Abstract
Introduction:
Over the past few years, direct oral anticoagulants (DOACs) have been gradually replacing warfarin. Inappropriate prescribing of DOACs in real-life practice settings can affect their perceived safety and efficacy, especially with the lack of a surrogate marker for guidance.
Objectives:
To describe the appropriateness of DOACs prescribing, compare dabigatran to rivaroxaban in terms of inappropriate prescribing, and determine other factors associated with inappropriate DOACs use.
Methods:
In this cross-sectional retrospective study, 5-year DOAC prescriptions data were extracted. Appropriateness was evaluated based on approved dosing and indications in Canada and the United States. Descriptive and inferential statistics were performed using SPSS.
Results:
From 2011 to 2015, there were 1049 DOACs prescriptions, among which 572 (54.5%) were for dabigatran and 477(45.5%) were for rivaroxaban. The DOACs were prescribed for inappropriate indication in 35 (3.3%) patients, while inappropriate dosing was found in 352 (33.6%) prescriptions. There were significantly more inappropriate dabigatran prescriptions compared to rivaroxaban both in terms of indication (4.7% vs 1.7%, P = .004) and dosing (50.9% vs 12.8%, P < .001). Logistic regression analysis confirmed that dabigatran prescribing was the only factor associated with inappropriate indications (odds ratio [OR] = 2.9, 95% confidence interval [CI]: 1.3-6.5). Factors associated with inappropriate dosing included dabigatran prescriptions (OR = 7.6, 95% CI: 5.5-10.5) and poor renal function (OR = 14.6, 95% CI: 3.6-58.4).
Conclusion:
Direct oral anticoagulants have been gradually replacing warfarin in Qatar; however, they are not always prescribed appropriately especially in patients on dabigatran and those with renal impairment. Educating health-care practitioners is necessary. Future studies comparing the clinical safety and effectiveness of the DOACs especially when used at an inappropriate dose are also warranted.
Introduction
Oral anticoagulation therapy is used to prevent thromboembolism in patients with a variety of disorders, including atrial fibrillation, prosthetic heart valves, coronary artery disease, and venous thromboembolism (VTE). 1,2 Since the 1940s, warfarin remained as the mainstay oral anticoagulant; however, a reshape in the oral anticoagulation landscape occurred when direct oral anticoagulants (DOACs) were introduced to the market in 2010. 3,4 Dabigatran, which acts by directly and selectively inhibiting thrombin, was the first approved DOAC and was then followed by 3 factor Xa inhibitors (rivaroxaban, apixaban, and edoxaban). 5 -7 Compared to warfarin, DOACs have more predictable therapeutic effect with a fixed-dose regimen, cause less intracranial bleeding, do not require routine monitoring, and have less drug–drug and drug–food interactions. 8 -10 These benefits should also be considered in light of some potential disadvantages such as the increase in gastrointestinal side effects (especially for dabigatran and rivaroxaban), lack of antidote (except for dabigatran), contraindication in patients with major renal dysfunction, and lack of superiority when compared to patients with well-managed warfarin therapy. 9 -12 Lastly, the cost of DOACs compared to warfarin may be prohibitive for some patients. 13 All current practice guidelines include DOACs as first-line or alternative treatment for VTE treatment and prophylaxis as well as for stroke prevention in atrial fibrillation. 14 -16 Since their approval, DOAC use started to rise and they are now widely adopted by clinicians. 4,17 -21 Although DOACs were shown to have comparable efficacy and superior safety compared to warfarin in phase 3 trials, 9,22 -26 factors such as adherence and appropriate utilization may affect their perceived efficacy and safety. What makes these factors even more important in DOACs is the lack of monitoring parameter and/or surrogate marker to indicate their therapeutic level. For a DOAC regimen to be appropriate, factors such as indication, renal function, and age have to be considered. To date, there is little knowledge on how appropriately DOACs are prescribed and what factors may lead to inappropriate prescribing of these agents.
In this study, we aim to evaluate DOAC appropriate use, compare dabigatran to rivaroxaban in terms of inappropriate prescribing, and determine other factors associated with inappropriate DOAC use.
Methods
Design and Setting
The study design was a retrospective cross-sectional study aiming to collect information on the appropriate usage of DOACs at Hamad Medical Corporation (HMC) and the Primary Health Care Corporation (PHCC) over the past 5 years. The HMC is the principal public health-care provider in the State of Qatar that encompasses 8 hospitals. The PHCC, on the other hand, comprises 21 ambulatory care health centers. Both HMC and PHCC serve more than 90% of patient needs in Qatar. At HMC, majority of anticoagulation follow-up is performed at specialized outpatient anticoagulation clinics run by either pharmacists or physicians. These clinics serve both patients on warfarin and those on DOACs. Description of the anticoagulation clinics in Qatar, its quality outcomes, and how pharmacist-managed is compared to physician-managed can be found in more details in the studies by Elewa et al. 27,28
Data Collection and Study Participants
At HMC and PHCC, dabigatran was added to the formulary in 2011 followed by rivaroxaban in 2014. To date, apixaban is a nonformulary medication and its use is very limited at HMC and PHCC. In this study, we collected reports from electronic medical records on both DOACs (dabigatran and rivaroxaban) dispensed as inpatient or outpatient prescriptions from 2011 to 2015 at HMC and PHCC. Prescriptions were stratified by the year and the DOAC agent. The following information was extracted from the reports: DOAC name and regimen; indication for anticoagulation; and patient demographics and baseline characteristics including age, gender, country of origin, renal function, liver function, concomitant use of antiplatelets, and other interacting medications. Each patient’s prescription was counted as a unit regardless of the number of refills for this patient except if the DOAC regimen was changed. Patients were eligible for inclusion if they were dispensed DOACs for at least 3 days from 2011 to 2015 at HMC and/or PHCC with an indication retrieved in the records.
Outcomes Measured
Outcomes included the prevalence of inappropriate prescribing of DOACs based on dosing and indication according the American and the Canadian labeled indications (Table 1). We also compared the inappropriate prescribing of rivaroxaban to dabigatran and the factors associated with inappropriate prescribing of DOACs.
Dabigatran and Rivaroxaban Indications and Dosing Based on the American and the Canadian Labeling.

Abbreviations: AF, atrial fibrillation; CrCl, creatinine clearance; DVT, deep venous thrombosis; PE, pulmonary embolism; VTE, venous thromboembolism.
Statistical Analysis
Both descriptive and inferential statistical analyses were applied for the collected data using IBM Statistical Package for Social Sciences (IBM SPSS 23 software). Categorical variables were expressed as frequencies and percentages, while continuous variables were expressed as means (standard deviation). Baseline characteristics were compared using either t test for continuous variables or χ2 test for categorical variables. Inappropriateness of rivaroxaban and dabigatran was compared using χ2 test. Multiple logistic regression test was used to assess factors associated with inappropriate prescribing of DOACs. A level of significance was set a priori at P < .05.
Ethics Approval
The institute of research board at Qatar University and HMC approved the study protocol.
Results
From 2011 to 2015, there were 1112 DOAC prescriptions (63 of which were excluded because of the inability to retrieve the indication on the medical records, which makes it very challenging to assess appropriateness). Among the remaining 1049 patients using DOACs, 477 (45.5%) used rivaroxaban while the remaining 572 (54.5%) were on dabigatran.
Patients prescribed rivaroxaban were younger than those in the dabigatran group, 53.1 (16) years versus 61.8 (14) years (P < .001), although there were significantly larger number of patients older than 75 years in the dabigatran group compared to the rivaroxaban, 84 (14.7%) versus 43 (9%), P < .001. Similarly, there were more patients with normal renal function in the rivaroxaban group, 437 (91.6%) versus 496 (86.7%), P < .001, although the number of patients with poor renal function (defined as creatinine clearance <30 mL/min) was larger in the rivaroxaban group compared to dabigatran, 8 (1.7%) versus 5 (0.9%), P < .001. More than 80% of patients using dabigatran had atrial fibrillation as the primary indication compared to 40% of patients prescribed rivaroxaban, while only 4% of the dabigatran group had VTE treatment as the primary indication compared to 44.2% in the rivaroxaban group. The remaining indications for DOAC prescriptions were for VTE prophylaxis or off-label use for other conditions. Table 2 summarizes the demographics of all patients prescribed DOACs included in the study.
Demographics and Baseline Characteristics of Patients on DOACs From 2011 to 2015.
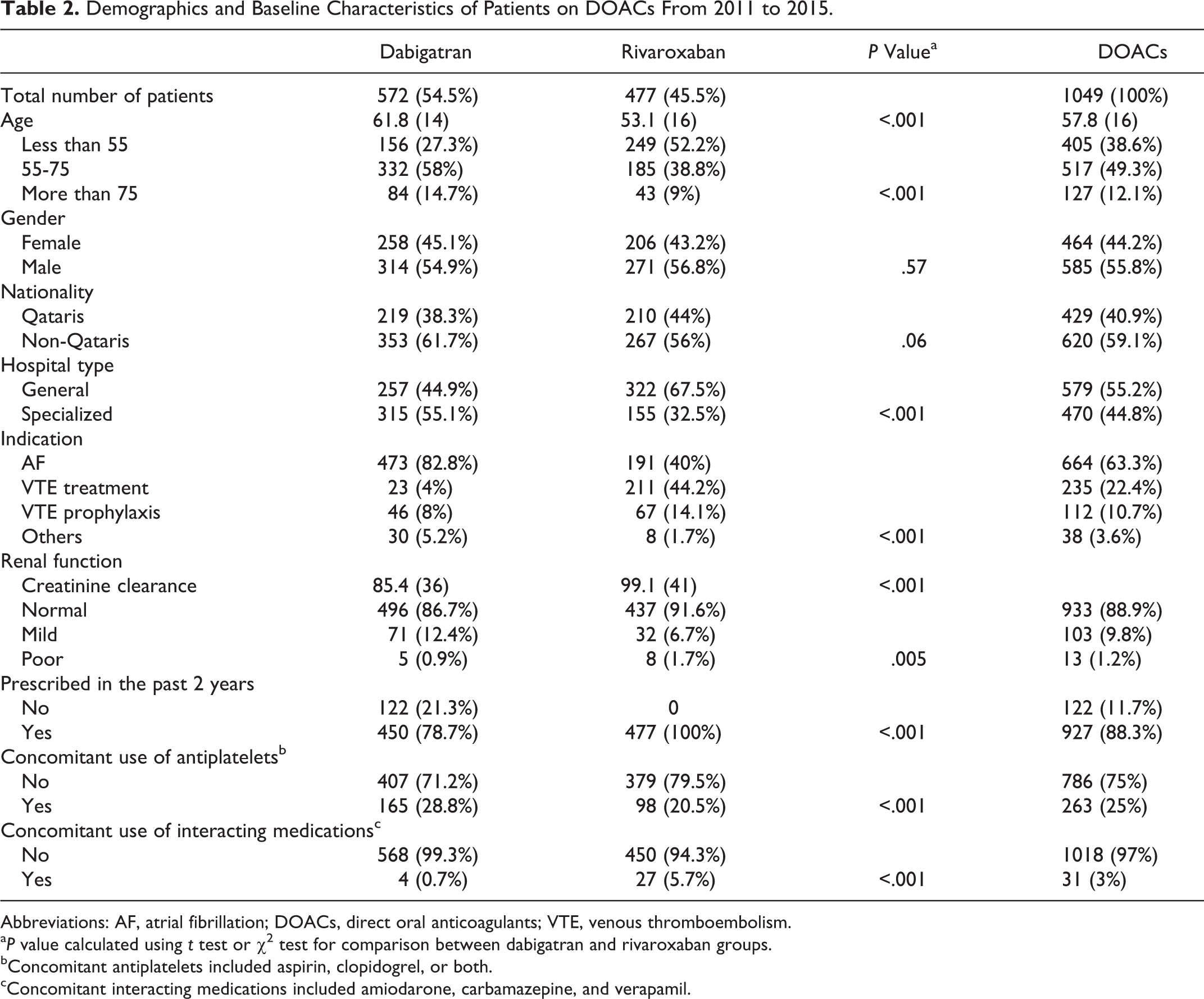
Abbreviations: AF, atrial fibrillation; DOACs, direct oral anticoagulants; VTE, venous thromboembolism.
aP value calculated using t test or χ2 test for comparison between dabigatran and rivaroxaban groups.
bConcomitant antiplatelets included aspirin, clopidogrel, or both.
cConcomitant interacting medications included amiodarone, carbamazepine, and verapamil.
Direct oral anticoagulants were prescribed with inappropriate dosing in about one-third of the patients (33.6%); however, it was mostly prescribed for the appropriate labeled indications (96.7%). When comparing inappropriate use of dabigatran versus rivaroxaban, we found that there were significantly larger number of inappropriate dabigatran prescriptions compared to rivaroxaban prescriptions both in terms of dosing (50.9% vs 12.8%, P < .001) and indication (4.7% vs 1.7%, P = .004; Figure 1). A stepwise logistic regression analysis indicated that the main factors associated with inappropriate dosing are the use of dabigatran (odds ratio [OR] = 7.6, 95% confidence interval [CI]: 5.5-10.5) and the poor renal function (OR = 14.6, 95% CI: 3.6-58.4). Additionally, the use of dabigatran was the only factor associated with inappropriate indication (OR = 2.9, 95% CI: 1.3-6.5; Table 3). Examples of inappropriate dosing included the use of wrong dose, wrong dosing interval, or both, while inappropriate indications included the use for antiphospholipid antibody syndrome, coronary artery disease, and pulmonary hypertension.

A, Direct oral anticoagulant (DOAC) dosing appropriateness: percentage of appropriate (light gray) versus inappropriate (dark gray) dose of DOAC prescriptions. B, Comparison of dosing appropriateness between dabigatran and rivaroxaban: percentage of prescriptions with appropriate (light gray) and inappropriate (dark gray) dose of rivaroxaban versus dabigatran. *P < .001 using χ2 test. C, The DOAC indication appropriateness: percentage of appropriate (light gray) versus inappropriate (dark gray) DOAC indication prescriptions. D, Comparison of indication appropriateness between dabigatran and rivaroxaban: percentage of prescriptions with appropriate (light gray) and inappropriate (dark gray) indication of rivaroxaban versus dabigatran. *P = .004 using χ2 test.
Factors Associated With Inappropriate Prescribing of DOACs.

Abbreviations: CI, confidence interval; DOACs, direct oral anticoagulants; OR, odds ratio.
Discussion
This research work indicates that DOACs are frequently prescribed at inappropriate dose, especially in patients with impaired renal function and in those prescribed dabigatran. The association with poor renal function is logical since precise dose adjustments are required in patients with low creatinine clearance in whom dabigatran or rivaroxaban are prescribed. Prescribers, however, did not always follow these dose adjustments appropriately. On the other hand, our finding regarding the presence of more inappropriate dabigatran prescriptions than rivaroxaban is not well understood. It could be interpreted, however, by the overuse of the dabigatran 110 and 75 mg in patients with atrial fibrillation who are not older than 80 years or with creatinine clearance between 15 and 30 mL/min, respectively. In fact, one may argue that the use of dabigatran 110 mg twice daily may be a suitable option for stroke prevention in patients with high bleeding risk since it was shown to reduce major bleeding while maintaining similar rates of stroke and systemic embolism when compared to warfarin in the Randomized Evaluation of Long-Term Anticoagulation Therapy (RE-LY) trial. 9 However, this strategy should be discouraged since it may be associated with higher rates of stroke and systemic embolism which have greater clinical significance than nonfatal bleeding. Additionally, subgroup analysis in the RE-LY looking at patients with high risk of bleeding such as those older than 75 years, with moderate renal impairment (creatinine clearance >30-50 mL/min), or previous bleeding was unable to find any improvement in the dabigatran’s benefit–risk ratio when using the 110 mg dabigatran dose. 29
It would be interesting to conduct an observational study on this cohort of patients to assess their stroke and bleeding risk compared to those using dabigatran at the appropriate dose. In alignment with this report, 2 recent studies conducted in the United States evaluated DOAC appropriateness and found that the dosing of DOACs was not optimal in 25% and 14% of the studied populations, respectively. 30,31 Similar to these studies, we found that baseline laboratory values were not always assessed prior to DOAC initiation, especially for the liver and renal function.
Our study must be taken in the context of several limitations. First, although these data likely represent over 90% of DOACs use in Qatar, it excluded private hospitals and community pharmacies, which may affect the generalizability of our results to the whole state of Qatar or to other countries. Further, the data captured from the medical records may have errors or inconsistencies that cannot be accounted for such as indication, risk of bleeding, or other factors that could at least partially explain the used dose in the DOAC prescriptions. The study did not investigate the correlation between inappropriate use of DOACs on clinical outcomes. Lastly, apixaban and edoxaban are not represented in this study, since edoxaban is not yet approved for use in Qatar while apixaban use is very limited at HMC and PHCC since it is nonformulary.
In conclusion, DOACs have been rapidly adopted in clinical practice; however, they are not always prescribed appropriately. Patients most likely to get an inappropriate prescription include those with poor renal function and patients prescribed dabigatran. Educating health-care practitioners is necessary to improve the appropriate use of DOACs. Future studies comparing the clinical safety and effectiveness of the DOACs especially when used at an inappropriate dose are also warranted.
Footnotes
Author Contributions
Hazem Elewa contributed to conception and design, contributed to interpretation, drafted the manuscript, and critically revised the manuscript. Honain El-Makaty contributed to data collection, interpretation, drafted the manuscript, and revised the manuscript. Zainab Ali contributed to data collection, interpretation, drafted the manuscript, and revised the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Qatar University internal grant # (QUST-CPH-SPRING-15/16-9).
