Abstract
Identifying adherence to direct oral anticoagulants (DOACs) plays a major role in treatment efficacy and safety. The DOAC Dipstick can detect DOACs in urine samples of acutely diseased patients at plasma thresholds of about 30 ng/mL. A prospective observational consecutive cohort study was performed on outpatients taking DOACs. The presence of direct oral factor Xa inhibitors (DXIs) in patient urine samples were independently evaluated by visual interpretation of the DOAC Dipstick pad colors. DOAC plasma concentration was assessed using STA®-Liquid Anti-Xa and STA®-Liquid Anti-IIa chromogenic substrate assays. Positive DOAC Dipstick results were compared with a threshold plasma of DOAC concentration ≥30 ng/mL. Of 120 patients (age 55.4 + 16.1 years, female n = 63), 77 were on rivaroxaban and 43 on apixaban. Plasma concentrations were 129 ± 118 ng/mL for rivaroxaban, and 163 ± 130 ng/mL for apixaban, DOAC Dipstick test has a sensitivity of 97.2% and a positive predictive value of 89.5% at 30 ng/mL. No differences occurred between DXIs. Specificity and negative predictive value could not be determined due to the low number of true negative values. There were no differences in the interpretation of rivaroxaban and apixaban pad colors between observers (Kappa 1.0). Results show that DOAC Dipstick may be a useful tool for identifying DXIs in urine samples in an outpatient setting at a plasma threshold ≥ 30 ng/mL. Further studies should include patients treated with dabigatran, vitamin K antagonists, or other anticoagulants.
Introduction
Direct orally active anticoagulants (DOACs) are the leading therapeutic choice for thrombosis, demonstrating a favorable benefit/risk ratio and convenience. 1 In some emergency medical situations, such as surgery for a hip fracture, DOACs may need to be detected quickly and accurately in order to schedule the most appropriate surgical strategy and thus, decrease the risk of perioperative hemorrhage.2–4 Nevertheless, outline detection of DOACs before urgent surgical interventions is considered both an expensive and unfeasible practice in many laboratories. 5 International guidelines6,7 recommend that DOACs should be determined depending on patients’ risk factors for bleeding such as advanced age, severe renal failure, dialysis, BMI > 40 kg/m2, known drug interactions, and prior elective surgery with high bleeding risk. In emergency situations, the presence and concentration of DOACs may need to be defined if a reversal strategy is required. Antidotes should be administered based on plasma DOAC concentrations. Threshold values of 30 to 50 ng/mL have been set for urgent medical interventions with a high bleeding risk. 6
The in vitro diagnostic DOAC Dipstick is an easy-to-use bedside tool that detects DOACs after they have been excreted into the urine. 8 With this tool, direct oral factor Xa inhibitors (DXIs) and direct thrombin inhibitors (DTIs) are detected on different DOAC Dipstick pads, each containing immobilized reagents that specifically interact with the respective DOAC. The DOAC Dipstick was shown to accurately detect DOACs in urine samples at a plasma threshold concentration of 30 ng/mL in cardiology, neurology, 9 and emergency care settings. 10 Since DOAC Dipstick is an easy-to-use, noninvasive test, it would be suitable for assessing adherence to DOAC treatment in outpatients. The present single-center prospective noninterventional study was conducted in order to assess the accuracy of DOAC Dipstick assessment in outpatients treated with apixaban or rivaroxaban and to evaluate the correlation between DOAC Dipstick measurement and DOAC plasma concentration.
Patients and Methods
This was an observational cohort study. Patients on DOAC treatment were consecutively enrolled at the Thrombosis Center of Tenon University Hospital, Assistance Public Hôpitaux de Paris (AP-HP). Patients were recruited from December 1, 2019, to July 31, 2021.
Inclusion and Exclusion Criteria
Inclusion criteria were (1) the ability to understand the study protocol and follow the instructions, (2) a dated and signed written informed consent, (3) age ≥18 years, and (4) receiving DXIs (apixaban and rivaroxaban) for at least 1 week. Patient adherence to treatment, as well as intake of the drug within the last 24 h from inclusion in the study, was confirmed by thorough interviews during consultation. Exclusion criteria were (1) an inability to provide spontaneous urine samples, (2) an inability to understand informed consent, and (3) compromised cognitive function. The study protocol was approved by the local ethics committee of the participating center according to the Declaration of Helsinki 1964 and patients gave written informed consent prior to participating in the study.
Sample size calculation: The number of patients included in the study (n = 120) provided sufficient statistical power for evaluating the accuracy of the assay DOAC Dipstick.
Data Documentation
Biographic data, indication for DOAC therapy, DOACs taken, inclusion and exclusion criteria, concomitant diagnoses and medications, visual DOAC Dipstick test evaluation, plasma DOAC activity, and renal function were extracted from the patient’s paper medical records and inserted into a digital patient database (ORBIS ®). Two documentation assistants entered data from the database into Excel 2010 according to the four-eye principle.
Blood and Urine Collection
Urine and blood samples were collected between 9 am and 1 pm at the end of the consultation. The maximal interval between urine and blood collection was 30 min. During the consultation interview the timing of the last dose intake of rivaroxaban or apixaban was also registered.
Urine samples were collected in a propylene container (100-mL sterile container with integrated transfer unit for closed urine transfer in the V-urine vacuum system, with lid and safety label, [SARSTEDT®, Ref 75.562.400]). The DOAC Dipstick was used to test the presence of DOACs in the urine sample at the time of collection by the site nurses as described below.
Blood was obtained by atraumatic antecubital venipuncture at baseline and collected in Vacutainer® tubes (5 mL tubes, containing 0.109 mol/L trisodium citrate;1 volume trisodium citrate to 9 volumes blood). They were used to extract platelet-poor plasma (PPP) by double centrifugation at 2000g for 20 min at room temperature and concentrations of rivaroxaban or apixaban were measured within 3 h after blood collection.
DOAC Dipstick Test Procedure
Each test strip was completely immersed in urine for 2 to 3 s. The strip was then incubated at room temperature for 10 min without contact with the table or other surfaces. After incubation, the colors of the factor Xa inhibitor pads were visually compared with those of a reference scale to determine the presence (positive result) or absence (negative result) of the respective DOACs. 11 Personnel interpreting the test results had completed a standardized training program on identifying the test pad colors. Individuals with known red-green blindness cannot perform the test. A digital photo of the test strip was taken and placed next to the reference scale for documentation. After interpretation, the test strips were discarded.
Measurement of DOAC Concentration in Plasma
DOAC plasma concentrations were quantified using STA®-Liquid Anti-Xa and STA®-Liquid Anti-IIa chromogenic substrate assays according to the manufacturer's instructions (Diagnostica Stago, Asnieres, France).
Threshold Concentration of DOAC in Plasma
We evaluated the accuracy of the DOAC Dipstick results at a plasma threshold concentration of > 30 ng/mL; this concentration has been proposed for safe hemostasis with low bleeding risk for programmed surgery.7,11,12 At plasma levels of DOAC ≥ 30 ng/mL, a positive and negative test was considered as true positive and false negative, respectively. At plasma levels of < 30 ng/mL, a negative DOAC Dipstick test was considered a correct negative, and a positive DOAC Dipstick result was considered a false positive.
Determination of Renal Function
Renal function was assessed using the Cockroft–Gault equation.
Primary Outcome
The primary outcome was the rate of true positive and true negative DOAC Dipstick results when determining the presence of DXIs in urine samples at a plasma DOAC threshold of ≥ 30 ng/mL.
Secondary Outcomes
Secondary outcomes were differences in true positive and true negative DOAC Dipstick results between patients taking rivaroxaban and apixaban at a plasma threshold of ≥ 30 ng/mL and the interrater agreement between observers.
Statistical Analysis
Statistical analyses were performed using SAS software, version 9.4 (Cary, North Carolina, USA). Qualitative data are described as absolute and relative frequencies and quantitative variables are presented as mean values and standard deviations or as median values with minimum and maximum. Qualitative DOAC Dipstick results were compared with quantitative DOAC plasma concentrations following dichotomization at a threshold of ≥ 30 ng/mL. True positive, true negative, false positive, and false negative values are presented for the DXIs rivaroxaban and apixaban. Sensitivity was calculated with 95% confidence intervals (CI) using the SAS procedure PROC FREQ. 13 Sensitivity of the DOAC Dipstick to detect DOAC in urine samples was defined as the proportion of true positive results in relation to the total number of patients taking DOAC at a threshold of ≥ 30 ng/mL and the positive predictive value (PPV) of true positive results at a threshold of ≥ 30 ng/mL in relation to the total number of DOAC Dipstick results at this threshold. Specificity and negative predictive values (NPV) were not determined because of limited data. Differences between the rivaroxaban and apixaban groups were analyzed using the Chi-square test or Fisher's exact test as appropriate. Quantitative values were compared using a two-sample t-test (in the case of normally distributed data) or a Mann-Whitney U-test. Kappa coefficients were calculated to assess interobserver variability. 14 Results were considered significant at a P-value of < 0.05.
Results
Between September 2019 and July 2021, 120 participants were included, whose data were analyzed: 77 patients were on rivaroxaban and 43 on apixaban treatment. Patient demographic data, treatment indications, DXI plasma concentration (mean ± SD), and creatinine clearance, are presented in Table 1. All patients had normal renal function with a mean creatinine clearance of 94 ± 18 mL/min/1.73 m2. Only 2 patients were taking dabigatran and these participants were not included in the analysis. There were no significant differences in demographic and laboratory data between the subgroups of patients treated with rivaroxaban and apixaban (Table 1). Patients with deep vein thrombosis or recurrent thromboembolism were treated more frequently with rivaroxaban and patients with pulmonary embolism or cancer-associated thrombosis were treated more frequently with apixaban (P < 0.015, Table 1). The frequencies of the other indications were too low to describe differences between rivaroxaban and apixaban.
Demographic and Biological Data of Patients in the DXI Group and Rivaroxaban and Apixaban Subgroups.

Abbreviations: DXIs, direct oral factor Xa inhibitors; DOAC, identifying adherence to direct oral anticoagulants.
Primary Outcome
In the DXI group, true positive DOAC Dipstick results were obtained in 102/105 patients and false negative results in 3/105 patients. True negative DOAC Dipstick results were seen in 3/15 patients and false positive results in 12/15 patients (Table 2A). The sensitivity was 97.1% (95% CI, 91.9-99.4) and the PPV was 89.5% (95% CI, 82.3-94.4). The specificity and negative predictive values cannot be reliably calculated reliably because of the low number of patients in our study with correct negative results of the factor Xa inhibitor pad and are therefore not reported.
Frequency of Positive and Negative DOAC Dipstick Results at a Plasma Threshold of ≥ 30 ng/mL in Patients Treated with DXI (2A), Rivaroxaban (2B), and Rivaroxaban (2C).
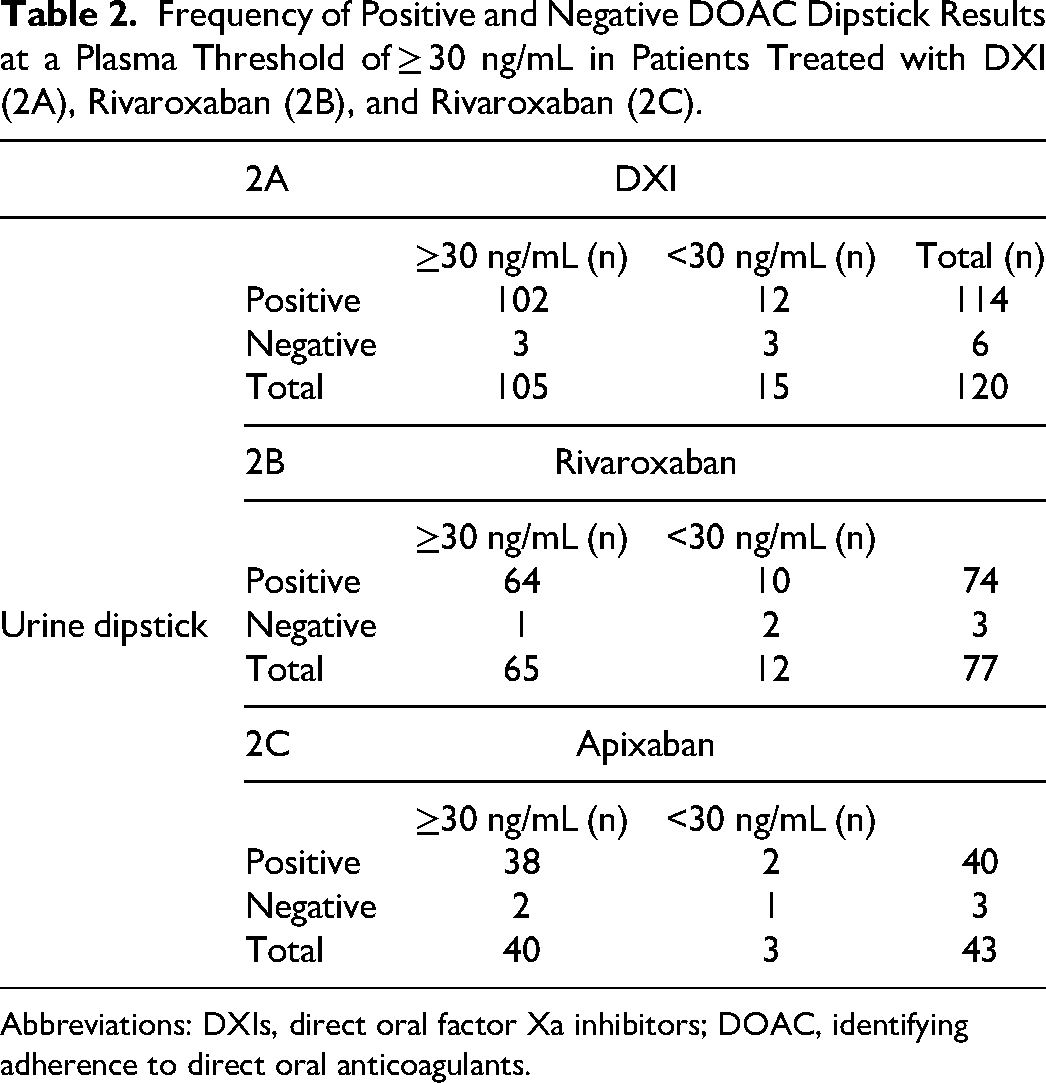
Abbreviations: DXIs, direct oral factor Xa inhibitors; DOAC, identifying adherence to direct oral anticoagulants.
Subanalysis
In the rivaroxaban group, true positive DOAC Dipstick results were obtained in 64/65 patients and false negative results in 1/65 patients. True negative DOAC Dipstick results were seen in 2/12 patients and false positive results in 10/12 patients (Table 2B). The sensitivity was 98.5% (95% CI, 91.7-100) and the PPV was 86.4% (95% CI, 83.2-89.2).
In the apixaban group, true positive DOAC Dipstick results were obtained in 38/40 patients and false positive results in 2/3 patients.
Interobserver Agreement
There was complete agreement in test strip interpretation between the 2 observers at a threshold of 30 ng/mL and there were no differences in true positive and negative and false positive and negative results between the observers (kappa value = 1).
Discussion
The present study shows that the DOAC Dipstick test can detect with accuracy the presence of DXIs in urine samples with high sensitivity at a plasma threshold of ≥ 30 ng/mL in outpatients treated with rivaroxaban or apixaban mainly for deep vein thrombosis, recurrent deep vein thrombosis, and pulmonary embolism. Other indications are infrequent due to the specialization of our outpatient care unit.
We demonstrate for the first time that the DOAC Dipstick methodology is a valuable, sensitive, and accurate tool for the patient-friendly evaluation of adherence to treatment with DOACs, that can be applied by general practitioners at the level of primary healthcare. In agreement with our findings, Margetić et al 9 reported a sensitivity of more than 95% for the DOAC Dipstick test at the same plasma threshold in patients treated with rivaroxaban, apixaban, and dabigatran in cardiology and neurology care units. Merrelaar et al 10 confirmed these results and demonstrated similar specificity for edoxaban in patients admitted to an emergency care department. Örd et al 15 also demonstrated the high sensitivity of the DOAC Dipstick at this plasma threshold in a smaller group of patients treated with rivaroxaban and apixaban. A metaanalysis strengthens the confidence in the adoption and the performances of the DOAC dipstick by comparing the prototype test strip results with its commercial version. 16 The present study increases the body of evidence that the DOAC Dipstick test can accurately detect DOACs in urine samples of patients at a plasma threshold concentration of ≥ 30 ng/mL in patient care units where the rapid and sensitive determination of DOACs may accelerate medical decision-making processes.
The detection of vitamin K antagonists (VKA) via point-of-care testing is well established, 17 but the detection of DOACs before urgent medical interventions is still under debate. 18 DOACs may need to be determined if the patient switches from VKA to DOAC therapy or if the patient has not adhered to their VKA therapy. 19 Patient adherence to VKA therapy and switching from VKA to DOAC treatment depends on a number of variables. For example, patient satisfaction scores were higher after switching to DOAC treatment. 20 The occurrence of false negative results in a patient adherent to therapy can be minimized by including clinical information such as a diary to monitor DOAC intake. Impairment of renal function is measured with the DOAC Dipstick creatinine to exclude false-negative DOAC test results.
Finding that DOACs can quickly and easily be detected in urine may improve patient satisfaction and outpatient care. Medication adherence is defined by the World Health Organization as “the degree to which the person's behavior corresponds with the agreed recommendations from a health care provider.” 21 In a study investigating reasons for nonadherence to DOACs, one important improvement strategy proposed by interviewed patients was to perform “a blood test to see that apixaban really works.’’ 22 The DOAC Dipstick test could be used as an easy-to-use, noninvasive rapid alternative to enhance patients’ adherence. Patients or carriers can assess by themselves strip's colors and have visual proof of DOAC’s presence or absence. This can be very useful for patients that forget to take their medication, such as elderly or poly-medicated patients. In the rare event of false-negative results, the test can be easily repeated in order to minimize errors such as not the adequate time from strip dip to lecture, wrong color interpretation, hematuria, etc. The use of an automatic reader is a step toward this direction. Clear instructions should be given that in case of discordance, patients should seek healthcare advice.
In patients with plasma concentrations below the threshold of 30 ng/mL, the DOAC Dipstick may still be positive, but this positivity does not correspond to a clinically relevant plasma concentration of a DOAC. The assessment of adherence should be based on a clinical evaluation: confirming the hour of last drug intake, the dose, and the absence of potential drug-drug interactions. DOACs are approved for patients with mild to moderate CKD.23,24 In case of a combination of a negative DOAC test result of the factor Xa and thrombin inhibitor pad and a test result “low” of creatinine pad, the dipstick result for DOAC is invalid as described in the instructions for use. If indicated, a plasma assay needs to be performed.
To date there are no clearly established therapeutic ranges for DOACs, therefore dose changes are based on clinical aspects, such as bleeding risk or hemorrhagic complications, recurrent thromboembolic disease, or patient request while on long-term treatment. 25 Certainly, more data are needed to define the need for dose reduction or dose escalation in specific cases such as extreme weight and suspicion of drug–drug interactions. In an outpatient setting, the DOAC dipstick test is a qualitative and easy test that can be used to confirm the chosen anticoagulation strategy and to alert treatment failure in the presence of recurrent events.
The present study has some limitations. The variability of the concentrations of rivaroxaban or apixaban in patients’ plasma is high. Theoretically, this could potentially affect the validity of the false negative results. Nevertheless, the data presented herein calculated the concentrations of rivaroxaban and apixaban independently of the dose and the interval between the intake of the last dose and blood collection. Nevertheless, the interindividual variability of the concentrations of rivaroxaban or apixaban in plasma is high even in homogenous groups regarding dose and interval between the last dose intake and blood collection. Moreover, it is well established that the clinical relevance of DXI concentration in plasma is limited. Thus, we consider that this variability does not influence the outcome of our study which is the evaluation of the capacity of DOAC Dipstick to get a positive or negative signal at the threshold of 30 ng/mL plasma. Plasma and urine samples were not collected before DOAC intake, which reduced the probability in our study to obtain plasma concentrations < 30 ng/mL. This reduced the possibility of negative DOAC Dipstick test results affecting the specificity and NPV values. In addition, specificity and negative predictive values cannot be calculated accurately because of the low number of patients in our study with correct negative results of the factor Xa inhibitor pad and are therefore not reported, in contrast to other studies.9–11 Increasing plasma threshold concentration from 30 to 50 ng/mL will result in more negative findings of DOAC Dipstick of patients under treatment DOACs. This means a somewhat lower sensitivity and a higher specificity at higher threshold values. It may be expected that the PPV will increase by increasing the threshold concentration to 50 ng/mL. However, these calculations will be associated with a high level of uncertainty as the number of patients in our study is rather small, and sensitivity, specificity, and predictive values are not performed at a higher threshold plasma concentration.
In conclusion, our study shows that the DOAC Dipstick detects rivaroxaban and apixaban with high sensitivity at a plasma threshold of ≥ 30 ng/mL in outpatients. Drug evaluation in urine is a reliable and clinically relevant methodology that allows a realistic, and patient-friendly monitoring of drug intake and patients’ adherence to the antithrombotic treatment. In addition, the DOAC Dipstick is a universal tool for the evaluation of patients on treatment with specific factor Xa inhibitors, that is, apixaban, edoxaban, and rivaroxaban. The introduction of DOAC Dipstick at the level of the primary health care system could improve patients’ adherence to the antithrombotic treatment and will allow general practitioners to improve the quality of healthcare in patients on long-term antithrombotic treatment with rivaroxaban or apixaban, particularly in those at high risk of recurrent thrombosis. The DOAC Dipstick realization and interpretation is easy and user-friendly; these conditions may allow self-assessment of the patients or by the family milieu in case of doubts on the drug intake. Moreover, the binary interpretation of the test (presence or absence of the DXI) allows easy introduction of the provided information in the digital personal medical records of the patients on long-term antithrombotic treatment.
Highlights
DOAC Dipstick accurately detects apixaban, rivaroxaban, and dabigatran in urine samples at a plasma threshold concentration of ≥ 30 ng/mL in hospitalized and emergency care patients.
The study shows that DOAC Dipstick detects rivaroxaban and apixaban with high sensitivity at a plasma threshold of ≥ 30 ng/mL in outpatients.
This point-of-care test is appropriate to detect adherence to DOAC therapy in outpatients.
Footnotes
Authors’ Contributions
The role of each investigator can be specified as follows: LP: study design, investigation, methodology, data analysis, writing original draft preparation. SH: data analysis, statistical evaluation, writing. JF: conceptualization, interpretation, reviewing. SA: conceptualization, methodology, writing. LT: investigation, methodology, writing. JH: study design, data analysis, writing, editing. CW: statistical evaluation, writing, editing. IE: conceptualization, interpretation, reviewing. GG: study design, methodology, editing, supervision.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Correction (August 2023):
The phrase “false negative” has been corrected to “false positive” in three instances and patient number corrected to “2 / 3” in the Subanalysis section on page 4.
