Abstract
To explore the risk factors for venous thromboembolism (VTE) in inpatients with colorectal cancer. The demographic factors, comorbidities, and hematological indices of patients with colorectal cancer treated in our hospital from 2016 to 2021 were collected and recorded. Venous thromboembolism events, including deep venous thrombosis and/or pulmonary embolism, were recorded and the patients were divided into the VTE group and the non-VTE group. We compared clinical data between the two groups and explored risk factors for VTE. Comparing the clinical data of 293 cases of non-VTE group and 235 cases of VTE group, we found significant differences in age, smoking, temperature, amount of blood loss, differentiation degree, peripherally inserted central catheter (PICC), radiotherapy, anemia, infection, white blood cell count, prothrombin time (PT), PT%, prothrombin ratio, international normalized ratio, thrombin time, CA199 and CEA between the two groups (P < 0.05). Logistic regression analysis showed that age (P = 0.0444), temperature (P = 0.0317), amount of blood loss (P = 0.0067), PICC (P < 0.0001), chemotherapy (P = 0.0459), anemia (P = 0.0007), international normalized ratio (P = 0.003) and CA199 (p = 0.0234) were independent risk factors for VTE. Receiver operating characteristic curve analysis showed that the amount of blood loss predicted thrombosis better (AUC = 0.778, P < 0.001), when the cutoff value was 20 mL, the sensitivity was 76.17%, and the specificity was 79.18%, respectively. And PICC predicted thrombosis better (AUC = 0.808, P < 0.001), the sensitivity was 70.21%, and the specificity was 91.47%, respectively. Clinical parameters are associated with VTE in inpatients with colorectal cancer, which will help to guide clinicians to take effective measures to improve the patients' prognosis.
Background
Colorectal cancer (CRC) refers to cancer of the colon or rectum, and is one of the most common malignant tumors in the world. CRC accounts for about 15.3% of malignant tumor-related deaths in the world every year, and is the second most common malignant tumor type in women and the third most common malignant tumor type in men.1,2 Cancer patients are at high risk for venous thromboembolism (VTE), and data show that the incidence of VTE in cancer patients is 4 to 7 times than that of non-cancer patients. 3 VTE is a plaque of blood that forms after abnormal coagulation in the venous system, causing the lumen to be partially or completely blocked. According to the different sites and stages of blood plaque formation, it mainly includes deep vein thrombosis (DVT) and pulmonary embolism (PE) in clinical practice. VTE is one of the most important causes of death in cancer patients.4,5 It has been reported that VTE increases the risk of death in cancer patients by 2 to 6 times, mainly within 30 days after surgery. 6 Previous evidence suggests that effective assessment and prevention can reduce the absolute risk of cancer in patients with VTE by 50%.7,8
Currently recognized risk factors for VTE include advanced age, obesity, surgery, hospitalization due to trauma or fracture, long time in bed, cancer and its treatment, infection, pregnancy, oral contraceptives, and hormone replacement therapy.9–12 A study of 5039 women who underwent breast surgery found that chronic hypertension, high body mass index, cancer type, and evidence of metastasis turned out to be the most significant risk factors for VTE. 13 A study had showed that independent predictors for VTE in metastatic CRC included female sex, African American race, prior VTE history, and right sided cancers. 14 However, there has not been a unified conclusion on the risk factors of VTE in inpatients with CRC in China and our study explored it to aid clinical prevention and treatment.
Materials and Methods
Patients
Patients with CRC who were hospitalized in Harbin Medical University Cancer Hospital from 2016 to 2021 were enrolled in the study group. Patients with lower limb pain, swelling or skin color change in the treatment process needed to immediately perform lower limb vascular Doppler ultrasound examination, using Philips EPIQ 7C, Mindray DC 8, Canon Aplio i900 and other ultrasound instruments, intravascular thrombosis was diagnosed as DVT (Figure 1). Patients presented with unexplained chest tightness, chest pain, hemoptysis, dyspnea, or fainting were performed computer tomography pulmonary angiography examination immediately using Siemens Definition Flash dual-source CT, filling defect of pulmonary artery and its branches was diagnosed as PE (Figure 2). Referring to the results of vascular ultrasound or computer tomography pulmonary angiography examination, the patients were divided into the VTE group and the non-VTE group. This retrospective study was in line with the Declaration of Helsinki and approved by the Ethics Committee of Harbin Medical University Cancer Hospital, the Institutional Review Board number of the approval for this study is KY2021-18.

The yellow arrow shows left popliteal vein thrombosis in a patient with colorectal cancer.

The yellow arrow shows right pulmonary artery embolism in a patient with colorectal cancer.
Data Collection
Demographic factors, comorbidities, and hematological indices were recorded on hospitalized patients with CRC. Parameters included age, gender, body mass index, smoking, temperature, heart rate, systolic blood pressure, diastolic blood pressure, amount of blood loss, transfusion, pathology, differentiation degree, metastasis, history of diabetes, hypertension, coronary heart disease, transfusion history, peripherally inserted central catheter (PICC), chemotherapy, radiotherapy, anemia, and infections. Laboratory results included white blood cell count, blood platelet count, prothrombin time (PT), PT%, prothrombin ratio (PTR), international normalized ratio (INR), activated partial thromboplastin time, fibrinogen, thrombin time (TT), CA199, and CEA. White blood cell count and platelet count were measured by Mindray BC-6800Plus automatic blood cell analyzer. PT, PT%, PTR, INR, activated partial thromboplastin time, fibrinogen, and TT were determined using Sysmex CS-5100 automatic blood clotting analyzer, PT, activated partial thromboplastin time, fibrinogen, and TT were measured by coagulation method. PTR is the ratio of PT to normal PT. INR is (the ratio of a patient's PT to a normal control's PT) international sensitivity index, the international sensitivity index of the reagent we used was 1.03. Serum tumor markers CA199 and CEA were detected by Roche cobas 8000 e801 automatic chemiluminescence immunoassay analyzer.
Statistical methods were used to compare the differences in clinical parameters between the VTE group and the non-VTE group, and to analyze the risk factors of VTE formation in CRC patients.
Statistical Analysis
SAS9.4 software was used for statistical analysis, Shapiro-wilk test was used for normality test. The measurement data of normal distribution were expressed as mean ± standard deviation (
Results
Two-hundred thirty five patients with DVT and/or PE were selected as the VTE group, and 293 patients without were selected as the non-VTE group. The demographic factors, comorbidities, and hematological indices of the 2 groups were compared, the VTE group was younger than the non-VTE group (P = .002). CRC patients who smoked were more likely to develop VTE (P = .001), and patients who experienced bleeding during surgery were more likely to suffer from VTE (P < .001). The differences in differentiation degree between the 2 groups were statistically significant (P = .002), the patients with poor differentiation in the VTE group were significantly more than those in the non-VTE group. The VTE group had more PICC than the non-VTE group (P < .001). Patients with CRC who received radiotherapy were less likely to develop VTE (P = .002), while the difference between the 2 groups in terms of chemotherapy was not statistically significant (P = .419). The CRC VTE group was more likely to present with anemia (P < .001), infection (P < .001), and increased white blood cell count (P < .001). The VTE group showed higher PT (P = .001), PTR (P < .001), INR (P < .001), and TT (P = .02), PT% (P < .001) was lower than the non-VTE group. Meanwhile, CA199 (P = .001) and CEA (P < .001) values were higher in the VTE group (Table 1).
The Demographic Factors, Comorbidities, and Hematological Indices Between the non-VTE Group and VTE Group.

VTE, venous thromboembolism; PT, prothrombin time; PTR, prothrombin ratio; INR, international normalized ratio; APTT, activated partial thromboplastin time; TT, thrombin time.
Univariate and multivariate analyses of VTE in patients with CRC showed that age (OR 0.9771, 95% CI 0.9554-0.9994; P = .0444), temperature (OR 4.2824, 95% CI 1.1361-16.1418; P = .0317), amount of blood loss (OR 1.0034, 95% CI 1.0009-1.0059; P = .0067), PICC (OR 26.75, 95% CI 15.2554-46.9053 ; P < .0001), chemotherapy (OR 1.7223, 95% CI 1.0099-2.9371; P = .0459), anemia (OR 2.6483, 95% CI 1.5068-4.6547; P = .0007), INR (OR 77.2943, 95% CI 4.3974-1358.6278; P = .003), and CA199 (OR 1.0024, 95% CI 1.0003-1.0045; P = .0234) were independent risk factors for VTE (Table 2). On receiver operating characteristic analysis, age, temperature, amount of blood loss, PICC, anemia, INR, and CA199 were all predictive for VTE in colorectal patients. The amount of blood loss predicted VTE better (AUC = 0.778, 95% CI 0.740-0.813, P < .001), when the cutoff value was 20 mL, the sensitivity was 76.17%, and the specificity was 79.18%, respectively. And PICC predicted VTE better (AUC = 0.808, 95% CI 0.772-0.841, P < .001), the sensitivity was 70.21%, and the specificity was 91.47%, respectively (Table 3, Figures 3 and 4).
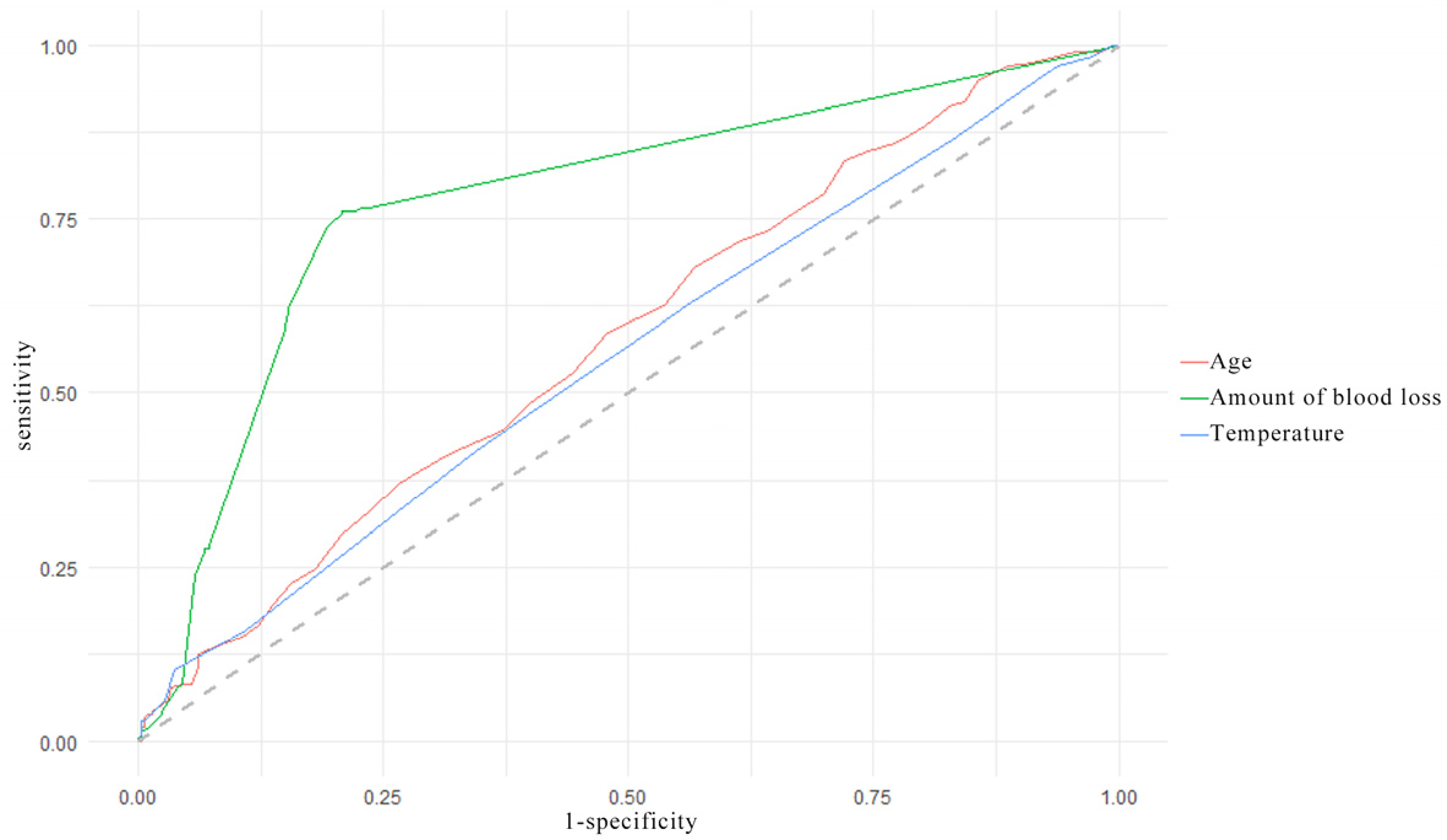
Receiver operating characteristic curve analysis of age, amount of blood loss, and temperature in predicting venous thromboembolism formation in colorectal cancer patients.

Receiver operating characteristic curve analysis of INR, CA199, PICC, chemotherapy, and anemia in predicting venous thromboembolism formation in colorectal cancer patients.
Logistic Regression Analysis of Risk Factors for Venous Thromboembolism in Patients With Colorectal Cancers.

PT, prothrombin time; PTR, prothrombin ratio; INR, international normalized ratio; APTT, activated partial thromboplastin time; TT, thrombin time.
ROC Curve Analysis of Clinicopathological Characteristics in Predicting Venous Thromboembolism in Patients With Colorectal Cancers.

INR, international normalized ratio.
Discussion
CRC is one of the most common abdominal cancers. 15 The changes in pelvic anatomy caused by surgery and the systemic hypercoagulability caused by the disease lead to a higher incidence of VTE in patients undergoing CRC surgery than those undergoing ordinary abdominal surgery, increasing the risk of death in cancer patients.6,16,17 However, there is no unified conclusion on the risk factors for VTE formation in inpatients with CRC in China, our study found that age, temperature, amount of blood loss, PICC, chemotherapy, anemia, INR, and CA199 were independent risk factors for VTE.
In this study, 235 patients in the VTE group and 293 patients in the non-VTE group were selected to better statistically compare the difference between the 2 groups, and to explore risk factors for VTE formation in inpatients with CRC in China. A total of 33 demographic factors, comorbidities, and hematological indicators were included. Our study showed that CRC patients in the VTE group were younger than those without VTE, which was similar to the previous research. 14 But the results differed from those of previous studies, possibly because of differences in the population and disease studied.18,19 Gender had no statistically significant difference between the 2 groups in this study, unlike the previous study, which may be due to the different populations and regions studied. 20 CRCs patients who smoked had a higher rate of VTE formation, which was different from previous studies.10,21 Perhaps because smokers have abnormal coagulation, the potential effect of inflammation on clotting offers further potential for thrombosis. CRC patients who had bleeding during surgery were more likely to develop VTE, bleeding instinctively activates the patient's coagulation system and increases the risk of VTE. The patients with poor differentiation in the VTE group were more than those in the non-VTE group, which may mean that the higher the malignancy of CRC, the higher the likelihood of thrombosis. The incidence of VTE in CRC patients with PICC was significantly higher than that in patients without PICC, which was consistent with the results of previous studies.22–24 The catheter was placed in the venous lumen, resulting in destruction of vascular endothelial cells, hemodynamic changes, and blood stasis. By upsetting the balance of the Virchow triad, these factors create a hypercoagulable state that increases the chance of thrombosis. In this study, cancer patients receiving radiotherapy were not prone to VTE, and there was no statistically significant difference between the VTE group and the non-VTE group in chemotherapy, these results were different from previous studies.25,26 The different cancers studied in these studies, and the different radiotherapy and chemotherapy regimens adopted, naturally lead to different conclusions.
In addition, we also explored differences in hematological indexes between the VTE and non-VTE groups in CRC patients. Anemia was more common in the VTE group, which paralleled the findings in past studies.27,28 CRC patients in the VTE group had higher rates of infection with higher white blood cell counts, which was similar to previous studies.29,30 Inflammatory responses are involved in thrombosis, leukocytes (mainly neutrophils), vascular endothelial cells, and blood plates, and their release of inflammatory mediators play an important role in thrombogenesis. 31 At the same time, we also collected and compared the parameters representing coagulation and bleeding between the 2 groups, and found that the VTE group had higher PT, PTR, INR, TT, and lower PT%. We also found that CA199 and CEA values were higher in the VTE group. These parameters had not been examined in previous studies, and these results suggested differences in the coagulation status of cancer patients and a greater likelihood of thrombosis when cancer was active.
Our research found that age, temperature, amount of blood loss, PICC, chemotherapy, anemia, INR, and CA199 were independent risk factors for VTE, which was not exactly the same as previous studies.10,13,22,32 PICC was recognized as an independent risk factor for thrombosis, the behavior leads to a hypercoagulable state of the blood and increases the risk of thrombosis by disrupting the Virchow triad.23,24 Chemotherapeutic drugs can inhibit the synthesis of prostacylin, thrombomodulin, heparan sulfate, tissue factor pathway inhibitors, and plasminogen activator, thus facilitating the formation of venous thrombosis. Furthermore after the tumor cells were damaged by chemotherapy, the release of pro-inflammatory and pro-angiogenic factors would have an effect on the vascular endothelial cells, and finally induced the endothelial cells to present the pre-thrombotic changes through the up-regulation of tissue factor, cell adhesion factor, and plasminogen activator inhibitor. Our results suggest that chemotherapy was an independent risk factor for VTE formation in CRC patients. We need to be highly alert to these risk factors in clinical treatment and timely give the patients corresponding intervention and treatment.
ROC curve analysis showed that the amount of blood loss and PICC were effective in predicting the occurrence of thrombosis, age, temperature, anemia, INR, and CA199 also had certain predictive evaluation efficiency, but their accuracy was slightly weakened compared with the former. So as stated earlier, clinicopathological characteristics of CRC had a certain degree of influence on the formation of VTE in patients, we need to always pay attention to the dynamic changes of relevant indexes, timely receiving primary signals emitted by the body, so as to make corresponding treatment to prevent further development of VTE.
In this study, the risk factors of VTE formation in CRC were obtained, which was conducive to the clinical prevention and treatment. However, this study was a single-center study with a small sample size, and the statistical results may be biased to a certain extent. Further multicenter studies with a large sample size are expected. On the other hand, the specific demographical conditions of our study in China affect the contribution of independent risk factors, and a worldwide study is urgently needed.
Conclusions
There were differences in demographic factors, comorbidities, and hematological indices between the non-VTE group and VTE group in CRC patients. Age, temperature, amount of blood loss, PICC, chemotherapy, anemia, INR, and CA199 were independent risk factors for the formation of VTE in the patients. Receiver operating characteristic curve analysis showed that the amount of blood loss and PICC predicted thrombosis better. Accurate identification of these risk factors was helpful for clinical diagnosis and treatment.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Fund of Health Commission of Heilongjiang Province numbered 20230303010309.
Ethical Approval
Ethical approval to report this case was obtained from Ethics Committee of Harbin Medical University Cancer Hospital.
Informed Consent
Written informed consent was obtained from the patients for their anonymized information to be published in this article.
