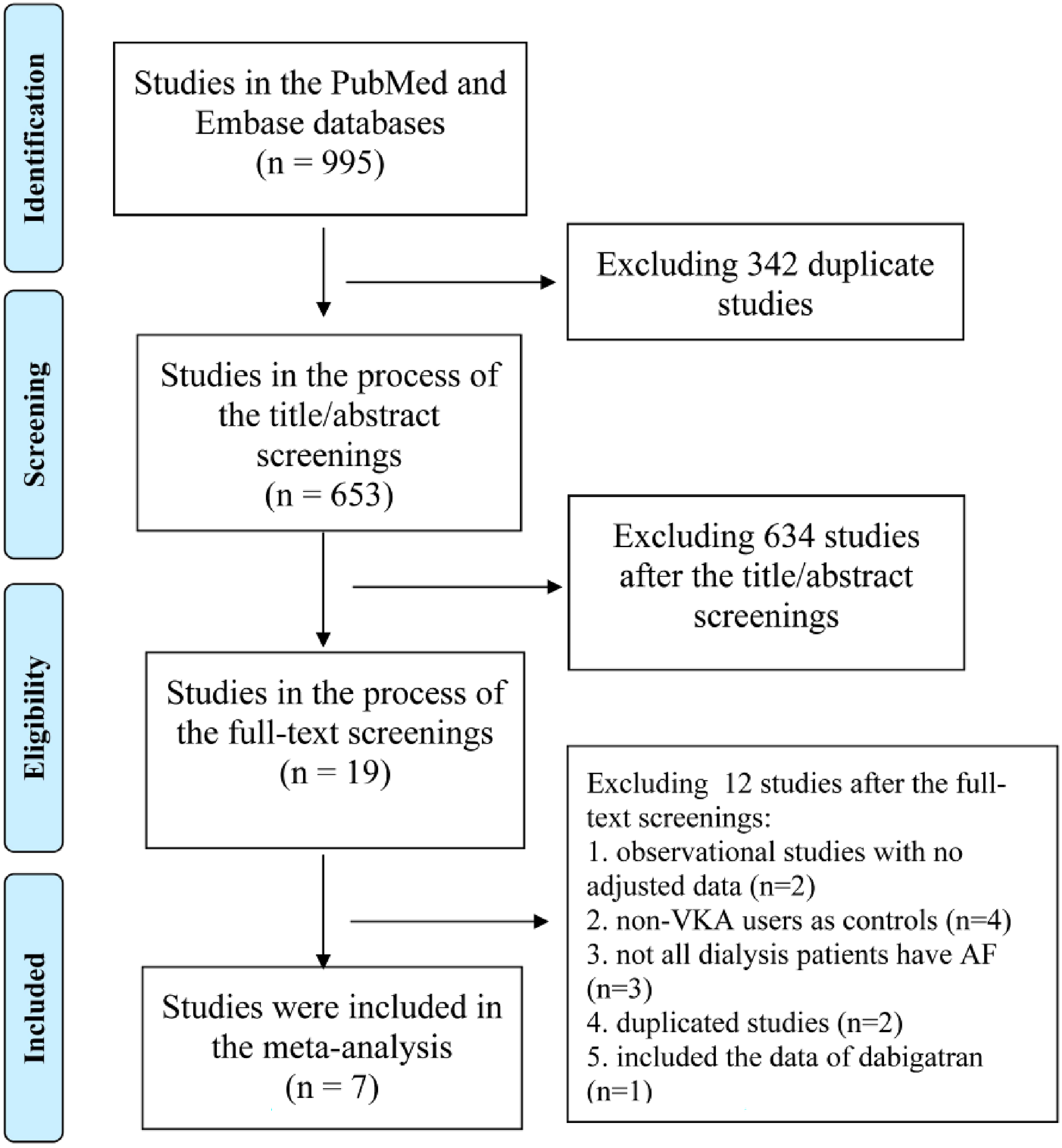
The PRISMA flow diagram of this meta-analysis.
The baseline characteristics of the included studies are summarized in
Baseline Characteristics of Randomized Controlled Trials and Observational Studies.
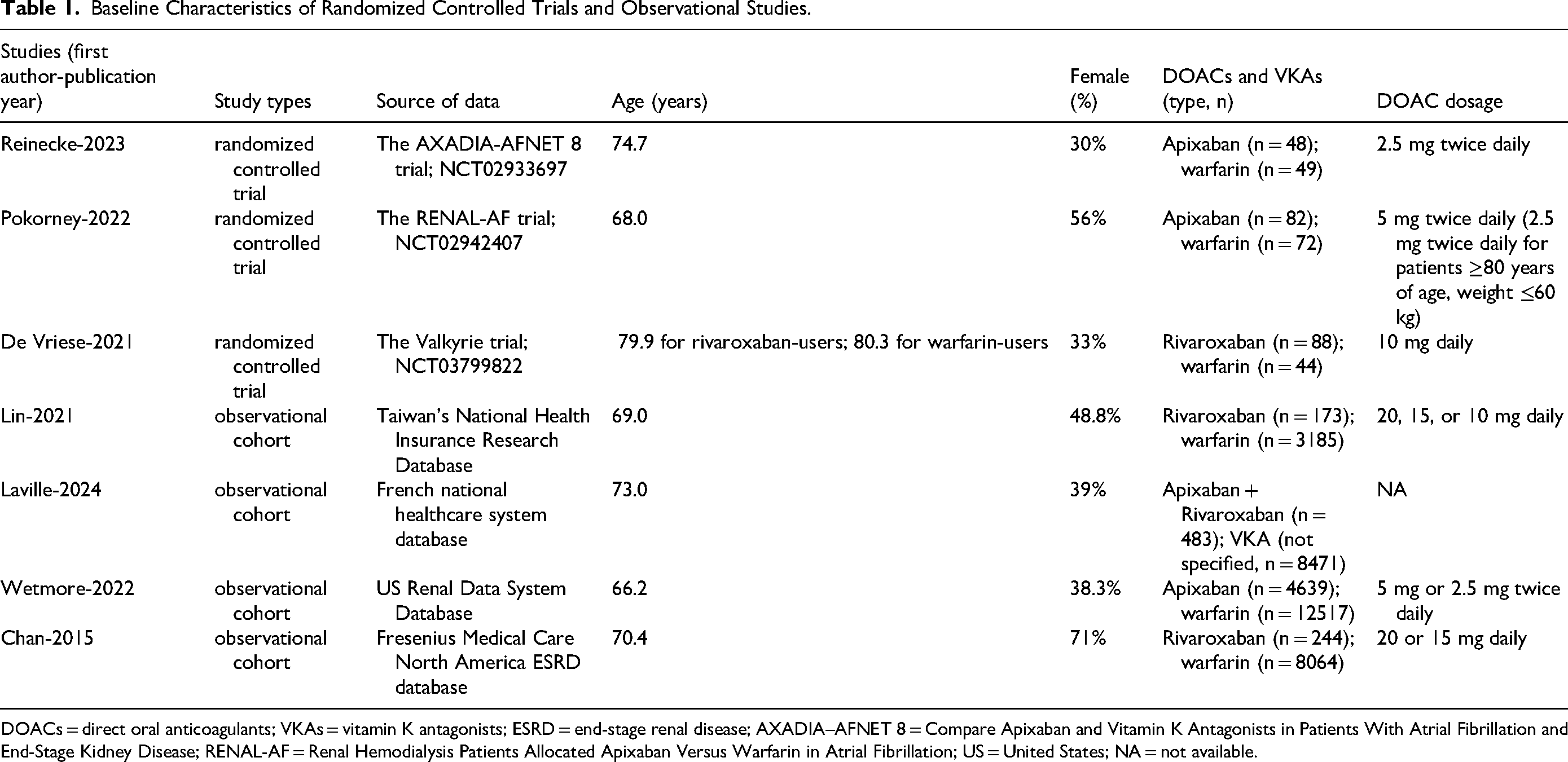
DOACs = direct oral anticoagulants; VKAs = vitamin K antagonists; ESRD = end-stage renal disease; AXADIA–AFNET 8 = Compare Apixaban and Vitamin K Antagonists in Patients With Atrial Fibrillation and End-Stage Kidney Disease; RENAL-AF = Renal Hemodialysis Patients Allocated Apixaban Versus Warfarin in Atrial Fibrillation; US = United States; NA = not available.
Efficacy Outcome: Stroke or Systemic Embolism
Pooled analysis of the RCTs demonstrated a significant reduction in the risk of stroke or systemic embolism in patients treated with factor Xa inhibitors compared to VKAs (OR = 0.37, 95% CI 0.15–0.93; p = 0.03). There was no evidence of significant heterogeneity (I2 = 0%). The forest plot for this analysis is depicted in
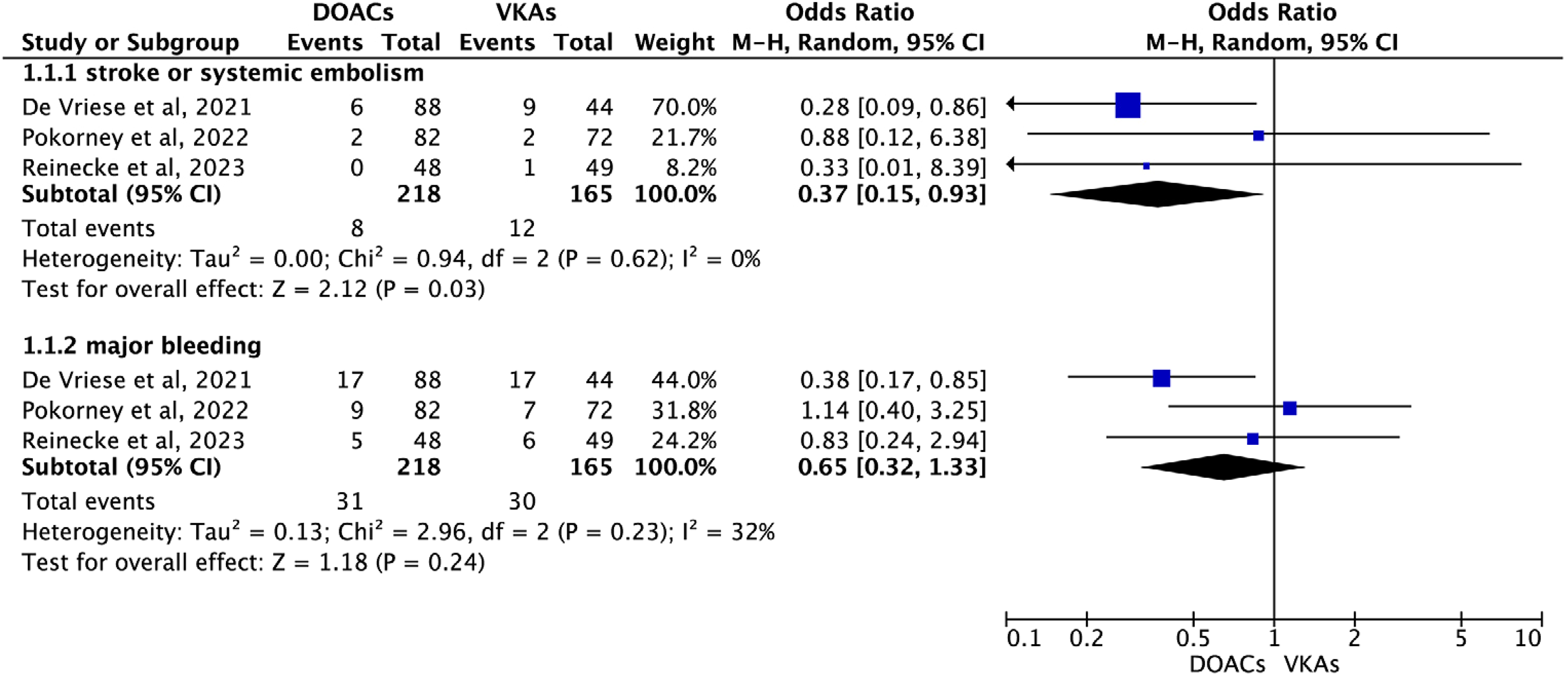
Pooled analysis of the randomized controlled trials in AF patients treated with factor Xa inhibitors compared to VKAs.
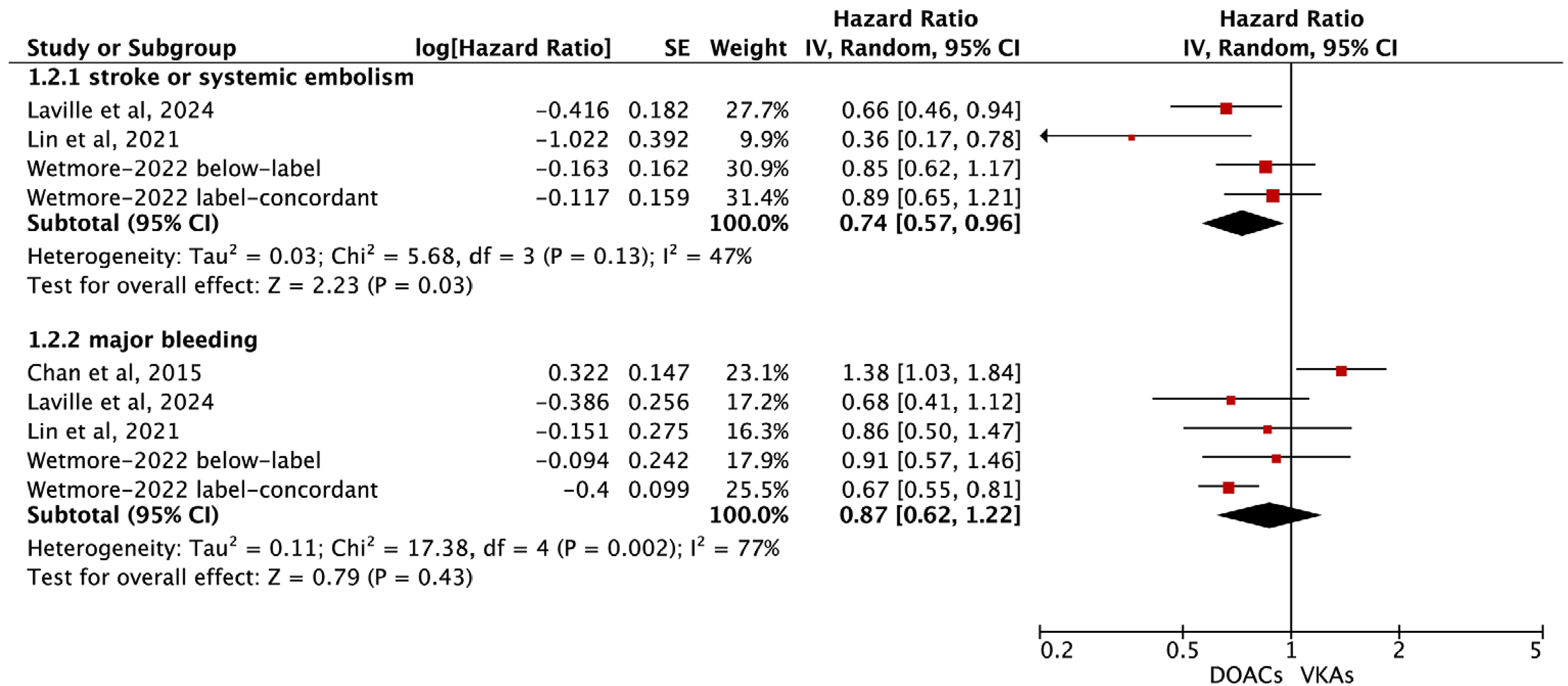
Pooled analysis of the observational cohort studies in AF patients treated with factor Xa inhibitors compared to VKAs.
Safety Outcome: Major Bleeding Events
Analysis of the RCTs revealed no significant difference in the risk of major bleeding events between patients treated with factor Xa inhibitors and those on VKAs (OR = 0.65, 95% CI 0.32–1.33; p = 0.24;
Subgroup Analysis
For observational studies, the subgroup analysis based on the type of factor Xa inhibitor (apixaban vs rivaroxaban) showed no significant interaction or difference in stroke or systemic embolism (rivaroxaban:adjusted HR = 0.55, 95% CI 0.26–1.15, and apixaban:adjusted HR = 0.75, 95% CI 0.55–1.03; p-interaction = 0.44; Supplemental Figure 3 ) or major bleeding (rivaroxaban:adjusted HR = 0.98, 95% CI 0.59–1.61, and apixaban:adjusted HR = 0.69, 95% CI 0.57–0.84; p-interaction = 0.21; Supplemental Figure 4 ). This suggests a similar therapeutic profile for both apixaban and rivaroxaban in the context of stroke prevention and bleeding risk.
Quality of Evidence
As shown in Supplemental Table 2, the RCTs yielded findings with high confidence levels for both the outcomes: stroke or systemic embolism and major bleeding. In the case of pooled observational studies, the findings indicated moderate confidence for the outcome of stroke or systemic embolism, while the confidence level for major bleeding was low.
Discussion
The findings of this systematic review and meta-analysis provide valuable insights into the comparative efficacy and safety of factor Xa inhibitors and VKAs in the management of AF in patients with ESKD on dialysis. The results indicate that factor Xa inhibitors, specifically apixaban and rivaroxaban, are associated with a significantly lower risk of stroke or systemic embolism without a significant increase in the risk of major bleeding events. These findings are particularly relevant given the high thromboembolic risk and the challenges associated with anticoagulation in this patient population.
One of the primary concerns with the use of VKAs in patients with ESKD on dialysis is the increased risk of bleeding.29,30 The narrow therapeutic window and the need for frequent monitoring and dose adjustments further complicate the management of VKAs in this setting. Our analysis suggests that factor Xa inhibitors may offer a safer and more effective alternative, with a favorable risk-benefit profile. This is supported by the observation of a reduced risk of stroke or systemic embolism and no significant difference in major bleeding events between the two treatment groups.
The results of our subgroup analysis based on the type of DOAC did not reveal any significant interaction between apixaban and rivaroxaban in terms of efficacy and safety outcomes. This suggests that both agents may be considered as viable options for stroke prevention in AF patients with ESKD on dialysis. However, further studies are needed to explore the comparative effectiveness of different factor Xa inhibitors and to identify patient-specific factors that may influence treatment selection. In addition, the included studies in our current meta-analysis employed a variety of dosing regimens, reflecting the ongoing clinical debate about the optimal dosing strategy for factor Xa inhibitors in ESRD patients on dialysis. Future research should aim to resolve this discrepancy and establish standardized dosing guidelines.
Patients with AF undergoing dialysis face unique challenges due to their compromised renal function, which can significantly influence the clearance and metabolism of medications, including dabigatran. This impaired drug clearance may result in increased risk of drug accumulation and consequently, a heightened risk of bleeding complications. The pharmacokinetic profile of dabigatran, characterized by its renal excretion and susceptibility to drug-drug interactions, warrants careful consideration for this patient population. In addition, it is also important to note that dabigatran is dialyzable to an extent of approximately 50–60%, which may influence its suitability as a treatment option for patients with ESRD on dialysis.
Several previous meta-analyses, including those by Kyriakoulis et al 31 and de Lucena et al, 32 have examined the impact of DOACs in AF patients requiring dialysis. Notably, de Lucena et al's study was restricted to RCTs and did not incorporate observational studies, potentially limiting the comprehensiveness of their findings. Meanwhile, Kyriakoulis et al's research, which did encompass both RCTs and observational studies, has raised significant concerns due to the inclusion of dabigatran, which may not be as suitable for patients with impaired renal function. Our current meta-analysis deliberately chose to focus exclusively on factor Xa inhibitors, which may present a more appropriate therapeutic option for AF patients undergoing dialysis. To our knowledge, this is the first meta-analysis to compare factor Xa inhibitors with VKAs in AF patients on dialysis, indicating that factor Xa inhibitors at least have comparable efficacy and safety outcomes to VKAs in this specific patient population.
Limitations
We acknowledged the potential limitations. First, the protocol for our current study has not been registered with PROSPERO. Second, the observed heterogeneity in the observational studies may influence the interpretation of the results, reflecting differences in study design, patient characteristics, and adjustment for confounding factors. 33 Additionally, the potential for residual confounding and selection bias in observational studies cannot be entirely ruled out. Third, there is an absence of studies reporting the use of edoxaban in patients with AF undergoing dialysis. Additional RCTs are required to validate the efficacy and safety of edoxaban 34 within this specific patient population. Lastly, future research endeavors should be directed towards elucidating the role of factor Xa inhibitors in secondary outcomes such as all-cause mortality, intracranial hemorrhage, and gastrointestinal bleeding.
Conclusion
Our meta-analysis suggests that factor Xa inhibitors, specifically apixaban and rivaroxaban, may be considered as effective and safe alternatives to VKAs for stroke prevention in AF patients with ESKD on dialysis.
Supplemental Material
sj-pdf-1-cat-10.1177_10760296241271423 - Supplemental material for Factor Xa Inhibitors Versus Vitamin K Antagonists in Atrial Fibrillation Patients with End-Stage Kidney Disease on Dialysis: A Meta-Analysis
Supplemental material, sj-pdf-1-cat-10.1177_10760296241271423 for Factor Xa Inhibitors Versus Vitamin K Antagonists in Atrial Fibrillation Patients with End-Stage Kidney Disease on Dialysis: A Meta-Analysis by Meimei Xiong, Linjuan Guo and Yun Wan in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Acknowledgment
None.
Author Contributions
Initials (Meimei Xiong), conceptualization; Initials (Yun Wan and Linjuan Guo), methodology; Initials (Meimei Xiong), investigation; Initials (Yun Wan) wrote the manuscript; Initials (Linjuan Guo), validation, revised the the manuscript. All authors read and approved the final manuscript.
Availability of Data and Materials
All relevant data and materials are presented in the paper.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Not required.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
