Abstract
Background
The presence of an outer shell has been recently described as a common feature of acute ischemic stroke (AIS) thrombi. We performed a systematic review of the current literature on shell genesis, structure, and clinical significance.
Methods
Following PRISMA guidelines, we searched Ovid Cochrane Central Register of Controlled Trials, Embase, Medline, Scopus, and Web of Science for studies reporting the composition and structure of AIS thrombi and clot analogs. Identified studies were added to Covidence software for primary screening. Two reviewers independently screened titles and abstracts followed by full-text screening.
Results
From 1290 identified studies, 10 were included in this review. Studies using histology/immunohistochemistry/immunofluorescence described fibrin, platelets, von Willebrand factor, and neutrophil extracellular traps as the main components of the shell. Scanning electron microscopy demonstrated a dense, compact fibrin/platelet-rich shell, and a core rich in polyhedrocytes. Microfluidics studies identified highly activated P-selectin-positive platelets and fibrin forming the core while secondary agonists adenosine diphosphate and thromboxane, along with loosely packed P-selectin-negative platelets constituted the shell.
Conclusions
The composition, compaction, and integrity of the shell may impact thrombolysis and revascularization outcomes. The preponderance of studies supported this conclusion.
Introduction
The development and integration of endovascular treatment for acute ischemic stroke (AIS) have provided the opportunity to study clots retrieved via mechanical thrombectomy (MT), allowing previously unanswered questions about clot composition and stroke etiology to be explored.1–6 Moreover, there has been a growing interest in understanding the factors responsible for the effectiveness of pharmacological and endovascular treatment.7,8
In general, thrombi have different compositions based on the thrombotic disorder: arterial thrombi obtained from patients with myocardial infarction contain mainly fibrin and other components such as platelets, red blood cells (RBCs), leukocytes, and cholesterol crystals while venous thrombi are composed of RBCs and fibrin with sparse platelets and leukocytes. 9 AIS thrombi are typically formed of RBC-rich and platelet-rich areas each of them incorporating fibrin and other components in different amounts and distributions. 8 Nevertheless, there is evidence that fibrin forms a film on the clot surface regardless of the formation site. 9
In this context, the presence of an outer fibrin crust shell in AIS thrombi was first described in 2016 by Ahn et al. 10 Further studies exploring the shell have been sparse, with not enough emphasis given to the shell in histological or immunohistochemical analysis. Di Meglio et al 11 have recently described the shell-core structure of AIS thrombi at length suggesting that thrombi shell may impair fibrinolysis.
This systematic review aimed to define and assess the current state of knowledge of the AIS thrombus shell genesis, structure and clinical significance, while providing possible avenues of research for future studies.
Materials and Methods
Eligibility Criteria
The current review considered all human studies investigating the composition and organization of retrieved thrombi and clot analogs.
We built our inclusion criteria in the PICO (Population, Intervention, Comparison, Outcome) format; the population included patients with AIS, the intervention was MT with/without prior thrombolysis, and no control/comparison was needed. For the outcomes of interest, we included studies investigating the thrombus shell formation, structure, or clinical significance. Articles were limited to English-language peer-reviewed journal articles to ensure quality was upheld.
We excluded all review articles, editorials, case reports, conference abstracts, and animal studies. In the case of studies with duplicate or overlapping patient populations, only the publication with the most complete dataset was included.
Literature Search Strategy
A full electronic search was conducted on July 19, 2022 by a medical librarian in Ovid Cochrane Central Register of Controlled Trials (1991+), Ovid Embase (1974+), Ovid Medline (1946+ including epub ahead of print, in-process, and other non-indexed citations), Scopus (1823+), and Web of Science Core Collection (Science Citation Index Expanded 1975+ and Emerging Sources Citation Index 2015+). Search strategies were created using a combination of keywords and standardized index terms. Full search strategies are provided in Supplemental Table 1. The search was not extended to unpublished studies or other sources of grey literature.
Selection and Screening Process
Once retrieved, studies were added to Covidence software for primary screening and management. Screening was guided by the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) flow diagram (Figure 1). Title and abstracts yielded by the search were independently screened by two reviewers blinded to one another using the inclusion criteria, and all duplicates were removed. Full-length studies were considered if the inclusion criteria were met or the title/abstract suggested that the content may be relevant. The reviewers then screened the full-text studies to evaluate whether these met the inclusion criteria. Reasons for excluding articles at this stage were recorded. Reviewer discrepancies were resolved by discussion or in conjugation with the senior author.

PRISMA flowchart of the electronic database search and selection of the studies.
Data Extraction
The following information was collected and extracted from the eligible studies by reviewers: general information: (1) first author name, year of publication, journal; (2) type of study; (3) sample type and size; (4) objective(s) of the study; and (5) main findings; methodology: (1) conventional histology; (2) immunohistochemical (IHC)/immunofluorescence (IF) analysis, and whole mount tissue immunostaining; (3) microfluidic assays and intravital microscopy; and (4) scanning electron microscopy (SEM). The data extracted were synthesized into tables and text to explain the characteristics and relevant findings for each study. The extracted data were double-checked for validation.
Methodology Used to Characterize the Mechanisms of Thrombus Formation and Composition
Traditional histology, IHC/IF, and whole-mount tissue immunostaining aimed to identify various components of AIS thrombi and offered descriptive and qualitative data. SEM revealed clot architecture and ultrastructural features (ie, intravital contraction) that cannot be observed using routine histological techniques. Microfluidic assays allowed to interrogate AIS thrombus formation and growth under flow as well as biological processes such as platelet activation. Details on the methodologies used and their advantages and limitations are presented in Table 1.
Summary of the Methodologies Used for the Assessment of Acute Ischemic Stroke Clot Composition and Structure.
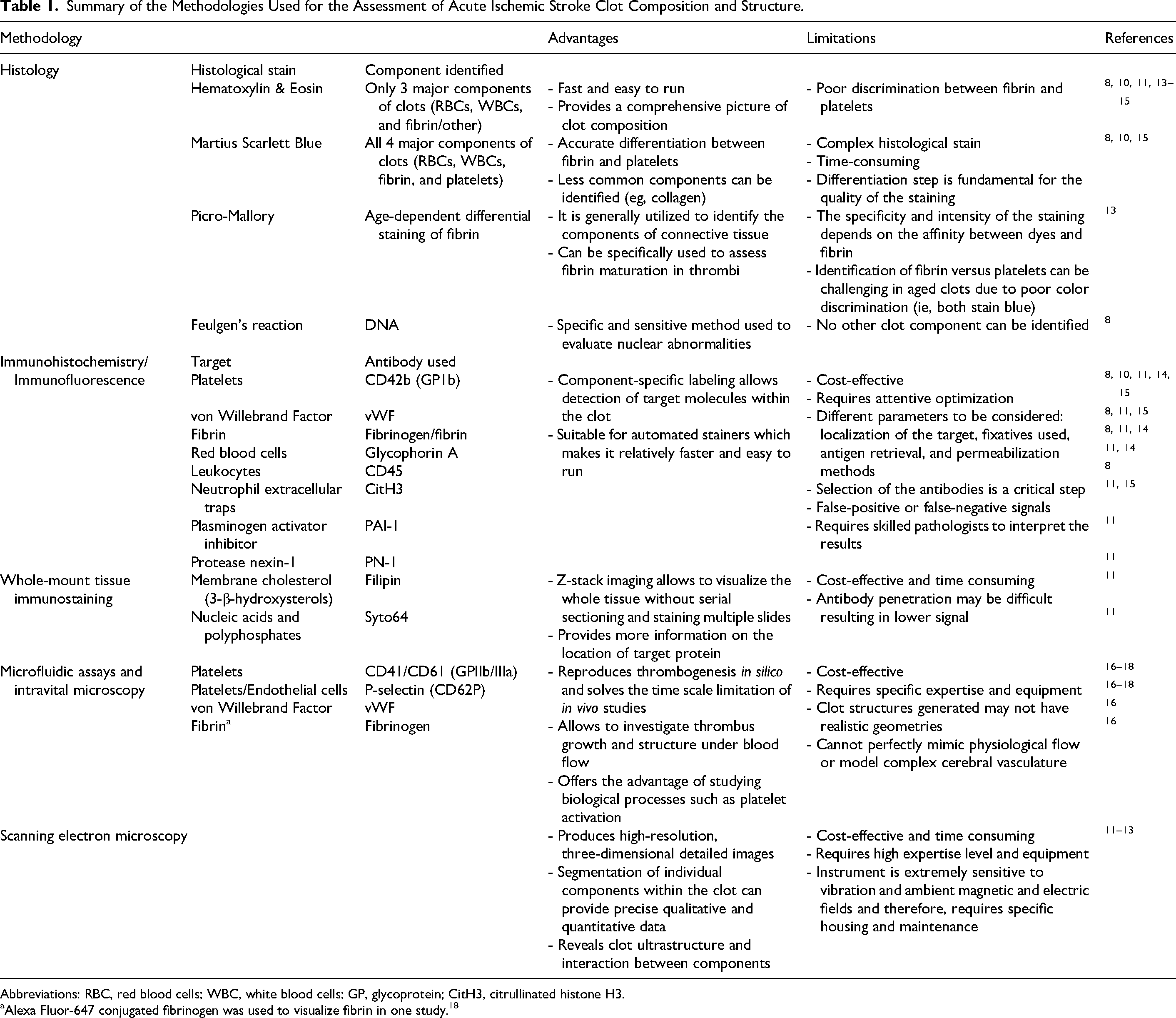
Abbreviations: RBC, red blood cells; WBC, white blood cells; GP, glycoprotein; CitH3, citrullinated histone H3.
Alexa Fluor-647 conjugated fibrinogen was used to visualize fibrin in one study. 18
Macroscopic Evaluation
Macroscopic evaluation of retrieved thrombi was carried out in two studies.8,12
Histology and IHC/IF
Six of the ten studies used conventional histopathology such as Hematoxylin & Eosin (H&E) or Martius Scarlett Blue (MSB) stainings as well as IHC/IF to describe the main clot components.8,10,11,13–15 Khismatullin et al 13 used Picro-Mallory stain for the age-dependent differentiation of fibrin. Another study employed Feulgen's reaction to stain extracellular DNA. 8
Five studies performed IHC/IF to better visualize and quantify specific components within the thrombi such as platelets via CD42b (Glycoprotein1b, GP1b) antibody, von Willebrand Factor (vWF), and fibrin.8,10,11,14,15 Two studies used glycophorin A to identify RBCs.11,14 Leukocytes were stained with CD45 antibody, 8 the serpins plasminogen activator inhibitor (PAI-1), and protease nexin-1 (PN-1) were labeled in one study. 11 Neutrophil extracellular traps (NETs) were targeted using citrullinated histone H3 (CitH3) in two studies.11,15 Di Meglio et al 11 performed whole-mount immunostaining using Filipin and Syto64 to target membrane cholesterol (3-β hydroxysterols) and nucleic acids/polyphosphates, respectively.
Microfluidic Assays
Three studies utilized microfluidic assays to test their hypotheses and describe the flow conditions under which the shell-core architecture of thrombi arises.16–18 Platelets were labeled with CD41/CD61 (GIIb/IIIa) antibody as well as with an antibody against the transmembrane cell adhesion protein P-selectin (CD62P), which is also expressed by the endothelial cells.
SEM Analysis
SEM was performed in three studies to gain a better understanding of the AIS clot ultrastructure and to describe the interaction between AIS thrombi and MT devices.11–13
Results
Search Results and Study Selection
The search from the electronic databases retrieved 1290 peer-reviewed journal articles (Embase: 318; Medline: 269; Scopus: 346; Web of Science: 330; CENTRAL: 27). A further 10 articles that appeared relevant were included manually. Upon removing duplicates and irrelevant records, 80 articles were assessed for eligibility by full-text screening. Of these, 10 articles were included in the review; the rest did not meet the inclusion criteria (Figure 1).
Summary of Study Characteristics
A summary of the studies included is presented in Table 2. The studies were published over a period of 6 years (2016-2022) in different journals.
Studies Included in the Systematic Review.
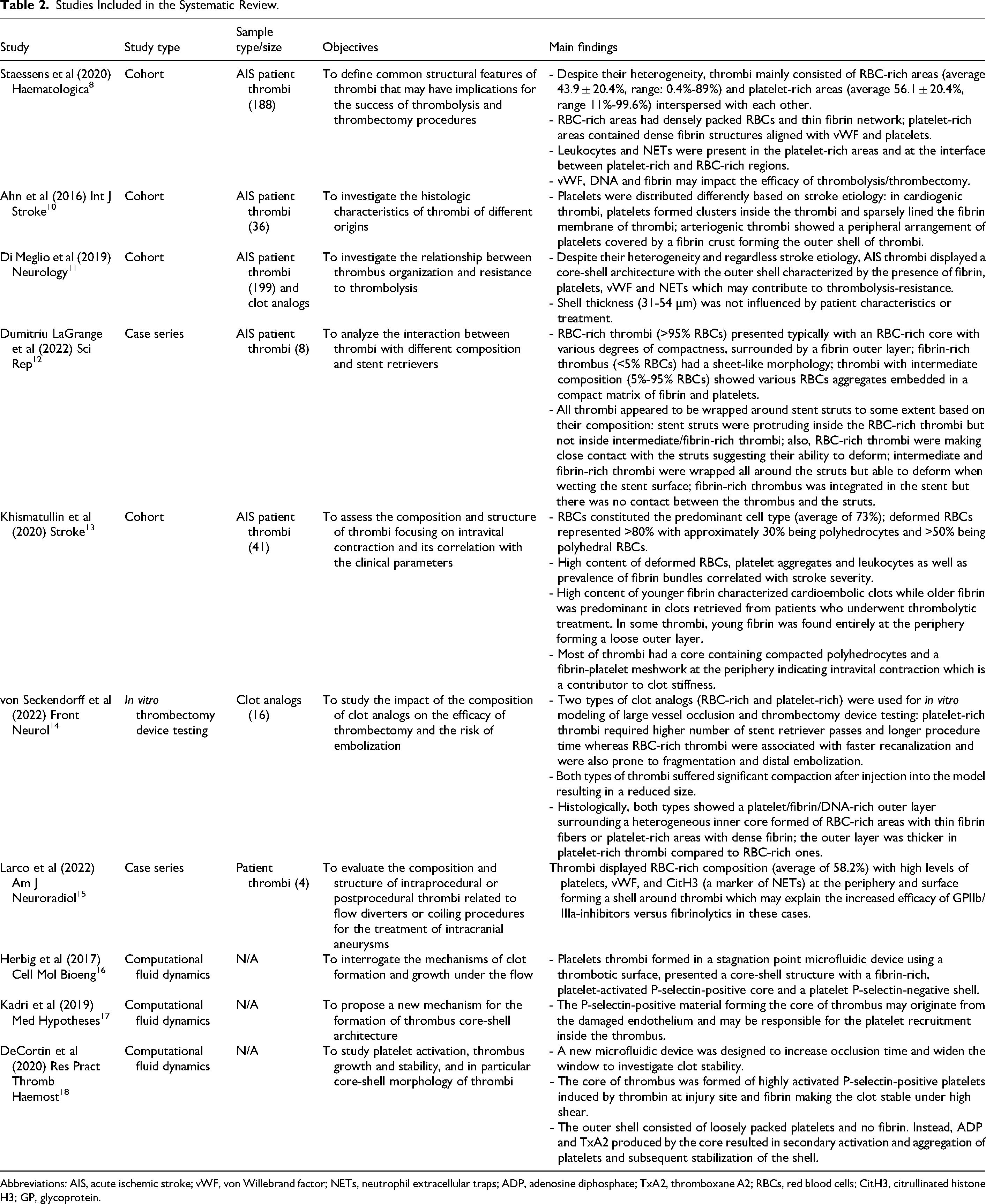
Abbreviations: AIS, acute ischemic stroke; vWF, von Willebrand factor; NETs, neutrophil extracellular traps; ADP, adenosine diphosphate; TxA2, thromboxane A2; RBCs, red blood cells; CitH3, citrullinated histone H3; GP, glycoprotein.
The majority were cohort studies (40%) with varying cohort sizes, ranging from 36 to 199 thrombi retrieved from AIS patients. Two case series of four and eight patients were also included. In addition to retrieved thrombi, clot analogs were used in two studies. In three studies (30%), computational fluid dynamics was performed to investigate clot formation under varying physical and flow conditions.
Characteristics of AIS Retrieved Thrombi and Clot Analogs: the Presence of Shell
Overall, the studies aimed to investigate the composition and organization of AIS thrombi and their impact on the resistance or sensitivity of thrombi to fibrinolysis and endovascular treatment.
Macroscopically, thrombi appeared to be heterogeneous in size, shape, and color. 8 Histologically, several studies classified clots based on the predominant component using different cut-off values and criteria to establish this. Staessens et al 8 found that most of the clots consisted of RBC-rich areas and platelet-rich areas interspersed with each other and therefore, classified them as RBC-rich/platelet-poor, mixed, and RBC-poor/platelet-rich. Furthermore, IHC/IF analysis showed that RBC-rich areas contained densely packed RBCs and thin fibrin strands while platelet-rich areas showed dense fibrin structures aligned with vWF and platelets. Leukocytes and NETs were found in the platelet-rich areas and at the interface between platelet-rich and RBC-rich regions.
In another study, AIS thrombi were classified as RBC-rich, fibrin-rich, and thrombi with intermediate composition. 12 Ahn et al 10 described the characteristics of thrombi based on stroke etiology. RBCs were the most abundant component in arteriogenic clots, followed by fibrin. In cardiogenic clots, fibrin was the most abundant component, followed by RBCs. The proportions of white blood cells (WBCs) and platelets did not differ significantly between the two types of thrombi. Thrombi with undetermined etiology had a composition similar to cardiogenic thrombi.
Despite the heterogeneity of AIS thrombi, the presence of a surface shell was a common feature that may represent the structural basis for resistance to recombinant tissue plasminogen activator (rt-PA) treatment.
Staessens et al showed that some AIS thrombi had an RBC-rich core surrounded by platelet-rich material that could hamper thrombolysis. 8 Ahn et al 10 demonstrated that arteriogenic thrombi present a “fibrin crust” constituting the outer shell and platelets distributed underneath which may inhibit the initial action of rt-PA on the surface of thrombi. Moreover, activated platelets may release rt-PA inhibitors such as PAI-1 or PN-1. The immunohistologic analysis performed by Di Meglio et al 11 showed high variability of the inner core in terms of proportion and organization of the fibrillary and cellular content whereas the shell typically contained fibrin with altered morphology forming dense matted deposits, platelets, vWF, NETs as well as PAI-1 and PN-1. Increased amount of fibrin, platelets, vWF, WBCs, and NETs within the shell was demonstrated in all 164 thrombi analyzed out of 199 total AIS thrombi (82%) included in the study. Moreover, the shell thickness varied between 31 and 54 µm and was not influenced by the AIS etiology, patient characteristics, or treatment. The authors also examined the shell-core structure of 30 AIS thrombi using SEM analysis and revealed that 24 thrombi (80%) had a compacted, sealed outer shell surrounding a loose RBC-rich core.
Overall, these important findings indicate that majority of AIS thrombi present an outer shell covering entirely or partially their surface.
Khismatullin et al 13 found that young fibrin was mostly located at periphery and characterized cardioembolic clots. Older fibrin was predominant in clots retrieved from patients who underwent thrombolytic treatment suggesting that rt-PA may have dissolved the young fibrin at the periphery of thrombi.
Larco et al 15 analyzed the composition of intraprocedural or postprocedural thrombi related to flow diverters or coiling procedures for the treatment of intracranial aneurysms. The authors found that all thrombi were predominantly composed of RBCs and had an outer shell. The expression of CD42b, vWF, and CitH3 was higher in the shell compared to the overall expression in thrombi. Therefore, these thrombi may respond better to GPIIb/IIIa-inhibitors rather than rt-PA.
The study of Khismatullin et al 13 assessed the intravital contraction of AIS thrombi and its correlation with clinical characteristics. The quantitative analysis of the scanning electron micrographs showed that RBCs were the predominant cell type with deformed RBCs representing more than 80%. The platelet-driven contraction and compression of the thrombus caused the formation of polyhedral RBCs in the core, while fibrin/platelets were found at periphery. Moreover, the high content of deformed RBCs, platelet aggregates, and leukocytes correlated with stroke severity. Fibrin content, specifically fibrin bundles, was significantly higher in cardioembolic thrombi than in atherothrombogenic ones.
Therefore, the tight compaction of shell components, the integrity of the shell and increased clot stiffness are factors capable of impairing thrombolysis.
The overall composition and shell-core organization of AIS thrombi may also impact the success of thrombectomy. Von Seckendorff et al 14 used two types of clot analogs (RBC-rich and platelet-rich) to simulate cerebral arterial thrombectomy in vitro and found that platelet-rich thrombi required more stent retriever passes to achieve complete recanalization than RBC-rich thrombi which were more prone to fragmentation and distal embolization. The retrieved thrombi displayed histological features similar to thrombi from AIS patients with a platelet/fibrin/DNA-rich outer layer encapsulating an RBC-rich core. This outer layer was thicker in the platelet-rich thrombi than in the RBC-rich ones, surrounding a heterogeneous inner core formed of RBC-rich areas with thin fibrin fibers or platelet-rich areas with dense fibrin networks.
Dumitriu LaGrange et al 12 investigated the interaction between AIS thrombi and stent retrievers using SEM. RBC-rich thrombi had a core rich in polyhedrocytes and an outer fibrin layer while fibrin-rich thrombi had a sheet-like morphology with layers of fibrin bundles forming aggregates and layers of porous fibrin. Thrombi with intermediate composition showed RBCs aggregates embedded in a dense matrix of fibrin and platelets. The core regions with compacted polyhedrocytes are less susceptible to fibrinolysis and did not allow stent struts to protrude inside the thrombus. The loosely packed regions of RBC-rich thrombi were attached to the stent struts suggesting their ability to deform. The intermediate and fibrin-rich thrombi were wrapped all around the struts but were able to deform when wetting the stent surface. The fibrin-rich thrombus was folded around the stent struts without making close contact.
Hypotheses of Shell Formation
Three microfluidic studies investigated the mechanism of thrombus formation under flow conditions and the potential factors responsible for the shell-core architecture of AIS clots.
Herbig et al 16 studied thrombus formation in stagnation points by using a microfluidic device with a thrombotic collagen/tissue factor (TF) surface that was then subjected to a perpendicular flow of whole blood. They found that thrombus morphology was influenced by the inlet flow rates, with a dendritic structure forming in low flow and an evenly spread thrombus in high flow, likely due to the higher shear gradients that deform and densify the platelet mass to an even mass. Thrombi formed at both flow rates had a distinct shell-core architecture with a highly activated platelet P-selectin-positive and fibrin-rich core and a less dense platelet P-selectin-negative shell suggesting that vWF found in the core could be a combination of plasma and platelet vWF. Platelet adhesion and vWF deposition near stagnation point at both flow rates were significantly reduced by adding N-acetylcysteine (NAC). Moreover, inhibiting the fibrin polymerization did not change thrombus structure or the accumulation of platelets/vWF.
Kadri et al 17 described the shell-core structure with the core responsible for attaching the clot to the vessel wall and stopping the blood loss while the shell represents a source of the pathogenic embolism leading to thrombosis and fatal complications. The core and shell of thrombi have a distinct molecular composition due to the limited diffusion of certain larger particles from the core toward the outer parts of thrombi. The core is formed by soluble agonists released at the site of injury, particularly thrombin which triggers platelet activation in the core. This results in a densely packed platelet core, fibrin deposition, and decreased solute transport. In contrast, the shell comprises loosely packed platelets, high solute transport, and no fibrin. The activated platelets in the core release α-granules causing a secondary gradient of smaller agonists, such as adenosine diphosphate (ADP) and thromboxane (TxA2) that recruit, activate and stabilize additional platelets within the shell. Furthermore, the authors proposed that the P-selectin positivity of the core compared to the shell may be due to P-selectin originating from multiple sources such as damaged endothelial cells and activated platelets.
DeCortin et al 18 designed a new microfluidic device with extended channel height to increase the occlusion time and investigate clot stability by perfusing whole blood over collagen/TF and buffer exchange. The authors found that TF, and thus thrombin, increased clot stability and the number of activated platelets at high and low shear rates. Thrombin inhibition reduced the number of P-selectin-positive platelets and thrombus stability confirming that thrombin is a key activator of platelets. Fibrin inhibition increased the number of P-selectin-positive platelets but did not reduce thrombus stability suggesting that fibrin resides only in the P-selectin-positive, shear-resistant core. P-selectin-positive platelets were found to be more stable than P-selectin-negative platelets. Soluble agonist inhibitors such as acetylsalicylic acid (ASA), which blocks the production of TxA2 and MRS-2179, an ADP inhibitor, were also used to test clot stability. ASA-treated thrombus eroded more under increased shear and buffer conditions but the P-selectin-positive platelets remained stable under high shear rates. ADP inhibition lowered P-selectin-positive platelets during the growth period but when switching to buffer, P-selectin-positive platelets remained stable suggesting that TxA2 production by the core is crucial in stabilizing shell platelets whereas ADP may be more important in the initial platelet aggregation. Therefore, secondary agonists stabilize platelets in the shell region that are more likely to be sheared off.
Discussion
In the present systematic review, we focused on the existing knowledge of the AIS thrombus shell through two broad lenses: histopathology/SEM and computational fluid dynamics. The studies employing histopathology and SEM described a dense, compact, fibrin-, and platelet-rich shell which may explain the resistance to fibrinolytic treatment. Moreover, the intravital contraction of thrombi compresses and deforms RBCs to polyhedrocytes forming dense clusters within the core and causing decreased clot porosity that may also impact thrombolysis. The studies using microfluidic models explored the genesis of the shell-core architecture that can be viewed in the context of fluid forces. These studies found that platelet activation and the behavior of different components (and their agonists) that comprise thrombi may be responsible for the formation of the shell.
Computational fluid dynamics and SEM require high expertise level and equipment; conventional histology and IHC/IF were employed by more groups as standard protocols to investigate thrombus composition and structure. All histopathology/SEM studies included in this review provided a comprehensive analysis of the AIS thrombus shell and unanimously described a dense, compacted, fibrin- and platelet-rich shell surrounding an RBC-dominant core mainly formed of polyhedrocytes.8,11,13 The shell and core represent two separate entities with distinct cellular characteristics as the core retains visible individual cells and fibers whereas the shell shows altered morphology characterized by the lack of clear cell borders due to compaction and densely matted fibrin with no identifiable fibers induced by oxidation, carbamylation, or exposure to platelet factor 4. 11 Therefore, the shell has reduced porosity and is more resistant to rt-PA treatment than the core. The presence of fibrin, platelets, WBCs, vWF, NETs (Figure 2), along with factors such as PAI-1 and PN-1 may explain the shell's increased structural and functional integrity which is another important aspect as thrombi with disrupted shell undergo lysis much faster than intact clots. 11

Shell-core structure of acute ischemic stroke thrombi revealed in three representative cases using histology/immunohistochemistry (IHC)/immunofluorescence (IF) and scanning electron microscopy (SEM). In the left panel, gross photo of a thrombus shows a thin white layer at the surface of the clot representing the outer shell which surrounds a red blood cell (RBC)-rich core. Hematoxylin & Eosin (H&E) and Martius Scarlet Blue (MSB) stainings were performed. IHC for platelets (CD42b), von Willebrand factor (vWF), fibrin/fibrinogen, and citrullinated histone H3 (CitH3) demonstrate their presence in the shell. IHC images are higher magnifications of the clot areas in rectangle in the H&E/MSB images. Arrows indicate the shell while the asterisk corresponds to the core. IF shows the presence of DNA (DAPI), CitH3, and vWF as well as their co-localization in the shell. IF images are higher magnification views of the area indicated by arrow in the MSB staining of another thrombus. In the right panel, SEM reveals the ultrastructure of a third thrombus presenting a dense, compacted shell (A-D) which surrounds an RBC-dominant core mainly formed of polyhedrocytes (E-H). Multiple folds and ridges are seen on the surface of the clot. Arrow (A, shell) and asterisk (E, core) correspond to the locations analyzed by SEM. (B-D) Higher magnifications of the surface show integration of components and mature organization. Some normal RBC that are not embedded in the thrombus are evident which may be due to insufficient rinsing of the fragment after retrieval. (F-H) Magnified view of the core displays compacted polyhedrocytes indicative of limited integration and immature structure. Scale bar (left panel) = 750 µm (gross photo), 500 µm (H&E, MSB), 200 µm (CD42b/vWF/CitH3 IHC), 100 µm (fibrin IHC), 2 mm (MSB) and 20 µm (IF); Scale bar (right panel) = 500 µm (A, E), 100 µm (B, F), 50 µm (C, G) and 10 µm (D, H).
SEM analysis showed the ultrastructural organization of AIS thrombi and demonstrated that intravital contraction of thrombi driven by activated platelets is critical for the formation of the shell through the redistribution of the platelet-fibrin meshwork to the periphery and compacted RBCs to the core. A higher proportion of deformed RBCs suggests greater compaction and is associated with increased stroke severity and lower fibrinolytic susceptibility. 13 While histology/IHC helped to study the impact of thrombus composition on the efficacy of thrombectomy, 14 SEM highlighted the interaction between various components (Figure 2).13,19 SEM also evaluated the interaction between thrombi and stent retrievers based on the composition of thrombi and the adhesive affinity for the stent surface. 12
Microfluidics uses and manipulates fluids at geometrically constrained scales, micrometers, to study the effect of surface forces on them. The studies using microfluidic models highlighted the importance of blood flow and vessel characteristics and inferred different theories of shell formation. The distinct shell-core structure of AIS thrombi formed at both low and high flow rates with a highly activated P-selectin positive, dense platelet and fibrin-rich core from which secondary agonists ADP and TxA2 flow outwards. P-selectin is proposed to confer the core its dense compaction and stability against erosion.16,17 In contrast, the shell consisted of a loose layer of P-selectin-negative platelets with little to no fibrin present.16,18
The presence of the shell in AIS thrombi has important therapeutic implications. First, the authors went into detail8,10,11,15 elucidating several shell components that are known to have specific inhibitors such as platelets (i.e, GPIIb/IIIa-inhibitors), vWF (ie, ADAMTS13 and NAC), NETs, and extracellular DNA (i.e, DNAse 1).20–22 Furthermore, PAI-1 and PN-1 may also represent potential therapeutic targets. Therefore, future reperfusion therapies may benefit from targeting the non-fibrin components of the shell, which could be particularly efficient in platelet/vWF-rich thrombi and may also help partially dissolve the shell. Moreover, targeting these factors in addition to rt-PA will benefit AIS treatment regardless the etiology since the presence of the shell and its thickness are not influenced by patient characteristics or the source of thrombi.
The microfluidic studies also proposed possible targets with platelets/vWF and secondary agonists being the most important. While NAC interferes with the platelet/vWF and vWF-vWF interactions reducing the overall platelet deposition, 16 the P-selectin-negative shell is more prone to erosion by shear stress when treated with ASA compared to the thrombus core. 18
Secondly, it has been recognized that overall composition of AIS thrombi impacts the efficacy of MT with platelet-rich thrombi being more resistant to MT and RBC-rich thrombi more prone to fragmentation.7,14 Other components such as NETs were shown to be associated with increased number of passes required for recanalization. 22 The incorporation of AIS thrombi into the stent retrievers is also influenced by thrombus composition. 12 These findings suggest that certain components of the shell (ie, association of vWF and NETs with fibrin) could make AIS thrombi more difficult to retrieve by MT but further investigations are necessary to better understand the mechanisms involved.
This systematic review points toward the importance of thrombus analysis in other thrombotic disorders. In addition, the choice of appropriate methodology to study thrombus composition requires careful consideration. Despite the compositional differences between arterial and venous thrombi, the presence of a fibrin film on the surface of thrombi is a common feature that may account for similar therapeutic strategies. Moreover, the surface of coronary thrombi contains more fibrin and platelets and few RBCs while the inner part is rich in densely packed polyhedrocytes. Notably, polyhedrocyte formation increases with ischemic time. 9 Polyhedrocytes and intermediate forms represent the main constituents of venous and pulmonary emboli. 23 Regardless of the origin of thrombi, the presence of polyhedrocytes is a sign of clot contraction and indicates a common mechanism of thrombus formation. Nonetheless, it is crucial to acknowledge the role of local conditions at sites of thrombus initiation and growth (ie, blood flow and vessel wall) in defining the final thrombus characteristics.
The studies included in this review had several limitations. The major limitation in analyzing retrieved thrombi is that only thrombi that have not dissolved, spontaneously or during thrombolysis, and were successfully retrieved can be assessed. The structural integrity of the thrombi may be impacted during MT leading to fragmentation in many cases, 10 hence evaluation of thrombi as a whole is not possible. In addition, the rt-PA treatment prior to MT may cause morphological changes. Furthermore, it is not possible to evaluate the presence of the shell in thrombi that rt-PA dissolved completely. The clot analogs used in these studies were representative for only two types of thrombi (RBC-rich and platelet-rich). Therefore, the heterogeneity and complete spectrum of AIS thrombi were not captured. The case series included in this review were single-center studies with small sample sizes. Multi-institutional registries and larger cohorts are warranted to confirm the findings presented in this review. Lastly, the microfluidic assays allow to investigate thrombus growth, stability and morphology but cannot perfectly model physiological flow or mimic more complex geometries of the cerebral vasculature.
Conclusions
The presence of the shell represents a common feature of AIS thrombi that may affect thrombolysis and MT. Further analysis is required to identify novel constituents of the shell and develop new pharmacological strategies for clot lysis. Advanced technologies such as proteomics and Fourier-transform infrared spectroscopy may represent valid tools for studying signaling pathways and detecting biochemical parameters that could provide a definite shell signature and advance AIS treatment.
Supplemental Material
sj-pdf-1-cat-10.1177_10760296231213632 - Supplemental material for Shell Versus Core Architecture and Biology of Thrombi in Acute Ischemic Stroke: A Systematic Review
Supplemental material, sj-pdf-1-cat-10.1177_10760296231213632 for Shell Versus Core Architecture and Biology of Thrombi in Acute Ischemic Stroke: A Systematic Review by Oana Madalina Mereuta, Tamanna Agarwal, Sherief Ghozy, Daying Dai, Santhosh Arul, Waleed Brinjikji, David F. Kallmes and Ram Kadirvel in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Data Availability
Data are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institutes of Health (R01 NS105853).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
