Abstract
The plasminogen activator inhibitor-1 (PAI-1) gene has been found to be associated with the pathogenesis and progression of vascular diseases including stroke. A 4G/5G, PAI-1 gene polymorphism has been found to be associated with the plasma PAI-1 levels in different ethnic populations but results are still controversial. The aim of this study was to determine the potential association of 4G/5G polymorphism and plasma PAI-1 levels in the development of ischemic stroke (IS) in young Asian Indians. One hundred patients with IS and an equal number of age- and sex-matched controls were studied. The 4G/5G polymorphism was genotyped in the study population through allele-specific polymerase chain reaction. Plasma PAI-1 levels were evaluated using a commercial kit. The PAI-1 levels were significantly higher in patients when compared to the controls (P = .03). The variant 4G allele for the PAI-I 4G/5G polymorphism showed both genotypic (P = .0013, χ2 = 10.303; odds ratio [OR] = 3.75) as well as allelic association (P = .0004, χ2 = 12.273; OR = 1.99) with IS. The homozygous variant 4G/4G also was found to be associated with the higher PAI-1 levels (0.005). The variant allele 4G of PAI-1 4G/5G polymorphism and higher plasma PAI-1 levels were found to be significantly associated with IS in young Asian Indians.
Introduction
The plasminogen activator inhibitor-1(PAI-1) is a serine protease inhibitor that plays an important role in the regulation of fibrinolysis. Its function involves the inhibition of the activity of tissues plasminogen activator (tPA) and urokinase plasminogen activator (uPA). 1 The fibrinolytic system involves the conversion of plasminogen into plasmin by the action of tPA and uPA, inhibited directly by the PAI-1 and indirectly by the Thrombin activatable fibrinolysis inhibitor (TAFI) and α2-antiplasmin. This, therefore, indicates a central role of PAI-1 in the regulation of the fibrinolytic system. 2 The PAI-1 is now well indicated as the main molecule linking pathogenesis and progression of various thrombotic vascular events including stroke. Also, PAI-1 is known to play varied roles in metabolic and vascular diseases, evolution of brain damage, and recovery after stroke, besides its effects in modulating the activity of tPA and uPA. 3 –6
There has been a well-documented association between prothrombotic disease conditions such as obesity, diabetes, insulin resistance, and hypertension and elevated PAI-1 plasma levels. 7 Studies involving both clinical studies and experimental ones have clearly indicated that higher PAI-1 plasma levels are associated with thrombosis, whereas deficiencies cause accelerated fibrinolysis and bleeding. 5
A number of studies confer that endothelial activation in small vessel brain injury is associated with low levels of PAI-1. Earlier studies also associate high levels of t-PA and differences in the activity of components of the fibrinolytic system with white matter lesion development. 8,9
The PAI-1 gene in humans is located on chromosome 7 (q21.3-q22). 10 A number of polymorphisms in the PAI-1 gene including, -844 G/A, 4G/5G, 978A/G, 11053T/G, and 11320I/D have been studied, however, the 4G/5G polymorphism in the promoter region is the most widely studied one. 11 –13 An in vitro study by Festa et al found that the 4G allele expresses almost 6 times more messenger RNA than 5G allele. 14 Also, in vivo study by Eriksson et al discovered that the lowest PAI-1 plasma levels were in participants’ homozygous for 5G allele and the highest PAI-1 levels were in 4G homozygotes. 15 This is explained because 4G allele is lacking a binding site but not 5G allele for a repressor protein. 16
A few studies have reported that individuals having 4G allele in the homozygote condition have increased PAI-1 levels and heterozygotes (4G/5G) have intermediate PAI-1 levels, whereas those homozygotes for the 5G allele have decreased PAI-1 levels. 17 The association of 4G/5G polymorphism and PAI-1 levels is also well described in patients with diabetes mellitus and myocardium infarction as well as in healthy participants. An association has also been observed between the 4G allele and cholesterol and low-density lipoprotein (LDL) plasma levels in patients with coronary artery disease (CAD). 18 An increased PAI-1 activity leads to decreased fibrinolytic activity in patients with CAD. 19
Presently, there are several studies representing the effect of 4G/5G polymorphisms on the risk of developing stroke, but the results remain controversial. 19 –21
In the present study, we explored the association of higher PAI-1 plasma levels and the prevalence of the 4G/5G polymorphism in the PAI-1 gene promoter region in young patients with ischemic stroke (IS).
Materials and Methods
Study Participants (Inclusion and Exclusion Criteria)
The participants for the study were young adults (18-45 years) and were recruited from the department of Hematology and Neurosciences at All India Institute of Medical Sciences, New Delhi, India. Patients (N = 160) presenting acute IS who were clinically and radiologically (computed tomography/magnetic resonance imaging) diagnosed were recruited for the study. Patients of North Indian origin were exclusively included in the study in order to maintain the geographical homogeneity. Patients presenting cardiovascular diseases, hemorrhagic stroke, hematological disorder, and major systemic diseases were excluded from the study.
Patients who were on treatment with oral anticoagulation, contraceptive, steroids, or antibiotics were included in the study only if they stopped the medication at least for 15 days from the time of sampling. Also, such patients were excluded from the study, who had trauma or had any surgery in the past 30 days. In the final study, only such patients were included who reported for at least on follow-up after 3 to 6 months. During sampling, 38 patients were lost to follow-up and 22 failed to fulfill the inclusion criteria. Finally, a total of 100 patient samples were included in the study. The control group comprised of 100 healthy participants, aged (±3 years) and sex-matched, with no relationship with patients but from the same demographic area. The control population had to fulfill a questionnaire to ascertain that they were stroke-free. 22
A standardized interview was conducted to get information on smoking habits, medication use, and personal history of the disease. Body mass index was measured as weight in kilograms divided by height in square meters. Persons who were aware of having hypertension, took antihypertensive medication, and/or had blood pressure values 160/90 mm Hg at baseline were defined as being actual hypertensive. A participant was defined as a regular smoker when at least 5 cigarettes per day were currently being smoked, and alcohol consumption habits were 2 to 4 days in a week. The study participants both patients and controls joined the study voluntarily and gave a written consent for the same. The local ethics committee of the All India Institute of Medical Sciences approved the above study and was conducted in accordance with the Declaration of Helsinki.
Sample Collection
Venous blood samples were collected 3 months after the episode of stroke. Samples were obtained from each participant after at least 12-hour fasting. Samples were drawn between 7:30
Plasminogen Activator Inhibitor-1 Measurement
Evaluation of PAI-1 antigen levels was done by a commercially available kit (Assay Pro, Truman Boulevard). Enzyme-linked immunosorbent assay was performed as per the kit manufacturer’s instructions. Plasma PAI-1 levels were evaluated for patients (postacute phase) as well as controls. The minimum detectable dose of PAI-1 is typically <200 pg/mL.
Genotyping the 4G/5G Polymorphism
The genotyping for 4G/5G polymorphism was performed through allele specific polymerase chain reaction (PCR). The primers for the 5G allele: 5′-GTC TGG ACA CGT GGG GG-3′ and for the 4G allele: 5′-GTC TGG ACA CGT GGG GA-3′ with the common downstream primer 5′-TGC AGC CAG CCA CGT GAT TGT CTA G-3. Polymerase chain reaction for the 4G and 5G allele was performed in separate tubes. The control upstream primer 5′AAG CTT TTA CCA TGG TAA CCC CTG GT-3′ was used to check the DNA amplification in the absence of the allele in the genomic DNA in a separate tube. 23 The gel for 4G/5G polymorphism is shown in Figure 1.
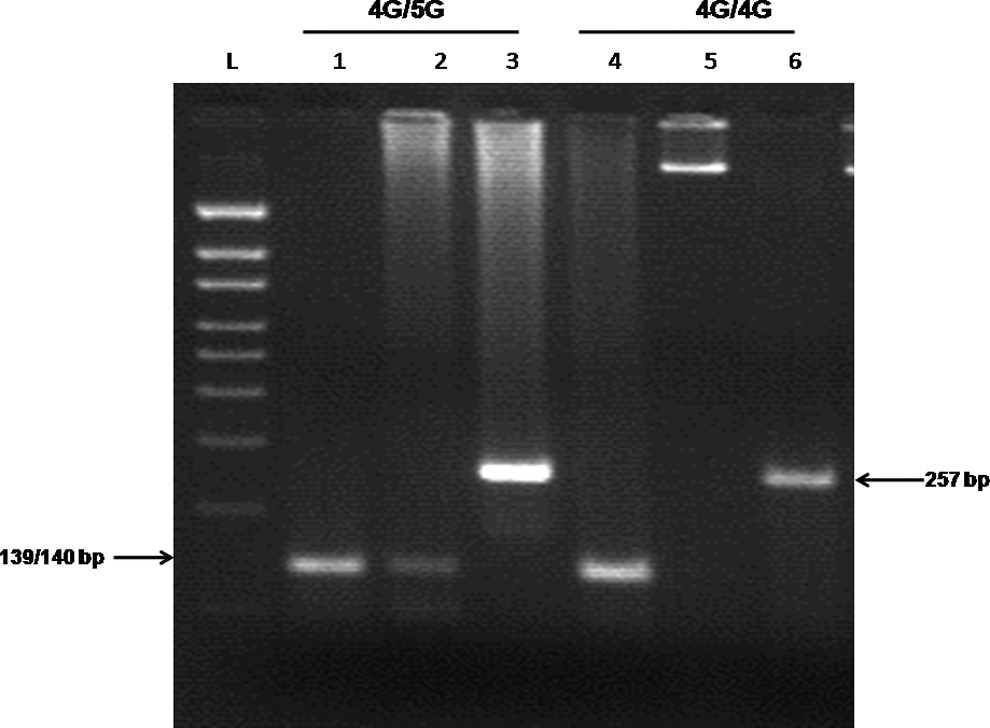
This is an allele specific polymerase chain reaction (PCR) run on 2.5% agarose gel showing amplified product of 4G/5G polymorphism. Lanes 1 and 4 display the 4G allele amplification product; lanes 2 and 5 the 5G allele, and lanes 3 and 6 controls. L denotes 100 bp ladder.
Results
The demographics and clinical characteristics of the patients and controls from the study are summarized in Table 1. Significantly, higher PAI-1 levels were observed in patients when compared to controls (Table 2 and Figure 2). The number of patients with high PAI-1 levels (defined as >75th percentile of study population, ie, ≥13.5 ng/mL) was significantly higher (χ2 = 8.33, P = .004) n = 8 than in controls (n = 0). The risk for stroke increases to almost 8-fold for participants in the fourth quartile (Figure 3). There were no significant differences in the PAI-1 levels between patients with recurrent (n = 33) and nonrecurrent (n = 67) stroke (recurrent strokes: 6.78 ± 4.18 μmol/L, nonrecurrent stroke: 5.89 ± 3.56 μmol/L; P = 0.8).
Clinical Baseline Characteristics and Demographic Data of the Study Participants.
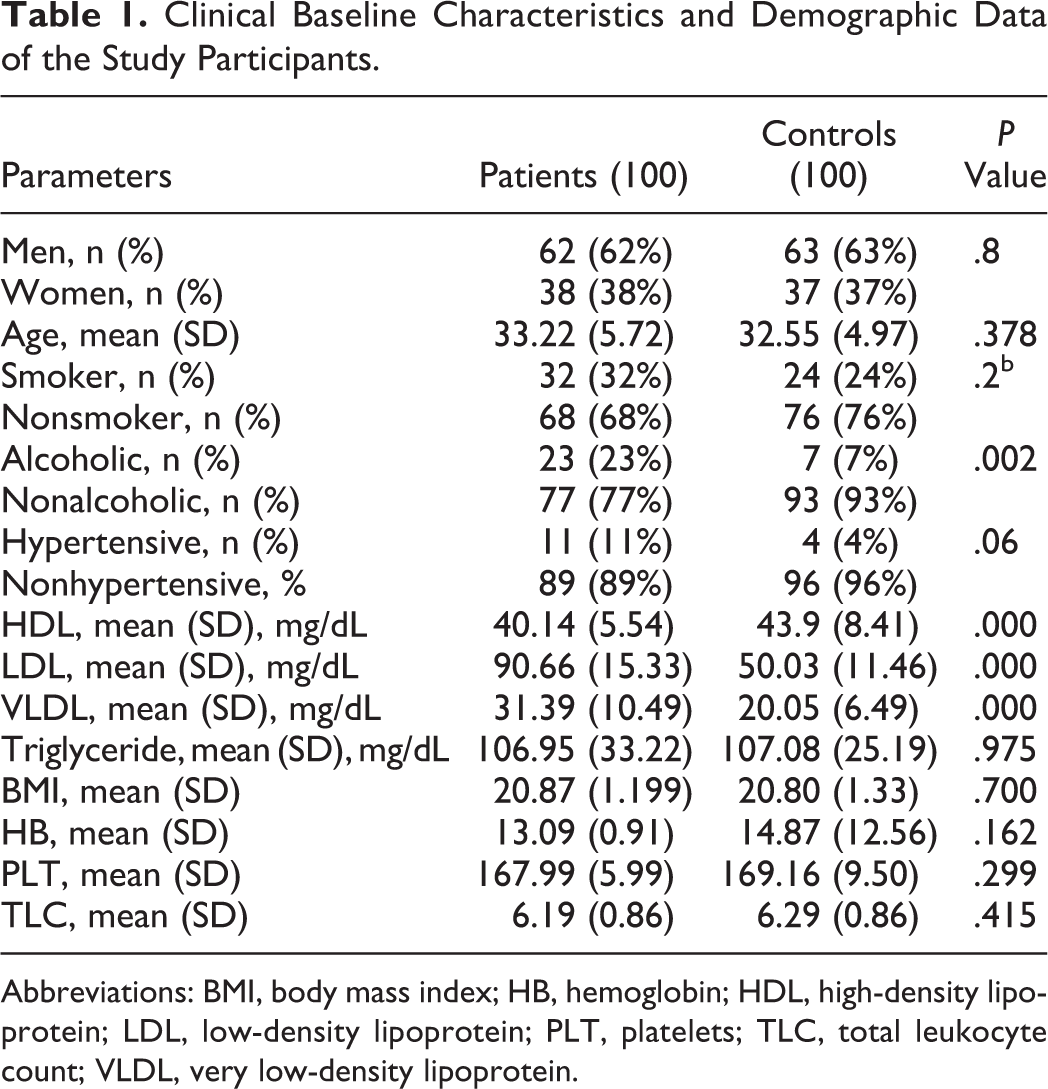
Abbreviations: BMI, body mass index; HB, hemoglobin; HDL, high-density lipoprotein; LDL, low-density lipoprotein; PLT, platelets; TLC, total leukocyte count; VLDL, very low-density lipoprotein.
PAI-1 Antigen Levels in Patients and Controls.a
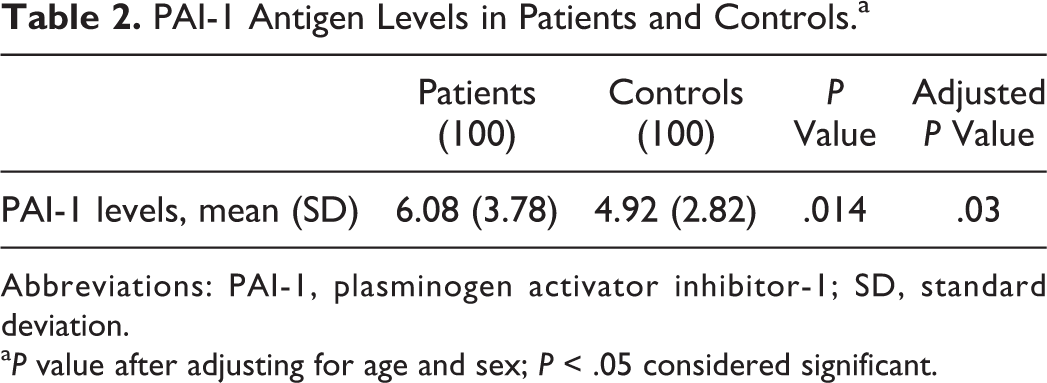
Abbreviations: PAI-1, plasminogen activator inhibitor-1; SD, standard deviation.
aP value after adjusting for age and sex; P < .05 considered significant.
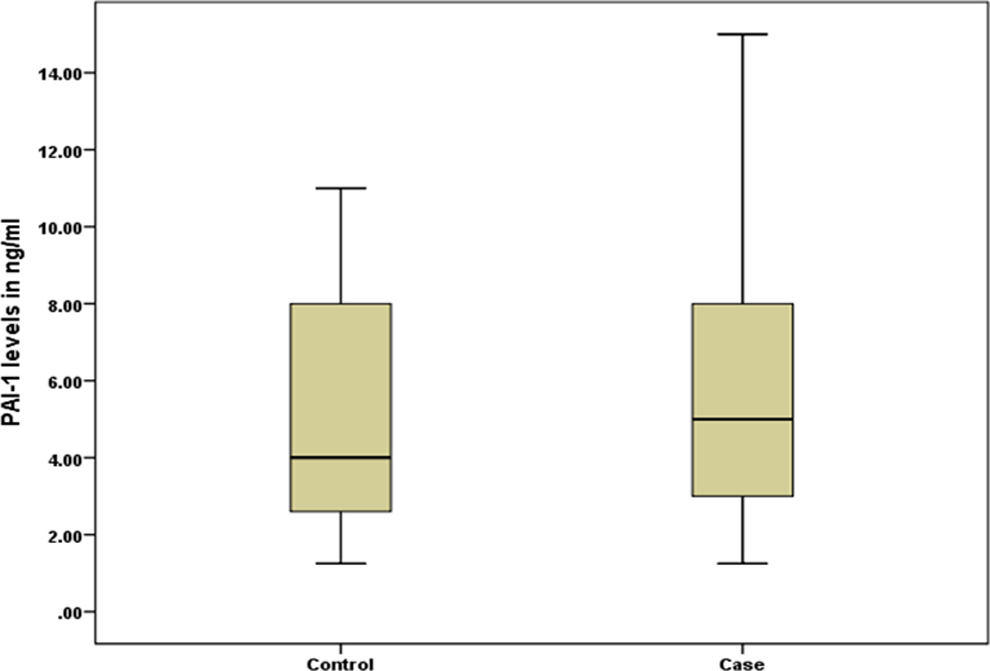
Comparative box-plot illustration for comparison of plasminogen activator inhibitor-1 (PAI-1) levels in patients and controls.
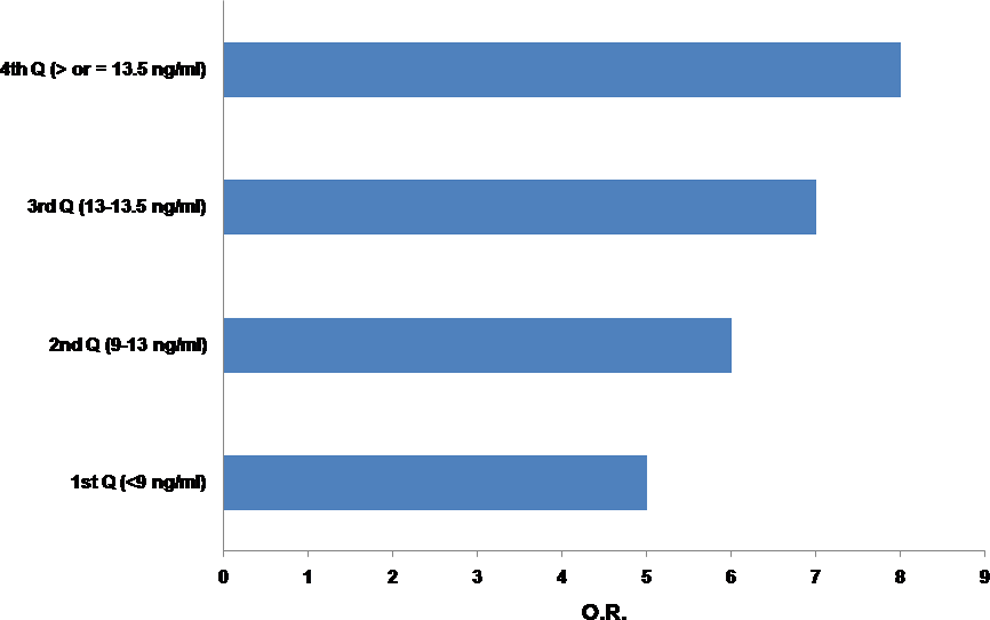
The increase in risk (signified by the increase in odds ratio (OR) as one enters the fourth quartile of plasminogen activator inhibitor-1 (PAI-1) level (ie, >75th percentile of the tested population).
In multivariate linear stepwise regression analysis, the variables such as smoking, hypertension, high-density lipoprotein, LDL, very LDL, triglycerides, and body mass index have poor correlation with PAI-1 levels, whereas sex (P = .042) and age (P = .046) were having a significant correlation. After adjusting for sex and age, we still observed a significant difference in the PAI-1 levels between stroke and control (Table 2). Intra-assay and interassay coefficients of variation for the PAI-1 levels were 5.0% and 7.0%, respectively.
Genotyping for 5G/4G polymorphism showed a prevalence rate of 34 (34%) and 24 (24%) for the 4G/4G genotype in patients and control population, which is significantly different (P = .020, χ2 = 5.35; odds ratio [OR] = 2.5). The allelic frequency of 4G allele was 124 (62%) and 102 (51%) in patients and control population, respectively, which is significantly different (P = .026, χ2 = 4.90; OR = 1.56). These results show that 4G allele has a highly significant association with patients with IS.
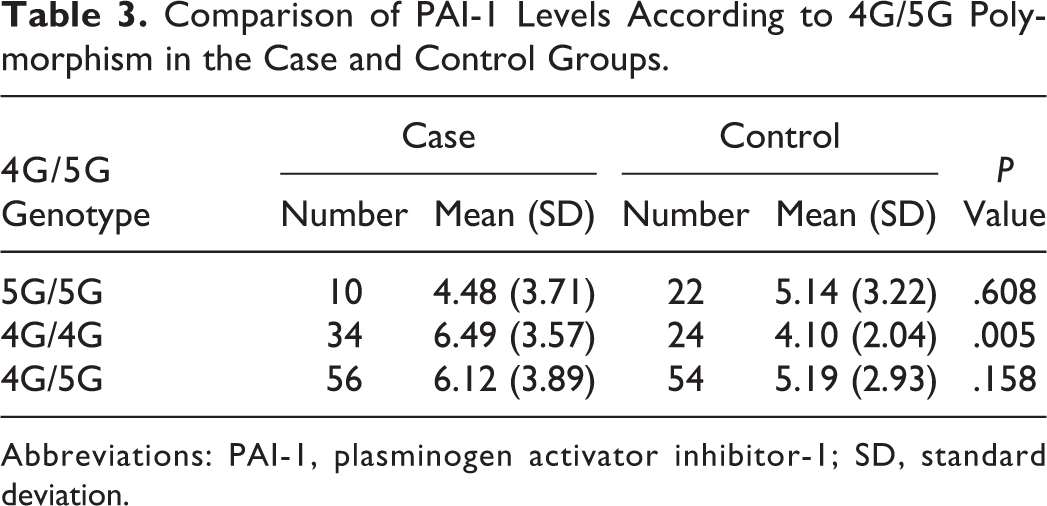
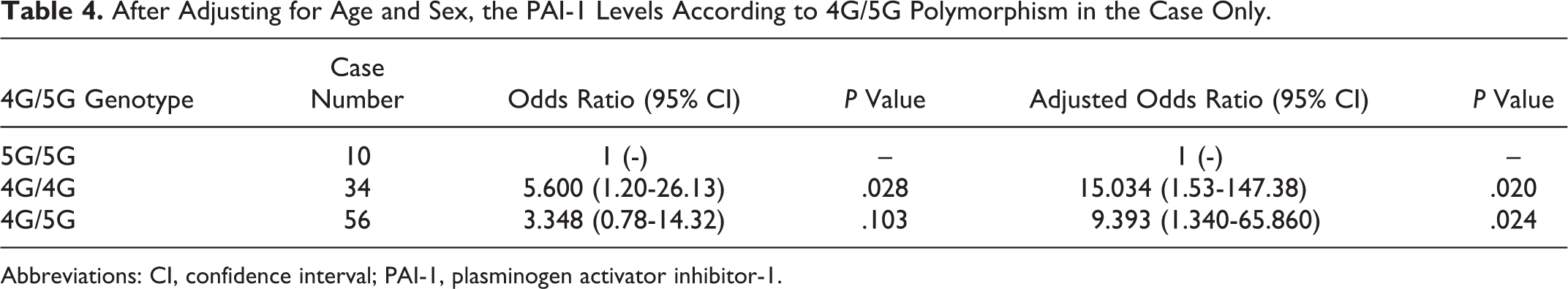
The 4G/5G polymorphisms were significantly associated with increased PAI-1 levels with the variant homozygous 4G/4G corresponding to mean values in patients versus controls (6.49 [3.57] vs 4.10 [2.04], P = .005, Table 3) whereas heterozygous 4G/5G genotypes were not showing significant association with increased PAI-1 levels in patients versus controls (6.12 [3.89] vs 5.19 [2.93], P = .158, Table 3). In this case only, after adjusting for age and sex, the elevated PAI-1 levels were still significantly associated with the 4G/4G genotype and the 4G/5G genotype (Table 4).
Comparison of PAI-1 Levels According to 4G/5G Polymorphism in the Case and Control Groups.
Abbreviations: PAI-1, plasminogen activator inhibitor-1; SD, standard deviation.
After Adjusting for Age and Sex, the PAI-1 Levels According to 4G/5G Polymorphism in the Case Only.
Abbreviations: CI, confidence interval; PAI-1, plasminogen activator inhibitor-1.
Discussion
A dysfunctional fibrinolytic system is known to lead to hemostatic imbalance, which may participate in the pathophysiology of thrombus formation. The PAI-1 gene plays a prominent role in the regulation of fibrinolysis, and an overexpressive PAI-1 may promote the occurrence of thrombotic events. In this study, we looked for the association of PAI-1 antigen levels and 4G/5G polymorphisms in the development of IS in young Indian population. Both clinical and experimental studies have earlier shown that PAI-1 deficiency causes accelerated fibrinolysis and bleeding, whereas elevated PAI-1 plasma levels are associated with vascular thrombosis. 5 In our study, we found that increased PAI-1 plasma levels were independently associated with IS, which is in agreement with other studies that have observed increased PAI-1 plasma levels with risk of IS. 24 –26 We also find in our study that the OR rises as we move from the first quartile to the fourth quartile of the PAI-1 levels (Figure 3), which represents that the participants having PAI-1 levels above the 75th percentile might have an increased risk for being a patient with stroke (almost 8-fold). However, no significant difference in PAI-1 levels between recurrent and nonrecurrent was seen (recurrent strokes: 6.78 ± 4.18 μmol/L, nonrecurrent stroke: 5.89 ± 3.56 μmol/L; P = 0.8), implying that it would not be possible for us to comment on the predictability of a stroke based even on PAI-1 level values. In a nested case–control study by Iacoviello et al, 27 it was observed that individuals in the highest compared to the lowest quartile of PAI-1 had significantly increased risks of ischemic stroke. Lindgren et al 28 found high PAI-1 antigen concentrations in patients with cerebral infarction represents increased fibrinolytic inhibition but not in the subgroups of patients with cerebral hemorrhage. However, in his study, Haapaniemi et al 29 assessed the PAI-1 levels with the neurological examinations using validated stroke scales, conducted at admission, 1 week, 1 month, and 3 months after stroke and reported that the neurological scores did not show a correlation with the PAI-1 levels. Whereas Kim et al 30 in his study found a significant role of PAI-1 in inducing resistance to thrombolytics chiefly through platelet-mediated mechanisms and suggested that increased plasma PAI-1 levels can be used as a biomarker for the prediction of thrombolysis failure in patients with stroke. Johansson et al 31 reported no significant association of PAI-1 antigen levels with the IS; however, they observed the plasma concentration of tPA/PAI-1 complex to be higher for the stroke cases than for the controls. Limitations of these studies include variations in PAI-1 plasma levels due to circadian changes and the acute phase response, as well as alterations due to common comorbid disease states. While in our study, we have tried to overcome such limitations to arrive at conclusive results. However, a recent study by Denorme et al, 32,33 on murine acute IS model demonstrated that targeting PAI-1 inhibition through a monoclonal antibody helps to attenuate fibrin deposition and improves reperfusion while the bispecific inhibitor against TAFI and PAI-1 results in a prominent profibrinolytic effect in mice without increased bleeding. 31
Genetic variations are quite determining in influencing PAI-1 levels, and the most studied genetic variant, impacting PAI-1 levels, is the 4G/5G promoter polymorphism. 34 We observed a higher frequency of 4G/4G genotype and a lower frequency of the 5G/5G genotype in young patients with IS compared to healthy controls (Table 3). The higher prevalence of 4G/4G genotype in our patient population with IS (compared to controls) supports the earlier reports. 35,36 Our present study has shown a significant association of the 4G allele with IS in young Asian Indian population. It is noteworthy that we have previously reported a significant association of 4G allele with deep vein thrombosis 23 in the same population. However, there are some studies presenting contradictory results, where no association of 4G allele with IS and negative association of 4G allele and IS was reported.18, 37 However, Jood et al 38 and de Paula Sabino et al 26 found a negative association between the 4G allele and IS. A recent meta-analysis 39 to arrive at a complete picture of 4G allele association with stroke risk and to derive at a precise estimation showed a significant association of 4G/5G polymorphisms with increased risk of developing IS. When stratified by ethnicity Asian population showed significant association of 4G/5G polymorphism with IS but Caucasian population showed no such association. Which again is supportive of our observation.
We also looked for the association of 4G/5G polymorphism with PAI levels in IS and control population. We found a significant association of 4G/5G polymorphism with the PAI-1 antigen levels. However, we also found age and sex variables to be associated with the PAI-1 levels and, after adjusting these variables, 4G/5G polymorphisms still was independently associated with the PAI-1 levels (Table 4). Our study represents that individuals carrying homozygous 4G alleles have the highest mean of PAI-1 antigen levels (6.49 [3.57] ng/mL in patients with IS and 4.10 [2.04] ng/mL in controls) and those carrying heterozygous alleles, 4G/5G have moderate mean of PAI-1 antigen levels (6.12 [3.89] ng/mL in patients with IS and 5.19 [2.93] ng/mL in controls), whereas those individuals who were carrying homozygous 5G alleles have lower mean levels of PAI-1 antigen (4.48 [3.71] ng/mL in patients with IS and 5.14 [3.22] ng/mL in controls). In vivo study by Eriksson et al discovered the association of 4G/5G polymorphism with PAI-1 levels and that both alleles bind to a transcriptional activator; however, 4G allele was lacking a binding site but not 5G allele for a transcriptional repressor protein. 15 Therefore, it might be the reason that PAI-1 4G alleles have higher PAI-1 level expression. Hence, the 4G allele is associated with the higher plasma PAI-1 levels and implicated in stroke pathogenesis. 12,40,41 However, a few studies have reported contradictorily, van Goor et al 18 showed neither the PAI-1 4G/5G polymorphism nor the PAI-1 antigen level is a strong risk factor for the development of IS. de Paula Sabino et al 26 found an association of increased PAI-1 plasma levels in the development of IS in Brazilian young patients; however, they were not influenced by the 4G/5G PAI-1 polymorphism. Both studies had a lower number of study participants, which would be a limitation of these studies.
This study is the first of its kind that examines the association of PAI-1 polymorphism along with the PAI-1 plasma levels with stroke in young Asian Indian population. An indication of a genetic contribution to the risk of developing IS may have been enhanced in the study by involving exclusively young stroke population, and the findings from the study may be important in the management of stroke particularly in Asian Indian population. Our study, however, had certain limitations, the primary being the small sample size that prevented us from the subtyping of stroke. Another limitation is that the study includes only survivors of IS, and the possibility of survival bias should have been taken into consideration. To overcome the limitations of the study, similar designs must be carried out in large multicentric population, taking into consideration the survival bias that will account for the genetic heterogeneity of the Indian population.
Footnotes
Authors’ Note
The work was conducted in the department of Hematology, All India institute of Medical Sciences, New Delhi, India.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was partially funded by Department of Science and Technology, New Delhi, India.
