Abstract
Objectives
To assess the efficacy and safety of rivaroxaban compared to warfarin after inferior vena cava (IVC) filter implantation.
Method
This retrospective analysis includes data from 100 patients with deep vein thrombosis (DVT) who underwent IVC filter implantation due to a free-floating thrombus (n = 64), thrombus propagation (n = 8), or acute bleeding (n = 8) on therapeutic anticoagulation, catheter-directed thrombolysis (n = 8), or had previously implanted filter with DVT recurrence. Patients were treated with warfarin (n = 41) or rivaroxaban (n = 59) for 3-12 months. Symptomatic venous thromboembolism (VTE) recurrence and bleeding events were assessed at 12 months follow-up.
Results
Three (7.3%) cases of VTE recurrence without IVC filter occlusion occurred on warfarin and none on rivaroxaban. The only (2.4%) major bleeding occurred on warfarin. Three (5.1%) clinically relevant non-major bleedings were detected on rivaroxaban. No significant differences existed between groups when full and propensity scores matched datasets were compared.
Conclusions
Rivaroxaban seems not less effective and safe than warfarin after IVC filter implantation.
Introduction
Venous thromboembolism (VTE), including deep vein thrombosis (DVT) and pulmonary embolism (PE), remains a significant contributor to the global disease burden. 1 PE ranks third in the mortality from cardiovascular diseases after ischemic heart disease and stroke, with an estimated mortality rate of 105.5, 42.3, and 9.4-32.3 per 100,000 person-years, respectively. 2 DVT is traditionally considered a primary cause of PE, which occurs silently in 30-66% of patients with DVT.3-6 The actual risk of symptomatic PE in proximal DVT is unknown but estimated to be about 50% at 3 months without treatment. 7 Despite emerging evidence of inferior vena cava (IVC) filter inefficacy to prevent fatal PE, it is still widely used.8,9 Among all patients with VTE, up to 20-40% in the USA and up to 20% in Russia undergoing IVC filter implantation.10-12 Since retrievable filters were introduced several decades ago, their current removal rate is not high enough, estimated from 34% to 45%.13,14 Thus, a lot of patients require anticoagulation in the presence of IVC filter, which raised concern about the reliability of direct oral anticoagulants (DOACs) in such circumstances. Although phase III trials showed similar efficacy and improved safety of DOACs in comparison with vitamin K antagonists (VKAs) in patients with DVT and PE, the evidence on their use after IVC filter implantation is still limited.15-17
To fill this knowledge gap, we designed a current analysis that assessed the efficacy and safety of routinely prescribed oral rivaroxaban compared to warfarin in patients after IVC filter implantation.
Methods
This retrospective cohort study analyzes prospectively collected data from patients’ medical records. Data on all individuals admitted at Clinical Hospital No. 1 of the President's Administration of the Russian Federation between 2012 and 2018 with confirmed DVT and implanted IVC filter were included.
According to the local protocol, all patients suspected of DVT were assessed with a Wells score and D-dimer at admission. 18 Those with a high clinical probability or low clinical probability and positive D-dimer started low-molecular-weight heparin (LMWH) injections before DVT confirmation. Certified physicians performed the whole leg duplex ultrasound scan (DUS) within 6 h after admission. It was aimed to confirm DVT, detect its proximal extension, and reveal a free-floating head. According to the current National guidelines, a free-floating thrombus of ≥4 cm (or less in patients with a low cardiopulmonary reserve) was suggested as an indication for IVC filter implantation. This recommendation was based on limited evidence that assumes an increased risk of symptomatic PE in patients with a free-floating thrombus.19-22 The other indications were major bleeding or thrombus propagation in the background of therapeutic anticoagulation and catheter-directed thrombolysis (CDT). Some patients were admitted to the hospital with previously implanted IVC filters and DVT recurrence. Computed tomography pulmonary angiography (CTPA) was used to rule out PE in those with suspicious symptoms at admission.
During the first 2-10 days, patients received initial anticoagulation with therapeutic doses of LMWH (enoxaparin 1 mg/kg twice daily or equivalent) followed by oral anticoagulants (OACs). Before 2013, warfarin with a target international normalized ratio of 2.0-3.0 was prescribed to all patients, while after 2013, most patients received rivaroxaban (15 mg twice daily for 21 days and 20 mg once daily after that).
When a free-floating thrombus was observed, IVC filter implantation was performed in the background of therapeutic anticoagulation with LMWH, followed by OAC 2-3 days after the intervention. Serial DUS assessed thrombus propagation 3-5 days after admission or early without symptom relief. The main criterion was an extension of a proximal end of the thrombus for ≥5 cm from the primary point. In such a case, an IVC filter was implanted in parallel with changes in anticoagulant therapy: LMWH dose increased by 30% if heparin-induced thrombocytopenia was not suspected or fondaparinux administrated when it was supposed to be. If major bleeding occurred, therapeutic anticoagulation was temporarily ceased while an IVC filter was implanted. Treatment with LMWH was resumed within 3-7 days after achieving a stable hemostasis. CDT was performed in patients with iliofemoral DVT and severe symptoms of ≤14 days duration in the background of a 50% therapeutic dose of LMWH to reduce the risk of bleeding. At the time of observation, CDT was always accompanied by IVC filter implantation due to a lack of experience. Within 3-5 days after the intervention, LMWH was followed by OAC. When the attempt for filter removal was performed, patients were switched back to parenteral anticoagulation 2-5 days before the intervention and received them for 2-3 days after that.
The clinical provocation of the index DVT drove the total duration of anticoagulation. When thrombosis was provoked by a major transient (surgical) risk factor, it was limited for 3-6 months. In all other situations, it was prolonged indefinitely: DVT provoked by a minor transient (non-surgical, except oral contraceptives) risk factor, provoked by a minor or major persistent risk factor, unprovoked DVT, or recurrent VTE. In most cases, a not-retrieved IVC filter did not affect the total duration of anticoagulation except for individual circumstances.
All patients were followed up for 12 months or until death after discharge with control visits at 1, 3, 6, and 12 months when clinically examined for recurrent VTE and bleeding events. A DUS was performed to exclude recurrent DVT or IVC occlusion if suspected. Patients’ history between visits was also evaluated through medical records. All relevant information was documented for further extraction.
The study's primary efficacy endpoint was a symptomatic VTE recurrence confirmed by appropriate medical imaging (DUS or CTPA), including PE, DVT, and IVC occlusion. The primary safety outcome was major bleeding, according to the International Society on Thrombosis and Haemostasis criteria. 23 The secondary safety outcomes include clinically relevant non-major (CRNM) and minor bleeding. CRNM bleeding was defined as any bleeding that does not meet the criteria of major but requires discontinuation of anticoagulation, medical intervention, or unscheduled face-to-face consultation. Any other bleeding that does not meet the major or CRNM criteria was classified as minor. All study endpoints were registered since discharge, so the analysis did not include bleeding events and DVT propagation within a period of initial anticoagulation that required IVC filter implantation.
All treatments were performed as a part of routine clinical practice. Due to the retrospective character of the analysis, no additional informed consent was required, as confirmed by the Institutional Review Board of the Pirogov Russian National Research Medical University.
Statistical Analysis
Absolute values are represented as mean with standard deviation (M±σ), and relative values as percentages with a 95% confidence interval (CI) calculated by Wilson. A t-test was used to compare absolute values, and relative values were compared with the chi-squared test or two-tailed Fisher's exact test. The Kaplan-Meier curves with the log-rank test were used to compare outcomes. A propensity score matching (PSM) was performed via SPSS extension. The main clinical and demographical parameters (age, sex, side of the affected limb, proximal extension of DVT, PE at the admission, history of VTE, previous implantation of IVC filter, provocation of the index DVT, indication for an IVC filter implantation, filter retrieval, length of a free-floating head, duration of initial anticoagulation, total duration of anticoagulation) were entered as predictors for using different OACs, and a match tolerance of 0.5 was applied. The analysis was performed using SPSS software package version 26 (IBM Corp, Armonk, NY). The P-value of <.05 was considered statistically significant.
Results
From 2012 to 2018, 867 patients were admitted to the hospital with the suspicion of DVT; in 462, the diagnosis was confirmed, and 100 of them underwent implantation of an IVC filter and were included in the analysis. The age varied between 26 and 101 years (mean of 62.2 ± 17.5); 54 were men, and 46 were women. Left-sided thrombosis was found in 48, right-sided—in 47, and bilateral lesion—in 5 individuals. The proximal extension of the thrombus was limited by a popliteal vein in 19, a femoral vein in 20, a common femoral vein in 25, an iliac vein in 30, and reached IVC in six cases. PE, according to the suspicious symptoms, was revealed in 10 patients by CTPA. A history of previous DVT was reported in 17 individuals, of which 12 had an already implanted IVC filter without evidence of occlusion.
The index DVT was provoked by a major transient risk factor (recent surgery or trauma with immobilization) in 10, by a minor transient risk factor (oral contraceptives) in 4, by a minor persistent risk factor (congestive heart failure, obesity, chronic kidney disease, leg paralysis) in 46, by a major persistent risk factor (cancer) in 16 patients; it was considered unprovoked in 24 individuals.
The most common indication for IVC filter implantation was detecting a free-floating thrombus with a high risk of PE in 64 patients. The length of a free-floating head varied between 10 and 150 mm (mean of 50.1 ± 27.4). At the same time, acute bleeding during initial anticoagulation with LMWH, thrombus propagation, and CDT required intervention in eight patients for each indication. The last 12 patients had a previously implanted IVC filter with DVT recurrence.
Filters were implanted percutaneously via the femoral or jugular approach and located in the IVC below the lowest renal vein. The types of implanted filters were as follows: OptEase in 54, TrapEase in 24, ALN in 7, and Cook Celect Platinum in three cases. The type of previously implanted filter was not reliably known in the remaining 12 patients. After implantation, the filter was retrieved in 30 subjects between 14 and 56 days (mean of 24.7 ± 7.9).
The total duration of anticoagulant therapy was limited by 3 months in 8, lasted from 3 to 6 months in 6, and was extended up to 12 months or longer in 86 cases.
After an intervention, 41 patients were switched to warfarin, while 59 received rivaroxaban. The groups were comparable according to the main clinical characteristics (Table 1). The median length of a free-floating head was significantly higher in the rivaroxaban group because, in 2015, the revised National guidelines suggested a new indication for IVC filter implantation in a length of ≥7 cm. Also, fewer filters were implanted in the rivaroxaban group because of bleeding, thrombus propagation, and more due to CDT. It may be associated with a shorter period of initial anticoagulation with LMWH in a rivaroxaban group and CDT introduction after 2013. A significantly higher rate of IVC filter retrieval was observed in the rivaroxaban group because of the implementation of a local institutional policy in 2014.
The Main Demographical and Clinical Characteristics of Patients who Received Warfarin and Rivaroxaban After IVC Filter Implantation.
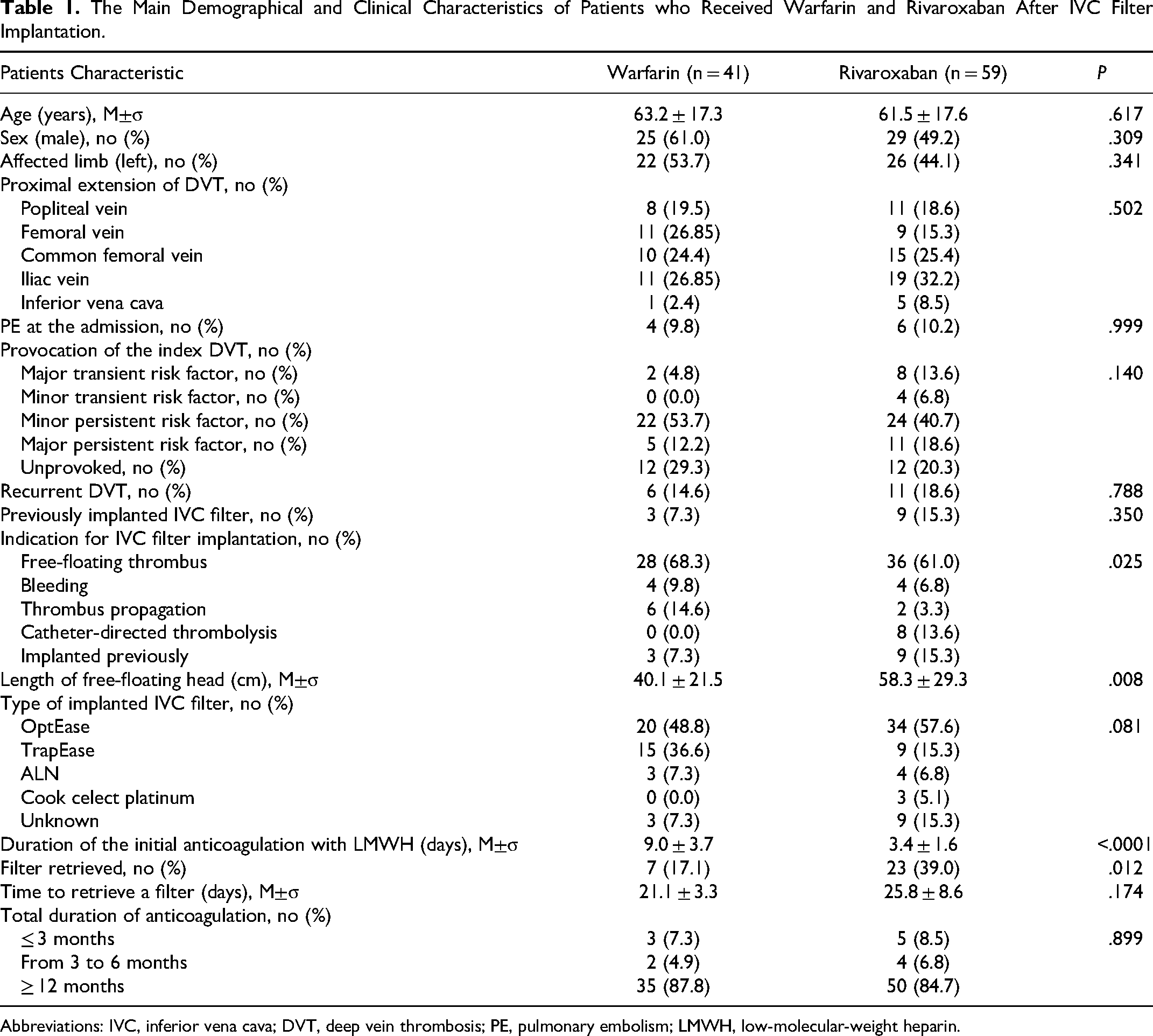
Abbreviations: IVC, inferior vena cava; DVT, deep vein thrombosis; PE, pulmonary embolism; LMWH, low-molecular-weight heparin.
All 100 patients were followed for 12 months or until death. Six of them were deceased at one (n = 1), three (n = 4), and six (n = 1) months after the intervention due to cancer progression (n = 5) or fatal PE (n = 1).
Recurrent VTE was detected in none cases in the rivaroxaban group and three (3.0%; 95% CI, 1.0-8.5%) cases in the warfarin group, of which two occurred on anticoagulation and one after discontinuation. They were represented by DVT in two patients and fatal PE in one patient. No evidence of IVC filter occlusion was observed in both groups. One episode of major intracranial hemorrhage was detected on warfarin (1.0%; 95% CI, 0.2-5.4%). CRNM bleeding occurred in three (3.0%; 95% CI, 1.0-8.5%) and minor bleeding in seven (7.0%; 95% CI, 3.4-13.8%) patients. Such CRNM bleeding as extensive skin bruising (n = 1), hemorrhoidal bleeding (n = 1), and hematuria (n = 1) occurred on rivaroxaban. Two and five episodes of minor epistaxis were observed on warfarin and rivaroxaban, respectively. No difference was found between groups by a direct comparison (Table 2). However, when Kaplan-Meier analysis was used, rivaroxaban appeared superior to warfarin regarding VTE recurrence, but not a combination of major and CRNM bleeding (Figures 1 and 2). A PSM produced two comparable groups of 21 patients without any differences (Table 2). As a part of a sensitive analysis, the outcomes were assessed in 70 patients without IVC filter retrieval, and no significant differences were found (Table 3).
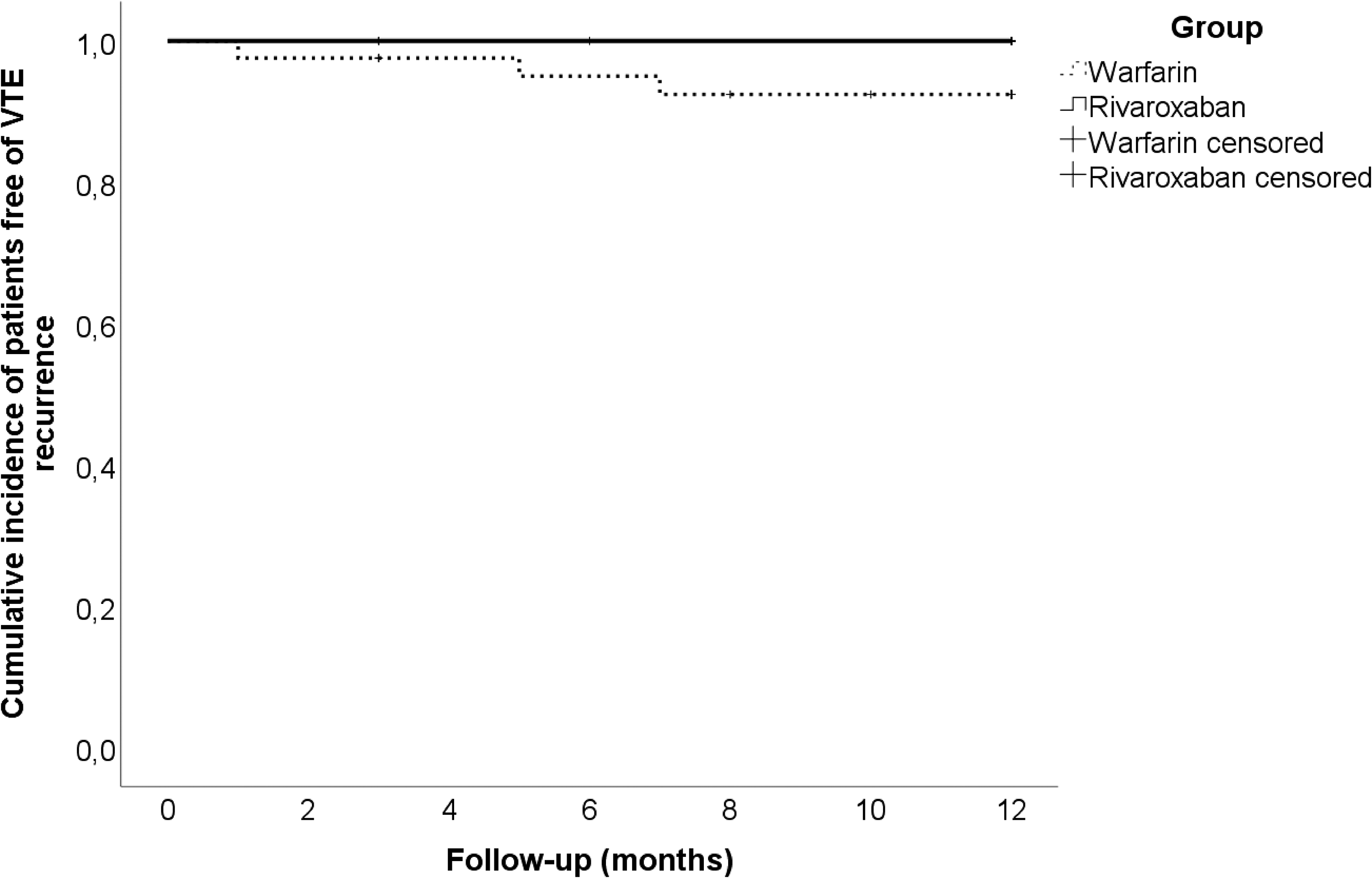
The Kaplan-Meier curves of symptomatic VTE recurrence in patients with IVC filter received rivaroxaban or warfarin (P = 0.035 by log-rank test). Abbreviations: IVC, inferior vena cava; VTE, venous thromboembolism.
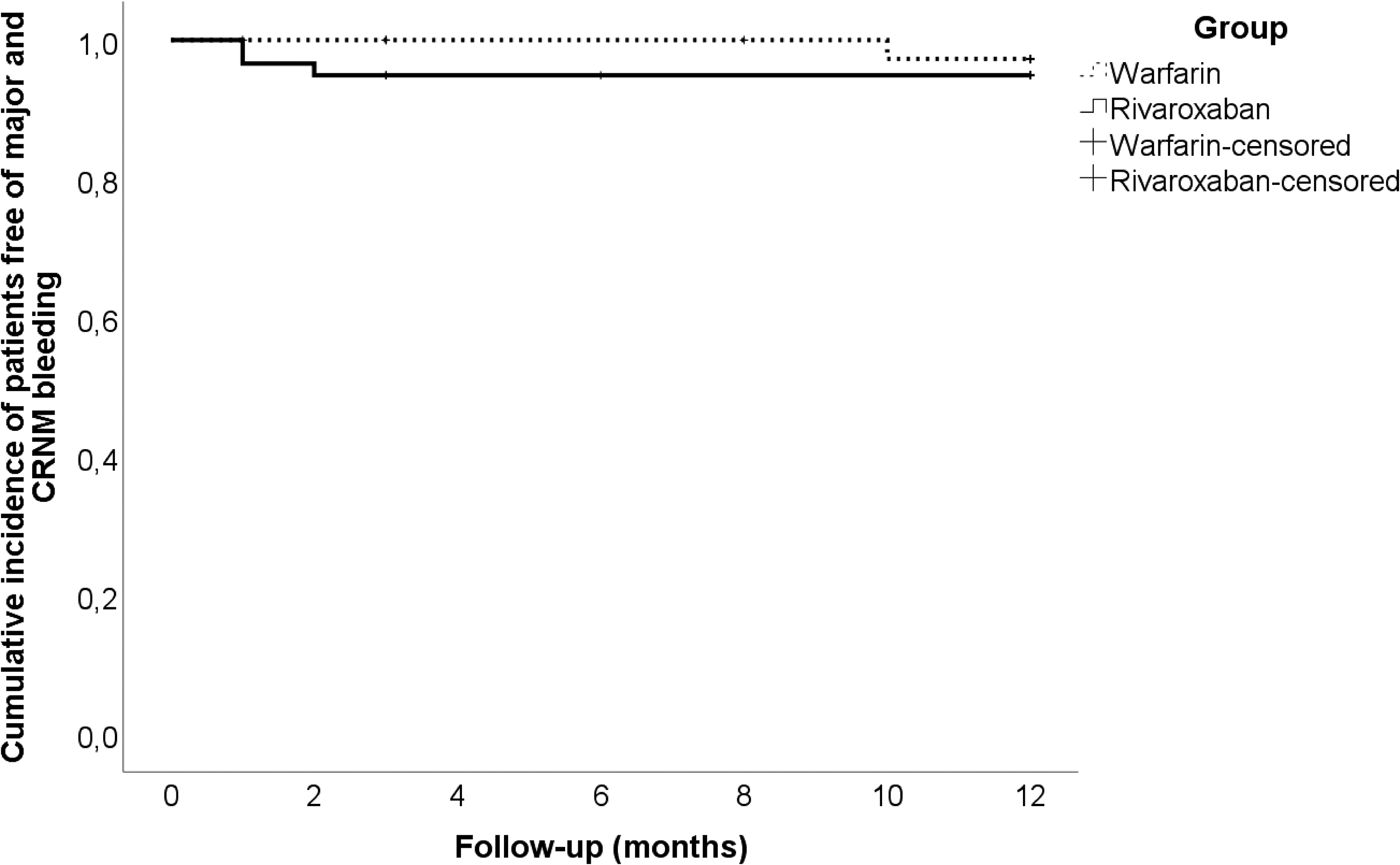
The Kaplan-Meier curves of major and clinically relevant non-major bleeding in patients with inferior vena cava (IVC) filter received rivaroxaban or warfarin (P = 0.512 by log-rank test).
Outcomes in Patients After Implantation of IVC Filter who Received Warfarin or Rivaroxaban.
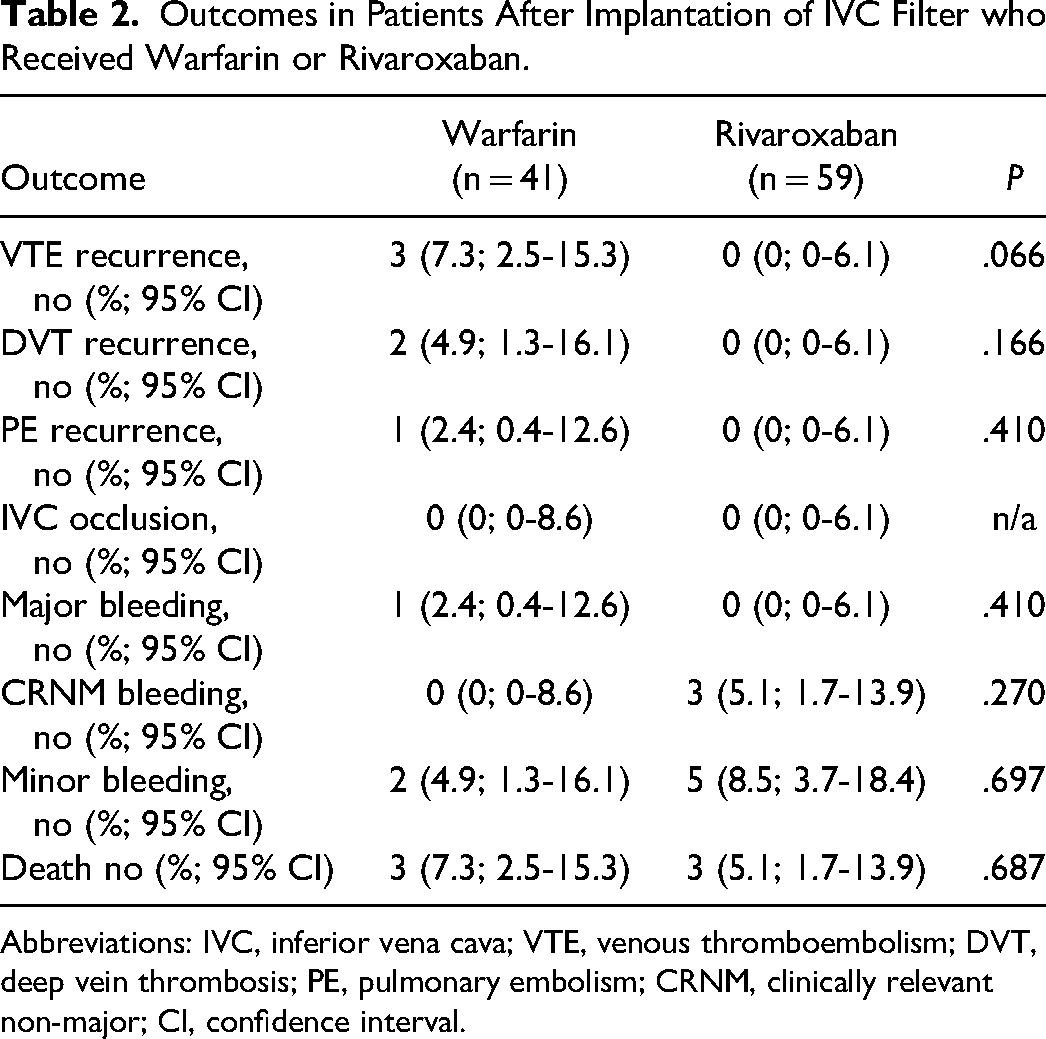
Abbreviations: IVC, inferior vena cava; VTE, venous thromboembolism; DVT, deep vein thrombosis; PE, pulmonary embolism; CRNM, clinically relevant non-major; CI, confidence interval.
Outcomes in Patients After Implantation of IVC Filter in PSM Cohort and Without Filter Retrieval.
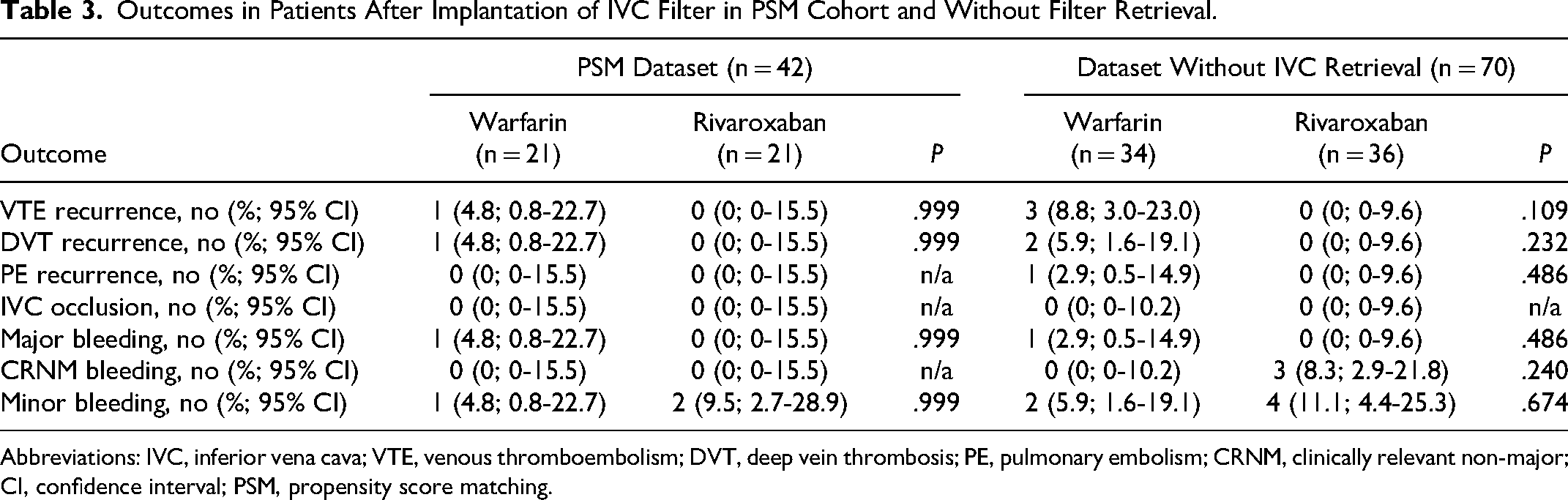
Abbreviations: IVC, inferior vena cava; VTE, venous thromboembolism; DVT, deep vein thrombosis; PE, pulmonary embolism; CRNM, clinically relevant non-major; CI, confidence interval; PSM, propensity score matching.
Discussion
Since the efficacy and safety of DOACs in comparison to warfarin were assessed in phase III randomized controlled trials, the patients after interventional treatment, including IVC filter implantation, were excluded. 15 Until recently, there was a gap in the evidence concerning the reliability of DOACs in such circumstances. Although the American College of Chest Physicians (ACCP) guidelines suggested the use of a conventional course of anticoagulation therapy if an IVC filter was inserted, many practitioners are concerned about the efficacy of DOACs and still prefer VKA. 24
In 2014, we reported outcomes in a limited number of 30 matched patients who received rivaroxaban or warfarin for 12 months after IVC filter implantation without evidence of recurrent VTE and IVC occlusion. 16 Recently, Wang B et al published outcomes in 202 patients with permanent IVC filters who received rivaroxaban or warfarin with a mean follow-up of about 60 months. 17 Authors found symptomatic filter thrombosis in 3.5% and 5.7% of patients who received rivaroxaban and warfarin, respectively, without significant differences. To our knowledge, no other studies compared DOACs and VKA after IVC filter implantation. One randomized controlled trial was designed to solve this issue but still has no outcomes. 25
Here, we present an extended analysis of patients who underwent IVC filter implantation in our institution and received rivaroxaban or warfarin afterward. In opposition to other studies, we did not see any filter thrombosis or IVC occlusion, previously reported in 2-30%. 26 The reason for such differences may be related to specific indications for filter implantation, type of devices, and limited follow-up.
For a long time in Russia, the main indication for IVC filter insertion was a free-floating thrombus, considered a high risk of symptomatic and fatal PE despite the absence of clear evidence. The earlier studies assumed an increased risk of primary and recurrent PE in patients with a free-floating thrombus, especially with a length of >5 cm.19-22,27 However, the other studies did not confirm this evidence.28-30 The controversial results were observed in the recent retrospective cohort study that found an increased risk of PE in occlusive thrombus when compared to free-floating (15.8% vs 6.7%), while embolism in IVC filter was detected more often with a free-floating head of >5 cm (40% vs 0%). 31 In 2010, National guidelines considered IVC filter implantation at a length of the free-floating head of ≥4 cm; in 2015, it was increased to ≥7 cm, and today, this parameter is no longer emphasized. The actual National and International guidelines support IVC filter implantation only in the case of active bleeding, which does not allow therapeutic anticoagulation and thrombus propagation despite adequate treatment.32-34 Patients with complications of conservative therapy compared to those representing free-floating thrombus may have more co-morbidities, particularly active cancer, which negatively impact the long-term outcomes of IVC filter implantation. In the current study, there was a lack of patients with thrombus propagation and contraindications for anticoagulation.
The number of device-related complications is also related to the type of IVC filter. 35 Particularly, when the retrievable filters are not removed, the complication rate appears higher than in permanent filters. 36 In the current study, the predominant type of filter was OptEase, with a low retrieval rate of 30%. However, this fact did not impact the long-term outcomes.
The crucial question is whether patients with permanent IVC filters need prolonged (life-long) anticoagulation. The current evidence suggests that anticoagulation does not impact the rate of VTE recurrence after IVC filter implantation. 37 Considering this fact and ACCP guidelines, we tried to limit the duration of therapeutic anticoagulation in those who had an index DVT provoked by a major or minor (oral contraceptive pills) transient risk factor. No recurrent VTE was observed among 14 patients who discontinued anticoagulation after 3-6 months. The only recurrent DVT without IVC occlusion was detected in a patient with an unprovoked index thrombosis who stopped taking warfarin after 6 months. However, we did not reduce the rivaroxaban dose to 10 mg for those who continued anticoagulation because such an indication was not registered at the time of observation. In contrast, Wang B et al used a reduced dose of rivaroxaban when they observed a 3.5% rate of filter occlusion. 17
The other side of prolonged anticoagulation is a bleeding risk. Sakai R et al reported 6 symptomatic DVT recurrences and 13 bleeding events in 158 patients with non-retrieval IVC filters within a mean follow-up of 5 years. 38 However, on rivaroxaban, the risk of major bleeding is lower than on VKA. 15 In the study of Wang B et al, the only major gastrointestinal bleeding was reported on rivaroxaban, while in the current study it was not observed. 17 Concerning CRNM bleeding, the figures in the current study were comparable with the previous one but without significant differences with warfarin. Potentially, it is associated with the use of full-dose rivaroxaban in contrast to its reduction after 6 months and the use of an alternative definition for CRNM bleeding.
In summary, the contemporary guidelines suggest only two main indications for IVC filter implantation: absolute contraindication to anticoagulation (predominantly active bleeding) and VTE recurrence despite therapeutic anticoagulation. In contrast, routine use of IVC filters in anticoagulated patients is not recommended, irrespective of the presence of a free-floating thrombus. The characteristics of the index VTE should drive anticoagulation's type, dose, and duration but not depend on the presence of an IVC filter.24,32-34
Limitations
The main limitation of this study is a historical control group of patients treated with warfarin, which raises a time bias. This suggests possible differences in standard care, accessibility to treatments, and variations in disease prevalence that may affect the outcomes and could not be overcome by PSM. The other significant limitation is that it is a relatively short follow-up related to the institutional policy that requires obligatory patient assessment during the first year after discharge. Also, the sample is relatively small to provide statistically significant differences and make a robust PSM. Patients on warfarin did not have reliable information on the dynamic of the international normalized ratio, so it could not be extracted for analysis.
Conclusion
Full-dose oral rivaroxaban seems not less effective and safe than warfarin in patients with DVT after the implantation of an IVC filter. Robust randomized controlled trials are required to confirm this statement.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
