Abstract
Heparin-induced thrombocytopenia (HIT) is a disorder originating from exposure to heparin and has a spectrum of presentation ranging from asymptomatic positive antibodies to thrombotic complications. When symptomatic, it is associated with morbidity and mortality. The incidence of HIT in the ESRD population is yet to be defined. End-stage renal disease (ESRD) patients are at particular risk due to constant exposure to heparin. The main treatment of HIT is to avoid heparin and pursue alternative anticoagulants. Since 1 of the main advantages of heparin in ESRD patients is the ease of its use due to non-renal clearance, the use of alternative anticoagulants poses yet another challenge for this population due to cost, availability, and adverse effects on ESRD patients. Argatroban seems like the best alternative to heparin in hemodialysis (HD) patients due to its liver clearance. Despite having limited studies in HIT, direct oral anticoagulants (DOACs) were added as a potential treatment for HIT, with apixaban favored in kidney dysfunction as it is the least dependent on kidney clearance. Other treatment modalities exist but are still being studied in ESRD patients. The presence of HIT antibodies is not always associated with clinical syndrome, and some studies suggested that heparin antibodies are transient, and the reintroduction of heparin is still being evaluated as a treatment option. Hence, HIT is a challenging diagnosis in ESRD patients, a population that has frequent exposure to anticoagulants, and a risk/benefit ratio should be weighed between the risk of progression to symptomatic HIT and the benefit of switching to a non-heparin anticoagulant bearing in mind the difficulties associated with the latter.
Keywords
Introduction
The widespread use of heparin products as a mean of anticoagulation has proven to be of high relevance in the clinical setting. An adverse event that can ensue after using heparin is the development of a serious condition called heparin-induced thrombocytopenia (HIT). 1 End-stage renal disease (ESRD) patients, especially those on dialysis, are considered a special population in terms of frequent exposure to heparin products. 2
HIT can be classified into 2 types: type I HIT, which is caused by non-immune-mediated platelet aggregation that leads to mild and transient thrombocytopenia, 3 and type II HIT, which is characterized by immune-mediated platelet activation and consumption. Heparin binds to a protein called platelet factor 4 (PF4) with high affinity, exposing neoepitopes on PF4. These neoepitopes can act as immunogens, triggering the production of heparin–PF4 antibodies. When these antibodies bind to the heparin–PF4 complex, they activate platelets, leading to the release of clotting factors and ultimately resulting in widespread platelet consumption, hypercoagulability, and thrombosis.4,5
Methodology
A comprehensive search of the PubMed, Embase, and Cochrane Library databases was performed from inception to December 2022 to identify relevant articles. A combination of keywords and medical subject headings (MeSH) terms, including “heparin-induced thrombocytopenia,” “end-stage renal disease,” “chronic kidney disease,”“heparin,” “anticoagulation,” “platelets,” “thrombocytopenia,” and “renal replacement therapy”, was used to generate the search strategy. In addition, the reference lists of included studies and relevant review articles were screened for potentially relevant studies.
Inclusion and Exclusion Criteria
Studies were included in this review if they met the following criteria: (1) conducted in patients with ESRD, (2) focused on HIT, (3) included information on anticoagulation therapy, (4) reported outcomes related to thrombocytopenia or bleeding events, and (5) published in English language. Studies that were not conducted on human subjects or were case reports, letters, or editorials were excluded.
Data Extraction and Analysis
Two reviewers independently screened the titles and abstracts of all identified studies to determine eligibility for inclusion. The full-text articles of potentially eligible studies were then obtained and reviewed in detail by the same reviewers. Any discrepancies between the 2 reviewers were resolved through discussion and consensus. Data were extracted from the included studies by 1 reviewer and independently verified by a second reviewer. The extracted data included study design, patient characteristics, interventions, outcomes, and adverse events. Data were summarized qualitatively due to the heterogeneity of the included studies.
Prevalence
The prevalence of HIT in ESRD patients is not well established, as there is limited data available specifically on this population. However, some studies have suggested that ESRD patients may be at an increased risk for HIT due to their underlying medical conditions and exposure to heparin during hemodialysis (HD) treatment.
One study by Kaur et al 6 reported a HIT prevalence rate of 4.6% in ESRD patients who received ultra-fractionated heparin (UFH) during hospitalization for various medical conditions. 6
Overall, the exact prevalence of HIT in ESRD patients remains unclear and may vary depending on factors such as the type of heparin used, patient comorbidities, and other individual risk factors. It is important for healthcare providers to be aware of the potential increased risk of HIT in ESRD patients and to monitor for signs and symptoms of this condition during treatment with heparin. 6
Pathogenesis
The specific mechanism of HIT in ESRD patients undergoing HD is not fully understood. HIT is a type of immune-mediated drug reaction, which occurs when heparin binds to PF4, forming a complex that can trigger the production of antibodies that activate platelets and cause thrombocytopenia. 7 The presence of ESRD and the exposure to heparin during HD may contribute to an increased risk for developing HIT.
Timing of HIT in ESRD/HD Patients
HIT can occur at any time during heparin exposure, including at the beginning of HD or after a long period of HD. However, some studies have suggested that the risk of developing HIT may be higher during the first few weeks of heparin exposure, particularly in patients who have not been previously exposed to heparin. 8
Risk of HIT with Heparin- or Heparin-Like Coated HD Membranes
The use of heparin or heparin-like coated HD membranes may increase the risk of developing HIT, as these membranes can release small amounts of heparin during use. However, the overall risk of developing HIT with these membranes is thought to be relatively low. Some studies have suggested that the use of non-heparin anticoagulants, such as citrate, may be a safer alternative in patients at high risk for HIT. 9
Etiologies
Low-molecular-weight heparin (LMWH) and UFH are the 2 agents most commonly used in the ESRD population. UFH is essential in the setup of dialysis as the lines and membranes are innately procoagulants and activate both pathways of the coagulation cascade—intrinsic and extrinsic. 10 The preferential use of UFH over LMWH in this population is owed to its relative lower cost, ease of use, rapid half-life, and lower risk of bleeding. 11 Newer studies comparing both in ESRD patients showed similar bleeding safety profile.12,13 Development of clinical HIT in the general population varies by the dose and type of heparin used. 14 Patients receiving LMWH have lower incidence of HIT than those receiving UFH when used prophylactically.14,15 This difference disappears when used at a therapeutic dose. 14 To date, there is not enough study power to draw any conclusions in terms of the use of both these agents in ESRD and the risk of clinical type II HIT. 12
Temporality
The study conducted by Maharaj et al in 2018 revealed a significant temporal pattern of heparin-induced antibodies (HIA) in outpatients undergoing HD using unfractionated heparin. Although positive HIA was not found to be significantly associated with the risk of thrombocytopenia or thrombosis, it is worth noting that subsequent thrombotic events occurred more frequently in the strong positive HIA group with an optical density (OD) > 1.000. Therefore, while the statistical significance was not achieved, the findings suggest the need for further research to explore the relationship between HIA and the risk of thrombosis in this population. 16
ESRD patients undergoing dialysis are considered to be at an increased risk for HIT due to continuous exposure to heparin, with up to 12% developing HIT. While HIT typically occurs in the early dialysis sessions when heparin is first administered, some patients who have antibodies to the PF4/heparin complex may be at risk for delayed-onset HIT. The clinical features of HIT in HD patients include acute thrombocytopenia, clotting of the extracorporeal circuit during heparin administration, resolution of clots with the use of an alternative anticoagulant, and, in rare cases, thrombotic occlusion of vascular access. 17
Morbidity
HIT can result in various adverse effects such as bleeding, thrombocytopenia, thrombosis, increased healthcare costs, prolonged hospitalization, and others. 18 Previous research has suggested that HIA in dialysis patients with ESRD may increase the risk of clinical thrombosis. 19 - 21 However, recent studies have shown that there is no significant association between HIA results and thrombotic events, vascular access thrombosis, bleeding, or thrombocytopenia.16,22- 27
Mortality
A review article was conducted to investigate the mortality rate of HIT in patients with ESRD, which synthesized the findings of previous studies. 15 The results showed that a positive test for HIA was linked with higher mortality rates and that mortality rates were further elevated in patients with higher titers of HIA. 28 - 30 However, it is worth noting that the study did not employ a functional assay in the diagnosis of HIT, which may lead to an underestimation of the population under study. These findings emphasize the need for further research to better understand the impact of HIT on the mortality rate of ESRD patients. 27
When to Suspect HIT
The diagnosis of HIT is guided by a strong clinical suspicion whenever a patient develops thrombocytopenia or manifests clinical evidence of thrombosis. HIT typically manifests within 5 to 14 days after the initiation of heparin. 31 The likelihood of HIT is usually estimated by using the 4T score that calculates the clinical probability of HIT. The diagnosis of HIT requires confirmation by demonstrating the presence of HIA and/or positivity of the functional assay. 32
Pre-Test Probability
Clinically, the 4T score is mostly used by clinicians to assess the pre-test probability of HIT diagnosis. 33 This test incorporates 4 factors: thrombocytopenia, time of onset of symptoms, presence of thrombosis, and the probability of other causes of thrombocytopenia. A total score of 4 and above should prompt for the diagnosis of HIT. Nevertheless, all these factors are affected in the ESRD population and may result in overdiagnosis of HIT. 17
A study done by Kelly et al found that higher PF4 ODs and high-suspicion 4T probability scores did not necessarily correlate with a serotonin-releasing assay (SRA)-positive HIT diagnosis. 34
New-onset thrombocytopenia in ESRD patients is difficult to evaluate owing to a plethora of possible etiologies. 35 ESRD patients on dialysis are at an increased risk for platelet consumption, platelet destruction from interaction with dialysis catheters and dialyzers, decreased platelet production and function, uremia, immunosuppression, and other comorbidities such as diabetes.17,36 Patients with ESRD on HD as renal replacement therapy are a subset of patients that suffer from both platelet dysfunction and impaired platelet–vessel wall interaction which further increases the risk of thrombosis or hemorrhage.37,38 Identifying HIT in this population is also challenging due to multiple causes that may usually result in thrombocytopenia such as dialyzer-induced thrombocytopenia, 39 - 41 pseudo-thrombocytopenia, 42 and infection-induced thrombocytopenia. 43
The HIT expert probability (HEP) score is a validated scoring system used to assess the pre-test probability of HIT. The score includes 4 clinical criteria and 1 laboratory test. While the HEP score was developed and validated in non-dialysis patients, it has been evaluated in the HD population.44,45
Several studies have compared the performance of the HEP score and the 4T score in HD patients. One study found that the HEP score had a higher negative predictive value (NPV) for HIT than the 4T score in this population, suggesting that the HEP score may be a more reliable screening tool in HD patients. However, other studies have reported that the HEP score has limitations in the HD population, such as overestimation of the risk of HIT and poor predictive accuracy. 46
Overall, while the HEP score has been evaluated in the HD population and may have some advantages over the 4T score, it still has some limitations, and further research is needed to determine its optimal use in this population. 47
Laboratory Diagnosis
The enzyme immunoassay (EIA) for detecting PF4/heparin is currently the most used laboratory marker for diagnosis of HIT in the hospital setting. This readily available test has shown to have a NPV of 98% making it valuable to rule out HIT in the inpatient population if the results are negative. 15 However, due to its variable specificity, a positive result does not rule in the diagnosis. 48
Most institutions use an OD threshold of >0.4 to diagnose HIT with EIA. However, it is worth noting that different institutions use different methods available with different sensitivities: the OD and OD thresholds may vary form 1 study/HIA assay to another.
The use of a certain cutoff for HIA has led to a significant number of false-positive HIT diagnoses. Studies have shown that higher OD values (>1.0) are associated with a more accurate diagnosis of HIT. 49 However, there is a lack of studies examining different OD thresholds for IgG PF4 antibodies in ESRD patients. 50
The presence of HIA does not reflect on the function of platelet and the possibility of HIT clinical manifestations. The results of 1 study done by Kelly et al showed equal rates of thrombocytopenia present in both HIA positive and negative groups in a subset of ESRD patients highlighting the fact that thrombocytopenia is a common comorbidity in ESRD patients. 34 In 1 study by Kelly et al, the presence of HIA did not correlate with the incidence of thrombocytopenia in a subset of ESRD patients. This study suggested that EIA is not a reliable diagnostic tool for HIT in ESRD patients. Another study examined the reliability of EIA in HD patients over a 2-year period and found it to be unreliable. Additionally, this study found that both the EIA and 4T pre-test score did not correlate with the rate of HIT diagnosis.
Functional Assays
Functional assays are commonly used to detect platelet activating HIA which results in the clinical syndrome of HIT. Most algorithms recommend performing this test when HIA testing is negative, but clinical suspicion for HIT is strong. The SRA is a highly specific functional assay for HIT diagnosis, but it is not practical for routine diagnosis in the clinical setting. 51 Despite its high sensitivity and specificity, the relative sensitivity/specificity and accuracy of the SRA have not been studied in ESRD. There have been reports of SRA-negative HIT, where clinical suspicion supersedes gold standard lab testing. 52 - 54
Another gold standard test for HIT diagnosis is the heparin-induced platelet activation (HIPA) assay, 55 which is faster and less cumbersome than the SRA and does not require radioactivity. 56 However, false-positive results can occur if not performed with strong clinical suspicion. 57 These functional assays have not been studied specifically in the ESRD population. To our knowledge, previous studies have not examined their use in ESRD patients.
Heparin Rechallenge
When a dialysis patient is affected by HIT, the main considerations are whether to use a heparin-free dialysis, which can be burdensome, or non-heparin anticoagulants, which are more practical. 17 However, alternative treatments may have drawbacks related to cost, availability, and safety profile.
In some cases, heparin rechallenge has been attempted successfully in ESRD patients on dialysis once serological and functional assays are negative, but controlled studies are lacking, and rechallenges should only be considered in exceptional circumstances with careful monitoring.15,58,59
Studies have shown that the level of HIT antibodies starts to decrease after 3 months of continuous use of heparin products, but there is a risk of earlier onset of clinical HIT upon re-exposure to heparin products, especially in patients with strong platelet activating profiles.60,61
It remains unclear whether both functional and serological assays are necessary to safely reinstitute heparin and requires further investigation. 59
Treatment
The management of HIT is centered around 2 main objectives: stopping platelet activation as quickly as possible and providing therapeutic anticoagulation with non-heparin products to address the initial risk of thrombosis. 62
It is crucial to immediately discontinue all heparin products to prevent further platelet activation and stop the formation of the heparin–PF4 complex. 63
Guidelines published in the chest recommend the use of argatroban or danaparoid over other non-heparin anticoagulants, and in patients with a history of HIT requiring ongoing renal replacement therapy or catheter locking, they suggest the use of regional citrate over heparin or LMWH. 64
It is important to note that there have been no head-to-head studies comparing the efficacy and safety of non-heparin anticoagulants during renal replacement therapy in patients with HIT.
Argatroban
Argatroban is the only medication approved by the Food and Drug Administration (FDA) for the treatment of HIT. This synthetic molecule inhibits thrombin directly, both soluble and clot-bound. Unlike other molecules, it does not form antibodies and is mainly cleared hepatically. Its anticoagulant effect is directly related to the dose, plasma concentration, and pharmacodynamic effect. 65 Argatroban is safe to use in patients with chronic kidney disease and ESRD on HD because it is hepatically cleared. 65
It takes 1 to 3 h for steady state, and drug levels increase relative to the dose of 40 μg/kg/min. A washout period of 39 to 51 min is observed after the infusion is stopped. During therapeutic use of argatroban, activated partial thromboplastin time (aPTT) is monitored every 3 h. Many institutions have implemented a nomogram to help achieve a faster steady state, with an initial dose of 2 μg/kg/min. Argatroban is metabolized by hepatic CYP3A4/4 oxidases, and its majority metabolites are excreted via the feces. 65
Renal insufficiency or failure does not affect the pharmacokinetic or pharmacodynamic parameters of argatroban, as it is generally well-tolerated in these individuals. 66
Argatroban produces safe, adequate anticoagulation in HD patients, and its clearance by high-flux membranes is clinically insignificant, necessitating no dose adjustment, according to a retrospective study by Guzzi et al. A prospective study done on 13 patients with different medication dosing over 38 dialysis sessions also demonstrated that argatroban is effective in providing anticoagulation. 67
Bivalirudin
Bivalirudin is a type of direct thrombin inhibitor that is derived from a semisynthetic 20-amino acid derivative of hirudin. Its half-life is approximately 25 min, and it can be used in patients with kidney dysfunction if the appropriate dose reduction is applied. Although bivalirudin is not approved for HIT treatment, it can be used in patients undergoing percutaneous interventions.
A retrospective analysis conducted on 135 patients who were treated with bivalirudin for HIT showed that patients with varying degrees of renal dysfunction required lower doses of bivalirudin to achieve the targeted anticoagulation goals compared to patients with normal kidney function. Patients on dialysis also required dose reductions compared to those with normal renal function but higher dosing requirements than patients not receiving dialysis with a creatinine clearance rate (CrCl) of less than 30 mL/min. 68
Lepirudin
Lepirudin is a direct thrombin inhibitor that binds irreversibly to both free and clot-bound thrombin. It was the first drug in its class to be used clinically. Because it is cleared by the kidneys, its half-life can be up to 316 h in patients on HD. It has been successfully used for thromboprophylaxis in patients with ESRD requiring HD. 69
The British Task Force on Thrombosis and Hemostasis recommends targeting an aPTT level 1.5 to 2.5 times the baseline value in patients diagnosed with HIT to minimize the development of clinical HIT syndrome and mortality. 70
The American College of Chest Physicians’ guidelines suggest that an initial bolus should not be given unless there is suspicion of threatening thrombosis, and the initial infusion rate should depend on serum creatinine levels. 71
However, lepirudin has been associated with a higher risk of bleeding in HIT patients, possibly due to its immunogenicity, as nearly half of the patients developed antibodies against it within 5 to 10 days of exposure, according to Greinacher et al.15,72
Danaparoid
Danaparoid is a medication composed of heparin sulfate, dermatan sulfate, and chondroitin sulfate. It has a half-life of 7 h and is unique compared to other non-heparin anticoagulants because it inhibits the formation of PF4–heparin complexes and interferes with PF4 binding to cell surfaces. Danaparoid mainly works by inhibiting factor Xa through antithrombin III, with less inactivation of factor IIa. It has a high anti-factor Xa to anti-factor IIA activity ratio and a low tendency to cause bleeding, making it a relatively safe and effective alternative antithrombotic for patients on HD. 73
However, patients with reduced kidney function may require lower doses.
Danaparoid can be given subcutaneously or intravenously, and its maintenance dosages should be reduced and titrated according to pre-dialysis plasma anti-factor Xa activity in patients undergoing HD. 74
Although it has been discontinued in the United States (US) due to a shortage in drug substance, it is still available in other countries.
Fondaparinux
Fondaparinux is a sulfated pentasaccharide that inhibits activated factor X and has a long half-life. The drug is mainly excreted by the kidneys, which is why it is contraindicated for use in patients with severe kidney dysfunction. However, a once daily injection has been used successfully in outpatient HD patients. The initial dose is 7.5 mg/d, but dosage titration is based on anti-Xa activity level measurement postdialysis.
A small prospective study done by Mahieu et al on 4 patients on HD and HIT found that fondaparinux used on these patients was safe, and bleeding risk did not increase; however, clotting risk increased with the dose reduction. At the end of the dose titration study, 3 patients were maintained on a dose of 0.03 mg/kg and 1 patient at 0.04 mg/kg of fondaparinux, with appropriate dose adjustment to body weight.
A small study found that fondaparinux was safe and efficient in patients with kidney failure and HIT. 75 Similarly, Linkins et al found that fondaparinux appears to be effective and safe for treating acute HIT. 76
Despite the lack of randomized trials, the drug is used off-label for HIT treatment. It is worth noting that fondaparinux has a small possibility of causing HIT postexposure and has not been FDA-approved for HIT treatment.
Vitamin K Antagonists
During the acute phase of HIT, the use of vitamin K antagonists is not recommended due to its reduction in protein C and S levels and increased risk of thrombosis. However, it may be resumed during the subacute phase after platelet counts have normalized and must be overlapped with a non-heparin anticoagulant for at least 5 days. 77
In HD patients, the use of vitamin K antagonists should be approached with caution as there is a significant risk of bleeding with full intensity anticoagulation. There has been extensive research on the use of vitamin K antagonists in HD patients. 78
Direct Oral Anticoagulants
Direct oral anticoagulants (DOACs), which include dabigatran, a direct thrombin inhibitor, and direct factor Xa inhibitors such as rivaroxaban and apixaban, are a safer and more effective alternative to HIT treatment for various patient populations, including ESRD on HD. 79 DOACs are easy to administer, do not require monitoring, and are less costly compared to other drugs. They can be used in the acute and subacute phases of HIT and are particularly useful in patients with low platelet counts. DOACs’ optimal dosing regimens and safety in patients with severe kidney dysfunction remain unclear. Among DOACs, apixaban appears to be the least dependent on renal clearance of the direct factor Xa inhibitors and may have the lowest bleeding risk. 80
Although evidence is limited, DOACs have been added as a potential HIT treatment by the American Society of Hematology (ASH).
Nucleic Acid Aptamers
Nucleic acid aptamers are a type of therapeutic oligonucleotide made of short, single-stranded deoxyribonucleic acid (DNA) or ribonucleic acid (RNA). They are designed to bind and inhibit coagulation factors with high specificity and affinity. 81 Although research has shown promise in using them as a treatment for HIT, current studies are still in phase II clinical trials. As a result, it is too early to determine their clinical efficacy in HIT patients, and their use in patients with kidney dysfunction or undergoing HD is uncertain. 82
Lipoxygenase- Inhibition
Platelets contain an enzyme called 12(S)-lipooxygenase (12-LOX) that produces an oxylipin. This oxylipin is responsible for the conversion of arachidonic acid into 12-hydroperoxy-5,8,10,14-eicosatetraenoic acid (12-HpETE), which plays a role in platelet aggregation. 83
Inhibiting 12-LOX could be a potential therapeutic target for HIT, as it may reduce platelet response. However, a 12-LOX inhibitor called ML-355 is still in the preclinical phase, and its effects on the kidneys have not been studied yet. 84
High-Dose Immunoglobulins
Autoimmune HIT refers to a type of HIT in which the patient's antibodies activate platelets both in the presence and absence of heparin, leading to atypical presentations such as delayed-onset HIT, refractory HIT, heparin “flush” HIT, fondaparinux-associated HIT, severe thrombocytopenia with disseminated intravascular coagulation, and spontaneous HIT syndrome.
Several case reports have been published regarding the use of high-dose immunoglobulins for the treatment of autoimmune HIT. A recent systematic review of intravenous immunoglobulin (IVIG) use in HIT patients suggested that IVIG should be used in patients who do not respond to non-heparin anticoagulants within 5 to 7 days of cessation of heparin. None of these have been reported in patients with ESRD. In most cases, IVIG was used as an adjunctive therapy alongside alternative anticoagulation, although this therapy is not commonly used. 85
Plasma Exchange
Vender et al first reported in 1986 that therapeutic plasma exchange (TPE) could be a potential therapy for HIT. Since then, 3 case reports and a retrospective review have suggested that TPE can be beneficial for HIT patients, particularly those undergoing cardiopulmonary bypass (CBP). The aim of this therapy is to reduce anti-PF4/heparin antibodies, enabling the use of heparin for CBP, and most patients have responded well to this treatment. TPE can be performed simultaneously with HD.86,87
A systematic review comparing the effectiveness of TPE and IVIG for the treatment of HIT found that 1 to 2 TPE procedures are generally sufficient to reduce or eliminate PF4/heparin antibodies/immune complexes, without any significant thrombotic complications (Table 1). 88
Alternative Anticoagulation in Patients with HIT.
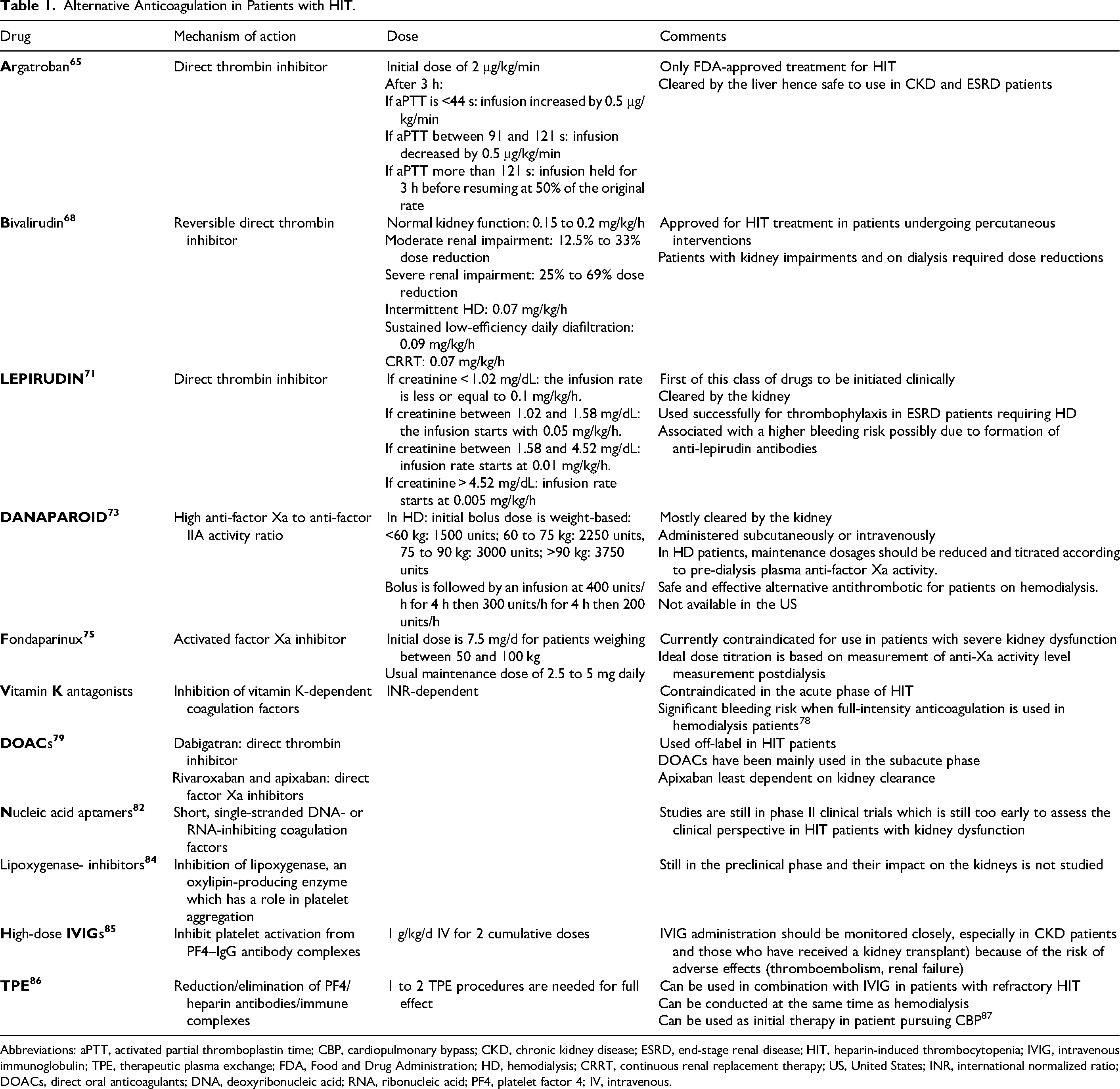
Abbreviations: aPTT, activated partial thromboplastin time; CBP, cardiopulmonary bypass; CKD, chronic kidney disease; ESRD, end-stage renal disease; HIT, heparin-induced thrombocytopenia; IVIG, intravenous immunoglobulin; TPE, therapeutic plasma exchange; FDA, Food and Drug Administration; HD, hemodialysis; CRRT, continuous renal replacement therapy; US, United States; INR, international normalized ratio; DOACs, direct oral anticoagulants; DNA, deoxyribonucleic acid; RNA, ribonucleic acid; PF4, platelet factor 4; IV, intravenous.
Conclusion
The diagnosis and treatment of HIT can be difficult in ESRD patients and requires careful consideration in order to provide appropriate care for this patient population. Alternative treatments are available, and clinical trials are ongoing to potentially prevent this condition from occurring in the first place. Therefore, a clinical approach should always be taken to address this issue.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
