Abstract
Thrombophilia in venous thromboembolism (VTE) is multifactorial. Von Willebrand factor (vWF) plays a major role in primary hemostasis. While elevated vWF levels are well documented in VTE, findings related to its cleaving protease (ADAMTS-13) are contradicting. The aim of this study was to determine vWF, ADAMTS-13, and the multifactorial Thrombospondin-1 (TSP-1) protein levels in patients after 3-6 months following an unprovoked VTE episode. We also explored a possible association with factor V Leiden (FVL) mutation. vWF, ADAMTS-13 and TSP-1 were analyzed using ELISA kits in 60 VTE patients and 60 controls. Patients had higher levels of vWF antigen (P = .021), vWF collagen-binding activity (P = .008), and TSP-1 protein (P < .001) compared to controls. ADAMTS-13 antigen was lower in patients (P = .046) compared to controls but ADAMTS-13 activity was comparable between the two groups (P = .172). TSP-1 showed positive correlation with vWF antigen (rho = 0.303, P = .021) and negative correlation with ADAMTS-13 activity (rho = −0.244, P = .033) and ADAMTS-13 activity/vWF antigen ratio (rho = −0.348, P = .007). A significant association was found between the presence of FVL mutation and VTE (odds ratio (OR): 9.672 (95% confidence interval (CI) 2.074-45.091- P = .004), but no association was found between the mutation and the studied proteins (P > .05). There appears to be an imbalance between vWF and ADAMTS-13 in VTE patients even after 3-6 months following the onset of VTE. We report that the odds of developing VTE in carriers of FVL mutation are 9.672 times those without the mutation, but the presence of this mutation is not associated with the studied proteins.
Keywords
Introduction
Venous thromboembolism (VTE) is a major health concern affecting thousands of individuals worldwide, with incidence rates differing by age, sex, and race.1,2 It encompasses two conditions: deep vein thrombosis (DVT) and pulmonary embolism (PE). The development of VTE is attributed to several acquired and inherited predisposing factors most of which increase blood coagulability and contribute to the formation of a thrombus. 3
In 1994, the factor V Leiden (FVL) mutation was characterized as a point mutation in the factor V gene (G1691A). This mutation leads to a change of arginine to glutamine at the amino acid 506 of the factor V molecule, which is one of the cleaving sites of factor V, used by the natural anticoagulant-activated protein C (APC) to inactivate factor V. 4 The presence of FVL mutation renders factor V resistant to cleavage by APC, which leads to factor V accumulation causing a prothrombotic environment and hypercoagulability, and this may eventually manifest clinically as VTE. 5
Among other coagulation proteins, von Willebrand factor (vWF) has been reported to play a significant role in venous thrombus formation.6,7 vWF is a multimeric glycoprotein synthesized in megakaryocytes and vascular endothelial cells, and functions as a moderator of platelets adhesion to the endothelium and to each other upon vessel wall injury. 8 Activated endothelial cells release high quantities of Ultra Large vWF (ULvWF) multimers, capable of spontaneously binding platelets and erythrocytes. 9 Several studies reported increased levels of vWF and factor VIII in VTE and thus were considered as possible risk factors for this disorder.10–12
Normally, vWF multimers are slowly and constantly cleaved by a plasma protease that degrades them into smaller multimers and prevents the formation of blood clots in normal circulation. 13 This protease was found to cleave the ULvWF multimers by cleaving the peptide bond between tyrosine at position 842 and methionine at position 843 in susceptible A2 domains of vWF monomeric subunits. In 2001, Zheng et al identified this protease as a new member of the ADAMTS (a disintegrin and metalloprotease with Thrombospondin-1 (TSP-1) repeats) family of metalloprotease and was named ADAMTS-13. 14
Another important regulator of vWF is the multifunctional glycoprotein TSP-1, which is released from platelets’ alpha-granules in response to activation by thrombin. 15 TSP-1 was reported to regulate the multimeric size of vWF and hence its hemostatic activity. 16 It was also reported that TSP-1 competitively binds to vWF cleaving sites causing inhibition of ADAMTS-13 mediated proteolysis of vWF.17,18 This protective mechanism by TSP-1 secures the attachment of a developed thrombus to damaged vessels in order to stop bleeding. 18
Maintaining a balance between vWF and its cleaving protease (ADAMTS-13) is important in maintaining hemostasis and normal blood flow. Deficiencies of ADAMTS-13 levels have been documented in many clinical situations including thrombotic thrombocytopenic purpura, heparin-induced thrombocytopenia, sepsis, immune thrombocytopenic purpura, and leukemia.19–23 The imbalance between vWF and ADAMTS-13 leads to accumulation of vWF which can induce a hypercoagulable environment and increase the risk of thrombus formation.
While levels of vWF are well documented in patients with VTE,24–28 studies about the expression of ADAMTS-13 protease are limited. In 2012, Mazetto et al reported increased levels of both vWF and ADAMTS-13 in VTE patients, stating that “high ADAMTS-13 levels represent a compensatory mechanism against persistently increased levels of vWF.” 29 More recent studies reported contradicting findings with VTE patients presenting with high vWF and significantly lower ADAMTS-13 levels.30,31
In regard to TSP-1, Riedl et al (2016) reported that sP-selectin, but not TSP-1, was associated with risk of VTE in cancer patients, 28 while Fujimura et al (2022) reported that the release of TSP-1 enhances the block on ADAMTS-13 function in COVID-19-related thrombosis. 32
The TSP-1, vWF, and ADAMTS-13 interaction had been investigated in several pathological conditions including sickle cell disease,33,34 cardiovascular disease 35 , and thrombosis related to COVID-19 32 but not in unprovoked VTE.
In this study, we proposed that levels of vWF, ADAMTS-13, and TSP-1 would return to normal and become comparable to those in health subjects after 3-6 months of the onset of a VTE episode. A different outcome may suggest continued endothelial activation that may possibly expose patients to new episodes. To test this hypothesis, we determined levels of vWF, ADAMTS-13 and TSP-1 in patients of Arab ethnicity in our local population after 3-6 months of the onset of unprovoked VTE. We also explored a possible correlation of TSP-1 with vWF and ADAMT-13 proteins, which would clarify the interaction of TSP-1 with these two proteins in VTE. An association between the presence of FVL mutation and vWF, ADAMTS-13, and TSP-1 was also evaluated.
Subjects and Methods
Subjects and Sample Collection
This study was conducted on 60 adult patients (36 males and 24 females) with unprovoked VTE (DVT or PE) and 60 normal healthy controls (23 males and 37 females). All subjects were of Arab ethnicity living in Kuwait.
The study was approved by the Kuwait University Health Sciences Center ethical committee, and the Kuwait Ministry of Heath ethical committee. Informed, signed consent was obtained from all participating subjects.
Patients’ blood samples were collected after at least 3 months of acute VTE onset and commencement of anticoagulant therapy. At the time of blood collection, all were receiving vitamin K antagonist therapy and on regular INR monitoring as part of their post-VTE follow-up. The diagnosis of VTE was based on clinical pre-probability scores, D-dimer assay, and radiological imaging. Patient exclusion criteria included smoking, pregnancy, malignancy, acute or chronic inflammation, major surgery and liver or kidney diseases.
Most of the control groups were volunteers in good health. Additionally, some healthy individuals who were receiving routine checkups at hospital outpatient clinics were enrolled.
Two blood samples were collected from each participant in the study: one in sodium citrate (3.8%) Becton Dickinson vacuum collection tube for the determination of vWF, ADAMTS-13, and TSP-1 using ELISA, and another sample in K3-EDTA (15%) Becton Dickinson vacuum collection tubes for DNA extraction and FVL mutation detection.
Plasma separated from sodium citrate tubes were stored at −20 °C until processed, while blood samples collected in EDTA were frozen (−20 °C) until DNA extraction and analysis.
vWF, ADAMTS13, and TSP-1 Analysis
Standard ELISA methodology was applied to determine levels of vWF, ADAMTS-13, and TSP-1 according to the manufacturer's guidelines. Kits for the determination of vWF Antigen (vWF:Ag), vWF: Collagen-Binding Activity (vWF: CBA), ADAMTS-13 antigen, and ADAMTS-13 activity were purchased from Technoclone, Vienna, Austria; while TSP-1 ELISA kits were from R&D Systems, Inc. Minneapolis, MN, USA. For all kits used, a series of five-seven standards (prepared as dilutions of a concentrated standard) were prepared to create a standard curve prior to patients’ and controls’ analysis. The standard curve was used to extrapolate the results of the unknown patients and healthy controls samples. Absorbance was measured at 450 nm using Clariostar microwell reader system (Clariostar (V.5.40), BMG Labtech, USA).
FVL Analysis
EDTA blood samples were used to extract nuclear DNA using QIAGEN kits for DNA extraction and purification from blood samples (Qiagen Company, Hilden, Germany), according to the manufacturer's instructions. For each DNA sample, real-time PCR was performed to detect the presence of FVL mutation (homozygous or heterozygous status) using specially designed primers and probes for the FVL (Solis BioDyne, Tartu, Estonia), following the manufacturer's protocol. Real-time PCR was performed on 7500 Fast Real-Time PCR System from Applied Biosystems (Foster City, CA, USA).
Statistical Analysis
Data were analyzed using statistical package for social sciences (SPSS; PC version 26.0, SPSS Inc. Chicago, IL, USA). Data were tested for normality using the Kolmogorov-Smirnov test. Parameters between patients and controls were compared using non-parametric Mann-Whitney Test and descriptive statistics are presented as median and interquartile range. For comparisons of more than two variables, the Kruskal-Wallis Test was performed. Spearman's rho correlation coefficient was used to describe the association between TSP-1 with vWF and ADAMTS-13. ADAMTS-13 activity/ vWF antigen ratio was obtained by dividing values of ADAMTS-13 activity by vWF antigen levels.
The association between FVL mutation and VTE was determined by calculating the odds ratio (OR) and 95% confidence interval (CI) using binary logistic regression. When the association was tested between the mutation and each tested protein, a cut-off point was determined (median levels of each protein was selected as a cut-off point). A two-tailed P-value <.05 was considered statistically significant.
Results
There was no statistically significant difference in age between patients [median (interquartile range): 46 years (34.75-54)] and controls [median (interquartile range): 44 years (35.25-53.75)] - (P = 0.746). When comparing ABO distribution, we report higher percentage of O blood group in control subjects (47%) compared to patients (12.5%) - (P = 0.006).
ELISA results showed higher levels of vWF:Ag (P = .021), vWF: CBA (P = .008) and TSP-1 antigen (P < .001) in patients compared to controls. On the contrary, ADAMTS-13 antigen levels were significantly lower in patients (P = .046), while ADAMTS-13 activity levels were not different between patients and controls (P = .172) (Table 1).
vWF, ADAMTS-13 and TSP-1 Levels in Venous Thromboembolism (VTE) Patients and Controls (C). Results are Expressed as Median (25th −75th Interquartile Range).
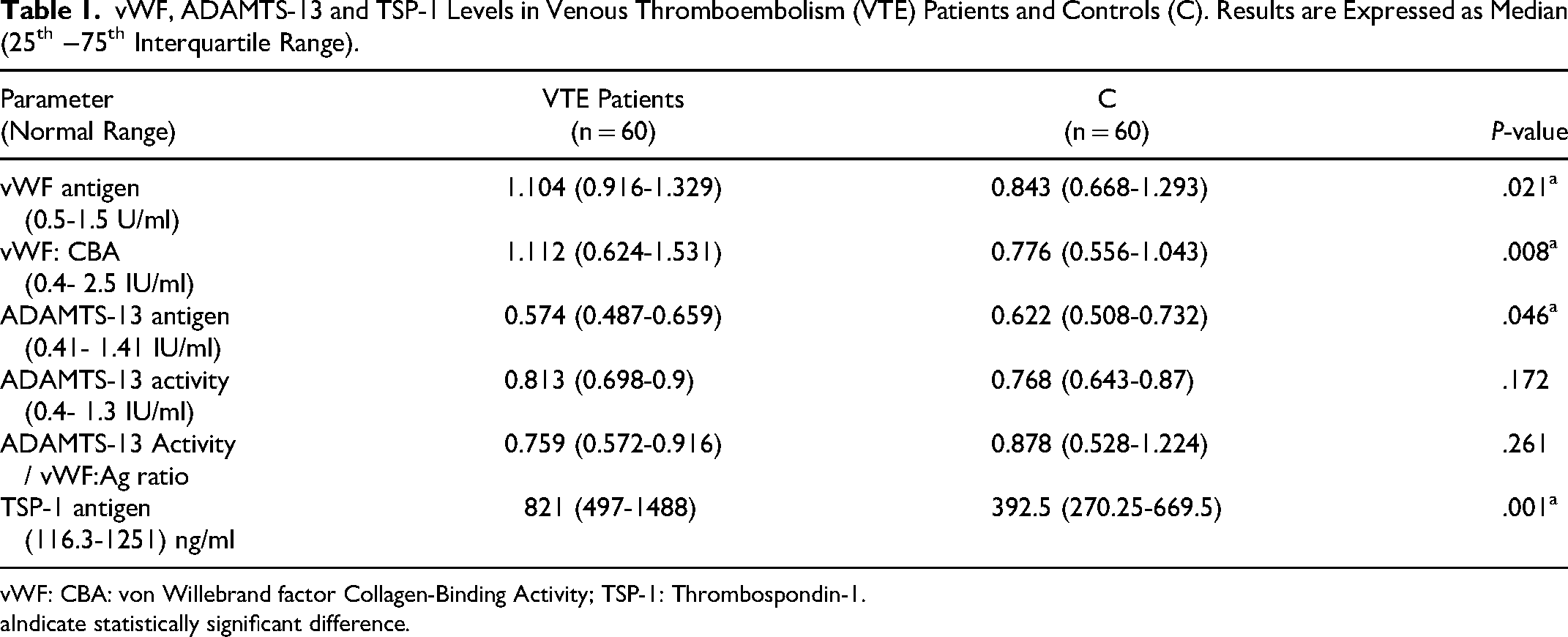
vWF: CBA: von Willebrand factor Collagen-Binding Activity; TSP-1: Thrombospondin-1.
Indicate statistically significant difference.
A non-parametric bivariate spearman's correlation was used to describe the association between TSP- 1 and vWF and ADAMTS-13. In VTE patients, TSP-1 had a significant positive correlation with vWF antigen (rho = 0.303, P = .021), but no correlation with vWF collagen-binding activity. Moreover, there was a significant negative correlation with ADAMTS-13 activity (rho = −0.244, P = .033) and ADAMTS-13 activity/ vWF antigen ratio (rho = −0.348, P = .007), but a weak and insignificant negative correlation with ADAMTS-13 antigen (Table 2 and Figure 1). On the other hand, no correlation was found between TSP-1 and any of the measured proteins in the control group (P > .05) (Table 2). Figure 1 shows the scatter plots of the significant association results.
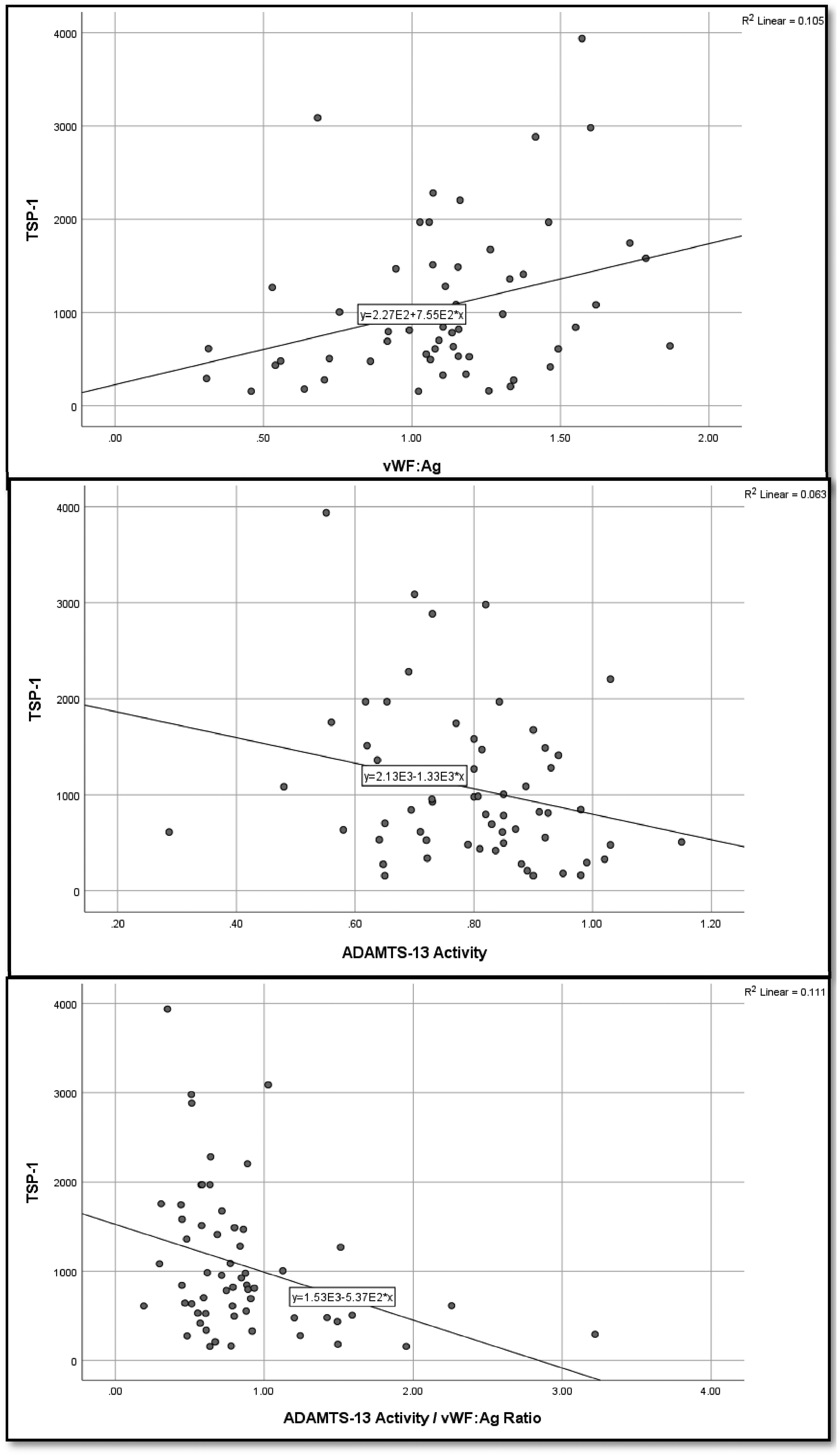
A scatter plot showing associations of Thrombospondin-1 with vWF:Ag, ADAMTS-13 activity and ADAMTS-13 activity/vWF:Ag ratio in venous thromboembolism (VTE) patients.
Correlation of Thrombospondin-1 with vWF and ADAMTS-13 in Venous Thromboembolism (VTE) Patients and Controls (C).
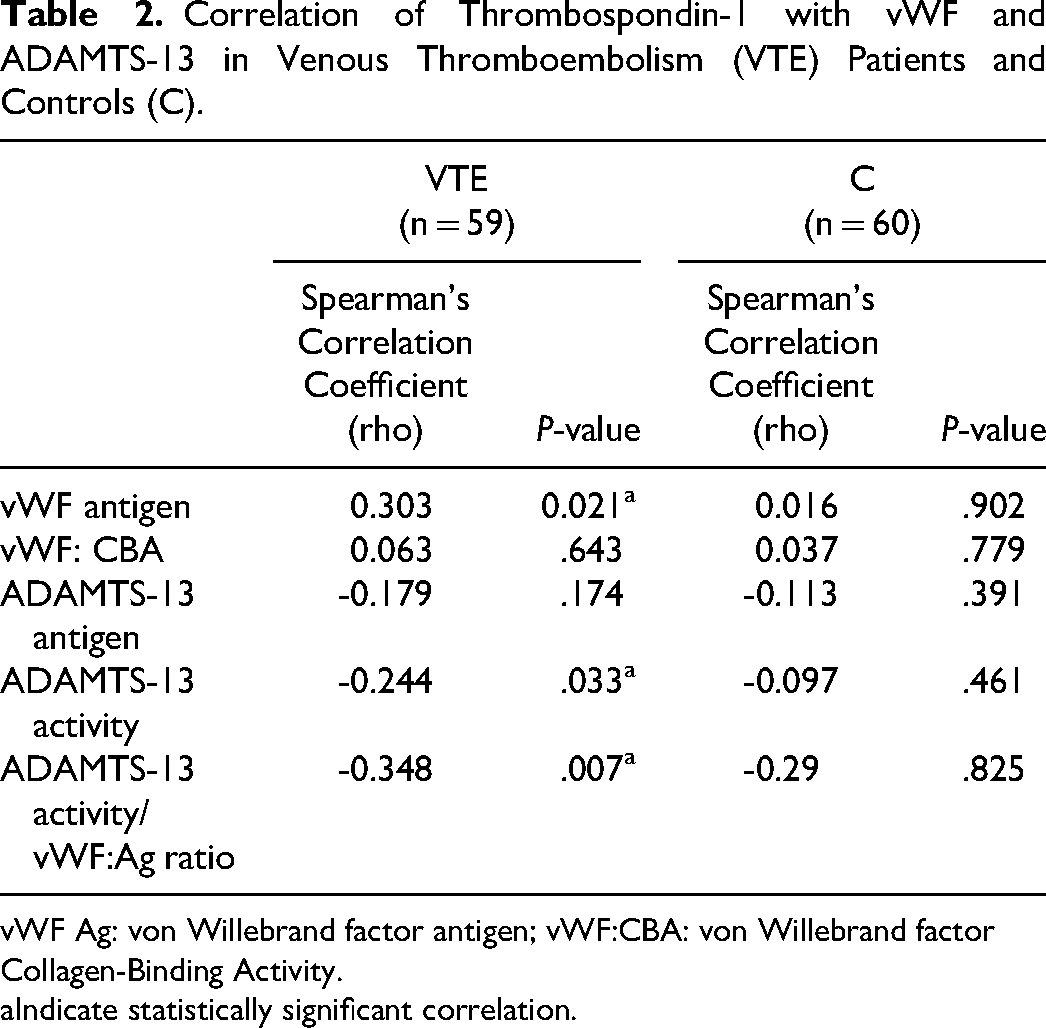
vWF Ag: von Willebrand factor antigen; vWF:CBA: von Willebrand factor Collagen-Binding Activity.
Indicate statistically significant correlation.
DNA analysis showed that 12 out of 60 VTE patients (20%) were heterozygous for FVL mutation, while five were homozygous (8.3%). On the other hand, only two out of 60 control subjects were heterozygous for FVL mutation (3.3%) and no case was homozygous. Using Binary Logistic Regression analysis, and VTE (Yes or No) as the dependent variable, we analyzed age, sex and FVL mutation (Yes = Homozygote + Heterozygote) or No = no FVL mutation) as independent variables. Due to low sample number, we opted to group homozygote and heterozygote cases together. The analysis showed a significant association between VTE and FVL mutation, with OR of 9.672 (95% CI 2.074-45.091 - P = .004). This indicates that the odds of developing VTE is 9.672 times in carriers of FVL mutation (Homozygote or Heterozygote) compared to subjects who are free from the mutation. No such association was found with age or sex (P = .667 and .183, respectively).
In the VTE patients, there was no statistically significant difference in vWF, ADAMTS-13 and TSP-1 between patients with or without FVL (homozygous and heterozygous)—(P > .05; results not shown). Also, no significant association was found between the presence of the mutation (homozygous and heterozygous analyzed together) in patients and the levels of vWF, ADAMTS-13, and TSP-1 (Table 3).
Associations Between the Presence of FVL Mutation (Homozygous and Heterozygous analyzed together) and vWF, ADAMTS-13 and TSP-1 Levels in Venous Thromboembolism (VTE) Patients (n = 60). Results are Expressed as Odds Ratios and 95% Confidence Interval (CI).
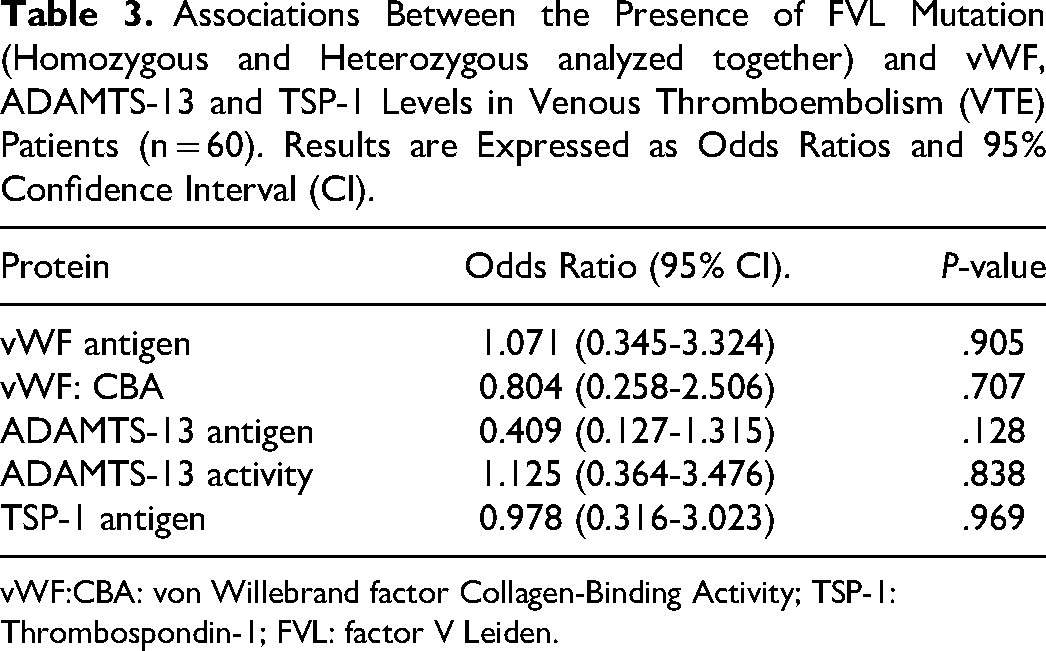
vWF:CBA: von Willebrand factor Collagen-Binding Activity; TSP-1: Thrombospondin-1; FVL: factor V Leiden.
Discussion
vWF is a large glycoprotein that plays a major role in platelet adhesion, aggregation, and thrombus formation. ADAMTS-13 acts as a protease that cleaves unusually large vWF multimers to smaller fragments leading to reduced coagulation activity of vWF and its removal from blood. Maintaining a balance between these two proteins is important for normal hemostasis, as loss of this balance has been reported to be associated with several pathological conditions.
In this study, we report higher levels of vWF antigen and activity in adult VTE patients compared to healthy controls (Table 1). Similar findings have been reported previously29,30 and were proposed to occur as a consequence of thrombus formation rather than being a risk factor for VTE. 30 We also found lower levels of ADAMTS-13 antigen in VTE patients compared to controls (Table 1). While a couple of studies reported similar findings,30,31 others had contradicting outcomes. 29
In 2016, Karakaya et al proposed that the low ADAMTS-13 level in VTE was a secondary phenomenon, “suggesting that it is a consequence of VTE rather than a risk factor.” 30 It is worth noting that sample collection in the Karakaya et al study was done between the first and second months after the diagnosis of VTE, signifying that proteins’ expressions were measured at a relatively early stage of thrombus formation. Our samples, however, were collected at least 3-6 months following the acute VTE event, and continued to show higher vWF antigen and activity levels in patients; suggesting that these concentrations may not be a consequence of the thrombotic episode but rather a possible risk factor for it, as has been reported recently by Edvardsen et al. 36
In the Edvardsen et al study, a dose-dependent association between plasma levels of vWF and future risk of incident VTE was reported and remained throughout the 13-year study period. 36 Our study had a similar outcome, as plasma levels of vWF remained high even after more than 3 months of the VTE episode; though it cannot be ruled out that the significantly higher percentage of type O in the control group might be a bias.
As stated by Mazetto et al in 2012, the increased levels of vWF in VTE patients is a result of “a persistence of inflammatory activity even a long time after the acute episode” 29 ; but unlike our study, Mazetto et al reported increased levels of ADAMTS-13, which were suggested to represent a compensatory mechanism against the persistently high levels of vWF. The contradicting findings between ours and that of others may be attributed to several factors including time of sample collection after onset of VTE, difference in patient management, subjects’ ethnicity, and ABO blood type.
Another parameter determined in this study was ADAMTS-13 activity/ vWF antigen ratio. This ratio explains the direction of the interaction between these proteins. When vWF antigen levels are high and ADAMTS-13 activity are low, this results in decreased ADAMTS-13 activity/ vWF antigen ratio (or increased vWF antigen / ADAMTS-13 activity ratio). Lower ADAMTS-13 activity/ vWF antigen ratio indicates insufficient cleavage of the released vWF antigen by ADAMTS-13 leading to an environment that enhances thrombi formation. In this study, VTE patients had a lower median level of ADAMTS-13 activity/ vWF antigen ratio compared to controls but the difference was not statistically significant (Table 1). It seems unlikely that this degree of decrease in the ADAMTS-13/vWF ratio would result in inadequate vWF cleavage. There might be an ADAMTS13 activity level preserved enough to balance the slight increase in vWF antigen levels. While we cannot rule out a tendency towards thrombosis, a larger sample size is needed to support this outcome.
Through its multidomain structure, TSP-1 was reported to bind to several receptors like CD36 as well as integrins and other adhesive glycoproteins including collagen, laminin, fibronectin, and vWF. 37 The plasma concentration of TSP-1 protein has been reported to increase 100 folds upon platelet activation, 38 which may explain the mirrored increase of TSP-1 with vWF and the positive correlation we found in our VTE patients (Table 1 and Table 2).
We also found a negative correlation between TSP-1 and ADAMTS-13 activity (Table 2 and Figure 1). This finding is consistent with the previously described protective role of TSP-1 against degradation of vWF by the cleaving protease ADAMTS-13. 18 To explain more, TSP-1 was reported to partially block ADAMTS-13 binding to A2 and A3 domains of vWF, and the full-length vWF, which restrained ADAMTS13 activity by up to 70%, leading to reduced vWF multimer processing by ADAMTS-13.17,39 This is further confirmed by the negative correlation we found between TSP-1 and ADAMTS-13 activity / vWF:Ag ratio indicating reduced degradation of vWF by ADAMTS-13 (Table 2 and Figure 1). Our results may suggest an inhibitory effect of high levels of TSP-1 on ADAMTS-13, although this requires further verification in a large-scale study due to the weak correlations.
Another aim of this study was to examine a possible association between vWF and ADAMTS-13 levels with FVL mutation; the presence of which was reported to be relatively high in the local population in Kuwait 5 ; and its association with VTE is well recognized. 40 We found a significant association between FVL mutation and VTE with odd ratio =9.672 (95% CI 2.074-45.091 - P = .004), suggesting that the odds of developing VTE in carriers of FVL mutation are 9.672 times those with wild-type Factor V. However, VTE patients carrying the heterozygote or homozygote mutation did not show difference in the expression of vWF, ADAMTS-13 and/or TSP-1 proteins compared to those with the wild type (P > .05). The lack of an association suggests that the risk factor imposed by FVL in VTE patients is independent of the studied proteins. It is possible that the relatively small number of the cohort made it difficult to obtain significant differences particularly when subjects are divided to subgroups.
The number of subjects is one limitation of this study. It was relatively small because of the “exclusion criteria” that limited the number of cases we could include in the study. Another limitation is the fact that there was only one point of sample collection for each subject, which made it difficult to compare findings at different intervals of the VTE episode. In the future, we hope to increase the sample number and collect samples at several time points; for example, directly at VTE onset, 3-6 months after onset and >12 months after onset. Assessing the trends of expression over time will further clarify the role of the studies proteins in VTE.
We conclude that the imbalance between vWF and ADAMTS-13 might be a continuing risk factor for possible future VTE episodes in the studied population; since such imbalance leads to accumulation of vWF which encourages a hypercoagulable environment in patients. The high levels of vWF and TSP-1 detected after more than 3 months of the VTE onset, highlight these proteins as possible markers for VTE. Whether these high levels are a consequence of low ADAMTS-13 or a reflection of an ongoing subtle endothelial activation is subject to further investigations.
We also conclude that FVL mutation, although imposes a significant risk to developing VTE, was not related to vWF, ADAMTS-13, or TSP-1. An epidemiological prospective study will provide better insight into this matter.
Footnotes
Acknowledgements
This work was funded by Kuwait University (Research Grant No. NM02/16). The authors wish to thank the Research Core Facility in Kuwait University Health Sciences Center (Research Project No. SRUL02/13) for providing help in using their different facilities. Special thanks for Mrs. Liena Abdulaziz, Mrs. Sicy Easow, Mrs. Jucy Gabriel, Mrs. Anju Nair, and Ms. Kefayah Al-Sabaan for excellent technical work. We are grateful for Dr Rasha Al-Khaldi for her help in the statistical analysis of the data and Prof. Indu Singh for reviewing the manuscript.
Data Availability Statement
Data is available on request from the authors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the Kuwait Ministry of Health's Research Ethical Committee and Kuwait University Health Science Centre Ethical Committee.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Kuwait University, (Grant Number NM02/16).
Informed Consent Statement
Subjects enrolled in the study signed a consent form that was approved by the Kuwait Ministry of Health's Research Ethical Committee and Kuwait University Health Science Centre Ethical Committee.
