Abstract
A high rate of thromboembolism and a high risk of death have been reported regarding hospitalized patients with coronavirus disease 2019 (COVID-19). Recently, we noticed that clinicians in some comparative studies used direct oral anticoagulants (DOACs) to prevent thromboembolism in patients with COVID-19. However, it is uncertain whether DOACs are better than recommended heparin for hospitalized patients with COVID-19. Therefore, a direct comparison of the prophylactic effects and safety between DOACs and heparin is needed. We systematically searched PubMed, Embase, Web of Science, and the Cochrane Library from 2019 to December 1, 2022. Randomized controlled trials or retrospective studies comparing the efficacy or safety of DOACs with that of heparin in preventing thromboembolism for hospitalized patients with COVID-19 were included. We assessed endpoints and publication bias using Stata 14.0. Five studies comprising 1360 hospitalized COVID-19 patients with mild to moderate cases were identified in the databases. Comparing the embolism incidence, we found that DOACs had a better effect than heparin, mainly low-molecular-weight heparin (LMWH), in preventing thromboembolism (risk ratio [RR] = 0.63, 95% confidence interval [CI] [0.43-0.91], P = 0.014). Considering safety, DOACs resulted in less bleeding than heparin during hospitalization (RR = 0.52, 95% CI [0.11-2.44], P = 0.411). Similar mortality was discovered in the 2 groups (RR = 0.94, 95% CI [0.59-1.51], P = 0.797). In noncritically hospitalized patients with COVID-19, DOACs are superior to heparin, even LMWH, in preventing thromboembolism. Compared with heparin, DOACs have a lower trend of bleeding and yield a similar mortality rate. Therefore, DOACs may be a better alternative for patients with mild to moderate COVID-19.
Introduction
Since the opening of the global coronavirus disease 2019 (COVID-19) epidemic, we have become more concerned about the adverse consequences of COVID-19. Thrombosis is an important piece in the puzzle of COVID-19, whose mechanisms are mainly related to endothelial inflammation, complement activation, thrombin generation, platelet and leukocyte recruitment, and the initiation of innate and adaptive immune responses in patients.
1
It is usually accompanied by increased levels of
A retrospective, multicenter cohort study 8 conducted in Japan showed that 43% of patients with COVID-19 received pharmacological thromboprophylaxis. The proportion of pharmacological thromboprophylaxis increased according to the severity of COVID-19 in 9.8% with mild COVID-19, 61% with moderate COVID-19, and 97% with severe COVID-19. Among anticoagulant drugs, heparins, and vitamin K antagonists (VKAs) have been the mainstay of prevention and treatment of potentially fatal thromboembolic events for over 80 years. Although they have a significant effect, there are some drawbacks. For example, both low-molecular-weight heparin (LMWH) and unfractionated heparin (UFH) require injections or drips, making them inconvenient for use outside of the hospital setting. Moreover, UFH requires monitoring because of a short half-life period and unpredictable plasma levels. In addition, bleeding has become the major concern of VKA-warfarin therapy due to the influence of environmental factors and drug interactions. Fortunately, a novel class of oral anticoagulants was approved for the prevention of thrombotic stroke, VTE, and PE in patients with nonvalvular atrial fibrillation in 2010. 9 Subsequently, these direct oral anticoagulants (DOACs) gradually developed into 2 categories, including direct thrombin inhibitors (dabigatran) and direct factor Xa inhibitors (rivaroxaban, apixaban, and edoxaban). Their pharmacokinetic and pharmacodynamic properties were more predictable than those of warfarin, and thus routine monitoring of the anticoagulant effect was not required. 10 Compared with traditional oral anticoagulants of VKAs, DOACs are not only effective in preventing and treating VTE but also safer in terms of bleeding. 11
Several studies showed that the anticoagulant of heparin was associated with a reduced in-hospital mortality risk in COVID-19 patients during hospitalization.12–16 Due to the higher risk of bleeding at therapeutic doses, the International Society on Thrombosis and Hemostasis guidelines 17 gave a strong recommendation for the use of a prophylactic dose of LMWH or UFH among noncritically ill patients hospitalized for COVID-19. However, the evidence was not truly persuasive enough from assessing only one randomized controlled trial (RCT) to explain whether DOACs were recommended for anticoagulation in hospitalized patients. In other words, it was still unclear whether DOACs were better than heparin for anticoagulation in hospitalized patients with COVID-19. Therefore, we conducted a systematic review and performed a meta-analysis to assess the efficacy and safety of DOACs compared with heparin for preventing thromboembolism in hospitalized patients with COVID-19.
Materials and Methods
Retrieval Strategy
We performed a comprehensive online search of PubMed, Embase, Web of Science, and Cochrane Library from 2019 to December 1, 2022. Relevant publications were selected that compared DOACs with heparin for the preventive treatment of thrombi in hospitalized patients with COVID-19. The following search terms were included: (‘Direct Oral Anticoagulants’ OR ‘DOAC’ OR ‘Factor Xa Inhibitor’ OR ‘rivaroxaban’ OR ‘apixaban’ OR ‘edoxaban’ OR ‘dabigatran’) AND (‘Heparin’ OR ‘Low Molecular Weight Heparin’ OR ‘LMWH’ OR ‘unfractionated heparin’ OR ‘UFH’ OR ‘Enoxaparin’ OR ‘Nadroparin’ OR ‘Dalteparin’ OR ‘Tinzaparin’) AND (‘COVID-19’ OR ‘2019 nCoV Disease’ OR ‘coronavirus disease 2019’ OR ‘novel coronavirus’ OR ‘SARS-CoV-2’). There were no country or language restrictions on the literature. To ensure that the work was of high quality, we performed the systematic review and meta-analysis according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement. 18 The registration number of our research on PROSPERO is CRD42022337955.
Inclusion and Exclusion Criteria
Potentially relevant published studies underwent a review of the entire published manuscript. For studies that evaluated the efficacy or safety of anticoagulation in patients with COVID-19, the selection criteria for inclusion in the meta-analysis included the following: (1) RCTs were mainly included. Retrospective studies were also considered when RCTs were insufficient; (2) Studies of hospitalized patients with confirmed COVID-19 aged ≥18 years were included; (3) Studies comparing the efficacy or safety of DOACs with that of heparin (LMWH or UFH) for preventive treatment were included; (4) Treatment studies for patients after embolism were excluded because they did not address the prevention of embolism; and (5) Duplicate publications, review articles, editorials, case reports, and animal experiments were excluded. There were no limitations on patient nationality or ethnicity. The decision to include or exclude the published studies was made separately by 2 researchers, and any disagreement was resolved by discussion until a consensus was reached or by consulting a third author.
Data Extraction and Quality Assessment
Two reviewers independently extracted the following information using a standardized data collection method: first author, year of publication, country, study design, sample size, age, treatment period, drug type and dose, embolism incidence (people with thromboembolic complications), bleeding incidence (people with major and clinically relevant nonmajor bleeding) and all-cause mortality. We used the Newcastle-Ottawa scale as the tool to estimate the quality of the retrospective studies. Studies with scores of 7 or higher were regarded as high quality through careful evaluation. Jadad scale was used to estimate the quality of RCTs, and scores of 4 or higher were regarded as high quality.
Data Analysis
A meta-analysis was performed using Stata 14.0 statistical software. The included studies were tested for heterogeneity using Cochran's Q test and I² test (when P < 0.10 was considered significant). According to the Cochrane handbook, the fixed-effects model was selected for no obvious or low heterogeneity (I² < 50%), and the random-effects model was deemed much more appropriate for moderate heterogeneity (75% ≥ I² ≥ 50%). When there was significant heterogeneity (I² > 75%) in the results, sensitivity analysis was performed using the leave-one-out method to determine the source. Estimated risk ratios (RRs) with 95% confidence intervals (CIs) were calculated to analyze variables. All P values were 2-tailed, and P < 0.05 was considered statistically significant in all tests. Finally, we selected the embolism incidence to examine publication bias using Egger's test.
Results
Literature Search and Screening
The electronic database and manual searches of the reference lists of relevant articles yielded 414 studies. Subsequently, 341 potentially relevant published studies remained after excluding duplicates. Then, 328 studies were excluded by screening titles and abstracts. After thoroughly reading the full text of the remaining 13 studies, 8 studies (including those with an inappropriate design, with unavailable data or without heparin as a control group) were subsequently excluded. Finally, 5 published studies19–23 fulfilled the inclusion criteria for the meta-analysis. Figure 1 illustrates the strategy and algorithm for study selection.

PRISMA flow diagram for study design and literature search.
Literature Characteristics and Quality Assessment
A total of 1360 hospitalized patients with COVID-19 (composing 5 trials) were enrolled in the meta-analysis, and the majority of them were in mild to moderate condition. Among the trials, 2 studies19,22 were retrospectively performed, and 3 studies20,21,23 were RCTs. They were carried out in 5 different countries, namely, Spain, India, Brazil, the USA and Egypt. All of them were published as full-text manuscripts from 2020 to 2022. Tables 1 and 2 show the baseline characteristics and primary outcome data of the 5 studies, respectively.
Characteristics of Literatures.

Abbreviations: RCT, randomized controlled trial; RETRO, retrospective study; Qd, once a day; Bid, twice a day; Q12h, every 12 h; T, trial group; C, control group; LMWH, low-molecular-weight heparin; UFH, unfractionated heparin; NOS, Newcastle-Ottawa scale.
Raw Data of Each Included Study.
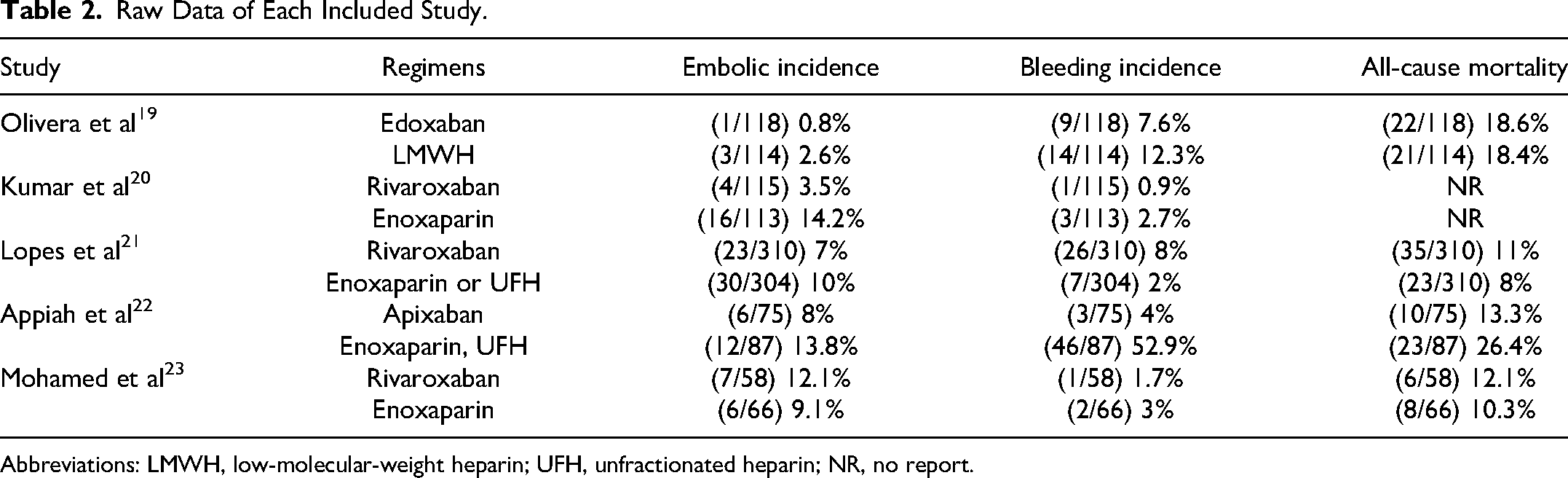
Abbreviations: LMWH, low-molecular-weight heparin; UFH, unfractionated heparin; NR, no report.
Embolism Incidence
Five studies19–23 including 1360 patients were included in the analysis. Among these patients, 108 individuals experienced thromboembolic events: 41 in the DOAC group and 67 in the heparin group. Low significant statistical heterogeneity among trials was detected using Cochrane's Q test (df = 4, I2 = 31.4%, P = 0.212). The results showed that DOACs afforded a lower embolism incidence than did heparin for anticoagulation in the included hospitalized patients with COVID-19, which reached statistical significance (RR = 0.63, 95% CI [0.43-0.91], P = 0.014) with a fixed model. The forest plots are shown in Figure 2.

Forest plot of embolism incidence.
Bleeding Incidence
Five studies19–23 reported the incidence of bleeding, including major and clinically relevant nonmajor bleeding, after preventive treatment of anticoagulation in hospitalized patients with COVID-19. The percentage of bleeding events in the DOAC group ranged from 0.9% to 8.0%, and that in the heparin group ranged from 2.0% to 52.9%. High heterogeneity was observed among the included studies (df = 4, I2 = 87.6%, P < 0.001). Nevertheless, we selected the random-effects model to pool the effect sizes. The analytical findings (Figure 3) showed no obvious significant difference between DOACs and heparin regarding bleeding incidence, but the former drug type was noninferior to the latter, with a smaller bleeding trend (RR = 0.52, 95% CI [0.11-2.44], P = 0.411).
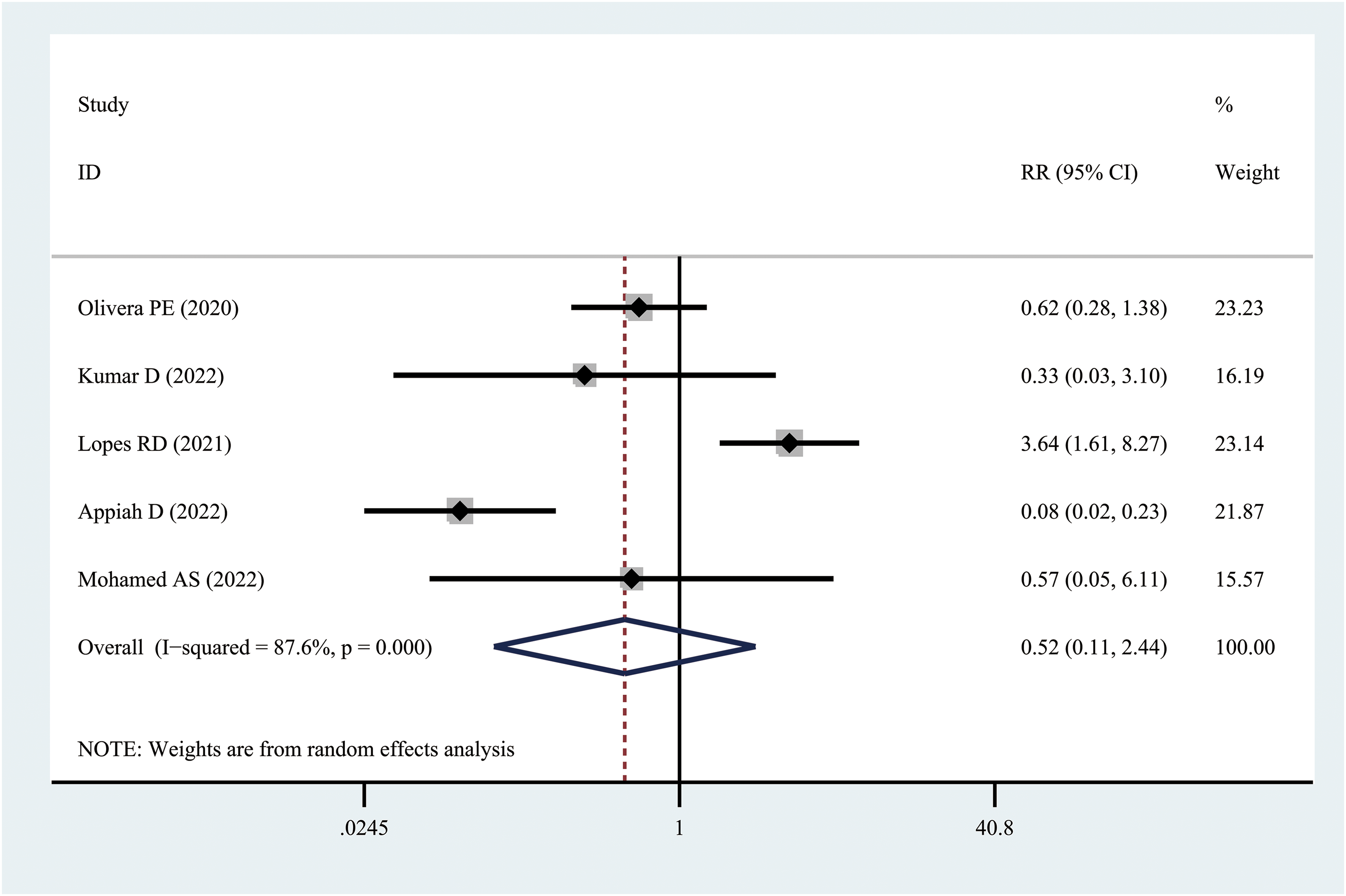
Forest plot of bleeding incidence.
All-Cause Mortality
Four studies19,21–23 consisting of 1132 patients were included, and 148 patients died during these trials. The mean mortality rates in the DOAC group and heparin group were approximately 13.0% and 13.1%, respectively. When we examined the 2 groups using Cochrane's Q test, moderate heterogeneity (df = 3, I2 = 53.8%, P = 0.09) was identified among the 4 studies. Therefore, we selected the random-effects model to pool the effect sizes. We observed that DOACs and heparin exhibited similar rates of mortality for hospitalized patients with COVID-19 (RR = 0.94, 95% CI [0.59-1.51], P = 0.797) (Figure 4).

Forest plot of all-cause mortality.
Sensitivity Analysis
As depicted by the forest plots, high heterogeneity (I2 = 87.6%) was determined for bleeding incidence. By reviewing the trials again, we noticed in one study 21 that 30 of 39 patients with clinically unstable conditions were assigned to the DOAC group before their first dose of study medication, which was significantly more than 9 of 39 in the heparin group. Therefore, we excluded that study and reanalyzed the bleeding incidence. The heterogeneity decreased from 87.6% to 71.2%, and the results also showed that DOACs were noninferior to heparin, with a corresponding RR of 0.29 (95% CI [0.08-1.10], P = 0.069).
Publication Bias
The embolism incidence was used to examine publication bias. The P value for Egger's test was 0.514 (P > 0.05), confirming the stability of our results with no significant publication bias.
Discussion
Prophylactic dosages of anticoagulant treatment may be more suitable for patients with COVID-19. 24 Traditionally, anticoagulant use, mainly heparin, reduced all-cause mortality in COVID-19 patients during hospitalization. 12 However, compared with prophylactic anticoagulation, therapeutic anticoagulation in hospitalized COVID-19 patients not only was not associated with a reduction in in-hospital mortality but also increased the risk of major bleeding.25–29 Especially in noncritically ill COVID-19 patients, the use of prophylactic dosages of anticoagulant is probably preferred due to the higher risk of bleeding at therapeutic doses. 12
Recently, although heparin, especially LMWH, seemed to perform well in anticoagulation and has been recommended in hospitalized COVID-19 patients.17,30–34 However, early evidence demonstrates that COVID-19 patients are at high risk for thrombosis, even those receiving standard or intensified thromboprophylaxis doses with LMWH or UFH. 35 In addition, heparin tolerance or resistance has become an ongoing problem in anticoagulation therapy for COVID-19, which makes it more difficult to monitor.36,37 Moreover, it cannot be ignored that the use of heparin for anticoagulation prophylaxis in hospitalized COVID-19 patients may place the patient at risk of developing heparin-induced thrombocytopenia,38–40 a potentially fatal complication that may combine with the thrombocytopenia syndrome caused by the COVID-19 vaccine. 41
Instead, the use of DOACs may ameliorate the situation. 42 With direct, selective, and reversible inhibitory effects, DOACs are an emerging alternative for the treatment of thromboembolic disease. 43 Currently, rivaroxaban is the most commonly used DOAC, followed by dabigatran and apixaban. A cost‒benefit analysis 44 showed that the treatment with DOACs resulted in fewer deaths due to bleeding, with 5 for dabigatran, 7 for apixaban, 7 for rivaroxaban, 9 for LMWH, and 15 for warfarin, per 10,000 patients. It was also demonstrated that DOACs incur lower costs than LMWH in prevention and treatment. Due to their well-known pharmacokinetic and pharmacodynamic characteristics, 43 DOACs exhibit many advantages compared with LMWH and VKA, including favorable safety and tolerance, which has gradually made them the preferred drugs for atrial fibrillation and venous thromboembolism treatment gradually. Consequently, they need to be evaluated for efficacy and safety in COVID-19 patients compared with the standard prophylactic treatment of heparin.
Toubasi's findings 45 suggested that previous use of DOACs reduced the mortality and severity among patients with COVID-19. It also substantiated the benefits of DOAC use in improving the outcomes of several diseases. In contrast, a meta-analysis by Dai et al 46 showed that the use of DOACs was not associated with a reduced risk of mortality in patients with COVID-19. Thus, controversy exists, and there is not enough evidence to prove either position. In addition, an analysis of thromboembolic events is needed to demonstrate the efficacy in more detail.
Our meta-analysis included hospitalized patients with COVID-19 from 5 different countries, most of whom were in noncritically ill conditions. In the intervention group of 676 patients, rivaroxaban, edoxaban, and apixaban were mainly used. In the control group of 684 patients, most were treated with enoxaparin, a kind of LMWH. On this basis, 2 studies partially added UFH. The results of our study demonstrated that DOACs had a much better capacity to prevent thromboembolic events than heparin, mainly LMWH. Considering the cost and benefit, it seems that DOAC anticoagulation is sufficient for noncritically ill patients with COVID-19.
Regarding safety, bleeding is one of the main adverse events in the anticoagulation process. It is well known that exposure to high doses of anticoagulant could lead to the occurrence of bleeding events, often resulting in a fatal outcome. Although the results of the present meta-analysis revealed no significant difference in bleeding incidence after preventive treatment in hospitalized patients with COVID-19, DOACs exhibited a better trend in the low-bleeding risk group than in the heparin group. Sensitivity analysis showed that after excluding one of the studies, the high heterogeneity (I2 = 87.6%) was decreased to a medium heterogeneity (I2 = 71.2%) at most, and there was no obvious change in the results. Coupled with convenience and no monitoring, DOACs seem to be a better option.
Finally, our studies have analyzed the all-cause mortality rate as the outcome in COVID-19 patients in relation to DOACs as an intervention of concern. The results demonstrate that, compared with heparin, DOACs had a similar influence on the mortality of COVID-19 patients receiving in-hospital preventive anticoagulant treatment. Because few critically ill patients were included, this may be related to the extent of COVID-19.
However, some limitations exist in our study. First, we found only 5 studies—2 retrospective studies and 3 RCTs. The data may not be sufficient to determine the efficacy and safety of DOAC preventive treatment, as additional RCTs are needed. Second, although all studies were conducted during hospitalization, more studies with the same time cycle need to be included to maintain their consistency and minimize treatment bias. Third, there is no comparative study of dabigatran and heparin in patients with COVID-19. Therefore, it is uncertain whether such data on dabigatran would be consistent with our research results. Finally, due to the limited available data in the included studies, we cannot make further comparative analysis in other aspects, including drug interactions, virus load, and inflammatory index.
Conclusion
Our findings suggest that among noncritically hospitalized patients with COVID-19, DOACs are superior to heparin, mainly LMWH, in the prevention of thromboembolism. Compared with heparin, DOACs have a lower trend of bleeding and similar mortality in mild to moderate patients. It seems that simple DOAC treatments are sufficient for these patients. In the future, more high-quality RCTs comparing DOACs (including dabigatran) with heparin are needed to verify this hypothesis in more aspects.
Supplemental Material
sj-jpg-1-cat-10.1177_10760296231164355 - Supplemental material for Efficacy and Safety of Direct Oral Anticoagulants Compared With Heparin for Preventing Thromboembolism in Hospitalized Patients With COVID-19: A Systematic Review and Meta-Analysis
Supplemental material, sj-jpg-1-cat-10.1177_10760296231164355 for Efficacy and Safety of Direct Oral Anticoagulants Compared With Heparin for Preventing Thromboembolism in Hospitalized Patients With COVID-19: A Systematic Review and Meta-Analysis by Yong Tian, MD, Tao Pan, Xudong Wen, Guangyu Ao, Yihan Ma, Xiaoxiao Liu, Rui Liu, and Hongmei Ran in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Acknowledgments
Thanks for the language editing service provided by Springer Nature.
Authors’ Note
YT designed the study, selected literature, collected and analyzed data, and drafted the manuscript. TP, XW, and YM collected and assembled the data. GA and XW contributed to interpreting the data and drafting the manuscript. XL and RL literature search and data analysis. HR contributed to study design, data interpretation, and finalizing the manuscript; all authors read and approved the final manuscript. All relevant data are presented in the review and supplemental material.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
