Abstract
Postoperative thrombotic thrombocytopenic purpura (TTP) shows clinical presentation similar to classical TTP, whereas exact pathophysiological contexts remain unexplained. In this study, we investigated intraoperative and postoperative changes in ADAMTS-13 (a disintegrin and metalloprotease with thrombospondin type 1 motifs, member 13), von Willebrand factor (VWF), large VWF multimers, and interleukin-6 (IL-6) in vascular surgery patients. The objective was to compare the impact of endovascular, peripheral, and aortic surgery on target parameters which are supposed to play a role in surgery-associated TTP. A total of 93 vascular surgery patients were included and divided into 4 groups according to the specific type of intervention they underwent. Blood samples were taken preoperatively, intraoperatively, and postoperatively on days 2 and 4. The ADAMTS-13 activity decreased significantly in 3 of the 4 groups during surgery (from median 81% to 49%,
Keywords
Introduction
Thrombotic thrombocytopenic purpura (TTP) is a life-threatening disease characterized by the classical pentad of microangiopathic hemolytic anemia with fragmented erythrocytes (schistocytes) in the peripheral blood smear, thrombocytopenia, renal failure, neurologic symptoms in various manifestations, and fever. 1 This clinical presentation is caused by thrombotic microvascular occlusions resulting in ischemic organ damage. Besides hemolytic uremic syndrome, TTP represents the epitome of thrombotic microangiopathies (TMAs). 2,3
One of the key factors in pathophysiology of TTP is the von Willebrand factor (VWF). The VWF is a multimeric glycoprotein which occurs in the blood in subunits of various molecular weights. It plays an important role in primary hemostasis by initiating the adhesion of thrombocytes at sites of vascular injury. Multimers of higher molecular weight are thereby more hemostatically active than smaller multimers. 4 –6
ADAMTS-13 (a disintegrin and metalloprotease with thrombospondin type 1 motifs, member 13) is the metalloproteinase specifically cleaving VWF into smaller subunits in the circulation by hydrolysis. 7 –10 High shear stress as occurring in the microcirculation seems to be needed to make the cleavage site of the VWF subunit accessible to ADAMTS-13. 3,8,11 When ADAMTS-13 no longer cleaves VWF into smaller multimers, unusually large von Willebrand factor (ULVWF) multimers occur in the plasma of patients with TTP. These ULVWF multimers are highly platelet adhesive and explain the microvascular thrombi rich in platelets and VWF leading to organ ischemia in patients with TTP. 3,12 –14 According to the current state of knowledge, acquired forms of TTP must be distinguished from hereditary ones. Congenital TTP is caused by an inherited deficiency of VWF-cleaving protease (ADAMTS-13), whereas circulating antibodies (immunoglobulin G) inhibiting ADAMTS-13 are responsible for acquired forms of TTP. 15,16
In the last years, several cases of TTP associated with different surgical procedures such as vascular or cardiac surgery have been reported. 17 –22 Patients developed microangiopathic hemolytic anemia and severe thrombocytopenia about 5 to 9 days postoperatively, this clinical syndrome was therefore named postoperative TTP (pTTP). 14,23
Postoperative TTP is a rare but life-threatening disease. As only a limited number of case reports on pTTP exists (one current review article by Sridharan et al 24 included 85 cases), indicating an incidence would be speculative, especially since the number of unrecognized cases might be significantly higher than the amount of cases correctly diagnosed and mentioned in literature. Common postoperative complications such as blood loss, infections, or heparin-induced thrombocytopenia present with a similar clinical picture and can delay or obscure the correct diagnosis. 23
The pathophysiology of pTTP is not yet completely understood. It is suspected that endothelial damage during surgical procedures releases high-molecular-weight VWF multimers in amounts exceeding circulating ADAMTS-13. 19,23 Vascular surgery naturally can be accompanied by considerable vascular damage and could therefore predispose patients to develop TMAs by direct endothelial cell injury. 19,25
Previous studies investigating ADAMTS-13 and VWF in patients undergoing cardiac surgery have shown a decrease in ADAMTS-13 and a significant increase in large VWF multimers and VWF antigen levels. 6,26,27
Our study was the first prospective investigation into detecting intraoperative and postoperative changes in ADAMTS-13, VWF, large VWF multimers, and interleukin-6 (IL-6) in vascular surgery patients. The intention was to compare the impact of endovascular, peripheral, and aortic surgery on the stated target parameters to identify possible contexts of the pathophysiology of pTTP. We also aimed to detect whether the changes were more pronounced in major vascular surgery than in minor or in endovascular surgery.
However, this study was not designed to detect a case of pTTP as this is an extremely rare condition and an unrealistic number of included patients would be necessary to discover one single case in a prospective investigation.
We included vascular surgery patients undergoing procedures of different extent because we hypothesized that major vascular surgery would lead to higher vascular/endothelial damage, with presumed higher VWF stimulation 6 than the previously performed investigations mainly including coronary artery bypass grafting (CABG) patients.
Furthermore, our study was the first to investigate the kinetics of IL-6 in relation to ADAMTS-13 and VWF in the perioperative setting, as previous studies reported about an IL-6-mediated inhibition of ADAMTS-13 in inflammatory episodes possibly linking inflammation with thrombosis. 28
Patients and Methods
Patients
This prospective, nonrandomized cohort study was performed with approval by the institutional review board (ethical committee, University Hospital Frankfurt, Germany, approval number 405/14). A total of 93 patients of the Clinic for Vascular and Endovascular Surgery of the University Hospital Frankfurt were included in the period from February 2016 until April 2017. To meet inclusion criteria, patients had to be undergoing different vascular procedures, at least 18 years old, and have given their written informed consent. Patients were excluded if they had known TTP in their medical history or were currently pregnant. Regarding the specific type of surgery they underwent, patients were divided into 4 groups (Table 1, Figure 1). Clinical data of the patients were obtained from the digital patient data management (Table 1). Intraoperative volume substitution, transfusion needs (erythrocyte concentrates, platelet concentrates, and fresh frozen plasma), the duration of surgery, and the use of an intraoperative blood salvage device (Cell Saver) were collected from the anesthetic protocols (Table 2).
Cohorts of Patients and Demographic Data.a
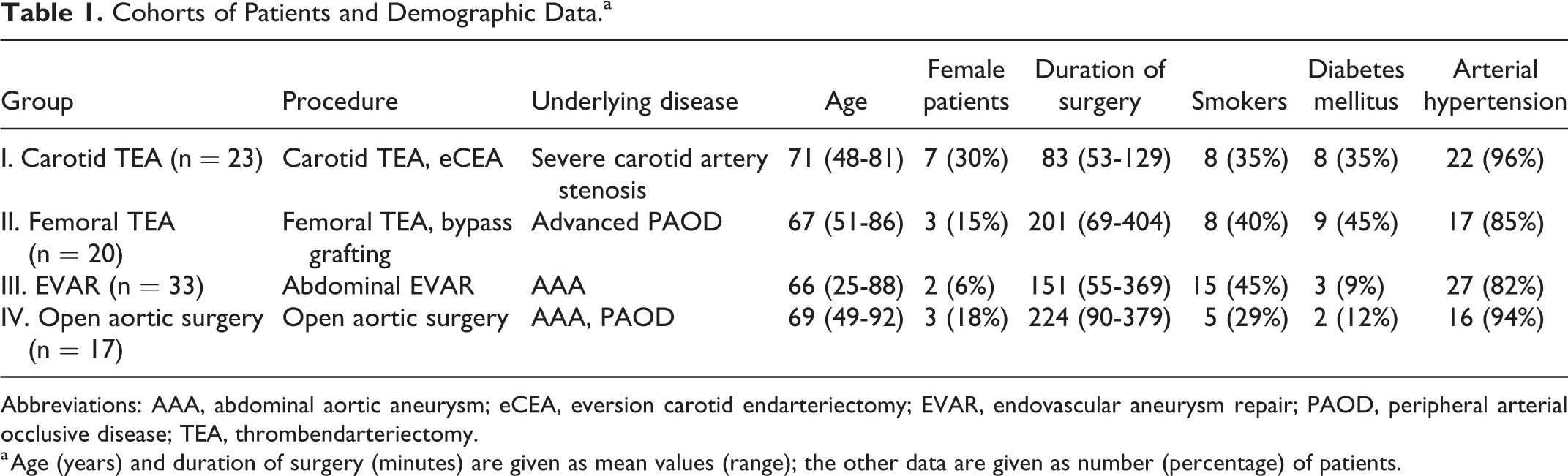
Abbreviations: AAA, abdominal aortic aneurysm; eCEA, eversion carotid endarteriectomy; EVAR, endovascular aneurysm repair; PAOD, peripheral arterial occlusive disease; TEA, thrombendarteriectomy.
a Age (years) and duration of surgery (minutes) are given as mean values (range); the other data are given as number (percentage) of patients.

Systematic overview of the study design. EVAR indicates endovascular aneurysm repair; intraop, intraoperative; preop, preoperative; postop, postoperative; TEA, thrombendarteriectomy; VWF, von Willebrand factor.
Intraoperative Transfusion Needs and Volume Loading.a

Abbreviations: EVAR, endovascular aneurysm repair; TEA, thrombendarteriectomy.
a Intraoperative blood loss (mL) and volume loading (mL; crystalloids and colloids) are given as mean values. The administration of ECs (erythrocyte concentrates), PCs (platelet concentrates), and FFP (fresh frozen plasma) is given as number (n) of patients receiving them (mean values of the number of transfusions for each patient). The use of a Cell Saver is presented as number (percentage) of patients treated.
Sample Collection
Blood samples were taken from the patients preoperatively, at 60 and 120 minutes during surgery, and postoperatively on days 2 and 4. Blood samples at 120 minutes were taken in the recovery room when surgery lasted <120 minutes. Postoperative samples were not taken on weekends or in cases where the patient was discharged before this time point. von Willebrand factor multimers were measured preoperatively and at 120 minutes intraoperatively; the other parameters were determined at all 5 time points.
Clinical Chemistry/Coagulation Assessment
Blood tubes with a capacity of 10 mL containing sodium citrate (1:10) as an anticoagulant (Sarstedt AG & Co KG, Nümbrecht, Germany) were used. These tubes were centrifuged for 15 minutes at 2500
The ADAMTS-13 activity was determined using Technozym ADAMTS-13 Activity ELISA (enzyme-linked immunosorbent assay; Technoclone).
von Willebrand factor multimers were analyzed and quantified with the help of densitometric gel analysis. Multimers were divided into small, intermediate, and large multimers as oligomers of 1 to 5, 6 to 10, and >10, respectively. 29
von Willebrand factor antigen was analyzed using a VWF Ag Testkit (BCS XP System, Siemens Healthcare Diagnostics).
von Willebrand factor activity was determined with the help of the Innovance VWF Ac test (BCS XP System, Siemens Healthcare Diagnostics).
Interleukin-6 and lactate dehydrogenase (LDH) were obtained using serum; hematologic parameters (platelet count, reticulocytes) were determined using EDTA blood.
Statistical Analysis
Statistical analyses were performed in cooperation with the Institute for Biostatistics and Mathematical Modelling, Department of Medicine, Goethe University Frankfurt, using BiAS (version 11.08), developed by Hanns Ackermann (Institute for Biostatistics and Mathematical Modelling, Department of Medicine, Goethe University Frankfurt). All tests were performed 2-sided, and a
Results
We included a total of 93 patients undergoing vascular surgery at the University Hospital Frankfurt from February 2016 until April 2017 (Table 1, Figure 1). Twenty-three of these patients underwent carotid TEA (thrombendarteriectomy) with severe carotid artery stenosis as underlying disease (group I); 20 patients having advanced peripheral arterial occlusive disease underwent femoral TEA or bypass grafting of the lower extremity (group II); 33 patients with abdominal aortic aneurysm underwent abdominal endovascular aneurysm repair (EVAR, group III); 17 patients underwent open aortic surgery due to abdominal aortic aneurysm or peripheral arterial occlusive disease (group IV).
We observed that ADAMTS-13 activity decreased significantly during surgery (at 120 minutes) in groups II (from median 71% to 63%,
ADAMTS-13 Activity (%) in Patients Undergoing Vascular Surgery.a

Abbreviations: ADAMTS-13, a disintegrin and metalloprotease with thrombospondin type 1 motifs, member 13; EVAR, endovascular aneurysm repair; TEA, thrombendarteriectomy.
a This table shows median (25th-75th percentiles) ADAMTS-13 activity (%) for each group at the respective pre-, intra-, and postoperative time point. n = number of patients (sample size),
b

ADAMTS-13 (a disintegrin and metalloprotease with thrombospondin type 1 motifs, member 13) activity in patients undergoing vascular surgery. median preoperative (preop), intraoperative (at 60 and 120 minutes during surgery), and postoperative (on postoperative days 2 and 4) ADAMTS-13 activity (%) is shown separately for each of the 4 groups (group I = carotid thrombendarteriectomy [TEA], group II = femoral TEA, group III = endovascular aneurysm repair [EVAR], group IV = open aortic surgery). Normal values: ADAMTS-13 (a disintegrin and metalloprotease with thrombospondin type 1 motifs, member 13) activity 40% to 130%.
The percentage of large VWF multimers (band >10) increased significantly in all groups of patients during surgery (from median 24.4% to 24.8%,

Large von Willebrand factor (VWF) multimers (band >10) in patients undergoing vascular surgery. The time course with 2 measuring points (preop = preoperative, 120 minutes = at 120 minutes intraoperatively) of large VWF multimers (%) is shown separately for each of the 4 groups (group I = carotid thrombendarteriectomy [TEA], group II = femoral TEA, group III = endovascular aneurysm repair [EVAR], group IV = open aortic surgery). Values are represented as a median with 25th/75th percentiles and minimum/maximum. *
von Willebrand factor antigen increased significantly on postoperative day 2 in all groups (from median 170% to 203%,

von Willebrand factor (VWF) antigen in patients undergoing vascular surgery. The time course with 5 measuring points (preop = preoperative, at 60 and 120 minutes intraoperatively, and on postoperative days 2 and 4) of VWF antigen (%) is shown separately for each of the 4 groups (group I = carotid thrombendarteriectomy [TEA], group II = femoral TEA, group III = endovascular aneurysm repair [EVAR], group IV = open aortic surgery). Values are represented as a median with 25th/75th percentiles and minimum/maximum. *
von Willebrand factor activity correlated significantly with VWF antigen in all groups.
Interleukin-6 (normal values: IL-6 <7 pg/mL) increased significantly during surgery in groups I (from median 4.3 to 10.6 pg/mL,
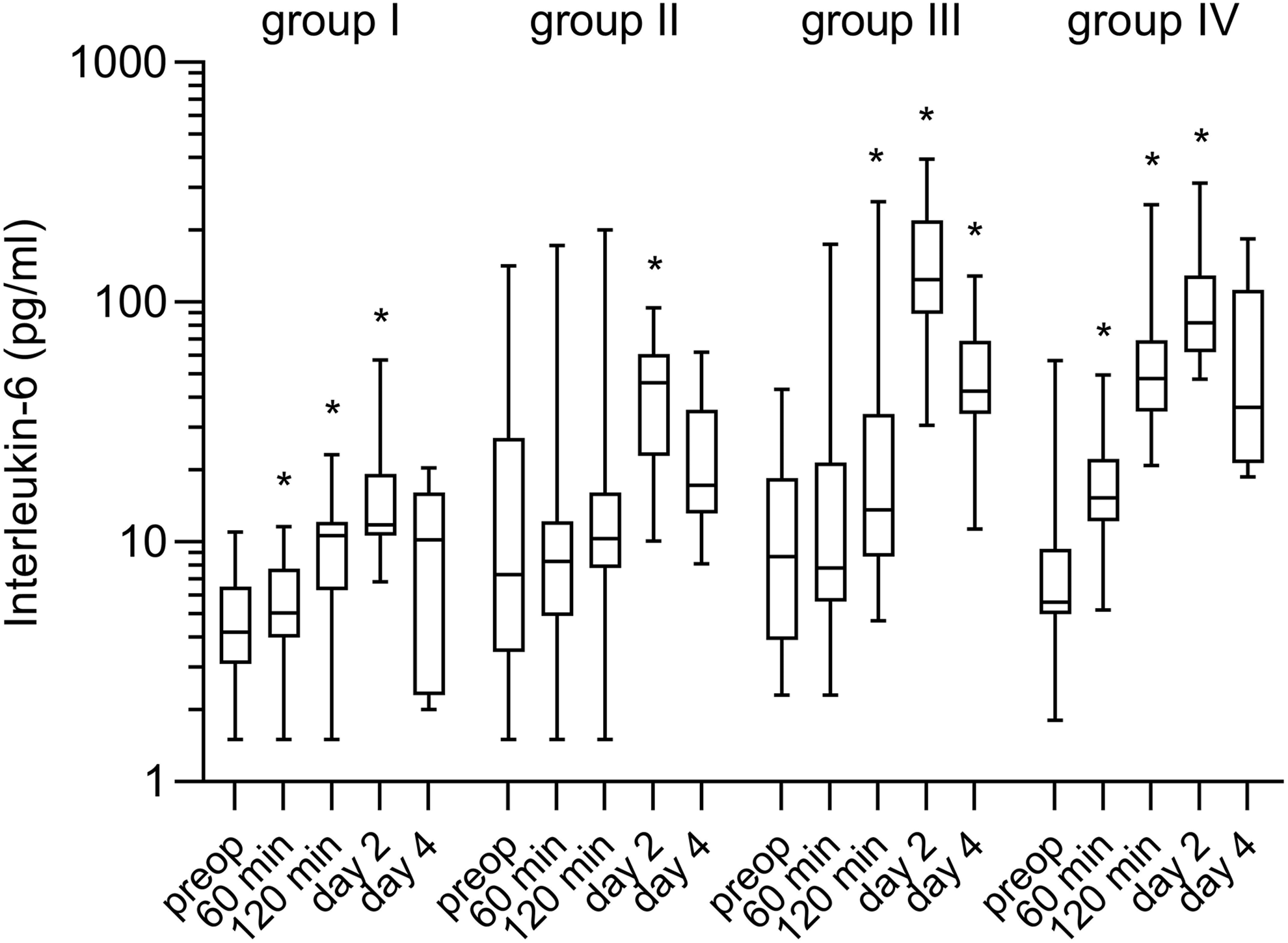
Interleukin-6 in patients undergoing vascular surgery. The time course with 5 measuring points (preop = preoperative, at 60 and 120 minutes intraoperatively and on postoperative days 2 and 4) of interleukin-6 (pg/mL) is shown separately for each of the 4 groups (group I = carotid thrombendarteriectomy [TEA], group II = femoral TEA, group III = endovascular aneurysm repair [EVAR], group IV = open aortic surgery). Values are represented as a median with 25th/75th percentiles and minimum/maximum. *
Regarding basic diagnostic parameters for hemolysis/TTP, LDH decreased during surgery in all groups. It increased in groups I, III, and IV on postoperative day 2 compared to preoperative levels. Platelet count decreased significantly during surgery in groups I, III, and IV. It decreased in groups III and IV on postoperative day 2. On day 4, platelet count was comparable to preoperative levels. The amount of reticulocytes decreased during surgery in groups I, III, and IV.
No significant correlation could be shown between the intraoperative decrease in ADAMTS-13 activity and the increase in large VWF multimers or VWF antigen. Furthermore, no significant correlation was noted between ADAMTS-13 activity and IL-6. A significant correlation was noted between IL-6 and VWF antigen during surgery (at 120 minutes) in group III (
There was no difference in ADAMTS-13 activity levels between patients with transfusion needs (erythrocyte concentrates, Table 2) and others without transfusions. Intraoperative blood loss (Table 2) did not correlate with ADAMTS-13 activity. There was no significant correlation between intraoperative volume loading (Table 2) and ADAMTS-13 levels.
There were 8 patients (n = 3 in group I, n = 2 in group II, n = 2 in group III, and n = 1 in group IV) who showed ADAMTS-13 activity <40% at 120 minutes with a median activity of 34.5% (32%-37%). In the postoperative period, these patients had a median activity of 43% (30%-48%), only 3 patients remained at low levels <40% on postoperative days 2 and 4. Regarding these 8 patients, they had a significantly lower median ADAMTS-13 activity before surgery than the other patients in this study (44.5% vs 76%,
Discussion
The objective of this prospective cohort study was to investigate intraoperative and postoperative changes in ADAMTS-13, VWF, large VWF multimers, and IL-6 in 93 patients undergoing vascular surgery. We aimed to compare the impact of endovascular, peripheral, and aortic surgery on target parameters which are supposed to play a role in surgery-associated TTP.
We detected an early decrease in ADAMTS-13 activity during surgery. Large VWF multimers increased in all groups in the intraoperative period, whereas VWF antigen increased belatedly in the early postoperative period in all groups. Interleukin-6 increased during surgery and again in the early postoperative period.
As mentioned above, several investigations focusing on cardiac surgery patients showed similar results of a decrease in ADAMTS-13 and an increase in large VWF multimers and VWF antigen levels in the context of cardiac surgery. 6,26,27 Only Lo et al 30 found an increase in both ADAMTS-13 and VWF antigen after on- and off-pump CABG.
Conversely, most case reports on pTTP do not provide measurement of ADAMTS-13 activity. In their review of 33 cases of pTTP in medical literature, Eskazan et al 14 found that mean ADAMTS-13 activity measured in 9 patients with pTTP was 46.5% (range: 23%-73%). Moreover, no patient in this review showed elevated ADAMTS-13 antibody titers. The absence of antibodies suggests a mechanism that is not immune mediated. 14 Recent investigations could not detect any appearance of new inhibitory ADAMTS-13 antibodies after cardiac surgery. 6 In contrast to these findings, Sridharan et al 24 identified 2 of the patients in their institutional review with immune-mediated pTTP. In both reviews, cardiovascular and vascular surgery were identified as the most common procedures leading to pTTP. 14,24 Less common procedures included, among others, abdominal, 25 gynecological, 31 and orthopedic 32 surgery.
It has not yet been established why surgery-associated TTP resembling classical TTP can occur even though ADAMTS-13 activity is only moderately reduced. Nor has the clinical relevance of mildly reduced intraoperative and postoperative ADAMTS-13 activity been clarified 24 ; no patient in this study showed clinical appearance of TTP. These findings may suggest that the level of ADAMTS-13 activity is not the only essential factor in the development of surgery-associated TMA.
The ADAMTS-13 activity was found to show a wide distribution in healthy human individuals from 40% to 170% in the literature, 33 patients in our study had ADAMTS-13 activity levels ranging from 30% to 107% (normal values: 40%-130%). Physiological or pathological conditions such as inflammatory and septic states, acute leukemia, chronic liver disease, uremia, autoimmune diseases, or pregnancy and even healthy test persons can show mildly to moderately decreased ADAMTS-13 activity, 27,33 –36 whereas ADAMTS-13 activity <10% or <5% (depending on the assay) seems to be specific to patients with acquired or congenital forms of TTP. 16
The mechanism of a decrease in ADAMTS-13 in the perioperative setting is not fully understood. One hypothesis is that the protease is consumed by high amounts of circulating high-molecular-weight VWF. 19,23,37 Other reasons could be inhibition of the protease by IL-6, 28 docking of the protease to VWF upon activated endothelial cells, 26 or hemodilution caused by intraoperative volume substitution. Another possible explanation could be preexisting marginal ADAMTS-13 activities before surgery, caused by medical history and genetic conditions. 23
In addition, the pathophysiology of pTTP is not completely explained. Surgical trauma and the following endothelial cell damage may lead to a release of high-molecular-weight VWF multimers in amounts exceeding circulating ADAMTS-13 levels and causing intravascular thrombi. 19,23,25 Decreased intra- and postoperative levels of ADAMTS-13 as found by us and other investigators may contribute to this imbalance. 26 According to this assumption, in our study group IV undergoing major aortoiliacal surgery, which is accompanied by higher wound surfaces and endothelial damage, showed more impressive changes in ADAMTS-13 and VWF compared to the other groups. The finding that most patients with pTTP described in the literature responded to plasma exchange may support the hypothesis that plasmatic imbalance is responsible for surgery-associated TMA. 24
Systemic inflammation—as potentially caused by major surgery—was shown to be associated with increased ULVWF and decreased ADAMTS-13 levels. 36,38 Investigating intensive care patients in inflammatory and septic states, Bockmeyer et al 39 found that acquired ADAMTS-13 deficiency was associated with the severity of inflammatory host response regardless of the underlying mechanism and an imbalance between levels of ADAMTS-13 and its substrate that was associated with the appearance of ULVWF multimers in the circulation.
Our investigation did not show a significant correlation between the intraoperative decrease in ADAMTS-13 and the increase in large VWF multimers or antigen, an observation that was also made by Reinecke et al. 6 In our study, the increase in VWF antigen was delayed with regard to the early intraoperative decrease in ADAMTS-13. In the 8 patients in our study showing ADAMTS-13 levels <40%, VWF antigen, large VWF multimers, and IL-6 did not differ from the rest of the patients included in this study. These findings may suggest that the increase in large VWF multimers and VWF antigen is rather due to endothelial cell activation and acute-phase response than to decreased ADAMTS-13 activity levels. 6,26
von Willebrand factor is known as an early marker of endothelial cell injury or dysfunction, and moreover, as an acute-phase protein with a half-life of 12 hours. 37,38,40,41 Inflammatory states such as infections, traumas, autoimmune diseases, or coronary and peripheral artery disease can cause increased VWF levels. 28 Surgical trauma may—by itself—cause a systemic dysregulation of the precisely regulated inflammatory response. A generalized activation of the endothelium and the shift to a prothrombotic state can be the consequence. This process can be seen as an inflammation-induced coagulation. 36,38
Recent investigations have shown that inflammatory cytokines released from endothelial cells or activated leukocytes in the early phase of systemic inflammation can affect the balance between ADAMTS-13 and its substrate, thus linking inflammation with thrombosis. 28 Interleukin-8, tumor necrosis factor-α, and to a lesser extent the complex of IL-6 and soluble IL-6 receptor, stimulated the release of ULVWF multimers in human umbilical vein endothelial cells in a dose-dependent manner. In addition, IL-6 inhibited the cleavage of ULVWF strings by ADAMTS-13 under flow. 28 Peigne et al 42 found a strong inverse correlation between ADAMTS-13 and IL-6 in septic shock patients and suggested an IL-6-mediated inhibition leading to a functional deficiency of ADAMTS-13.
Regarding the kinetics of IL-6 in our study, this pro-inflammatory cytokine increased during surgery and in the early postoperative period. Group IV undergoing open aortic surgery showed the most pronounced changes in IL-6 levels, probably as these patients experienced the highest extent of intraoperative tissue damage. Our investigation did not show clear correlations neither between IL-6 levels and ADAMTS-13 activity nor between IL-6 and VWF antigen or large VWF multimers. These findings suggest that IL-6 increased according to inflammatory reactions as part of the vascular procedure; accordingly the acute-phase protein VWF increased in the perioperative setting. However, the absence of correlations implies that the changes in IL-6 and ADAMTS-13 or IL-6 and VWF were not mutually dependent in our investigation.
Although the aim was to observe changes in ADAMTS-13, VWF, and IL-6 in the context of vascular surgery, we intended to detect possible mechanisms with respect to the pathophysiology of pTTP. This indicates certain limitations of our study. The limited number of patients included in this study cannot serve as the basis for general conclusions. We did not register the long-term clinical outcome of the patients; end point of this study was postoperative day 4. Conceivable hemodilution caused by intraoperative volume substitution and the administration of fresh frozen plasma containing ADAMTS-13 are confounding factors. Despite the absence of a clear correlation between ADAMTS-13 and intraoperative volume substitution or blood loss, hemodilution is one possible explanation for the intraoperative decrease in ADAMTS-13 which was most impressive in group IV where patients received more volume substitution than in all other groups.
No patient in this study was diagnosed with pTTP. As explained above, this is an extremely rare phenomenon and it was not intended to identify the occurrence of surgery-associated TTP during the observation period of this investigation. As the incidence of pTTP after vascular and cardiac surgery is very low, and inversely many patients with decreased ADAMTS-13 activity do not show symptoms of TMA, there may be further predisposing conditions for the development of pTTP. It needs to be elucidated if and by what mechanisms preexisting conditions, certain drugs, and the nature of surgical intervention with the resulting endothelial cell injury 24 contribute to pTTP.
Finally, the results of this study indicating an early decrease in ADAMTS-13 in patients undergoing major surgery comply with previous investigations and with the assumption that inflammatory states caused by major surgery can be seen as a stimulus for the release of endothelial cell–derived ULVWF in amounts exceeding marginal intra-/postoperative ADAMTS-13 levels in some patients. Further investigations focusing on the pathophysiology of pTTP and predisposing factors could lead to a better understanding and alertness of this rare but potentially lethal disorder in the perioperative setting.
Footnotes
Authors’ Note
Ethical approval to report this case series was obtained from the ethical committee of the University Hospital Frankfurt (approval number 405/14). Written informed consent was obtained from the patients for their anonymized information to be published in this article. This observational study was registered retrospectively at the German Clinical Trials Register (registration number DRKS00020821; ![]() ).
).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This investigator-initiated research was supported by a grant by Shire Deutschland GmbH, Berlin, Germany, a member of the Takeda group of companies (grant number IIR H14-23621).
