Abstract
Background/Objectives
Venous thromboembolism (VTE) is a frequent complication of cancer, namely lung cancer. Many studies have been done searching for risk factors for VTE, but most don’t address the topic of inherited thrombophilia. The aim of this study is to evaluate the contribution of inherited thrombophilia to VTE in patients with Non-Small Cell Lung Cancer (NSCLC).
Methods
This is an observational, prospective, case-control study, involving 40 patients with NSCLC, stages IIIB or IV, 20 with a diagnosis of VTE, and 20 with no VTE. Blood samples were collected for factor V and prothrombin genotyping, functional assays of antithrombin, protein C and protein S, and activated protein C resistance. All patients gave a signed informed consent, and the study was approved by the Ethics Committees of the Institutions involved.
Results
The case and control groups were similar in terms of gender, age, Body Mass Index (BMI), stage or type of cancer. No patients were found with deficiencies of antithrombin or protein C, or with the prothrombin mutation. Factor V Leiden (FVL) was found in 2 patients from the case group, both with activated protein C resistance, and in none of the control group. Low levels of protein S were found in 2 patients from the control group and 1 from the case group.
Conclusions
We conclude that FVL may be an important risk factor for VTE in patients with NSCLC. If these patients were screened for FVL, we could probably reduce the prevalence of VTE.
Keywords
Introduction
The association between cancer and venous thromboembolism (VTE) is well known since the first description by Trousseau in 1865 1 The pathogenesis of cancer-associated VTE is multifactorial and complex involving multiple overlapping pathways. 2 Some studies suggest inherited thrombophilia as an important risk factor for cancer-related VTE,3–5 but, apart from Acute Lymphoblastic Leukemia in children,6–8 there is much controversy on the contribution of thrombophilia to VTE in patients with malignancy. 9
Lung cancer is among those with the highest rate of VTE (5.1%), next to pancreas (8.1%), and kidney (5.6%). 10 Various studies have been conducted aiming at identifying high-risk groups 11 or risk factors 12 for VTE in lung cancer patients but, as far as we know, very few address specifically the relation of inherited thrombophilia to VTE.
The purpose of our study is to evaluate the contribution of inherited thrombophilia to VTE in patients with non-small cell lung cancer (NSCLC), particularly factor V Leiden (FVL), prothrombin mutation G20210A (PTG20210A), and the deficiencies of the natural anticoagulants antithrombin, protein C, and protein S.
Methods
Study Design and Patient Selection
For our study, we performed an observational, prospective, case-control study involving 2 centers, Unidade Local de Saúde de Trás-os-Montes e Alto Douro (ULSTMAD) and Unidade Local de Saúde de Santo António (ULSSA).
Selected patients were 18 years or older, able to give informed consent, diagnosed with NSCLC, stage IIIB or IV. Inclusion criteria for the case group was VTE, either deep venous thrombosis of the lower limb, pulmonary embolism or any other venous thrombosis in unusual location, diagnosed by ultrasonography, computed tomography scans or other imaging technics. Inclusion criteria for the control group was patients diagnosed with NSCLC, stage IIIB or IV, but clinically stable and with no history of thrombosis, either symptomatic or incidental. Exclusion criteria, for both groups, was: 1) patients taking anticoagulants before the diagnosis of the neoplasm; 2) age < 18 years; 3) patients unable to give informed consent; and 4) pregnancy, or with pregnancy ended at less than 8 weeks before.
Patient Evaluation
All patients were evaluated for: thromboembolic events, personal or family history of thromboembolism, Khorana risk score, and histologic type of neoplasm.
Laboratory Investigation
Blood samples were collected from all patients for factor V and prothrombin genotyping, functional assays of antithrombin, protein C and protein S, and activated protein C resistance. Samples were collected before the beginning of anticoagulant therapy or when anticoagulants were stopped for the purpose of invasive procedures.
Samples were collected in 2.8 ml potassium EDTA tubes for genotyping and in 2.9 ml sodium citrate tubes for the coagulation assays.
Factors V and II Genotyping
Genomic DNA was extracted from peripheral blood using a DNA isolation kit (QIAamp DNA Blood Mini Kit - Qiagen) according to the manufacturer's protocol.
The G1691A mutation in factor V gene (factor V Leiden) mutation was detected by the real time Polymerase Chain Reaction (PCR) Method with the GeneProof Factor V Leiden PCR kit. This method is based on amplification and detection of the target sequence using allele specific fluorophore labelled probes. The target sequence is a single nucleotide guanine/adenine polymorphism in site 1691 (G1691A).
The G20210A mutation in prothrombin (factor II) gene was detected by the real time Polymerase Chain Reaction (PCR) Method with the GeneProof Factor II Prothrombin PCR kit (GeneProof). The approach consists in amplification and detection of the target sequence with probes labeled with allele-specific fluorophores. The target sequence involves a single nucleotide guanine/adenine polymorphism at position 20210 (G20210A).
Functional Assays of Natural Anticoagulants and Activated Protein C Resistance Assessment
Samples were centrifuged at 2500 g for 15 min, then plasma was separated and frozen at −70 °C less than 3 h after collection. Functional Antithrombin, Protein C and Protein S assays were performed using the STA-Stachrom ATIII, the STA Stachrom Protein C, and the STA Staclot Protein S kits, respectively, all from Diagnostica Stago®. The assessment of the Activated Protein C Resistance (APC-R) was performed with the STA Staclot APC-R kit from Diagnostica Stago®.
Ethical Statements
The study was approved by the Ethics Committees of both hospitals (ULSTMAD and ULSSA) and all patients signed informed consent. After signing the informed consent, recruited patients were given an identification code number that was further used for registering all data collected.
Statistical Analysis
Case and control groups were compared using the Chi-square test for categorical variables and the student t test for continuous/numerical variables. Relative Risks and Odds Ratios were calculated for protein S and factor V Leiden. A probability P value < .05 was considered as statistically significant.
Results
Patients’ Characteristics
Forty-six patients were initially recruited but 6 were excluded after reviewing the clinical files; 1 died before colleting blood, and 5 did not fulfil the inclusion criteria (Figure 1). Among those 5 patients, 2 were taking anticoagulants before diagnosis of NSCLC for other reasons than VTE, 1 was stage IIIA, 1 had not lung cancer but laryngeal cancer with extension to bronchi, and in 1 the diagnosis of VTE could not be confirmed by imaging techniques.

Study Flow Chart.
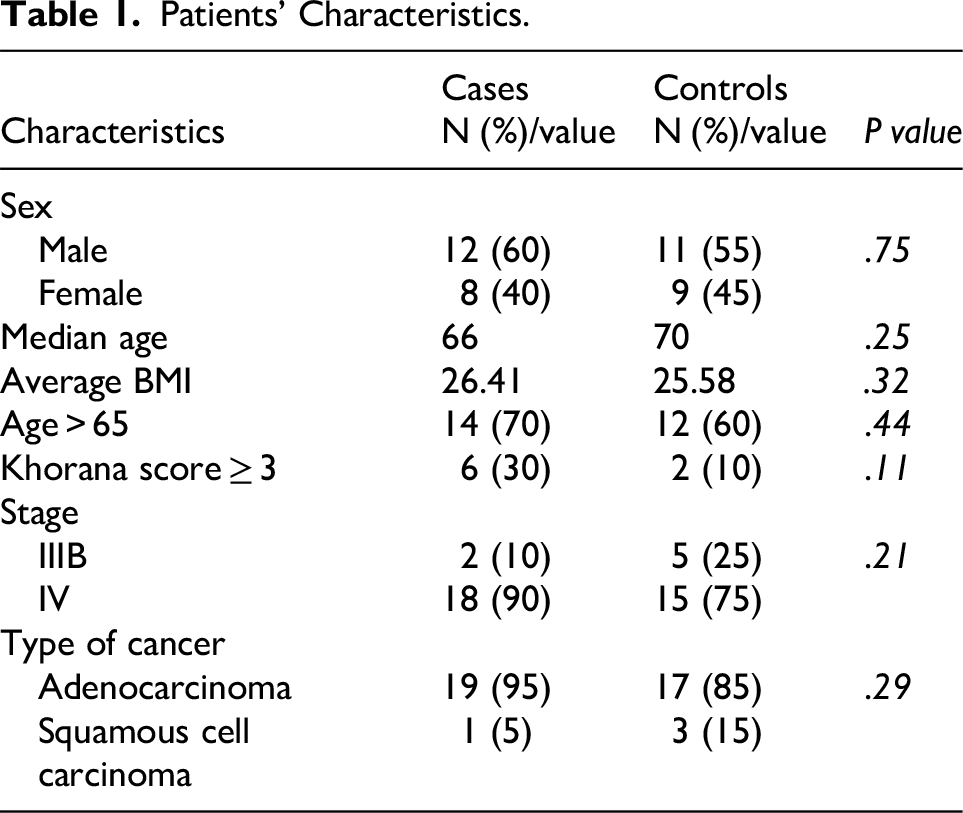
Patients’ characteristics are shown in Table 1. There is no significant difference in terms of gender, age, Body Mass Index (BMI), stage or type of cancer, between the case and the control groups. Also, the number of patients aged more than 65 years or with a Khorana score more than 3, in each group, does not differ significantly. Tables 2 and 3 show detailed patients’ characteristics and Table 4 shows types of cancer and VTE found.
Patients’ Characteristics.
Detailed Patients’ Characterization (Cases).

Detailed Patients’ Characterization (Controls).

Types of Cancer and VTE.

Thrombophilia Investigation Results
Results of the thrombophilia investigation are depicted in Table 5.
Results of Thrombophilia Investigation.

* Not confirmed
§ Confirmed
RR – Relative Risk
OR – Odds Ratio
ND – Not determined; calculation yields the value of infinite
No patients were found with deficiencies of antithrombin or protein C, or with the prothrombin mutation. Factor V Leiden was found in 2 patients from the case group, both with activated protein C resistance, and in none of the control group. Low levels of protein S were found in 2 patients from the control group and 1 from the case group. All 3 cases of low protein S had values between 40% and 46% and the normal range is 55%–140% (Table 6). However, due to the design of the study and, for ethical reasons, these results could not be confirmed with a second sample. Detailed results of the thrombophilia investigation can be found in Tables 7 and 8.
Reference Values of the Thrombophilia Tests Performed.

Detailed Results of Thrombophilia Investigation (Cases).

ATIII – Antithrombin III, PC – Protein C, PS – Protein S
APC – R - Activated Protein C, FVL – Factor V Leiden, PTG20210A – Prothrombin mutation G20210A
Abnormal results in bold
Detailed Results of Thrombophilia Investigation (Controls).

ATIII – Antithrombin III, PC – Protein C, PS – Protein S
APC – R - Activated Protein C, FVL – Factor V Leiden, PTG20210A – Prothrombin mutation G20210A
Abnormal results in bold
Relative risk (RR) and Odds Ratio (OR) for FVL could not be calculated because, in the control group, there is no FVL. As for PS deficiency, both RR and OR are similar and inferior to 1.
Discussion
Our study, being a prospective case-control study limited to patients with NSCLC stages IIIB and IV, is probably the only one addressing this topic that compares two very homogenous groups of patients differing only on the presence or absence of VTE. Large studies, including patients with various types of cancer, conclude or suggest that FVL or PTG20210A increase the risk of developing VTE compared with patients with cancer but no thrombophilia.5,13,14 However, results include all types of cancer and large populations with variable risk factors. On the opposite, other studies conclude for a lack of association between thrombophilia and the risk of VTE in cancer patients.15–20 A more recent study 21 from China includes 214 patients with all types of lung cancer, 92 with VTE and 122 without. Patients were evaluated for the mutations of FVL, methylenetetrahydrofolate reductase C677T (MTHFR C677T), and Plasminogen Activator Inhibitor- 1 (PAI-1) 4G/5G; the results suggest the PAI-1 polymorphism to be an increased risk factor for development of VTE, and that patients with the double homozygous mutations of MTHFR C677T and PAI-1 4G/5G were those more prone to VTE. In this study, no patients were found with the FVL, which is not unexpected as this mutation is very rare in the Asian population.
It is well known that the risk of VTE in cancer patients, either lung or other types of cancer, increases in metastatic cancer. 22 In our study, all patients were stages IIIB and IV, stages where the contribution of inherited thrombophilia to VTE would be less, eventually negligible, compared with the much higher risk brought about by metastatic cancer by itself. So, this study was designed as the worst-case scenario to show evidence of the contribution of inherited thrombophilia to VTE in these patients. Even though, 2 patients in the case group had FVL while none of the control group had this mutation. Moreover, the 2 patients with FVL were the only ones that had positive APC-R. We think this is evidence that FVL is an important contributory factor for VTE in patients with NSCLC, although this must be confirmed by studies involving larger cohorts. Considering the prevalence of FVL in Caucasians, it seems that screening patients with lung cancer for this condition, would enable identification of those at risk, and institution of early adequate prophylaxis. Indeed, the American Society of Hematology guidelines suggest ambulatory thromboprophylaxis for patients with thrombophilia. 23 This could reduce the risk of VTE and improve survival. Although FVL genotyping is not a routine laboratory test, APC-R is not a so expensive test, and is easy to perform for most hemostasis clinical laboratories.
We didn't find any patients with deficiencies of protein C or antithrombin, as expected considering the low prevalence of these two conditions24,25 and the small number of patients recruited. We also didn't find patients with the prothrombin mutation G2021A, besides being the second most common inherited thrombophilia in Caucasians. We found 3 patients with low protein S, 2 in the control group and 1 in the case group. As we couldn’t obtain second samples from these patients, we cannot conclude they had PS deficiency and, given the low prevalence of PS deficiency, this is highly unlikely. Moreover, low levels of protein S may be an acquired phenomenon in various clinical situations, such as pregnancy, use of oral contraceptives, 26 and many other, 27 such as inflammatory conditions and cancer, in which there is an elevation of C4b-binding protein (C4b-BP). C4b-BP binds PS in plasma leaving less free PS and so PS functional levels decrease. 28
Our study has some important limitations. The most important is the small number of patients recruited. The second is the fact that, the design of the study prevented us from taking second samples for confirmation of results, leaving some unexpected results, such as those of low protein S, unexplained.
Conclusions
Our study strongly suggests that FVL is an important risk factor for VTE in patients with NSCLC. Due to the small number of patients of our study, this conclusion needs to be validated with studies involving large cohorts. As for other inherited thrombophilia we cannot draw any conclusions due the small number of patients involved and to limitations of the study.
If patients diagnosed with NSCLC were screened for APC-R and, if positive, for FVL, probably, we could reduce the prevalence of VTE and improve survival. This could be an interesting topic for other studies.
Footnotes
Acknowledgments
The authors acknowledge Stago Diagnostics and Liga Portuguesa Contra o Cancro (Portuguese League Against Cancer – LPCC) for the support given to this study.
Ethical Considerations
The study was conducted according to the guidelines of the Declaration of Helsinki and was approved by the Ethics Committees of both Hospitals (ULSSA 137-18; ULSTMAD 347/2019). All participants gave written informed consent to participate and for their anonymized information to be published in this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Stago Diagnostics, Liga Portuguesa Contra o Cancro, (LPCC).
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The study was done with the support of Stago Diagnostics and LPCC. However, the sponsors had no role in the design, execution, interpretation or writing of the study.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author, J.C., upon reasonable request.
