Abstract
Epidemiological studies have suggested a potential relationship between the transforming growth factor-β1 (TGF-β1) gene and ischemic stroke (IS) risk; however, the current results are inconsistent. Therefore, we performed this meta-analysis to assess the precise association between TGF-β1 polymorphisms and IS risk. Online databases were searched for themes related to TGF-β1 polymorphisms and ARE risk. Quantitative calculations of odds ratios (ORs) and confidence intervals (CIs) were performed using 5 genetic models of each variant locus. Heterogeneity tests, cumulative analyses, sensitivity analyses, and publication bias were conducted to examine statistical power. Moreover, changes in the secondary structure and minimum free energy (MFE) were explored using in silico analysis. Nineteen case-control studies were included in our meta-analysis on rs1800468 G>A, rs1800469 C>T, and rs1800470 T>C polymorphisms and IS risk. Overall, only a marginal association was found between the rs1800469 C>T polymorphism and IS risk (T vs C: OR = 1.12, 95%CI = 1.00-1.46, P = .05, I2 = 77.0%). Otherwise, no significant association was observed between the rs1800468 G>A and rs1800470 T>C polymorphisms and IS risk in general and stratified analyses. Moreover, no significant changes in secondary structure and MFE were found in any of the 3 polymorphic loci. Current evidence cautiously suggests that TGF-β1 polymorphisms are not associated with IS susceptibility.
Introduction
Ischemic stroke (IS) is the most common cerebrovascular disease and is caused by a thrombus, embolus, or hypoperfusion due to blood flow blockage and typically results in an insufficient or even interrupted blood supply to the local brain, causing brain cell damage or death, leading to permanent brain injury and loss of neurological function. 1 With the aging of the population, the number of patients with IS has increased to more than 11.92 million worldwide in 2016, accounting for approximately 87% of all stroke types. 2 IS is the leading cause of death and disability, has a major impact on the quality of life of patients, and places a great burden on the family and society, especially in developing countries. 3 To the best of our knowledge, many pathophysiological studies have focused on IS. An increasing number of factors, such as hypertension, high cholesterol levels, smoking status, obesity, and unhealthy lifestyle, have been proven to be driving factors for IS. Moreover, in recent decades studies have shown that racial diversity and inheritance factors, such as genetic mutations and abnormal expression, are closely related to IS risks. 4
Vascular damage, fibrosis, and lipid-rich atherosclerosis are the most important steps in IS development. Transforming growth factor-β (TGF-β) is a class of secretory polypeptide signal molecules that is not only an auxiliary factor for connective tissue growth but also a major factor affecting epithelial cell regeneration and the hematopoietic system. TGF-β modulates cell proliferation, cell migration, matrix synthesis, wound contraction, calcification, and the immune response, and is involved in all major steps of tissue reconstruction, repair, and other functions.
The human TGF-β1 gene is located on chromosome 19q13 and contains 7 exons that encode a 25-kDa peptide, expressed in endothelial and connective tissue cells. TGF-β1 constitutes a high proportion of TGF-β family members and is its most active member. Some studies have demonstrated that TGF-β1 plays a pivotal role in maintaining the balance between inflammation and fibrosis in atherosclerotic plaques. 5 Recent research also indicated that TGF-β1 could suppress the expression of vascular smooth muscle cell (VSMC) proinflammatory genes by blocking both the STAT3 and NF-KB pathways and inhibiting the pathogenesis of atherosclerosis . 6 Increasing evidence suggests that some single nucleotide polymorphisms (SNPs) in the TGF-β1 gene play an important role in the pathogenesis of several cardiovascular and cerebrovascular diseases, such as stroke, coronary heart disease, angina pectoris, and myocardial infarction. In 2006, Sie et al conducted the first case-control study on the association between TGF-β1 gene polymorphisms and IS risk and found that mutations in both the rs1800469 C>T and rs1800470 T>C loci might contribute to increased IS susceptibility. 7 Since then, many studies have focused on the association between TGF-β1 polymorphisms and IS risk, but the results are either inconsistent or contradictory. Therefore, we conducted this meta-analysis of all related studies to further explore the association between TGF-β1 polymorphisms and IS risk.
Materials and Methods
This meta-analysis was conducted in accordance with the guidelines of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses statement. 8 All included information was collected from published studies and no ethical issues were involved.
Search Strategy
A comprehensive literature search was conducted in 5 online databases (PubMed, Embase, Web of Science, CNKI, and Wanfang) for all relevant studies published up to September 1, 2022 that focused on the association between TGF-β1 polymorphisms and IS risk using the following search terms: “transforming growth factor,” “transforming growth factor-β1,” “TGF-β1,” “rs1800468,” “rs1800469,” “rs1800470,” “polymorphism,” “variant,” “mutation,” “ischemic stroke,” “IS.” The relevant papers that were selected were further reviewed manually. Search strategy was used in PubMed for example:
#1 transforming growth factor
#2 transforming growth factor-β1
#3 TGF-β1
#4 rs1800468
#5 rs1800469
#6 rs1800470
#7 #1 OR #2 OR #3 OR #4 OR #5 OR #6
#8 polymorphism
#9 variant
#10 mutation
#11 #8 OR #9 OR #10
#12 ischemic stroke
#13 cerebral infarction
#14 #12 OR #13
#15 #7 AND #11 AND #14
Eligibility Criteria
All included studies in this meta-analysis met the following criteria: (1) observational studies involving the association between TGF-β1 polymorphisms and IS risk; (2) sufficient information regarding the genotypes both in the case and control groups, could be extracted to assess the crude odds ratios (ORs) and 95% confidence intervals (CIs); (3) published in English or Chinese; and (4) the largest or most recent samples on the same data were adopted when overlapping or duplicate samples were reported. Other studies were excluded when they were: (1) reviews, letters, or conference reports; (2) early publications or small sample studies with duplicate themes; (3) molecular biology research with animal models or cell lines; or (4) studies without sufficient data.
Data Extraction
All included studies were reviewed and the following information was extracted for each study by 2 independent investigators (Peng and Cheng): surname of first author, published year, country or region where the study was conducted, races of the research participants, control design, sample sizes of patients and controls, data of each genotype distribution and genotyping method.
Statistical Analysis
Crude ORs and 95% CIs were calculated to assess the statistical strength of the relationship between TGF-β1 polymorphisms [rs1800468 (−800 G>A), rs1800469 (−509 C>T), and rs1800470 (869 T>C)] and IS risk in the general population. Subgroup analyses were conducted based on the HWE status, race diversity, and genotyping methods with at least 3 studies on the same theme. Five genetic models, including allele contrast (M (mutative) versus W (wild)), co-dominant model (WM vs WW and MM vs WW), dominant model (WM+MM vs WW), and recessive model (MM vs WW+WM) were examined. Heterogeneity among the included studies was examined using Cochran's Q-test and the I2 statistical method. A fixed-effects model was adopted when the value of the I2 test was less than 40%; otherwise, a random-effects model was selected. Furthermore, meta-regression was conducted to explore the potential factors contributing to existing heterogeneity. Cumulative meta-analyses were conducted to assess change tendency in results with the addition of studies. Sensitivity analyses were also conducted to examine the stability of the results when studies were removed. Publication bias was examined using Egger‘s linear regression and Begg‘s funnel plots. Statistical analyses were performed using STATA software version 14.0 (Stata Corporation, College Station, TX, USA). A 2-sided Pvalue < .05 indicated statistical significance.
Bioinformatic Analysis
The secondary structure and variant effects of TGF-β1 polymorphisms were predicted using RNAsnp (http://rth.dk/resources/rnasnp/, accessed June 1, 2022).
Results
Study Characteristics
A total of 289 articles were identified based on the search criteria. The selection procedure is shown in Figure 1. During the subsequent steps, 277 articles were removed according to the eligibility criteria of duplicate checks, title and abstract screening, and text reviews. Finally, 12 articles reporting 19 independent case-control studies were included in this meta-analysis.7,9–19 In general, 3 studies focused on the rs1800468 G>A polymorphism and IS risk,7,9,10 8 studies focused on the 1800469 C>T polymorphism and IS risk ,7,10–16 and 8 studies focused on the rs1800470 T>C polymorphism and IS risk.7,10,12–14,17–19 Moreover, only 2 articles were based on Caucasian participants, and the rest were based on Asian descendants. Furthermore, in terms of genotype distribution in the controls, only one study deviated from the HWE in the 1800469 C>T polymorphism. All extracted information is shown in Table 1.

Statistical analysis of the association between TGF-β1 rs1800468 G>A polymorphism and IS risk in the dominant model. (A) ORs and 95% CIs; (B) cumulative analysis; (C) sensitivity analysis; (D) publication bias.
Characteristics of Included Studies on TGF-β1 Gene Polymorphisms and IS Risk.

HWE in control.
Abbreviations: ASSP-PCR, allele-specific sense primers-polymerase chain reaction; ARMS-PCR, amplification refractory mutation system polymerase chain reaction; PCR-RFLP, polymerase chain reaction-restriction fragment length polymorphism; PB, population-based control; HB, hospital/healthy-based control.
Quantitative Analysis
TGF-β1 rs1800468 G>A Polymorphism and IS Risk
Three case-control studies involving 748 patients with IS and 6756 controls were used to determine the association between the F-β1 rs1800468 G>A polymorphism and IS risk. No significant association between this locus and IS risk was identified in the pooled data of the 5 genetic models (A vs G: OR = 1.40, 95%CI = 0.59-3.29, P = .44, I2 = 90.8%; GA vs GG: OR = 1.41, 95%CI = 0.55-3.59, P = .47, I2 = 90.1%; AA vs GG: OR = 1.35, 95%CI = 0.57-3.20, P = .50, I2 = 30.1%; GA+AA vs GG: OR = 1.43, 95%CI = 0.57-3.63, P = .32, Figure 1A, I2 = 90.8%; AA vs GG+GA: OR = 1.26, 95%CI = 0.53-3.03, P = .60, I2 = 13.4%) (Table 2).
Summary ORs and 95% CI of TGF-β1 Gene Polymorphisms and Ischemic Stroke Risk.

Abbreviations: PB, population-based; HB, healthy/hospital-based.
Numbers of comparisons.
Test for heterogeneity.
Cumulative (Figure 1B for dominant model) and sensitivity (Figure 1C for dominant model) analyses were conducted according to publication date and consistently showed a negative result when each study was added or deleted. Publication bias was evaluated and no significant asymmetrical patterns were found (A vs G: P = .72; GA vs GG: P = .78; AA vs GG: P = .16; GA+AA vs GG: P = .74, Figure 1D; AA vs GG+GA: P = .11).
TGF-β1 rs1800469 C>T Polymorphism and IS Risk
Eight case-control studies on the association between the TGF-β1 rs1800469 C>T polymorphism and IS risk involving 3003 IS patients and 14,558 controls were collected. In general, only a marginal association was found in allele contrast, but not in other models (T vs C: OR = 1.21, 95%CI = 1.00-1.46, P = .05, I2 = 77.0%, Figure 2; CT vs CC: OR = 1.10, 95%CI = 0.88-1.38, P = .39, I2 = 56.5%; TT vs CC: OR = 1.22, 95%CI = 0.84-1.76, P = .30, I2 = 67.6%; CT+TT vs CC: OR = 0.65, 95%CI = 0.24-1.78, P = .40, I2 = 98.0%, Figure 2A; TT vs CC+CT: OR = 1.31,95%CI = 0.95-1.82, P = .08, I2 = 73.7%) (Table 2). In addition, similar negative results were found in the subgroup analysis based on HWE status, race diversity, and genotyping methods.
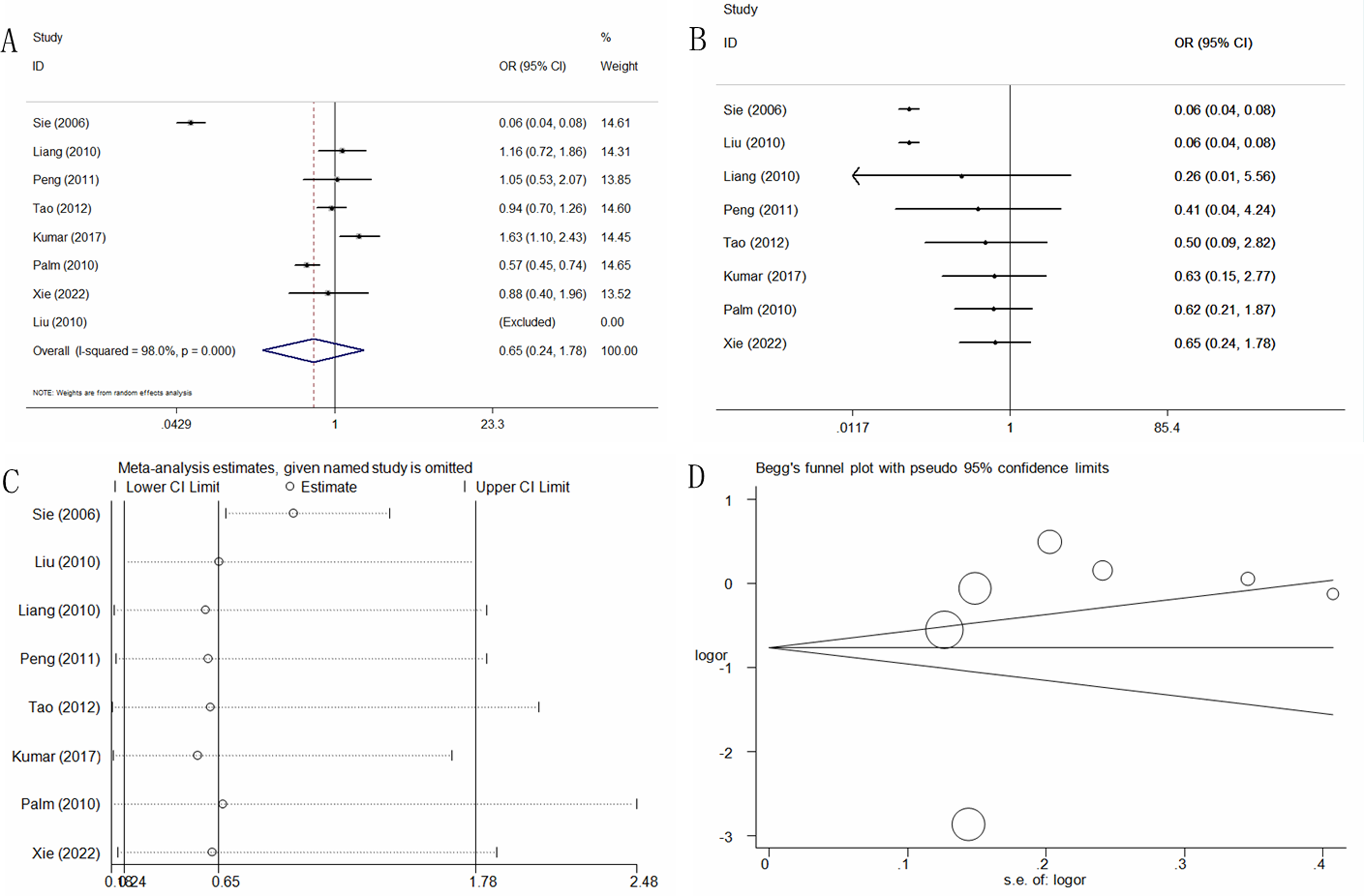
Statistical analysis of the association between TGF-β1 rs1800469 C>T polymorphism and IS risk in the dominant model. (A) ORs and 95% CIs; (B) cumulative analysis; (C) sensitivity analysis; (D) publication bias.
Cumulative analyses according to publication date presented continuous negative results when new studies were added (Figure 2B for dominant model). Sensitivity analysis also indicated consistent results of negative results only when the study by Tao et al was deleted in the T versus C, TT versus CC and TT versus CC+CT models (Figure 2C for dominant model). Publication bias was evaluated, and no significant asymmetrical patterns were found (T vs C: P = 0.08; CT vs CC: P = .88; TT vs CC: P = .14; CT+TT vs CC: P = .45, Figure 2D; TT vs CC+CT: P = .06).
TGF-β1 rs1800470 T>C Polymorphism and IS Risk
Eight case-control studies involving 2392 patients with IS and 11 017 controls were included to determine the association between the TGF-β1 1800470 T>C polymorphism and IS risk. No significant association was found between the rs1800470 T>C polymorphism and the risk of IS (C vs T: OR = 1.16, 95%CI = 0.93-1.44, P = .18, I2 = 87.0%; TC vs TT: OR = 1.04, 95%CI = 0.75-1.44, P = .83, I2 = 84.2%; CC vs TT: OR = 1.15, 95%CI = 0.81-1.64, P = .43, I2 = 77.6%; TC+CC vs TT: OR = 1.11, 95%CI = 0.79-1.55, P = .55, I2 = 86.5%, Figure 3A; CC vs TT+TC: OR = 1.19, 95%CI = 0.94-1.51, P = .15, I2 = 66.0%) (Table 2). No other significant associations were identified in the subgroup analysis based on HWE status, race diversity, and genotyping methods.

Statistical analysis of the association between TGF-β1 rs1800470 T>C polymorphism and IS risk in the dominant model. (A) ORs and 95% CIs; (B) cumulative analysis; (C) sensitivity analysis; (D) publication bias.
Cumulative (Figure 3B for dominant model) and sensitivity (Figure 3C for dominant model) analyses were conducted according to publication date and always showed a negative result when each study was added or deleted. Publication bias was evaluated, and no significant asymmetrical patterns were found (C vs T: P = .19; CC vs CT: P = .93; CC vs TT: P = .36; TC+CC vs TT: P = .65, Figure 3D; CC vs TT+TC: P = .16).
Bioinformatics Analysis
According to the RTH data, the RNAsnp indicated that the TGF-β1 rs1800468 G to A allele substitution results in a minimum free energy (MFE) change from −171.20 to −169.30 kcal/mol (Figure 4); the rs1800469 C to T allele substitution does not result in any MFE change of −127.30 kcal/mol (Figure 5); and the rs1800470T to C allele substitution results in a MFE change from −183.70 to −185.90 kcal/mol (Figure 6). Such changes, which may result in thermodynamic alterations in the RNA secondary structure ensemble, were not statistically significant (Prs1800468 = .08, Prs1800469 = .94, Prs1800470 = . 31).
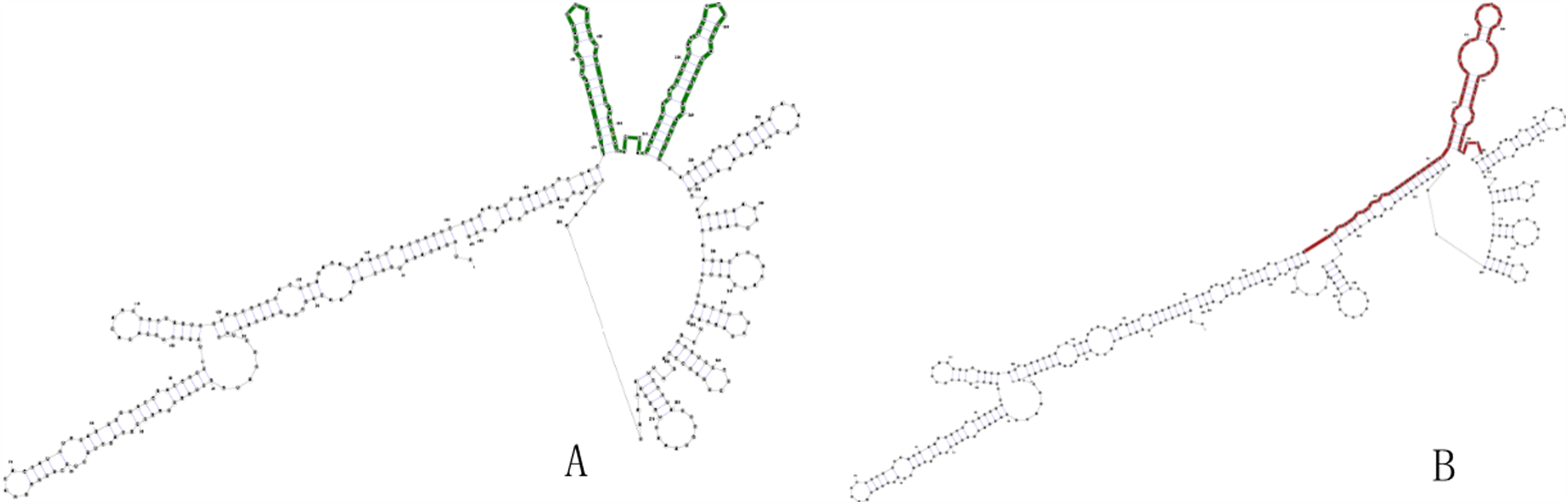
The predicting secondary structure of TGF-β1 rs1800468 G>A. (A) The secondary structure of wild-type sequence with G allele. (B) The secondary structure of mutant-type sequence with A allele.

The predicting secondary structure of TGF-β1 rs1800469 C>T. (A) The secondary structure of wild-type sequence with C allele. (B) The secondary structure of mutant-type sequence with T allele.

The predicting secondary structure of TGF-β1 rs1800470 T>C. (A) The secondary structure of wild-type sequence with T allele. (B) The secondary structure of mutant-type sequence with C allele.
Discussion
To our knowledge, stroke is still the leading cause of death and disability worldwide, resulting in an annual mortality rate of approximately 5.5 million. 20 IS is the most common category and represents more than 80% of stroke patients.21,22 IS, which originates from thrombotic or embolic occlusion of the brain vessels, leads to neural dysfunction, brain impairment, and even death. 23 Epidemiological studies have identified several risk factors for IS, including aging, hypertension, high cholesterol, cigarette and alcohol consumption, diabetes, stress, obesity, and poor diet.
TGF-β1 is an important cytokine with multiple-functions and is involved in the formation and stability of fibrin clots, as well as the adhesion and aggregation of platelets, which are strongly upregulated in the central nervous system following chronic ischemia-induced brain damage. 24 TGF-β1 is a key modulator of osteogenic transdifferentiation of VSMCs, resulting in the transformation of vascular calcification. 25 Active TGF-β1 promotes osteoinductive signaling pathways by combining with a transmembrane receptor and initiates the phosphorylation of regulatory Smad2/3 proteins. These processes drives the transcription of osteogenic genes and upregulation of MMP expression, leading to arterial calcification, 26 which accelerates arterial aging, and increases the occurrence of arterial embolism.27,28
After brain injury, active TGF-β1 promotes tissue regeneration and neurological recovery by regulating the TGF-β/Smad2/3 pathway, which is expected to stimulate both angiogenesis and tube formation in endothelial cells and inhibit neuronal cell injury. 29 In this complex process, IS is always accompanied by a severe inflammatory reaction, TGF-β1 is activated, and the abundant expression of TGF-β1 displays sustained anti-inflammatory effects on microglial cells by persistently inhibiting endogenous Toll-like receptor ligand-induced IκB . 30 TGF-β1 is an important inflammatory inhibitor that can inhibit the aggregation of leukocytes in the vascular wall, especially in inflammatory cells, such as macrophages and T cells. It can also inhibit the proliferation of VSMCs, secretion of inflammatory cell adhesion molecules by endothelial cells, and formation of foam cells to interrupt the pathological process of the vascular wall.5,31 TGF-β1 can inactivate T cells, protect the inner walls of blood vessels, and maintain the stability of atherosclerotic plaques. 32 Moreover, TGF-β1 combines with the extracellular matrix, causing collagen deposition in the vascular wall, decreased vascular elasticity, vascular remodeling, and elevated blood pressure, and participates in the pathological process of vascular development and remodeling in stroke patients.33,34
Several studies have suggested that mutations in TGF-β1 are associated with susceptibility to IS. In 2006, Sie et al conducted the first case-control study and found that individuals with the rs1800469TT genotype had an increased IS risk in Dutch populations (OR = 1.29, 95%CI = 1.02-1.63, P = .03), but not in the other 2 loci. 7 In 2011, Peng et al reported that the frequencies of the rs1800469 TT and rs1800470 CC genotypes were significantly higher in the cerebral infarction group and were associated with IS susceptibility in Chinese patients. 13 In contrast, negative associations with IS were reported in the latest research both by Palm et al 15 on the rs1800469 locus in Germans and by Kim et al 19 on the rs1800470 locus in Koreans. These results are inconsistent with or contradict with comparable research studies published later.
In 2013, Peng et al published the first meta-analysis and did not find any significant association between the rs1800470 T>C polymorphism and IS susceptibility . 35 However, this meta-analysis only focused on the rs1800470 T>C locus and the results were based on 6 case-control studies. In 2016, Kumar et al conducted another brief meta-analysis on the rs1800469 T>C polymorphism and IS susceptibility in only 2 case-control studies and no significant association was reported. 36
We comprehensively collected all current evidence and performed a meta-analysis to assess the association between TGF-β1 polymorphisms and IS susceptibility. Overall, no significant association was observed between the rs1800468 G>A, rs1800469 C>T, and rs1800470 T>C polymorphisms and IS risk with the combined data. Based on the RTH database, bioinformatics analysis indicated that there were slight changes in the spatial secondary structure of the 3 SNP loci, but no significant difference in the thermodynamic MFE between allele changes was observed, which further indicated that mutation of the above loci did not substantially change susceptibility to IS.
However, some limitations of our meta-analysis need to be addressed. First, only Caucasians and Asians were included in our meta-analysis, and the pooled results are difficult to apply to other races. Second, the above 3 polymorphic loci were assessed independently; thus, the haplotype among these loci and the interactive effects with other genetic loci could not be examined. Third, some environmental factors, such as infection, unhealthy drinking or smoking habits, and sleep disorders, could not be examined simultaneously due to the limited original data in the collected studies. Finally, heterogeneity was observed in several genetic models in our meta-analysis, but no significant factors were identified that might have contributed to the heterogeneity in the meta-regression analysis. Fortunately, the following scientific methods were adopted to ensure the quality of our meta-analysis: (1) scientific retrieval strategies and systemic inclusion and exclusion criteria were used to identify all relevant studies; (2) rigorous statistical methods including analysis for sensitivity, cumulative and meta-regression analyses, and tests for publication bias were used to guarantee the current results; (3) stratified analysis based on HWE status, ethnic diversity, and genotyping method was conducted to comprehensively assess the potential relationships; and (4) bioinformatics analysis with RTH data was conducted to explore the underlying mechanism of the change in spatial secondary structure and MFE due to gene mutations.
Conclusion
In conclusion, our meta-analysis indicates that the TGF-β1 rs1800468 G>A, rs1800469 C>T, and rs1800470 T>C polymorphisms are not determining factors for IS susceptibility. Based on this information, the current results should be accepted cautiously, and more case-control studies with larger sample sizes worldwide are required to further investigate the potential relationship between TGF-β1 polymorphisms and IS susceptibility.
Footnotes
Author Contributions
Xiao-Qin Peng and Lan Zhou designed the study, searched the literatures, collected the data, evaluated the qualities and analyzed data. Xiao-Qin Peng and Fan Cheng wrote the manuscript. Lan Zhou reviewed and revised the manuscript. All authors edited the manuscript and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics and Patient Consent
All included information was collected from published studies and no ethics or patient consent issues were involved.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
