Abstract
In 2018, Refacto AFR, a B-domain-deleted third-generation FVIII concentrate, became our preferential product. After the introduction, the development of inhibitors was prospectively monitored; retrospectively, we sought for risk factors in the patients who developed a de-novo inhibitor. Over a period of 15 months, 4/19 adult patients with non-severe haemophilia who were treated on demand for surgery, developed high titer antibodies to FVIII after administration of Refacto AFR; 5/52 mostly severe patients on prophylaxis, developed an inhibitor (3 ≥ 0.1 BU; 1 > 0.6 BU, 1 high titre) after they switched to Refacto AFR; all were children <14 years of age and with >100 exposure days, none related to surgery or intensive treatment; all received KovaltryR before. In conclusion: inhibitors were encountered in on demand patients and previously treated prophylaxis patients; this observation might be a coincidental finding, but also risk factors like genotype and surgery and/or that Refacto AFR is more immunogenic should be considered. For the patients on prophylaxis we hypothesize that loss of tolerance by preceding KovaltryR might have contributed to inhibitor development.
Introduction
Although nowadays hemophilia A patients can be treated with emicizumab, they usually receive supplementation of factor VIII (FVIII), either as prophylaxis or on demand. FVIII is usually produced with DNA-recombinant techniques and can be obtained from several pharmaceutical companies that use different production procedures. Due to competition between pharmaceutical companies producing FVIII, more often patients may switch from one product to another. In general, switching from one FVIII product to another is not considered to be a risk factor for developing an inhibitor. 1 For reasons of national contracting, our Hemophilia Treatment Center (HTC) switched in 2018 to use preferentially a third-generation recombinant B-domain-deleted (BDD) FVIII (Refacto AFR). After the switch, we monitored the occurrence of inhibitors closely. In this paper, we share our observations and discuss the factors that might have contributed to the elicitation of these inhibitors.
Materials and Methods
After the decision in 2018 to use preferentially Refacto AFR, all adult and pediatric patients already on prophylaxis with >50 exposure days (ED) switched to this product, except when they received or were to receive a longer acting FVIII product (FVIII-Fc, EloctaR). Children with severe hemophilia not yet on prophylaxis or <50 ED continued AdvateR supplementation. In the period June 2018–September 2019, adults and children >12 years of age with non-severe hemophilia in need of on demand treatment received also Refacto AFR. All patients that were switched to Refacto AFR were prospectively followed for de-novo inhibitor development as part of their standard patient care. No ethical committee approval for this observational study was necessary. Patients were verbally informed for use of their aggregated and anonymized data published in this article.
Inhibitor testing was done after a washout period of at least 48 h with the standard Nijmegen modification of the Bethesda assay, with a cut-off of 0.1 BU determined as positive. A positive result was always confirmed after an interval of at least 1 week. In the prophylaxis patients inhibitor testing was performed before and 2 months after the switch to Refacto AFR, and/or when the prophylactic treatment was clinically less efficacious. In the on demand patients, inhibitor testing was performed before and 6 weeks after surgery or trauma, earlier in case of increased bleeding.
Retrospectively, the charts of the inhibitor patients were analyzed for factors (possibly) associated with increased inhibitor risk. 2 We looked at age; FVIII gene mutation (genotype); family history of inhibitor development; recent (4 weeks before inhibitor demonstration) surgery, infection or vaccination; exposure days and FVIII treatment intensity (high intensity defined as >45 IU/kg/ED); in addition, the preceding FVIII product was reviewed.
Results
On Demand Patients
In the period June 2018–September 2019, 25 patients with moderate or mild hemophilia received factor VIII supplementation because of trauma or surgery. Of these, one adult had a pre-existing inhibitor and five children stayed on AdvateR; the other 19 (2 children and 17 adults) switched to Refacto AFR and were followed: four developed an inhibitor. Table 1 shows characteristics of these patients in comparison to those that did not develop an inhibitor. All inhibitors occurred in adults, between 33 and 69 years of age; they had an FVIII activity 8%–26% and a maximum of five previous exposure days; all occurred after surgery with continuously administered FVIII and in three after FVIII supplementation in combination with desmopressin (DDAVP). All were supplemented till an FVIII level of 100%; none needed >45 IU/kg/ED. Genotype was known in three patients, two male patients showed an almost identical missense mutation in exon 26 (c.6956 C > T, p.Pro2319Leu and c.6955 C > T; p.Pro2319Ser); in the female carrier (with a factor VIII level of 22%) the intron 22 inversion mutation was demonstrated.
Characteristics of Inhibitor Versus Non-Inhibitor on Demand Patients (n = 19).

The inhibitors were all high titre resulting in FVIII levels of <0.5% and the clinical picture of severe haemophilia. One patient (inhibitor titer 91 BU) received immunosuppressive therapy (steroids and cyclophosphamide) as early as 6 weeks after inhibitor onset because of ongoing serious bleedings which resulted in disappearance of the inhibitor; in the other three patients the inhibitor persisted for over 1 year with titers >100, >100 and 19 BU respectively; because of this persistence, all 3 have ultimately been treated with rituximab 3 resulting in decline of the inhibitor titer in two and disappearance in one. All 4 patients started with emicizumab prophylaxis which at the time of writing, could be stopped in two; none has been rechallenged with FVIII.
Patients on Prophylaxis with FVIII
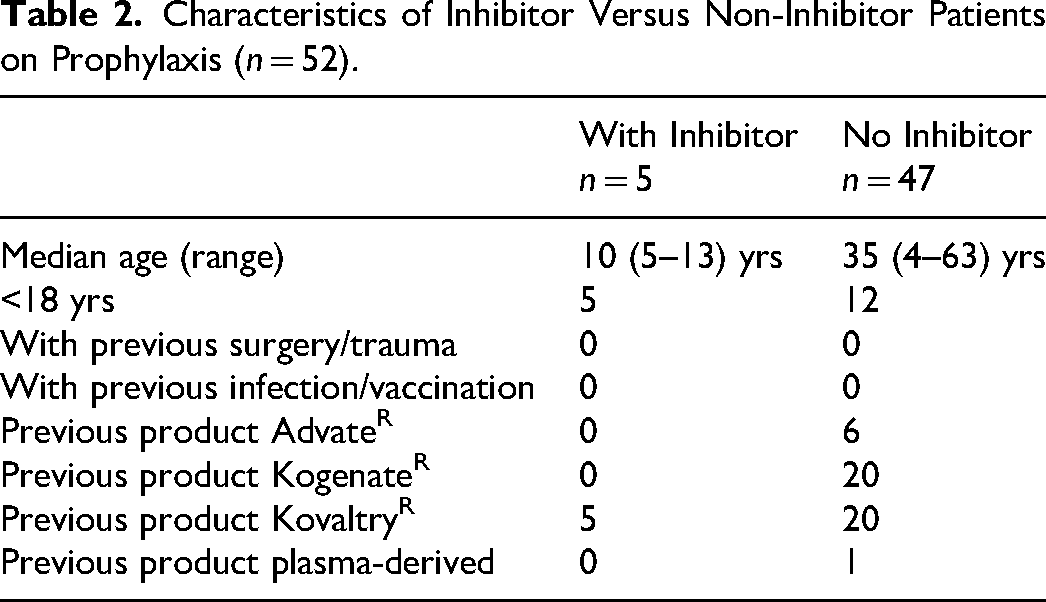
A total of 60 patients with severe or moderate haemophilia were on prophylactic treatment. Of these four were on study medication, three stayed on EloctaR and 1 switched to EloctaR; the remaining 52 patients (14 children and 38 adults) switched to Refacto AFR and were followed: 5 developed an inhibitor. Table 2 shows some characteristics of the inhibitor patients in comparison to those in whom no inhibitor occurred. The inhibitor patients were all children, 4–14 years of age, with severe hemophilia, all after >100 EDs; none had surgery, trauma, intensive FVIII treatment, vaccination, or infection before the inhibitor occurrence. In all inhibitor patients, KovaltryR was the preceding product. Two of the patients are brothers, the other three are without a family member known for an inhibitor. One inhibitor patient had previously developed a high titer inhibitor (max 48 BU) on AdvateR but after the disappearance of this inhibitor, it could not be demonstrated for more than 7 years. Another inhibitor patient had been exposed as a PUP to Refacto AFR for over 50 EDs without developing an inhibitor, but did so after the switch in 2018. With respect to FVIII gene mutation: one patient had an intron 22 inversion and 4 had a FVIII null mutation: the brothers had an insertion (after c.2640 resulting in p.Lys881Glyfs*22) and the other 2 a point mutation (c.36C > A, p.Cys12* and c.568C > T, p.Gln190*).
Characteristics of Inhibitor Versus Non-Inhibitor Patients on Prophylaxis (n = 52).
Four patients had low titer inhibitors (max 0.6, 1.0, 0.2, and 0.2 BU, respectively) which resulted in mild increase of bleeding tendency (more bruises and mucosal bleeds) that could be managed with intensification of the prophylactic regimen; with continuation of the prophylaxis the inhibitor disappeared after a maximum period of 5 months. The maximum inhibitor titer of the fifth patient was 32 BU; Refacto AFR was interrupted and he was treated with emicizumab; immune tolerance induction with Refacto AFR started 9 months after inhibitor occurrence without any demonstrable effect on the inhibitor titer; administration of Refacto AFR was continued resulting in disappearance of the inhibitor after 1.5 years and cessation of emicizumab after 2 years.
The occurrence of the inhibitors in both patient groups was reported in the national (KWARK) and international (EUHASS) complication registers.
Discussion
We have observed a number of inhibitors after the introduction of Refacto AFR in our HTC, specifically in two groups of patients.
The first group consists of four adults out of 19 patients with non-severe hemophilia after on demand treatment with Refacto AFR. Herewith, the prevalence of inhibitors in this type of patients in our center doubled over a short period of time. Comparing these four patients with the 15 that did not develop an inhibitor (Table 1), they had less EDs and always surgery; the type of surgery was not different (data not shown) between both subgroups nor the combined use of FVIII with DDAVP. In all four inhibitor patients the titer increased despite cessation of the administration of exogenous FVIII. In the three patients that not received immediate immune suppression, the inhibitor persisted for 12–28 months before the individual was decided to start rituximab. 3
From the INSIGHT studies, it has become clear that inhibitor development in non-severe hemophilia patients has a cumulative incidence of 5.3% at a median number of exposure days of 28 and increases with the number of exposure days. 4 After a median time of 18 months, the inhibitor disappeared in 71% of the patients, spontaneously (50%) or after eradication treatment (ITI and/or immunosuppressive treatment; (21%). 5 Considering these results from the INSIGHT studies, our observations seem not to fit: the number of patients that had developed an inhibitor was high (4/19), after ≤ 5 EDs, and no spontaneous decline or resolution of the inhibitor occurred after cessation of FVIII exposure. Moreover, inhibitors in carriers of hemophilia have been described only twice.6,7 The genotype is known in three of our on demand inhibitor patients: the missense mutations of the two male patients are found to be associated with an increased risk for inhibitor development 8 and the female carrier is heterozygous for the intron 22 inversion mutation; the latter mutation has been described in one of the two other described carriers with an inhibitor 7 and is also associated with increased risk of inhibitor development in PUPs. Additionally from the INSIGHT studies, it is known that a high dose of FVIII and surgery increases the risk for inhibitor development in non-severe hemophilia 7.5 and 4.2 times, respectively. 9 All our patients had surgery and were supplemented till a FVIII level of 100%, but none received high-intensity FVIII defined as >45 IU/kg/ED. Three of our four inhibitor patients received desmopressin during surgery; from this observation, one might get the impression that DDAVP co-administration could be a risk factor for inhibitor development while DDAVP purposely is administered to diminish the exposure to high doses of FVIII and reduction of the inhibitor risk 10 ; however, 6/15 of the non-inhibitor patients also received desmopressin. Also, the fact that our patients received all their FVIII substitution by continuous infusion is nowadays not considered to be associated with increased incidence of inhibitors anymore. 11 In the INSIGHT studies, product type was not found to be a risk factor for inhibitor development, either comparing plasma derived with recombinant FVIII concentrates or for specific types (first, second, and third generation) of recombinant concentrates. 12 However, in this latter report, the numbers of subgroups of patients that were treated with a specific recombinant FVIII concentrate were rather low, and thus this analysis might have been underpowered. Moreover, the INSIGHT study nor other studies have compared specific brands of products for their inhibitor risk in non-severe hemophilia patients.
From the above, the occurrence of inhibitors in the on demand patients with mild hemophilia should be considered coincidental or caused by known risk factors like surgery and/or genotype but might in addition be explained by increased immunogenicity of Refacto AFR (see below). Coincidence, however, seems unlikely in our second group which consists of five children out of a total of 52 patients with severe hemophilia A on prophylaxis, all with >100 previous exposures to other FVIII products. This incidence is rather high and can be explained in 3/5 inhibitor patients by our inhibitor definition with a threshold of 0.1 BU, where in general 0.6 BU is used. However, also our patients with low and transient inhibitor titers had symptoms of increased bleeding tendency resulting in increased anxiety. All these inhibitor patients were children <14 years of age, and all after a switch from KovaltryR to Refacto AFR. In a meta-analysis the incidence of inhibitors in previously treated patients (PTP) per 1000 patient years is calculated to be 3.0. 13 In a more recent study of the natural history of inhibitors in PTPs, 14 they usually appeared to be low titer and recovered spontaneously or after ITI. Also in 4/5 of our patients, the titers were low and transient (yet, requiring adaptation of treatment policy); in the fifth patient, a high titer inhibitor was found that indeed disappeared after ITI. In this study of the natural history of inhibitors, 14 risk factors were surgery in 22% of the cases and/or a period of intensive treatment with FVIII in 17%. 14 In none of our prophylaxis inhibitor patients, such a known risk factor could be shown except for the switch of FVIII product. As mentioned in the introduction, switching is not considered a risk factor 1 but the above cited study on the natural history of inhibitors found that switching could have been a factor in up to 10% of the 52 investigated cases. 14
Thinking of possible explanations for our observation that inhibitors occurred after switching FVIII concentrates in PTPs on prophylaxis, we suppose that the immunogenic difference between the previous product and the product switched to, is more relevant than generally considered. In our prophylaxis inhibitor patients, this means that KovaltryR from which our patients switched, is less immunogenic and/or that Refacto AFR to which our patients switched, is more immunogenic. KovaltryR is a relatively recently developed full-length FVIII concentrate produced by a BHK (baby hamster kidney)-cell line that has increased branching and sialylation of N-linked glycans as well as less aggregates (compared to KogenateR), both factors that are supposed to reduce immunogenicity.15,16 However, to our knowledge, there is no clinical study supporting this reduced immunogenicity of KovaltryR in clinical practice. Refacto AFR is a third-generation BDD recombinant FVIII concentrate produced by a CHO (Chinese hamster ovary)-cell line, that has been implicated to be increased immunogenic: a (much debated) meta-analysis of prospective clinical trials showed an increased incidence of inhibitors in PTPs receiving a BDD FVIII product 17 ; in addition, a more recent systematic review found an inhibitor incidence rate ratio (IRR) for Refacto AFR of 4.81 (95% CI 0.99-23.34), AdvateR being the reference. 18 Moreover, after switching to Refacto AF from other products than KovaltryR, often inhibitors have been found: in a non-random comparison from the United Kingdom, a not statistically significant (P = .06) difference in inhibitor incidence between switchers to Refacto AFR (7.8 per 1000 treatment years) and non-switchers (1.5 per 1000 years; IRR 5.3, 95% CI 0.5-260.3) was reported 19 and in a Canadian surveillance two inhibitors among 125 switchers to Refacto AFR were found. 20
If, indeed, KovaltryR is relatively less and Refacto AFR relatively more immunogenic this difference might explain (in part) the occurrence of inhibitors in our patients. Considering that antigen persistence for immunologic unresponsiveness is a necessity,21,22 KovaltryR may have caused loss of tolerance to exogenous FVIII, especially in our young patients because of less long and less variable exposition to (different) FVIII products than older patients. When this tolerance indeed is lost, inhibitors in PTPs may occur when exposed to a more immunogenic FVIII concentrate like Refacto AFR. Following this thought, in the era of registration of emicizumab for inhibitor and non-inhibitor patients, not inducing and maintaining tolerance to FVIII by additional regular intermittent FVIII exposure, may increase the risk of inhibitor development when exposure to FVIII is needed at the time of “high immunologic danger” such as a breakthrough bleed or surgery, 21 a situation that is comparable to that in non-severe patients in whom a linear relationship exists between increasing exposure days and risk of inhibitor development without a reduction of this risk after 50 EDs. 4
From our observation that we encountered inhibitors after a switch to Refacto AFR, we conclude that this might be coincidental or due to known risk factors like surgery and genotype in the on demand patients although one might argue that Refacto AFR is more immunogenic, and that in the young patients on prophylaxis this might be caused, in addition, through loss of tolerance by preceding KovaltryR.
Footnotes
Authors’ Note
1. L. H. Hooimeijer: design, analysis and interpretation of data, drafting and revision of the manuscript. M. A. Stein-Wit: collection and analysis of data, revision of the manuscript. M. A. J. Voskuilen: collection and analysis of data, revision of the manuscript. M. V. Lukens: analysis and interpretation of data, drafting and revision of the manuscript. K. Meijer: concept and design, analysis and interpretation of data, drafting and revision of the manuscript. A. B. U. Mäkelburg: collection and analysis of data, revision of the manuscript. R. Y. J. Tamminga: concept and design, analysis and interpretation of data, drafting, and revision of the manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Meijer reports speaker fees from Alexion, Bayer, and CSL Behring, participation in trial steering committee for Bayer, consulting fees from Uniqure, and participation in data monitoring and endpoint adjudication committee for Octapharma. All fees are paid to her institution. None of the other authors has a potential conflict of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
