Abstract
Although dual antiplatelet therapy for secondary prevention in acute coronary syndrome (ACS) is highly recommended by current guidelines, P2Y12 inhibitor non-adherence often occurs and devastates prognosis. To evaluate whether the ischemic risk during the early period of clopidogrel noncompliance was increased among ACS patients, a comprehensive search of PubMed, Embase, and Web of Science was conducted to identify studies reporting early ischemic risk after clopidogrel noncompliance in ACS patients. The primary endpoint was a composite of death or myocardial infarction (MI). Effect sizes were synthesized in patients with or without revascularization. A total of 7 observational studies focusing on clopidogrel noncompliance were included in this meta-analysis, whereas no studies involving ticagrelor or prasugrel were retrieved. A significantly increased risk of death or MI 0 to 90 days after clopidogrel noncompliance was found compared with that during 90 to 180 or 90 to 360 days regardless of revascularization (incidence rate ratio [IRR]: 2.01, 95% confidence interval (CI): 1.62-2.49, P < .001, I2 = 9%) or not (IRR: 1.61, 95% CI: 1.05-2.48, P < .001, I2 = 74%). Patients undergoing percutaneous coronary intervention had a higher risk of death or MI 0 to 90 days after clopidogrel noncompliance compared with 90–180 or 90–360 days irrespective of drug-eluting stent or bare metal stent implantation (P < .05 for both). The early ischemic risk after clopidogrel noncompliance is significantly higher than the late risk in ACS patients. Antiplatelet noncompliance remains a serious concern.
Keywords
Introduction
As the cornerstone for the secondary prevention of recurrent ischemic events, the administration of dual antiplatelet therapy (DAPT), including aspirin and P2Y12 receptor inhibitors, up to 12 months after the index event is highly recommended by current guidelines for patients with acute coronary syndrome (ACS) irrespective of revascularization.1,2 However, planned or unplanned noncompliance to P2Y12 inhibitors during the period of DAPT administration seems to be fairly common in long-term practice 3 despite the updated guidelines. P2Y12 inhibitor noncompliance due to various reasons could increase the risk of ischemic events in patients with ACS. 4 A recent study stratified the P2Y12 inhibitor compliance trajectory into several categories (including consistent noncompliance, rapid decline, delayed initiation and gradual decline, and persistent compliance) and found that each noncompliance category increases ischemic events in ACS patients with percutaneous coronary intervention (PCI). 5 To date, P2Y12 inhibitor noncompliance is a lingering concern for improving ACS prognosis.
Moreover, suddenly stopping P2Y12 inhibitors may cause a “rebound effect” and lead to a poor prognosis.6,7 Whether the risk of ischemic events will significantly increase during the early period after P2Y12 inhibitor noncompliance in patients with ACS remains uncertain, although multiple studies have focused on regimens with shortened DAPT duration followed by aspirin monotherapy without combining P2Y12 inhibitors.8–10 To address this issue, we conducted a meta-analysis reporting ischemic risk after P2Y12 inhibitor noncompliance in ACS patients and compared the incidence of death or myocardial infarction (MI) between different periods after noncompliance.
Methods and Materials
Study Screening and Outcome Assessment
This meta-analysis was conducted and reported according to the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) statement. 11 The PubMed and Cochrane databases were comprehensively searched on July 14, 2022, to identify studies reporting data on P2Y12 inhibitor noncompliance and clinical outcomes in ACS patients. The inclusion criteria of studies included the following items: (1) study population compromising ACS patients, (2) studies comparing the incidence of ischemic events during 0 to 90 days versus 90 to 180 or 90 to 360 days after P2Y12 inhibitor noncompliance, and (3) studies reported in English. Applying these inclusion criteria and the patient, intervention, comparison, outcome, and study type (PICOS) principle, we employed the following search terms: acute coronary syndrome, myocardial infarction, ST-elevation myocardial infarction, non-ST-elevation myocardial infarction, unstable angina, clopidogrel, ticagrelor, prasugrel, noncompliance, stopping, cessation, compliance, withdrawal, interruption, disruption, and termination. We did not retrieve terms about study type and clinical outcomes to ensure complete and comprehensive search results (PubMed search strategies are provided in the Supplemental Materials).
Titles and abstracts of records were reviewed. If relevant, the full text of these records was screened. Additionally, references of eligible articles were manually searched and reviewed. Conference abstracts were excluded due to insufficient data and other required details. The primary endpoint of our study was defined as a composite of ischemic events mainly including death and MI.
Data Extraction and Quality Evaluation
The following details of each eligible study were extracted: study type, author identification, year of publication, sample size, patient population, antiplatelet drugs, P2Y12 inhibitor treatment duration before noncompliance, follow-up duration, primary endpoint, and effect sizes. Given that all the studies finally included in the present analysis were observational studies, we used the modified Newcastle–Ottawa Scale (NOS) 12 to evaluate the quality of these studies and judged them as high-quality studies if the NOS score was at least 7 points. Two reviewers (Y. Li and R. Zhao) independently performed all of the processes associated with study selection and data extraction, and discrepancies were resolved by the third reviewer (Q. Zhang).
Data Synthesis and Sensitivity Analysis
Unadjusted incidence rates of a composite of death or recurrent MI per 1000 or 10 000 person-days were calculated for patients stopping P2Y12 inhibitors in each eligible study. Subsequently, adjusted incidence rate ratios (IRRs) and 95% confidence intervals (CIs) produced by Poisson regression were calculated to compare the risk of death or MI during the initial 90 days (0-90 days) after P2Y12 inhibitor noncompliance with the risk in the next 90 (90-180 days) or next 270 days (90-360 days). Then, we synthesized the effect sizes using a fixed-effect model or a random-effect model if significant heterogeneity was observed. Heterogeneity between studies in each synthesis was measured by the I2 method and was significant if I2 > 25%. The synthesis of effect sizes was separately conducted for either revascularized patients or medically managed patients without revascularization during hospitalization. Funnel plots and linear regression tests of funnel plot asymmetry were implemented to evaluate publication bias, and a P value > .10 indicated no publication bias.
Sensitivity analyses were conducted among different P2Y12 inhibitor durations (<6 months or ≥6 months), stent-type drug-eluting stents (DESs), or bare metal stents (BMSs). A level of P < .05 was considered statistically significant. All analyses were performed using R Studio (version 1.2.1335) meta-packages.
Results
A total of 2758 records were initially identified by comprehensive research. After removing reduplicated studies and screening titles and abstracts, 183 studies remained for full-text review and manual search of references. Finally, 7 observational studies were identified as eligible studies in our meta-analysis (selection process in Figure 1). One study was excluded due to the absence of IRR. 13 However, only clopidogrel noncompliance was reported by all 7 studies, whereas IRR after noncompliance to ticagrelor or prasugrel has not been reported to date.
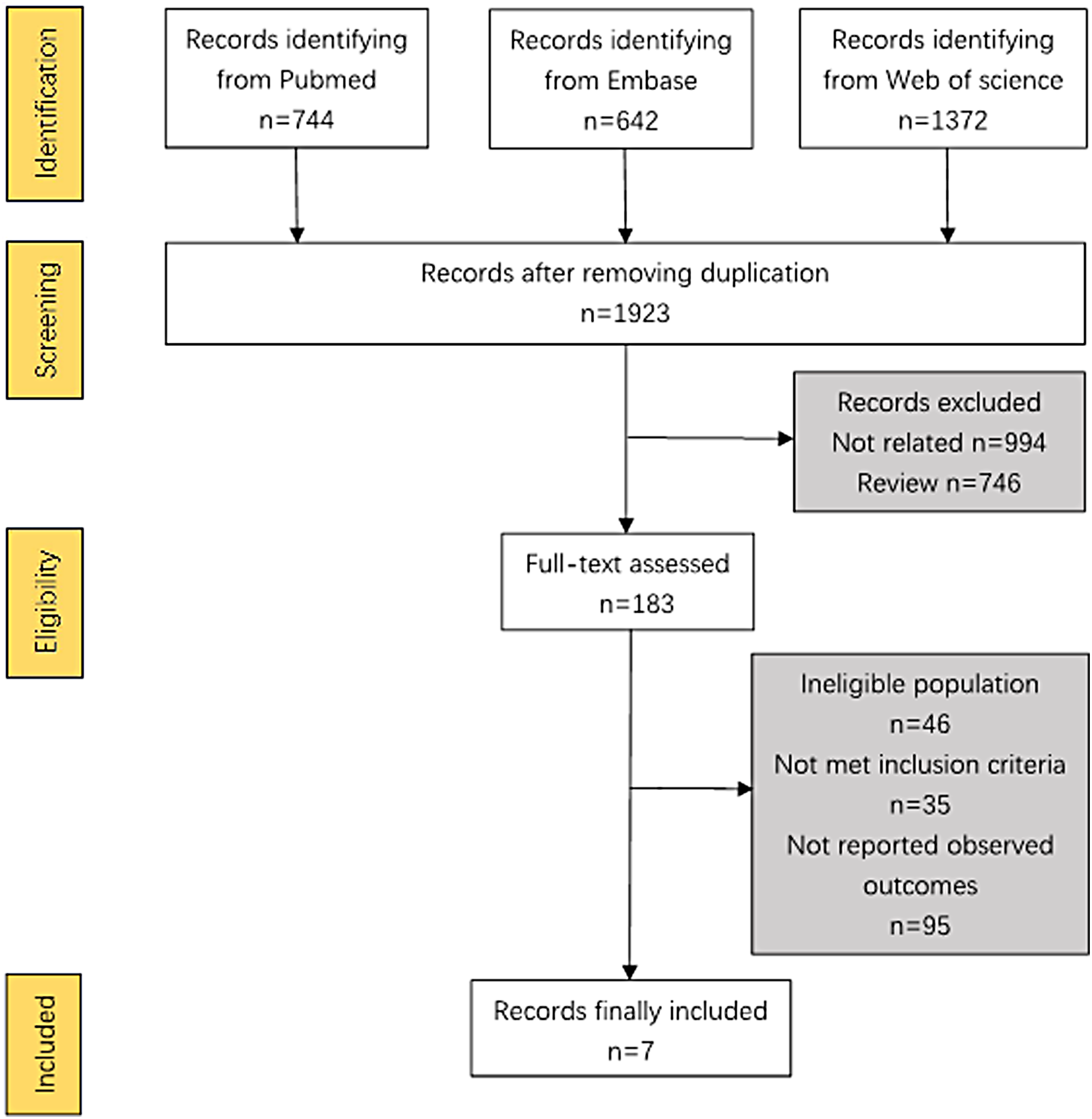
Flow diagram of selection process of eligible records.
Among the 7 included studies with a total of 15 326 patients recruited, 5 studies compared the incidence of a composite of death or MI during 0 to 90 days versus 90 to 180 days after clopidogrel noncompliance,14–18 and 3 studies conducted the comparison during 0 to 90 days versus 90 to 360 days after clopidogrel noncompliance.18–20 The baseline characteristics of these trials are shown in Table 1. All of these trials scored at least 7 points on the NOS and were identified as high-quality studies (Table 2).
Characteristics of Studies Included in the Meta-Analysis.

ACS, acute coronary syndrome; AMI, acute myocardial infarction; MI, myocardial infarction; SVG, saphenous vein graft; PCI, percutaneous coronary intervention; LM, left main; TIA, transient ischemic attack.
NOS Score of Studies Included in Meta-Analysis.
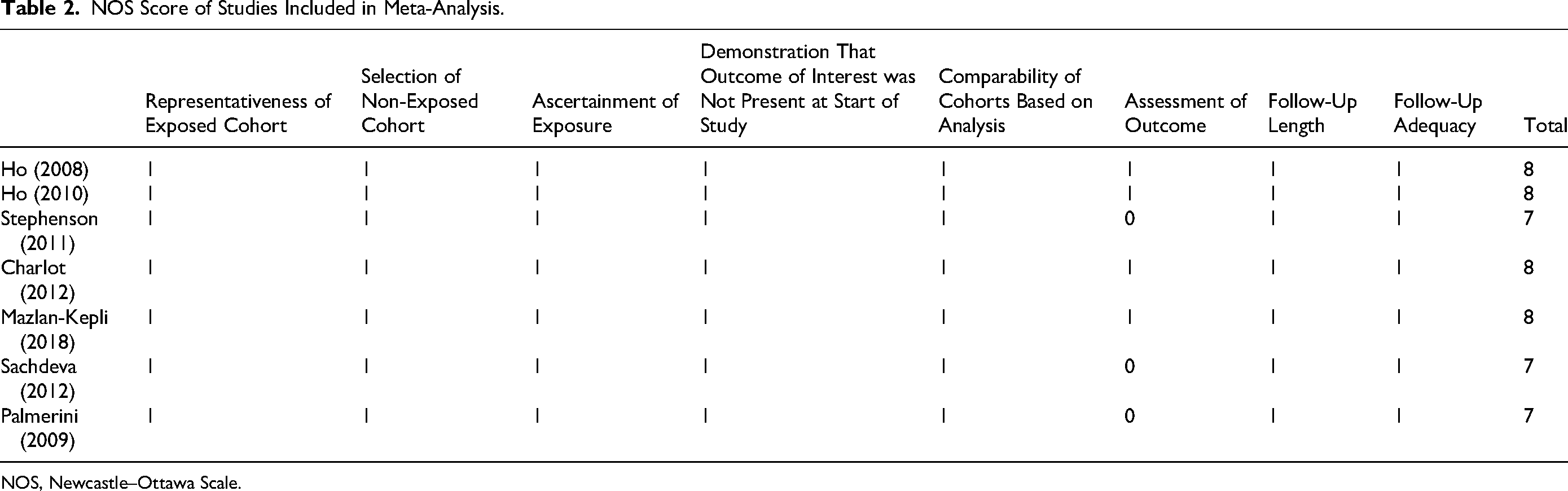
NOS, Newcastle–Ottawa Scale.
Main Outcomes
A total of 5 studies were included in the analysis among medically managed ACS (MMACS) patients.14,15,17–19 After effect size synthesis, a significantly higher incidence of death and MI was found during the period of 0 to 90 days after clopidogrel noncompliance in comparison with 90 to 180 or 90 to 360 days (IRR: 1.61, 95% confidence interval (CI): 1.05-2.48, I2 = 74%, random-effect model, P < .001, Figure 2A). In terms of patients undergoing PCI reported in 6 studies (including saphenous vein graft PCI in one study), the risk of death or MI during 0 to 90 days after clopidogrel noncompliance was also significantly higher than that during 90 to 180 or 90 to 360 days after noncompliance (IRR: 2.01, 95% CI: 1.62-2.49, I2 = 9%, P < .001, fixed-effect model, Figure 2B).
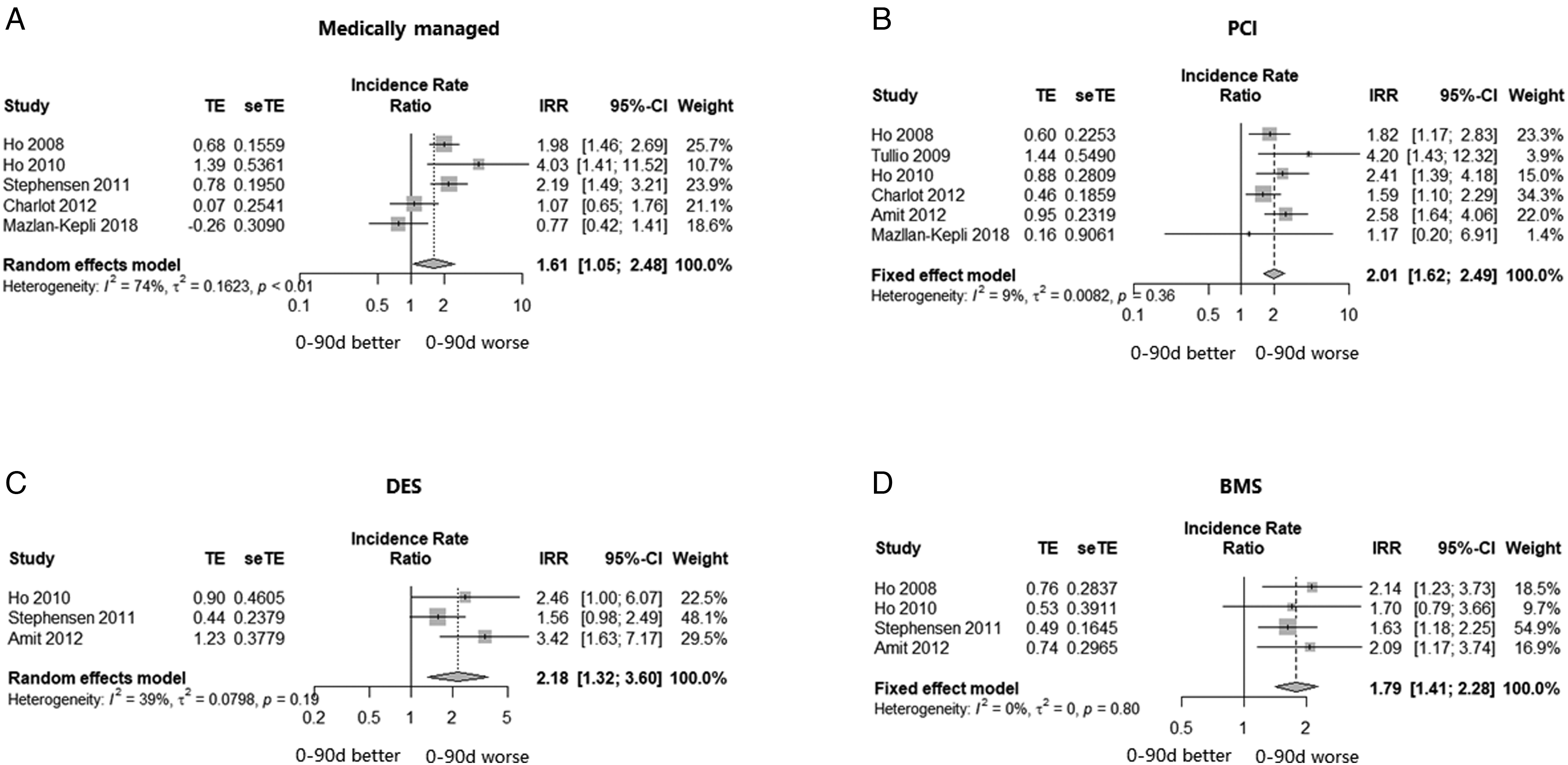
Forest plots of IRRs of primary endpoint during 0 to 90 versus 90 to 180 or 90 to 360 days after clopidogrel noncompliance. IRRs comparing the risk of death or MI during 0 to 90 days after clopidogrel noncompliance versus 90 to 180 or 90 to 360 days in ACS: (A) medically managed patients; (B) patients undergoing PCI; (C) patients with DES implantation; (D) patients with BMS implantation. IRR, incidence rate ratio; PCI, percutaneous coronary intervention; DES, drug-eluting stent; BMS, bare metal stent; MI, myocardial infarction; CI, confidence interval.
Subgroup analyses were conducted according to the stent type (DES or bare metal stent [BMS]) among patients with PCI. After effect size synthesis of 3 studies,17,19,20 patients with DES implantation had a significantly higher rate of death or MI during 0 to 90 days in comparison with that during 90 to 180 or 90 to 360 days after clopidogrel noncompliance (IRR: 2.18, 95% CI: 1.32-3.60, I2 = 39%, random-effect model, P = .0024, Figure 2C). Regarding patients with BMS from 4 studies,14,15,19,20 a significantly higher rate of death or MI during 0 to 90 days after clopidogrel noncompliance was found compared with that during 90 to 180 or 90 to 360 days (IRR: 1.79, 95% CI: 1.41-2.28, I2 = 0%, fixed-effect model, P < .0001, Figure 2D). No publication bias was observed in these results based on funnel plot and linear regression tests of asymmetry (P > .1 for all, Supplemental Figure S1).
Sensitivity Analyses
Sensitivity analyses were performed when patients were stratified by the clopidogrel duration (<6 months or ≥6 months) before noncompliance. We found that the risk of death or MI during 0 to 90 days after clopidogrel noncompliance was significantly higher than that during 90 to 180 days or 90 to 360 days irrespective of <6 months (IRR: 2.51, 95% CI: 1.90-3.33, I2 = 0%, fixed-effect model, P < .0001, Figure 3A) or ≥6 months (IRR: 2.15, 95% CI: 1.20-3.84, I2 = 0%, fixed-effect model, P = .01, Figure 3B). However, funnel plots and linear regression tests of asymmetry showed that these results had potential publication bias (<6-month P value for asymmetry < .10, and ≥6-month P value for asymmetry was not available due to the limited study sample size, Supplemental Figure S2).

Subgroup analyses of primary endpoint during 0 to 90 versus 90 to 180 or 90 to 360 days after clopidogrel noncompliance. Subgroup analyses comparing the risk of death or MI during 0 to 90 days after clopidogrel noncompliance versus 90 to 180 or 90 to 360 days in ACS: (A) <6-month clopidogrel duration before noncompliance; (B) ≥6-month clopidogrel duration before noncompliance; (C) 0 to 90 days versus 90 to 180 days after clopidogrel noncompliance among patients with PCI; (D) 0 to 90 days versus 90 to 360 days after clopidogrel noncompliance among patients with PCI; (E) 0 to 90 days versus 90 to 180 days after clopidogrel noncompliance among medically managed patients; (F) 0 to 90 days versus 90 to 360 days after clopidogrel noncompliance among medically managed patients. IRR, incidence rate ratio; PCI, percutaneous coronary intervention; MM, medically managed; MI, myocardial infarction; CI, confidence interval.
We conducted sensitivity analyses for both 0 to 90 days versus 90 to 180 days and 0 to 90 days versus 90 to 360 days among patients either medically managed or revascularized. Patients with PCI had a higher rate of death or MI during 0 to 90 days than during either 90 to 180 days (IRR: 1.76, 95% CI: 1.35-2.31, I2 = 1%, fixed-effect model, P = .001, Figure 3C) or 90 to 360 days (IRR: 2.38, 95% CI: 1.69-3.35, I2 = 0%, fixed-effect model, P < .0001, Figure 3D) after clopidogrel noncompliance. While different from the outcome, no significant difference was found among medically managed patients comparing the risk of death or MI during 0 to 90 days versus that during either 90 to 180 days or 90 to 360 days after clopidogrel noncompliance (random-effect model, P > .05 for both, Figure 3E and F).
Discussion
This meta-analysis was conducted to evaluate the early ischemic risk after P2Y12 inhibitor noncompliance during secondary prevention among ACS patients. The results showed that the risk of death or MI during 0 to 90 days after clopidogrel noncompliance was significantly greater than that during 90 to 180 or 90 to 360 days irrespective of revascularization. This relatively higher ischemic risk during 0 to 90 days after noncompliance also existed in patients implanted with both DES and BMS.
Although current guidelines strongly recommended 12-month DAPT after index events for second prevention in ACS patients, previous studies reported that planned or unplanned clopidogrel noncompliance for 12 months still occurs in 10%∼20% of patients with PCI.3,21,22 Reasons for noncompliance differ. Some patients have to stop clopidogrel because of adverse events, including bleeding, whereas others may choose to discontinue due to surgery or even individual unwillingness. 23 Moreover, unplanned or premature cessation of antiplatelet treatment due to any reason could increase the risks of ischemic events and poor prognosis.24,25 Our meta-analysis found that the initial 90 days after clopidogrel noncompliance within 12 months of the index event was associated with a higher incidence of all-cause death or MI compared with the next 90 or 270 days in ACS patients undergoing PCI irrespective of whether DES or BMS was implanted. Several studies demonstrated that patients treated with PCI experienced more ischemic events after prematurely stopping DAPT.26,27 However, a previous meta-analysis including 6 randomized controlled trials demonstrated that during 0 to 90 days following clopidogrel discontinuation, there was no significant increase in the risk of ischemic events between patients randomized to 3- or 6-month DAPT compared with at least 12-month DAPT. Notably, the DAPT durations in these clinical trials involved in the above meta-analysis were all prespecified and at physicians’ discretion. Nevertheless, our study focused on clopidogrel noncompliance, indicating that patients stopped taking medications without physicians’ guidance. This large difference in reasons for clopidogrel discontinuation led to a discrepancy between these 2 meta-analyses. To some extent, this discrepancy also suggests the extreme importance of prescription compliance and compliance.
Although guidelines recommend a revascularization strategy including PCI and coronary artery bypass grafting as the first-line treatment for ACS patients,1,2 a proportion of patients still do not undergo revascularization during hospitalization and have a worse prognosis; these patients are referred to as MMACS patients.28,29 In total, 12 months of DAPT after the index event is also recommended for MMACS patients.30,31 We found that MMACS patients still experienced an increased risk of death or MI during the initial 90 days after clopidogrel noncompliance. Further studies should be conducted to focus on this special population and guide clinical practice.
Currently, a series of clinical trials have focused on exploring the efficacy and safety of shortened DAPT duration versus long-term DAPT duration to maximally diminish the increased bleeding risk without discounting anti-ischemic efficacy, demonstrating that shortened DAPT had a better benefit-risk tradeoff.32–34 Of note, the DAPT strategy needs to be individually tailored under comprehensive evaluation, and some novel evaluation methods, such as the PRECISE-DAPT score, could be used to assist in decision-making. 35 Sensitivity analyses from our study found that clopidogrel noncompliance after both <6-month and ≥6-month DAPT was associated with a higher risk of death or MI. In addition to prescription noncompliance rather than prespecified discontinuation, potential publication bias detected by linear asymmetry may also explain these results. Despite a huge demand for exploring the optimal antiplatelet strategy, our meta-analysis implies that eliminating antiplatelet noncompliance is also an important issue for improving ACS patient prognosis.
Our meta-analysis has several limitations. First, the total number of included studies was small. However, the results of the quality assessment of these studies were acceptable, which made our outcomes reliable. Second, due to limited literature, only clopidogrel noncompliance was evaluated, and other P2Y12 inhibitors could not be included. Third, the reliability of some results may be weakened by publication bias.
Conclusion
The findings of our meta-analysis indicated that the early ischemic risk after clopidogrel noncompliance is significantly increased compared with late ischemic risk among ACS patients during the recommended DAPT duration. Improving antiplatelet compliance should be taken seriously.
Supplemental Material
sj-docx-1-cat-10.1177_10760296231196477 - Supplemental material for The Increased Ischemic Risk During the Early Period After Clopidogrel Noncompliance in Patients with Acute Coronary Syndrome: A Meta-Analysis
Supplemental material, sj-docx-1-cat-10.1177_10760296231196477 for The Increased Ischemic Risk During the Early Period After Clopidogrel Noncompliance in Patients with Acute Coronary Syndrome: A Meta-Analysis by Yushan Li, Ruting Zhao, Peng Yu, Yan Xu, Quanyu Zhang and Yaling Han in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Author Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by Yushan Li and Ruting Zhao. The first draft of the manuscript was written by Yushan Li; Peng Yu and Yan Xu commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by the National Natural Science Foundation of China (NSFC: 32071116), and the LIAONING S&T Project (2022JH2/101500028).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
