Abstract
Introduction
Chronic thromboembolic pulmonary hypertension (CTEPH) and venous thromboembolism (VTE) are thought to share many common risk factors. Our study aimed to determine the frequencies of 5 thrombosis-related gene single nucleotide polymorphisms (SNPs) associated with VTE in patients with CTEPH (n 129) compared with a control group of healthy individuals without a history of VTE (n 2637).
Methods
The SNPs of the following genes were investigated: F5 (F V Leiden, rs6025), F2 prothrombin (rs1799963), fibrinogen gamma (FGG, rs2066865), F11 (rs2289252) and ABO (non-O, rs8176719) in both groups.
Results
The study found that the rs1799963 variant was more common in patients with chronic thromboembolic pulmonary hypertension (CTEPH) compared to the control group (p < .0001). The GA heterozygous variant showed a significant increase with an odds ratio (OR) of 4.480 (95% CI: 2.344-8.562) or a finding by maximum likelihood analysis (MLA) with p < .0001. Additionally, there was a notable increase in the rs8176719 variant with p < .0001 in CTEPH patients. Both the homozygous G/G variant and the heterozygous -/G variant also showed an increase, with OR of 4.2317 (95% CI: 2.45571-7.2919) and 2.4324 (95% CI: 1.46435-4.0403) respectively, or MLA (p < .0001 and p .0006). The study also revealed a higher prevalence of the heterozygous C/T variant of rs2289252 in CTEPH patients, with an OR of 1.5543 (95% CI: 1.02503-2.3568) or MLA (p .0379).
Conclusion
The study suggests that the observed gene polymorphisms F2 (rs1799963), ABO (rs8176719), and F11 (rs2289252) may play a role as independent heritable risk factors in the development of CTEPH.
Introduction and Study Aim
Acute pulmonary embolism (PE) is a severe form of venous thromboembolism (VTE) with an incidence of approximately 39–115 per 100 000 population per year. 1 Chronic thromboembolic pulmonary hypertension (CTEPH) is a rare complication of previous embolisation, occurring with a time lag in 0.1–11.8% of patients affected by PE. 2 In the Czech Republic, a recent study 3 showed a 2year cumulative incidence of 2.3% for CTEPH, which was associated with a higher risk of rehospitalisation and death. The percentage of patients with a history of PE or deep vein thrombosis (DVT) was 78.1% and 41.9%, respectively. CTEPH is characterised by recurrent pulmonary thromboembolism and inadequate clot resolution, and lifelong therapeutic anticoagulation is recommended for these patients. It is, therefore, considered a complication of VTE.4,5 However, VTE is a multifactorial disease 6 with many acquired or inherited risk factors.7,8 Since 2008, newly published results from genome-wide association studies (GWAS) have begun to screen hundreds of thousands to millions of known single nucleotide polymorphisms (SNPs) in cohorts of tens of thousands to hundreds of thousands of individuals with VTE and controls without VTE.9–13 As the number of detected SNPs and participants in new GWASs has increased, dozens of new genetic associations with VTE have been discovered. However, most of these associations only had a tiny effect on VTE risk. Therefore, the selection criterion for their use requires that alleles associated with VTE achieve higher statistical significance in GWAS. 14 Apart from the F 5 Leiden polymorphisms (rs6025) and the prothrombin gene 20210G˃A mutation (rs1799963), the newly discovered SNPs of the fibrinogen gamma’ gene (FGG, rs 2066895), the F11 gene (rs2289252) and the ABO (non-O, rs8176719) met this requirement.
Therefore, our study aimed to investigate the prevalence of these five SNPs associated with a higher risk of VTE in patients with CEPTH compared to a control group of healthy individuals without a history of VTE.
Methods
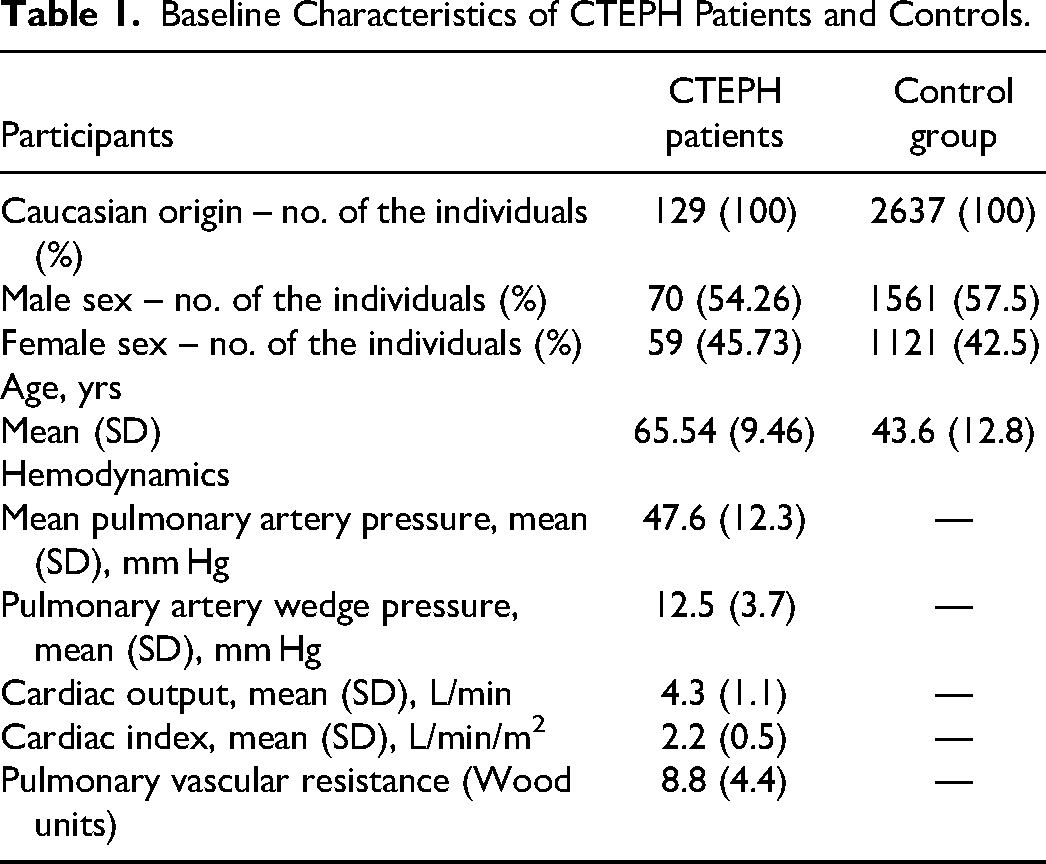
The cohort of 129 patients with CTEPH was registered at the Centre for Pulmonary Hypertension of the General University Hospital and the 1st Faculty of Medicine of Charles University in Prague. The registry of CTEPH patients has been described in detail previously. 3 In summary, the registry included adults (aged 18 years or older) newly diagnosed with CTEPH between years 2003 to 2018. Diagnostic criteria for CTEPH included a mean pulmonary arterial pressure ≥ 25 mm Hg, mismatched perfusion defects on lung ventilation perfusion scintigraphy, and diagnostic signs for CTEPH on multidetector CT angiography and conventional pulmonary angiography. The Ethics committee of the General University Hospital in Prague approved the registry. According to the completed questionnaire, the control group consisted of 2637 healthy individuals who did not have a VTE. This group included 1512 blood donors or healthy volunteers, 15 and 1125 participants in the Czech post-MONICA study. 16 The two control groups were combined to obtain representative samples from the entire Czech Republic. The first group was collected from Prague and Central Bohemia. CTEPH patients and controls gave written informed consent for DNA testing by the Declaration of Helsinki. The General University Hospital vin Prague Ethics Committee approved the genetic study focusing on risk factors for VTE (No: NT 11176-5). The Czech post-MONICA study, which was a population study, was approved by the Ethics Committees of the Institute of Clinical and Experimental Medicine and the Thomayer University Hospital in Prague. All study participants, including the CTEPH patients and the control group, were of Caucasian origin and resided in the Czech Republic. The baseline characteristics of both groups are presented in Table 1.
Baseline Characteristics of CTEPH Patients and Controls.
In the laboratory analyses, the SNPs of the following genes were investigated: F5 (F V Leiden, 1691 G > A, rs6025), F2 prothrombin (20210 G > A, rs1799963), FGG, fibrinogen gamma’ (10043 C > T, rs2066865), F11 (22 771 C > T, rs2289252) and ABO, non-O (rs8176719) in anonymous samples of uncoagulated blood.
Table 2 presents data on the incidence of thromboembolic events in patients with chronic thromboembolic pulmonary hypertension (CTEPH).
Pulmonary Embolism (PE) and Deep Venous Thrombosis (DVT) Manifestation in Patients with Chronic Thromboembolic Pulmonary Hypertension (n = 129).

Genome DNA was extracted from their leukocytes in peripheral blood and isolated using the MagNA Pure LC Nucleic Acid Extraction system™ with the MagNA Pure DNA Isolation Kit I™. DNA was isolated according to the MagNA Pure High-Performance DNA Extraction™ protocol (all products supplied by Roche Diagnostics, Mannheim, Germany). Mutations were determined using real-time polymerase reaction in a process called FRET (Fluorescence Resonance Energy Transfer). Tests were performed using the LightCycler® 480 System with LC® 480 Genotyping Master kits (all products supplied by Roche Diagnostics, Mannheim, Germany). Specific primers and fluorescently labelled probes were designed in cooperation with TIB MOLBIOL (Berlin, Germany), where they were custom-made.
Categorical variables were expressed as numbers and percentages, while continuous variables were presented as means and standard deviations according to their distribution. Group comparisons were made using Fisher's exact test. Logistic regression and maximum likelihood estimation (MLA) analyses were performed to assess the clinical impact of alleles and genotypes. Results were presented as odds ratios (OR) with corresponding 95% confidence intervals (CI) or as the results of chi-squared (ChiSq) tests with p-values in MLA analysis. Statistical analysis was performed using SAS statistical software (SAS version 9.4, SAS/Genetics™ 13.1, SAS Institute Inc., NC, USA) with population genetics tools. Results with a p-value <.05 were considered statistically significant differences in the representation of risk alleles and genotypes.
Results
The results of the tests in the CTEPH patients (n = 129) and controls (n = 2637) are shown in Table 3. All genotype findings in the CTEPH patients and controls fulfilled the Hardy-Weinberg equation (HWE), except for the finding of Factor V Leiden genotypes in the CTEPH patients (HWE pr˃ ChiSq 0.0119). Therefore, our statistical evaluation of the differences in the prevalence of Factor V Leiden between CTEPH patients and controls cannot be considered statistically significant. Fisher's exact test showed an increased prevalence of rs1799963 with p .0001 in patients with CTEPH compared to controls without a history of VTE. The heterozygous variant GA showed an increase with an OR of 4.480 (95% CI: 2.344-8.562) or MLA (p < .0001). In addition, only rs8176719 showed a significant increase with p < .0001 in CTEPH patients. We observed an increase in both the homozygous G/G variant with OR of 4.2317 (95% CI: 2.45571-7.2919) or MLA (p < .0001) and the heterozygous -/G variant with OR of 2.4324 (95% CI: 1.46435-4.0403) or MLA (p .0006). In CETPH patients, only the higher prevalence of the heterozygous C/T variant of rs2289252 was significant, with OR of 1.5543 (95% CI: 1.02503-2.3568) and MLA (p .0379).
Results of Alles and Genotypes Frequencies, Fisher’s Exact Test, Logistic Regression (Odds Ratio with 95% Wald Confidence Limit) and maximum Likelihood Estimation (MLE) Analysis Between CTEPH Patients (n 129) and Controls (n 2637) of the Five VTE Risk SNPs.
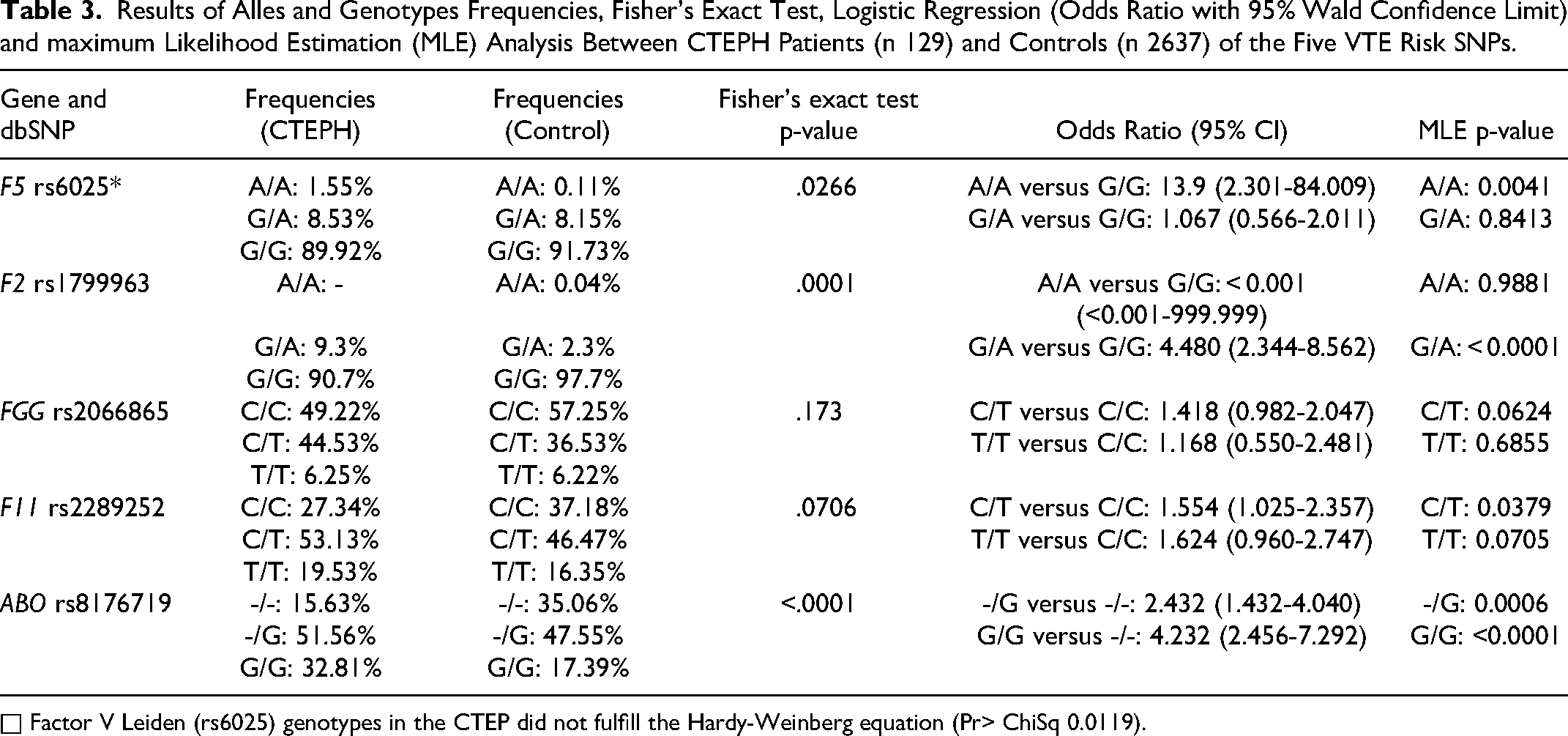
□ Factor V Leiden (rs6025) genotypes in the CTEP did not fulfill the Hardy-Weinberg equation (Pr> ChiSq 0.0119).
Discussion
The GWAS studies mentioned in the introduction showed statistically significant associations between the five tested SNPs rs6025, rs1799963, rs2066865), rs2289252 and rs8176719 and VTE. The likelihood of first VTE 17 and the likelihood of recurrent VTE after cessation of anticoagulation therapy 18 were also determined using these five SNPs. In both studies, the odds of first VTE and recurrent VTE increased with the number of risk alleles in the subjects.
The mutation in the coagulation F5 gene (FV Leiden, rs6025) is associated with resistance to activated protein C (APC). 19 The international prospective registry of four European centres 20 found that FV Leiden or APC resistance testing was present in up to 7.7% of patients with CTEPH. However, other studies have not found a significant increase in FV Leiden in patients with CTEPH compared to healthy controls.21,22 The US-CETPH registry in the United States reported APC resistance/FV Leiden in 9.9% of patients without genotype distribution. 4 However, in our study, testing for the FVL mutation in the group of patients with CTEPH did not adhere to the Hardy-Weinberg equilibrium (HWE) criteria. This exception was due to the discovery of 2 homozygous Factor V Leiden variants in a relatively small cohort of 129 CTEPH patients. The homozygous Factor V Leiden variant, in contrast to the heterozygous variant, is scarce in the population, approximately 1 in 5000 in the general population. 23 The frequency of the heterozygous variant of FV Leiden in patients with CTEPH (8.53%) was similar to that in the control group (8.15%), as well as to the prevalence of the heterozygous variant of FV Leiden of 8.91% in the Czech population. 15 Therefore, it can be concluded that the heterozygous FV Leiden variant does not play as significant a role in the aetiopathogenesis of CTEPH as it does in patients with VTE.
The rs1799963 prothrombin F2 20210G˃A mutation is associated with elevated plasma prothrombin levels and an increased risk of venous thrombosis. 23 In cohorts of patients with CTEPH, the prothrombin F2 20210 G > A mutation has been repeatedly described,20,22,24,25 but mostly without comparison to control group. However, Dodson MW et al 26 reported a higher frequency of prothrombin F2 20210 G > A mutation (8.5%) in a cohort of CTEPH patients who had acute PE before CTEPH diagnosis. Our study results show a significant increase in the incidence of the heterozygous GA variant (9.3%) in patients with CTEPH compared to the control group (2.3%) (p < .0001). The frequency of the heterozygous GA variant in the control group is consistent with the established frequency of 2.3% in the Dutch population 27 or 2.4% in the Czech population. 15
The investigated SNP rs2066865 is associated with reduced levels of fibrinogen gamma’ and reduced ratio of fibrinogen gamma’ to total fibrinogen. Decreased fibrinogen gamma’ levels and increased total fibrinogen levels are associated with an increased risk of venous thrombosis.26,28–30 However, our study did not show a statistically significant association between CTEPH and the FGG mutation.
In addition, our study showed a trend of association between rs2289252 in the F11 gene and CTEPH (p .07), which is independently associated with VTE.9–13,31 However, the prevalence of the heterozygous F11 C/T variant was significantly higher in CTEPH patients compared to controls, with an OR of 1.5543 (95% CI: 1.02503-2.3568) and MLA (p .0379). Previous studies have reported higher levels of F XI activity and antigen in VTE patients with both homozygous and heterozygous rs2289252 variants.32,33 However, this was not analysed here.
ABO, non-O genotyping allows improved differentiation between heterozygous and homozygous variants compared to serological determination. SNP rs8176719 represents a site in the ABO gene, commonly known as c.261delG, which is a critical SNP in determining blood group O status. 34 An allele encoding either blood group A or B will have a (G) at this SNP site. ABO blood group, especially non-O groups, is a common finding in both VTE and CTEPH patients.24,35 In our group of CTEPH patients, we found a significant increase in ABO, non-O with a p˂.0001. The study found an increased association between the homozygous non-O G/G variant and the outcome measure with an OR of 4.2317 (95% CI: 2.45571-7.2919) or MLA (p < .0001). In addition, the study also found an association between the heterozygous -/G variant and the outcome measure with an OR of 2.4324 (95% CI: 1.46435-4.0403) or MLA (p .0006). ABO, non-O rs8176719 is one of the essential polymorphisms associated with increased levels of coagulation factor VIII and its carrier vWF. 36 These increases in FVIII and vWF have also been described in patients with CTEPH.37,38 Based on the finding of an increase in vWF and a decrease in its cleaving enzyme ADAMTS13 in CTEPH patients, it is suggested that this dysregulation of the ADAMTS13-vWF axis is also related to the ABO blood group. 39
A limitation of the study is the small CTEPH cohort. In addition, the incidence of VTE in the control group was determined solely based on completed questionnaires without objective verification. Therefore, it would be helpful to extend the study of the significance of the 5 SNPs in the ABO, F2, F5, F11 and FGG genes in a prospective study to a more significant number of patients with PE and possibly to patients with chronic thromboembolic disease without pulmonary hypertension.
Conclusions
We suggest that the observed risk factors for VTE and its recurrence F2 (20210 G > A, rs1799963), F11 (22 771 C > T, rs2289252) and ABO, non-O (c.261 del G, rs8176719) play an important role as independent heritable risk factors in the pathogenesis of CTEPH.
Footnotes
Acknowledgements
The authors thank the staff and participants of the study for their important contributions. This study was supported by grant MH CZ DRO VFN 64 165 (given by the Czech Ministry of Health).
Addendum
All authors contributed to the critical revision of the manuscript and approved the final version. In addition, Jan Kvasnička contributed to concept and design study and drafted the manuscript; Pavel Jansa participated in the study design and performed the classification of CTEP cases; Renata Cífková classified control cases; Daniela Duškova classified control cases; Petra Bobčíková analysed the SNPs; Martin Ševčík and Zuzana Zenáhlíková provided statistical advice and Tomáš Kvasnička contributed to concept and design study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Grant MH CZ DRO VFN 64 165 (given by the Czech Ministry of Health),
