Abstract
Introduction and objectives
The present study aimed to investigate different peripheral lymphocyte subsets in patients with severe hemophilia A (HA) and factor VIII (FVIII) inhibitor production. For this, age-matched cases of 19 FVIII inhibitor-positive (IP), 21 FVIII inhibitor-negative (IN) and 45 healthy controls were selected for study.
Methods
Flow cytometry was used to analyze the peripheral lymphocyte subsets, including T, B, natural killer (NK) and NKT cells. The T cell subsets included CD3 + CD4-CD8- [double negative T (DNT)], CD3 + CD4 + CD8+ [double-positive T (DPT)], CD3 + CD4 + CD8- and CD3 + CD4-CD8+ T cells. Pairwise comparisons of absolute lymphocyte subset values were conducted among the three groups. The cut-off value for absolute lymphocyte counts was determined using receiver operating characteristic curve analysis.
Results
The results demonstrated that the absolute values of DPT cells in the IN and IP groups were significantly lower than those in the healthy control group (P = 0.007). The DNT values were also lower in severe HA patients with or without inhibitor than those in healthy subjects, but these differences were not statistically significant (P = 0.053). In addition, the absolute value of CD4+ Th cells in the IP group was lower than that in the healthy controls (P = 0.013). Although not statistically significant (P = 0.064), the absolute values of NKT cells were higher in the IN group compared with the IP group, and higher in the IP group compared with the healthy control group. There were no statistically significant differences in total T, B, CD8 + and NK cells among the IN, IP and healthy control groups. The cut-off value for absolute CD4+ Th cells in the IN group was < 598/µl.
Conclusion
The decrease in absolute values of CD4+ Th cells in severe HA patients may contribute to the establishment of infused FVIII immune tolerance. If the CD4+ Th value remains > 598/µl, clinicians should be vigilant for possible FVIII inhibitor production, especially on days prior to FVIII exposure.
Introduction
Hemophilia A (HA) is an X-linked recessive hereditary bleeding disorder resulting from a deficiency in or a dysfunction of coagulation factor VIII (FVIII). HA affects ∼1 in 5000 male infants and is characterized by recurrent joint and muscle bleeding. 1 The severity of HA is classified based on FVIII activity as mild (≥5–<40%), moderate (≥1–<5%) or severe (<1%). The primary treatment of HA is FVIII replacement therapy, but up to 20%–30% of severe cases and 13% of mild to moderate cases develop neutralizing antibodies (inhibitors) against exogenous FVIII, 2 leading to uncontrolled or life-threatening bleeding and subsequently increased treatment costs. 3 FVIII inhibitors represent the most severe complication of FVIII replacement therapy and can be managed with immune tolerance induction (ITI) therapy, which involves regular administration of high doses of FVIII over months or years.4,5 However, the treatment burden and high costs limit its use, particularly in economically disadvantaged.
The induction and maintenance of immune tolerance to exogenous FVIII is critical for all patients with HA. Research progress has been made in elucidating the mechanisms of tolerance induction using ITI therapy, both via patient studies and studies including cell culture and animal models.6–9 However, further human studies are needed to identify reliable prognostic biomarkers and improve mechanistic understanding of immune tolerance to exogenous FVIII. The development of FVIII inhibitors is influenced by both inherent genetic factors and external environmental factors, intensive FVIII treatment of hemorrhages or surgery at the start of treatment was associated with an increased inhibitor risk. Some findings suggest that in previously untreated patients with severe HA, high-dosed intensive FVIII treatment increases inhibitor risk and prophylactic FVIII treatment decreases inhibitor risk, especially in patients with low-risk F8 mutations.10,11 However, the mechanisms underlying FVIII inhibitor development in patients with HA require further elucidation. Since the immune system may be altered in patients with HA and FVIII inhibitors, investigating changes in immune cells may help identify risk factors and elucidate the underlying mechanism of FVIII inhibitor development.
In the present study, the absolute values of peripheral blood lymphocyte subsets were compared among three age-matched groups: FVIII inhibitor-positive (IP) patients with severe HA, FVIII inhibitor-negative (IN) patients with severe HA and healthy males. These comparisons aimed to further understanding of the potential immune mechanisms underlying the development of FVIII inhibitors in patients with severe HA.
Patients and Methods
Study subjects. The inclusion criteria for the IP study group were as follows: i) Patients with severe HA (FVIII: C < 1%); ii) IP (FVIII inhibitor titer ≥ 0.6 BU/ml), including high titer (≥5 BU/ml) and low titer (<5 BU/ml) ; iii) absence of bleeding events within the 2 weeks preceding blood collection; and iiii) did not receive any treatment including corticosteroids or immunosuppressants. 12 It should be noted that the peripheral blood lymphocyte subsets differed markedly according to age and sex.13–15 We chosed healthy individuals from age-matched males who had no previous diagnosis of HA or history of bleeding. If the exposure days of IN patients with severe HA were not enough, they still possibly developed inhibitor in the future. So the age-matched IN patients with severe HA were selected as the control groups whose exposure days of FVIII were not shorter than 300 days which was the longest exposure day we observed in IP group. The exclusion criteria for all subjects included infection, inflammation, immune disorders, cancer or psychological trauma. The present study was approved by The Ethics Committee of Peking University First Hospital Taiyuan Branch (Taiyuan Central Hospital of Shanxi Medical University), and informed consent was obtained from all participants.
Flow cytometry. Whole blood of the 85 participants was collected and assayed by flow cytometry (BD CantoTM :V657338000706, USA) following standard operating procedure of the stain-lyse-wash protocol. In brief, 5 μL of each antibody reagents [CD45-PerCP (catalog no.: 20190218), CD8-APC-cy7 (catalog no.: 20210024), CD4-PE-cy7 (catalog no.: 20210614), CD16-PE (catalog no.: 20190239), CD56-PE (catalog no.: 20190241), CD3-FITC (catalog no.: 20190216), CD19-APC (catalog no.: 20220048), all purchased from RAISE CARE company, China] was added to the bottom of the tube, 50 μL of well-mixed whole blood was added to the bottom of tube by the reverse pipette technique, mixed and incubated for 20 min. Then, 450 μL of lysing solution was added into the tube, mixed, and incubated for 15 min in dark at room temperature. Finally, the tube was washed twice by 1×PBS solution for detection.
The Diva 9 software (BD, USA) was used to analyse the peripheral blood lymphocyte subsets data, including CD3 + CD4-CD8- double negative T (DNT) cells, CD3 + CD4 + CD8 + double positive T (DPT) cells, CD3 + CD4 + CD8- T helper (Th) cells, CD3 + CD4-CD8 + cytotoxic T lymphocytes (Tc), total CD3+ T lymphocytes, CD19+ B lymphocytes, CD3 + CD16 + CD56 + natural killer T (NKT) cells and CD3-CD16 + CD56+ NK cells. The total lymphocyte counts yielded from routine blood examinations, not those of peripheral lymphocyte subsets including T, B, and NK lymphocytes were utilized to calculate the absolute values of lymphocyte subsets.
Statistical analysis. Calculations were performed using R software (version 4.1.1) (https://www.R-project.org/), and P < 0.05 was considered to indicate a statistically significant difference. Quantitative variables are presented as the median and the interquartile range (IQR). Given the relatively large number of variables, quantitative variables were initially transformed into ordinal variables using quartiles. Logistic regression was then used for initial screening, followed by tests for normality and homogeneity of variance on selected variables. Based on these results, one-way ANOVA or Kruskal-Wallis rank sum tests were conducted. Pairwise comparisons were then made when differences among the three groups were identified, with the function TukeyHSD or pairwise.wilcox.test in R. Receiver operating characteristic (ROC) curves were utilized to determine the cut-off values for the absolute lymphocyte counts.
Results
General Patient Information
In total, 19 IP patients with severe HA were enrolled in the present study, with ages at the emergence of FVIII inhibitor ranging from 3.5 to 59 years, and a median age of 30 years at enrollment (range, 4–62). Among them, 3 patients were pediatric (aged 4–18 years) and 16 were adults (aged 19–62 years). In the IP group, there were 8 low-titer patients with severe HA (0.6–3.4 BU/ml) and 11 high-titer patients (8.4–512 BU/ml). The control groups comprised 21 age-matched IN patients with severe HA, with a median age of 33 years (range, 7–60 years), and 45 age-matched healthy males, with a median age of 30 years (range, 4–61 years). No statistically significant differences were observed among the IN, IP and healthy control groups (P = 0.961). All IP patients with severe HA received on-demand treatment, while the IN patients with severe HA included 14 cases administered prophylactic treatment and 7 cases administered on-demand treatment. The exposure days at enrollment for IP patients with severe HA ranged from 15 to 300 days, which was significantly lower than that for IN patients with severe HA (300–2100 days). Among the IP group, 12 cases had genetic mutation results according to the historical medical record, including 8 cases of intron 22 inversion (Inv22), 1 case of nonsense mutation, 1 case of frameshift mutation and 2 cases of missense mutation. Among the IN group, 6 cases had genetic mutation results, including 3 cases of Inv22, 1 case of intron 1 inversion (Inv1) and 1 case of splice site mutation. The detailed general patient results are shown in Table 1.
The General Information of Enrolled Patients.
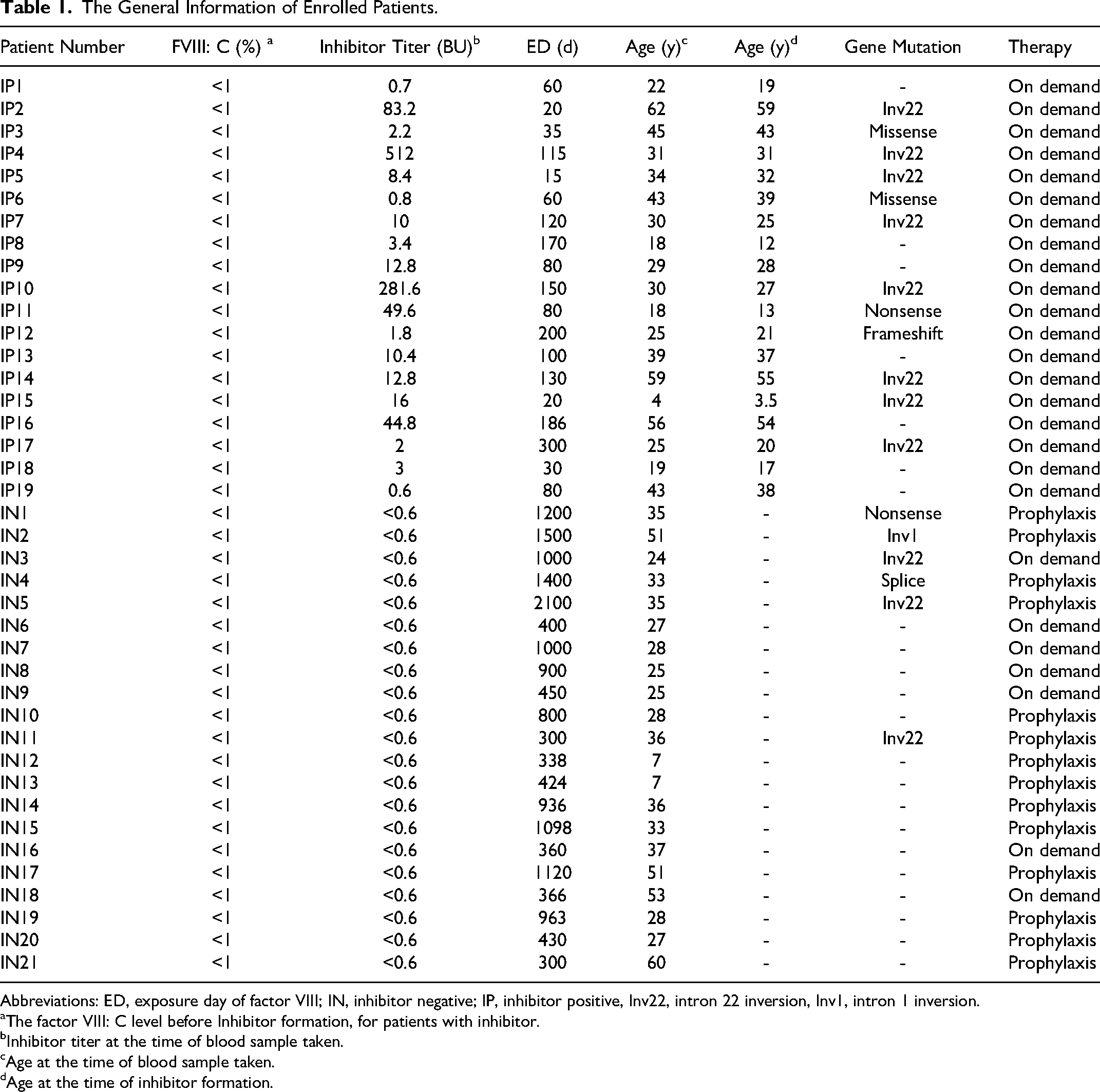
Abbreviations: ED, exposure day of factor VIII; IN, inhibitor negative; IP, inhibitor positive, Inv22, intron 22 inversion, Inv1, intron 1 inversion.
The factor VIII: C level before Inhibitor formation, for patients with inhibitor.
Inhibitor titer at the time of blood sample taken.
Age at the time of blood sample taken.
Age at the time of inhibitor formation.
Comparison of the T Lymphocyte Subsets Between the IP, IN and Control Groups
The assessed lymphocyte subsets included total T, B, NK and NKT cells, and the T cell subsets comprised DNT, DPT, Th and Tc cells. The descriptive statistical data on the absolute values of these subsets among the IP patients with severe HA, IN patients with severe HA and healthy controls are presented in Table 2.
The Absolute Value Reported as Median (IQR) of Peripheral Lymphocyte Subsets in IP, IN and Healthy Males.
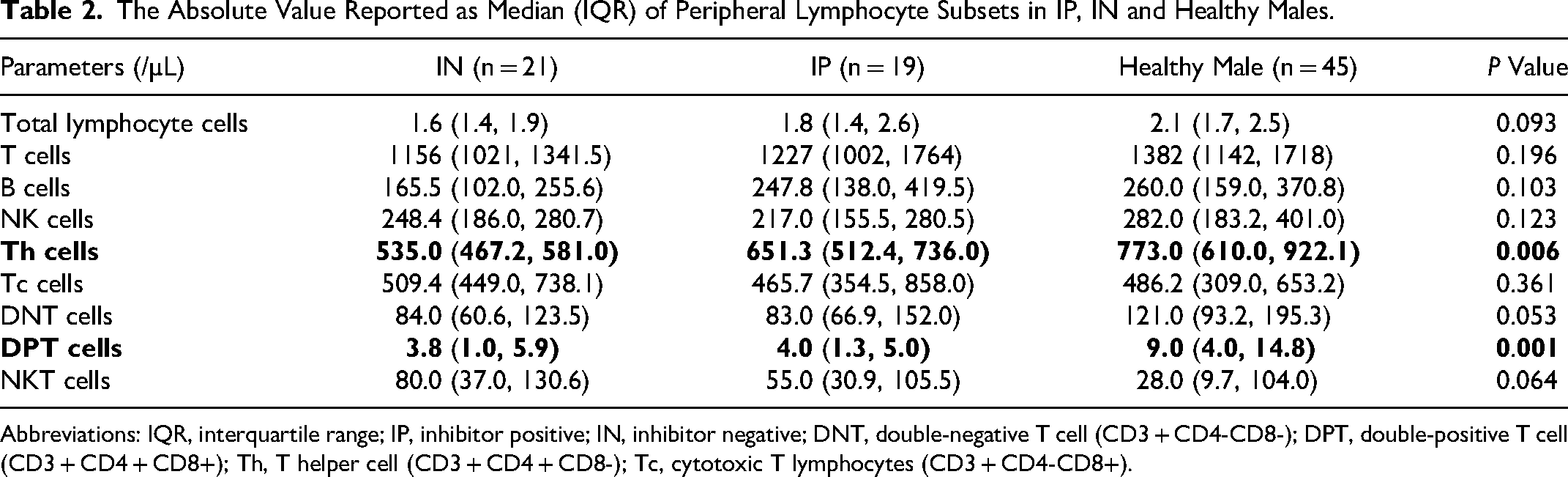
Abbreviations: IQR, interquartile range; IP, inhibitor positive; IN, inhibitor negative; DNT, double-negative T cell (CD3 + CD4-CD8-); DPT, double-positive T cell (CD3 + CD4 + CD8+); Th, T helper cell (CD3 + CD4 + CD8-); Tc, cytotoxic T lymphocytes (CD3 + CD4-CD8+).
Pairwise comparisons of absolute values for each lymphocyte subset were conducted among the three groups. The absolute DPT cell values in the IP and IN groups were significantly lower than that in the healthy control group (P = 0.007). Although the absolute DNT cell values in the IN and IP groups were lower than that in the the healthy control group, no statistically significant differences were observed (P = 0.053). The absolute values of CD4+ Th cells in IN group were significantly lower than that in healthy control group (P = 0.013). No statistically significant differences were found in the absolute values of CD4+ Th cells between the IN and IP groups or between the IP and healthy control groups. Additionally, pairwise comparisons revealed that there were no statistically significant differences in CD8+ Tc cells among the three groups (Figure 1).
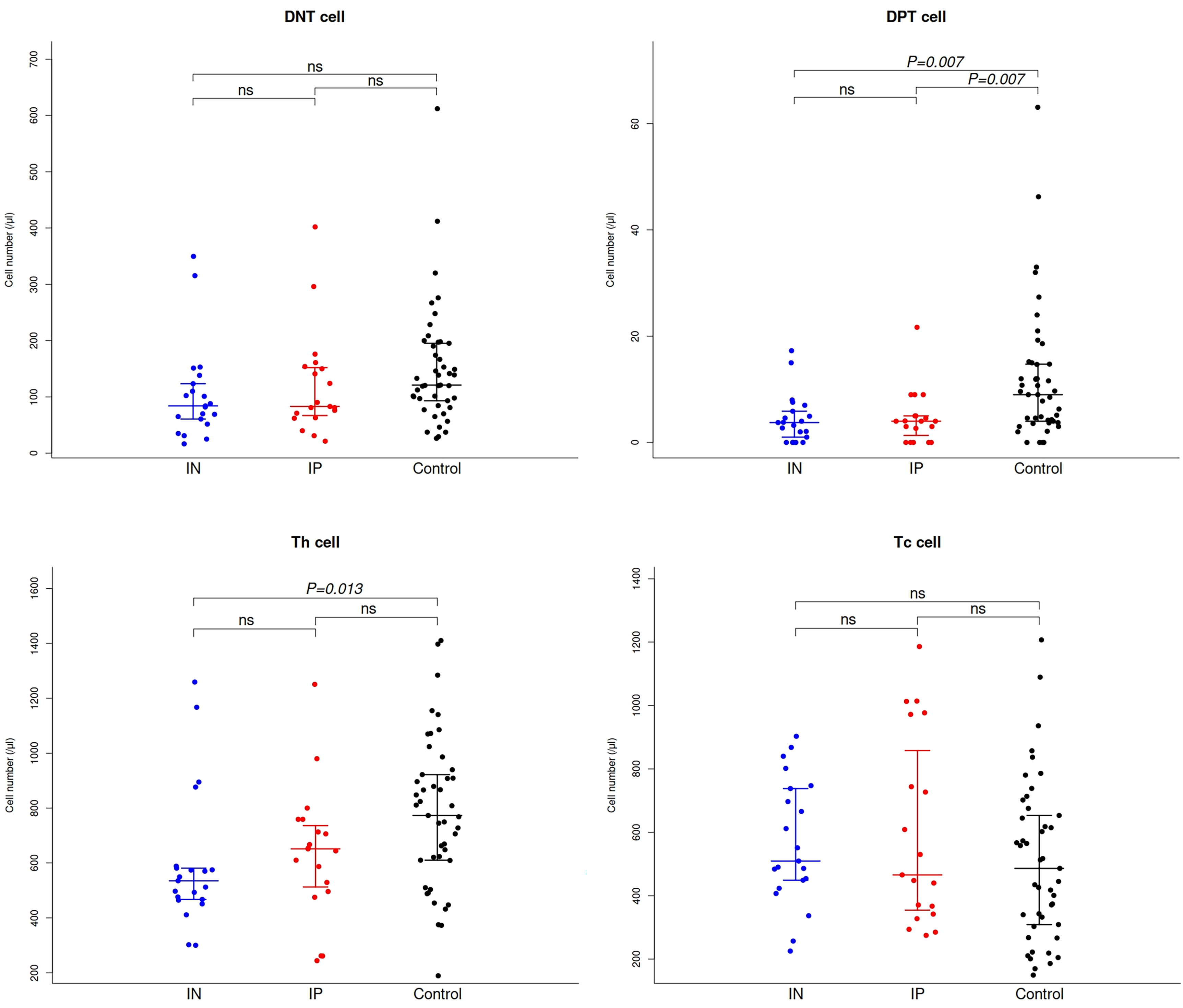
Number of DNT, DPT, Th and Tc lymphocytes in patients with hemophilia A and the age-matched healthy males. ns, non-significant; FVIII, factor VIII; IP, FVIII inhibitor-positive; IN, FVIII inhibitor-negative; DNT, double-negative T cell (CD3 + CD4-CD8-); DPT, double-positive T cell (CD3 + CD4 + CD8+); Th, T helper cell (CD3 + CD4 + CD8-); Tc, cytotoxic T lymphocytes (CD3 + CD4-CD8+).
Although not statistically significant (P = 0.064), the absolute values of NKT cells were higher in the IN group compared with the IP group, and higher in the IP group compared with the healthy control group. There were no statistically significant differences in total T, B, CD8+ and NK cells among the IN, IP and healthy control groups. Although significant differences were not observed, the absolute values of total T cells tended to be lower in the IN group compared with the IP group, and lower in the IP group compared with the healthy control group. No differences were noted for B cells and NK cells among the IN, IP and healthy control groups (Figure 2).
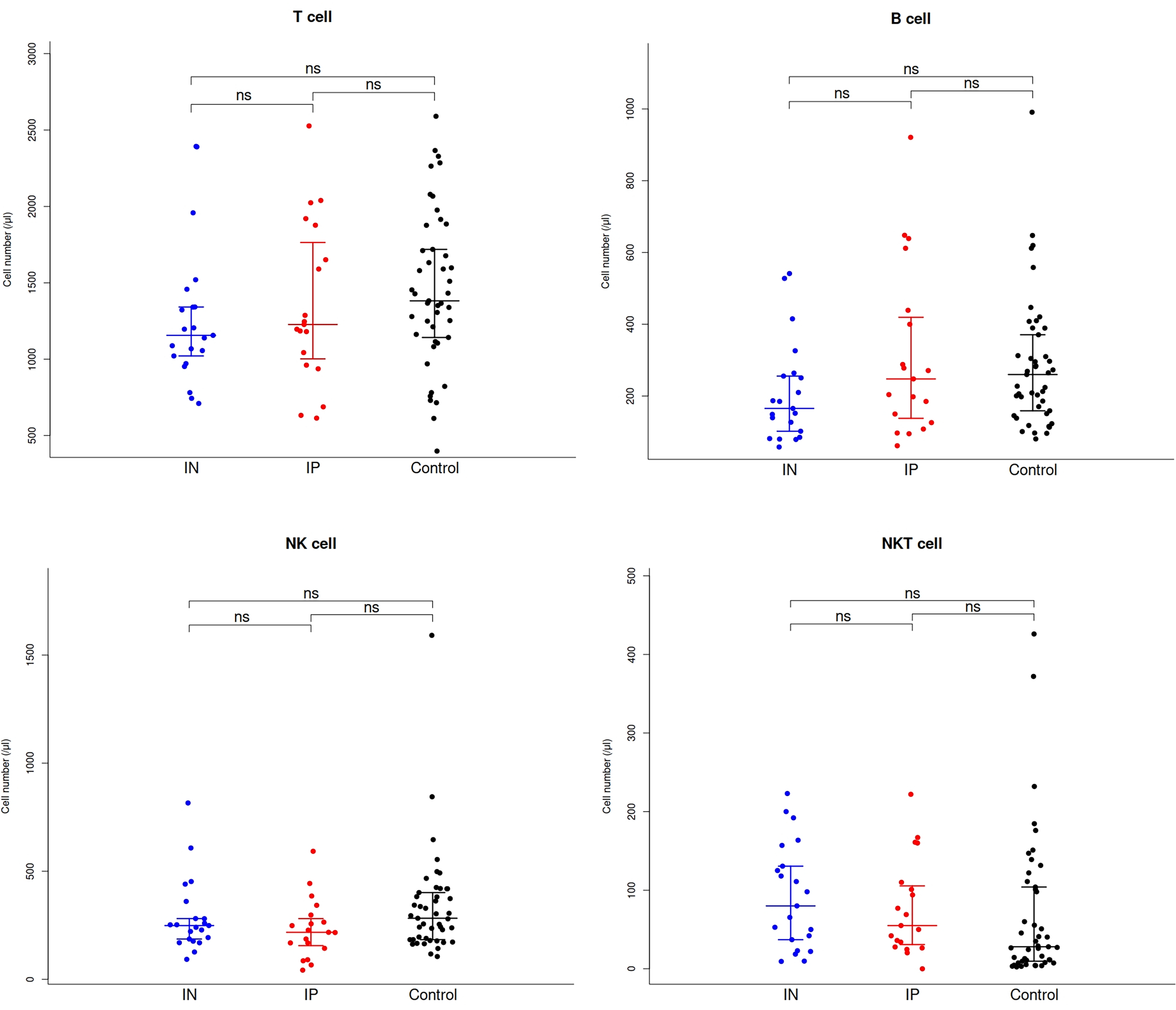
Number of T, B, NK and NKT lymphocytes in patients with severe hemophilia A patients with and without inhibitor and the age-matched healthy males. ns, non-significant; NK, natural killer.
Th cells cut-off value.
ROC curve analysis was conducted to determine the cut-off value for Th cells in the IN patients with severe HA group, which revealed a value of 598/µl (Figure 3). This cut-off value demonstrated a sensitivity of 81%, specificity of 77.8% and an AUC of 0.717 (95% confidence interval, 0.575–0.859). These findings suggested that when the absolute value of Th cells is < 598/µl, patients with severe HA are less likely to develop FVIII inhibitors during exogenous FVIII infusion. Therefore, only careful monitoring is warranted in such cases. Conversely, if the absolute value of DNT cells remains > 598/µl, heightened vigilance for the possible emergence of FVIII inhibitors is advised, particularly on the days prior to exposure (Figure 3).
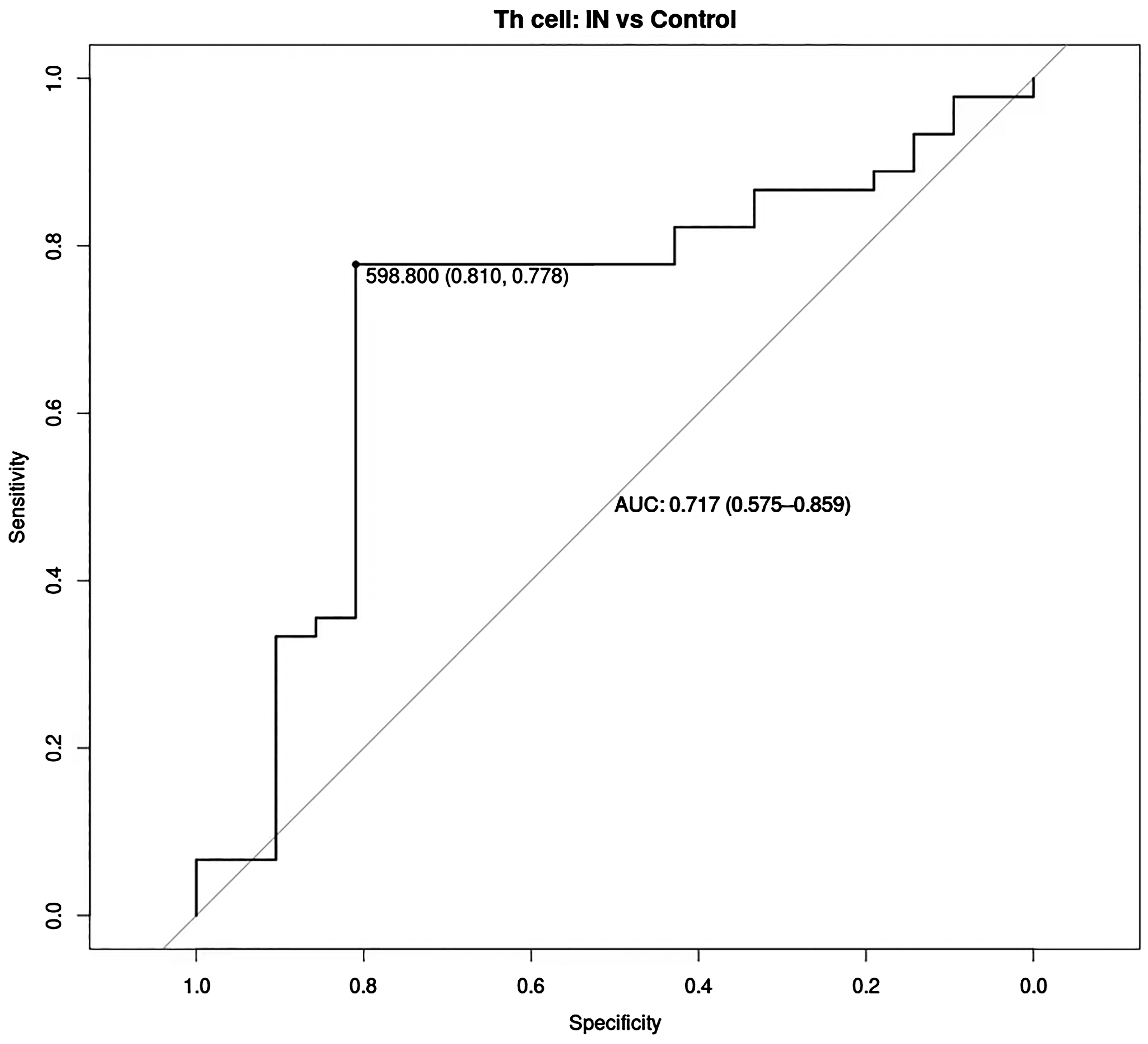
ROC curves indicating the CD4+ Th cut-off score in the IN group. Results: optimal cut-off score, 598/µl; sensitivity, 81%; specificity, 77.8%; AUC, 0.717; CI, 0.575–0.859. ROC, receiver operating characteristic; AUC, area under curve; CI, confidence interval; IN, factor VIII inhibitor-negative.
Discussion
Early thymic precursor cells initially exhibit a DNT phenotype and subsequently differentiate into DPT cells.16,17 Central immune tolerance, a developmental process occurring in the thymus, ensures the elimination of self-reactive T cells, undergoing negative selection to prevent autoimmunity. 18 During positive selection in the thymus, interaction between major histocompatibility complex (MHC) class I molecules and T cell receptors leads to the selection of CD4-CD8+ or CD4+CD8- single-positive T cells capable of recognizing polymorphic antigenic segments presented by MHC class I and II molecules. 19 Patients with FVIII cross-reactive material (CRM)-negative status are at a higher risk of developing FVIII inhibitors, 20 due to the complete absence of endogenous FVIII, leading to failure to establish central immune tolerance. Therefore, peripheral immune tolerance mechanisms must be reinforced and maintained to prevent the escape of self-reactive T and B cells into the periphery, and thereby avert unnecessary immune activation.21,22 FVIII inhibitors are only produced when peripheral immune tolerance mechanisms fail.23,24
Healthy individuals typically exhibit central immune tolerance to endogenous FVIII, and IN patients with severe HA may establish central tolerance to endogenous FVIII polypeptides. Moreover, even if central tolerance fails, peripheral immune tolerance to exogenous FVIII is typically established during FVIII infusion. However, both central and peripheral tolerance mechanisms to FVIII may fail in IP patients with severe HA, resulting in the production of FVIII inhibitors during exogenous FVIII infusion. In our study, a cross-sectional comparison of immune profiles between healthy controls and HA patients with and without inhibitor were analyzed. Severe HA patients with and without inhibitor have both demonstrated lower DPT cells than healthy subjects. Decrease of DPT counts is not consequential to the development of inhibitors, but to the naturally occurring the response to infused FVIII. There was also a tendency of decline of DNT counts in the HA patients with and without inhibitor than healthy controls.
CD4+ T cells are critical to the production and maintenance of inhibitors, as demonstrated by the observation that HIV+ HA patients experienced declines in inhibitor titers that were, unfortunately, reversed when T-cell levels rebounded with effective antiretroviral therapy. 25 FVIII-specific CD4+ T-cells are key players of the allo-immunization to infused FVIII. They supported B-cells to differentiate into FVIII-specific memory B-cells and anti-FVIII producing plasma cells.26,27 FVIII-specific CD4+ T-memory cells are also the main targets of ITI.28,29 Our results demonstrated that successful immune tolerance to exogenous FVIII depends on the decrease of CD4+ T cell counts. The cut-off for the absolute value of CD4+ Th cells in IN patients with severe HA was determined to be 598/µl, suggesting that when the absolute value of DNT cells is < 598/µl, patients with severe HA are less likely to develop FVIII inhibitors during exogenous FVIII infusion. Therefore, monitoring the absolute value of CD4+ Th cells in patients with severe HA during FVIII administration, especially in the days following prior FVIII exposure, is recommended. Not enough reduction in the number of Th cells may lead to insufficient peripheral immune tolerance of infused FVIII, ultimately resulting in the emergence of FVIII inhibitors. In conclusion, a decrease in the absolute value of CD4+ Th cells during FVIII replacement therapy could facilitate the establishment of FVIII immune tolerance in patients with severe HA.
To the best of our knowledge, the present study is the first to report on alterations in peripheral blood lymphocyte subsets in IN and IP patients with severe HA. Despite the limited sample size, relatively reliable results were obtained through the design of a matched case-control study. Future research should involve an expansion of the sample size to study lymphocyte subsets, including initial, helper, effector and memory T and B cells, to elucidate the immune mechanisms underlying FVIII inhibitor production.
Footnotes
Authors’ Contributions
LZ, XG and ZG were major contributors to designing the work and writing the manuscript. ZL and WL analyzed the patient data. JH was responsible for specimen collection. YG and ZP collected the patient data. JS and YZ were responsible for the patient follow-up. ZG made the final revision of the manuscript. All authors read and approved the final version of the manuscript.
Availability of Data and Materials
The data generated in the present study may be requested from the corresponding author.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval
The present study was approved by The Ethics Committee of Taiyuan city Central Hospital (2023073).
Funding
This work was supported by the grants from Natural Science Foundation of Shanxi Province (Grant No. 202103021224001) and Science and Technology Program Project of Taiyuan City (Grant No. 202239).
Informed Consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
