Abstract
Introduction
Hemorrhagic early death (HED) remains a major cause of treatment failure among patients with acute promyelocytic leukemia (APL). We aimed to investigate the prognostic potential of rotational thromboelastometry (ROTEM) for bleeding in patients with APL.
Materials and Methods
31 newly-diagnosed APL patients (median age of 40 years; 14 female/17 male) that underwent treatment at the Clinic of Hematology UCCS from 2016-2020 with all-trans retinoic acid and anthracyclines were recruited. CBCs (complete blood count), conventional coagulation tests (CCTs), and ROTEM parameters obtained before treatment initiation were evaluated.
Results
All patients demonstrated at least one ROTEM parameter out of the reference range. ROTEM parameters associated with significant hemorrhage were EXTEM clotting time (CT) (P = 0.041) and INTEM amplitude 10 (A10) (P = 0.039), however, only EXTEM CT (P = 0.036) was associated with HED. Among CBCs and CCTs, only platelets were associated with significant bleeding (P = 0.015), while D-dimer was associated with both bleeding and HED (P = 0.001 and P = 0.002, respectively).
Conclusion
Our results indicate that ROTEM parameters may reveal hypocoagulability in APL patients and have the potential to improve current hemorrhage prognostic methods. Additionally, these results suggest the combination of ROTEM and CCTs might be useful in identifying patients at risk for HED.
Keywords
Introduction
In the era of all-trans retinoic acid (ATRA) and arsenic trioxide (ATO), acute promyelocytic leukemia (APL) has become the most curable subtype of acute myeloid leukemia with a complete remission rate of > 90%.1–4 However, despite advancements in patient management, severe adverse hematologic outcomes, such as hemorrhagic early death (HED), continue to pose significant risks. According to data, early death rate varies between 17 to 30%, with severe hemorrhage as a cause in 5% in controlled clinical trials to 41% in the real world setting.3–5 Furthermore, previous studies have suggested that certain distinctive patterns of coagulopathy seen in APL patients may lead to potentially fatal bleeding events.4,5 Thus, preventing hemorrhage in APL may prove to be crucial in reducing the morbidity and mortality of APL patients.
Although numerous baseline parameters, such as elevated white blood cell count (WBC), poor performance status (PS), high serum lactate dehydrogenase level (LDH), prolonged prothrombin (PT), activated partial thromboplastin times (APTT), decreased fibrinogen concentration, elevated D-dimer, and a high International Society on Thrombosis and Hemostasis (ISTH) DIC score were previously observed as prognostic for both bleeding and HED in APL, none have been found to be reliably and consistently predictive.3,6–9
Recently, rotational thromboelastometry (ROTEM) has been found to be useful in predicting mortality and bleeding risk as well as guiding transfusion therapy in critically ill patients, such as patients with DIC, trauma, liver transplant, or surgery.10–14 However, despite increasing interest for the use of ROTEM in hematological disorders, studies investigating the utility of ROTEM in patients with acute leukemia are lacking and, based on our search, nonexistent regarding the utility of ROTEM in APL patients specifically.
Consequently, the aims of this study were to assess ROTEM pattern in APL patients, correlations between ROTEM, complete blood counts (CBCs), and conventional coagulation tests (CCTs) in patients with APL, and also to identify ROTEM parameters that are potentially associated with both bleeding and HED.
Patients and Methods
Patients and APL Diagnostic Procedures
After obtaining written informed consent, thirty-one adults diagnosed with APL were recruited at the Clinic of Hematology UCCS between January 2016 and May 2020. Morphological diagnosis was made according to the WHO 2016 revision of classification of myeloid neoplasms and acute leukemia. 15 Conventional G-band karyotyping was employed for cytogenetic analysis. 16 The PML-RARA fusion transcript was detected using reverse transcriptase polymerase chain reaction (RT-PCR). 17 Immunophenotyping was performed by direct multicolor immunofluorescence applied to the whole bone marrow specimen. 18 All procedures and protocols were approved by an accredited ethics committee and institutional review board.
Clinical and Treatment Data
ED was defined as death from any causes that occurred up to 30 days after hospitalization and initiation of therapy. 1 Patients were monitored for bleeding episodes from the date of admission until the last day of hospitalization with a median follow up of 27 days (range 0−42). Bleeding events were graded according to the modified World Health Organization (WHO) grading system.19,20 Bleeding grades III and IV, defined as bleeding associated with moderate hemodynamic instability requiring red blood cell transfusion and bleeding associated with severe hemodynamic instability, fatal bleeding, or CNS bleeding, were classified as severe.19,20 All patients were treated in accordance with the PETHEMA APL 2012 protocol. 21 Transfusions of platelet concentrates were provided at platelet count of < 50 × 109/L, while fresh-frozen plasma (FFP) and cryoprecipitate were transfused at a fibrinogen level of < 1.5 g/L. These supportive measures were provided until correction of coagulopathy, previously defined as normalization of fibrinogen and PT and bleeding cessation. 22
Laboratory Assays
APTT, PT, and fibrinogen levels were measured via Automated Instrumentation Laboratory (ACL, Werfen, Italy). D-dimer was determined immuno-turbidimetrically with the Siemens BCS XP System (Siemens, Germany). The referenced normal ranges for PT, APTT, fibrinogen, and D-dimer are listed in Table 1. DIC was diagnosed according to the International Society on Thrombosis and Haemostasis scoring system. 23
Patient CCT and ROTEM Parameters.

Abbreviations: A10, amplitude 10; APTT, activated thromboplastin time; CCT, conventional coagulation tests; CT, clotting time; CFT, clot forming time; MCF, maximum clot firmness; ML, maximum lysis, PT, prothrombin time; ROTEM, rotational thromboelastometry; WBC, white blood cell count.
ROTEM delta (TEM Systems Inc., Munich, Germany) was performed by a trained physician. Whole blood samples were drawn into citrate tubes (BD-Plymouth, PL6 7BP, UK) and processed within 1 h of collection at 37 °C. Four ROTEM standard assays were performed for each patient: EXTEM, INTEM, APTEM, and FIBTEM and the following parameters recorded: clotting time (CT), clot formation time (CFT), maximum clot firmness (MCF), amplitude 10 (A10), and maximum lysis (ML). EXTEM assays were conducted with 0.2 mol/L calcium chloride (STARTEM), recombinant tissue factor, phospholipids, and 300 µL of blood. INTEM assays were conducted with 0.2 mol/L calcium chloride and addition of ellagic acid and phospholipids. APTEM tests were performed in the presence of 20 µL of the antifibrinolytic tranexamic acid. Mild activation with tissue factor was used to standardize the coagulation process in vitro and produce a more rapid result. FIBTEM assays consisted of a modified EXTEM assay with the addition of a potent platelet inhibitor, cytochalasin D, which functioned to block platelet activation, shape change, and expression and activation of glycoprotein IIb/IIIa receptors. 24 Reference ranges for ROTEM parameters are shown in Table 1. Hypocoagulability was diagnosed if > 1 parameter was out of normal range, defined as prolonged CT, prolonged CFT, decreased A10 or decreased MCF in any ROTEM assay.
Statistical Analyses
Depending on the type of variable and the normality of the distribution, results are presented as proportion of patients (%), median, and mean ± SD. In order to test statistical hypotheses, we used the t-test, Mann-Whitney test, chi-square test, and Fisher’s exact test where appropriate. The correlations between variables were estimated using Spearman's correlation coefficient. We used false discovery rate (FDR) method to adjust P-values for multiple comparisons. 25 Statistical hypotheses were analyzed at a significance level of 0.05.
Statistical data analysis was performed using IBM SPSS Statistics 22 (IBM Corporation, NY, USA), Software package of R software environment (R Core Team, 2021).
Results
Patient Characteristics and Treatment Course
Thirty-one patients were enrolled in the study, 17 males (55%) and 14 females (45%), with a median age of 40 years (range 18 − 73). Median follow-up, defined as the time from admission until the last day of hospitalization, was 27 days (range 0 − 42). During the follow-up period, bleeding events were observed in all patients, with 9/31 (29%) having experienced severe hemorrhage. ED occurred in 5/31 patients (16.1%), and the only cause of death was intracranial hemorrhage (ICH). According to ISTH, 26/31 (83.8%) of patients met criteria for DIC (ISTH DIC score ≥ 5).
Conventional Coagulation Tests and ROTEM Parameters
Low platelet counts and increased D-dimer were observed in all patients. PT and APTT were elevated in 83.9% and 38.7% of patients, respectively. However, only 35.5% of patients had decreased fibrinogen level. Among ROTEM parameters, prolonged CT was observed in a majority of patients in EXTEM (93.5%) and in all patients in FIBTEM (100%), while only 45.5% had prolonged CT in INTEM. Similar to CT, prolonged CFT was seen in 89.5% and 92.9% of study population in EXTEM and INTEM, respectively. Very poor clot stability and firmness were observed among EXTEM and INTEM parameters. Shorter than normal EXTEM MCF and A10 were observed in 96.8% and 93.3% of patients, respectively. Decrement of INTEM MCF and A10 was observed in 100% and 90.9% of patients, respectively. FIBTEM demonstrated reduced MCF and A10 in 62.1% and 63.3% of patients. The results of CCTs and ROTEM parameters at baseline are shown in Table 1.
Correlations
Significant positive correlations were found between (1) EXTEM CT and WBC (P = 0.007, ρ = 0.57), D-dimer (P = 0.016, ρ = 0.46), and ISTH DIC score (P = 0.021, ρ = 0.51); (2) FIBTEM CT and WBC (P = 0.037, ρ = 0.43); and (3) INTEM MCF and platelet count (P = 0.007, ρ = 0.65). Moreover, significant negative correlations were observed between: (1) EXTEM MCF and WBC (P = 0.017, ρ = −0.49), (2) INTEM, FIBTEM CT and fibrinogen levels (P = 0.031, ρ = −0.54; P = 0.028, ρ = −0.50, respectively), (3) EXTEM CT and PT (P = 0.014, ρ = −0.54) and (4) EXTEM, INTEM, and FIBTEM MCF and D-dimer (P = 0.007, ρ = −0.53; P = 0.016, ρ = −0.54; P = 0.007, ρ = −0.60, respectively). There were no correlations between APTT and any ROTEM parameter, as shown in Table 2.
Correlations between ROTEM, CBCs and CCTs.

Abbreviations: A10, amplitude 10; APTT, activated thromboplastin time; CBC, complete blood count; CCT, conventional coagulation tests; CT, clotting time; CFT, clot forming time; MCF, maximum clot firmness; ML, maximum lysis, PT, prothrombin time; WBC, white blood cell count; ρ, Spearman's rank correlation coefficient.
*P < 0.05.
Bleeding Events, HED, and Risk Factors
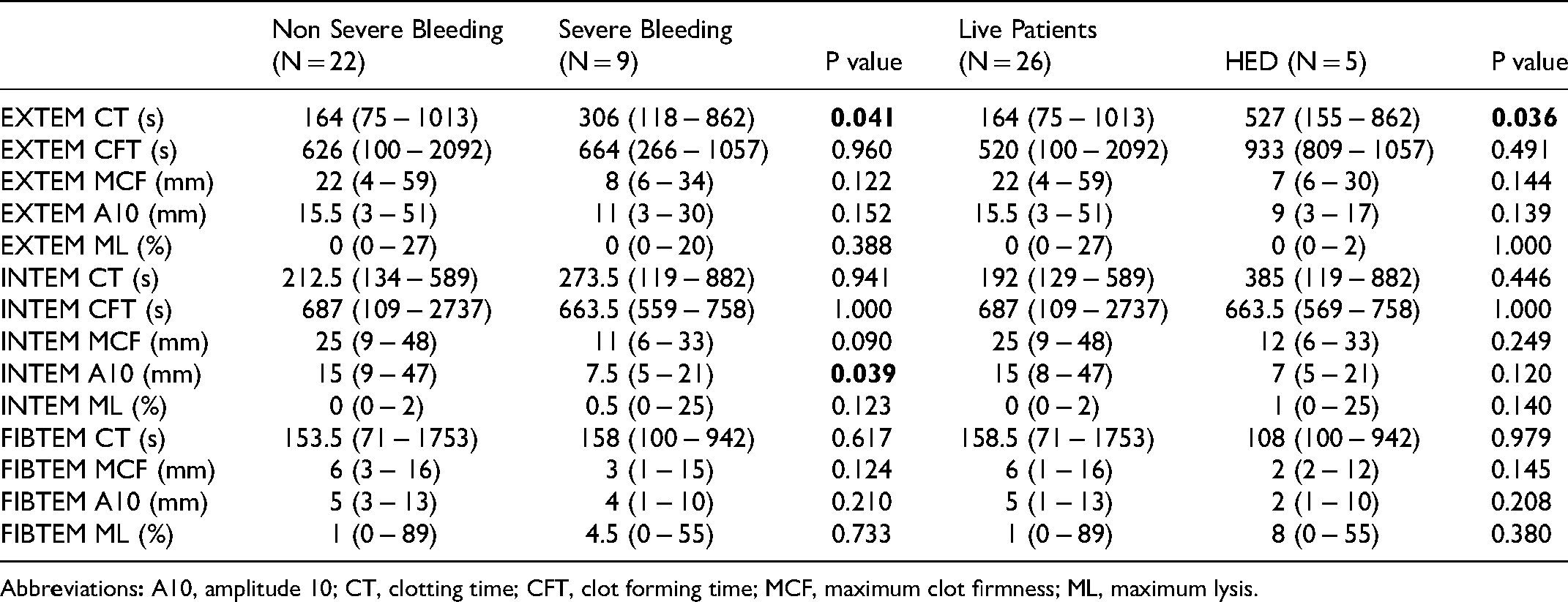
Among CBCs and CCTs, only platelet counts (P = 0.015) and D-dimer (P = 0.001) were associated with severe hemorrhage. However, among ROTEM parameters, EXTEM CT (P = 0.041) and INTEM A10 (0.039) were associated with severe bleeding.
Analysis also found that, among all CCTs, higher D-dimer levels and prolonged EXTEM CT were significantly associated with death (P = 0.002 and 0.036, respectively). A comparison of CCT values and ROTEM parameters for patients with and without severe bleeding, as well as those with HED, is shown in Tables 3 and 4.
Differences between Patients with Clinically Non-Significant and Significant Hemorrhage, and Patients with HED and Alive Patients.

Abbreviations: APTT, activated thromboplastin time; PT, prothrombin time; WBC, white blood cell count.
ROTEM Parameters in Patients with Clinically Non-Significant and Significant Hemorrhage, and Patients with HED and Alive Patients.
Abbr
Discussion
Persistence of a high HED rate despite improved specific and supportive APL therapy has led to attempts to identify predictors of hemorrhage leading to early death. Although numerous parameters were previously observed as prognostic for bleeding/HED, none of them were consistently predictive.
In our study, all APL patients’ blood samples expressed ROTEM patterns of hypocoagulability. More specifically, prolonged EXTEM and FIBTEM CT, both of which were observed in the majority of tested blood samples, suggested inadequate initiation of coagulation due to thrombocytopenia, DIC, and fibrinolysis. 26 Furthermore, the decreased MCF values observed via EXTEM, INTEM and FIBTEM in the majority of APL patients’ blood samples implied poor clot stability. Additionally, a positive correlation between WBC and CT in EXTEM/FIBTEM and a negative correlation between WBC and EXTEM MCF were observed. These results suggest that, in high-risk APL patients with elevated WBC, clot initiation is delayed (decreased EXTEM/FIBTEM CT) and the eventually-formed clot is inadequate and weak (decreased EXTEM MCF), likely contributing to an increased bleeding susceptibility.
In the studied cohort, fibrinogen levels were below the normal range in 35.5% of APL patients’ blood samples while FIBTEM CT was prolonged in all tested blood samples. This observation suggests FIBTEM is more reflective of actual functional fibrinogen than standalone fibrinogen levels. Nonetheless, despite varying CCTs and ROTEM parameter correlations being reported in both the current study and previous studies of patients undergoing liver transplantation, critically ill patients, or children undergoing surgery, the aforementioned correlations have not been consistently demonstrated, suggesting that CCTs and ROTEM cannot be used interchangeably, a notion corroborated by previous groups.10,12,27,28
Our study found that, among ROTEM parameters, EXTEM CT and INTEM A10 were associated with hemorrhage. Furthermore, EXTEM CT was also the only ROTEM parameter associated with HED. Despite this, studies addressing the association between thromboelastometry and bleeding in patients with hematological malignances are lacking and consistent results have yet to be demonstrated. For example, in their study of 50 patients with acute leukemia, lymphoma, myeloma, and other hematological malignances, Estcourt et al found a significant association between low EXTEM and INTEM MCF and a WHO bleeding grade of ≥ 2. However, this association was no longer observed after adjustment for total platelet count. 29 In another study, Russell et al investigated the ability of thromboelastography (TEG) to predict bleeding in patients with hematological malignancies and sepsis, however, none of the TEG parameters were found to be significantly predictive. 30
Thus, treatment in the form of prophylactic platelet transfusion remains a common practice in patients with hematological malignancies and thrombocytopenia performed with the intention of preventing bleeding events. 31 However, platelet count cut-offs for transfusion are largely arbitrary and, expectedly, outcomes are variable and difficult to predict. 32 Indeed, a recent study largely focusing on AML showed a significant decrease in EXTEM CFT and an increase in EXTEM MCF 24hours post-platelet transfusion. 33 Moreover, a study conducted by Akay et al showed no changes in magnitude of ROTEM response after transfusion of either “high” or “low” dose platelet transfusions, suggesting that ROTEM could be a promising tool in reducing the number of a prophylactic platelet transfusion and the associated potential adverse effects and financial expenditures. 34 Even though our study was not designed to determine a ROTEM cut-off values that could be used as transfusions thresholds, for example, if the cut-off values of FIBTEM MCF (< 10 mm) and A10 (< 5 mm) ROTEM parameters for the indication of cryoprecipitate transfusion in trauma are applied to our APL patient population sample, we hypothesize that almost 60% of them could have received a cryoprecipitate or FC transfusion that would be potentially lifesaving. However, by using a transfusion threshold of fibrinogen <1.5 g/L as a current guide, only 35% of our patients received cryoprecipitate or FC transfusion. Thus, ROTEM-guided transfusion in patients with APL could possibly lead to a reduction in the rates of severe bleeding and HED, but further well designed studies would be needed.
Although several studies have emphasized fibrinolysis as a major cause of coagulopathy in APL, the therapeutic use of tranexamic acid remains controversial.4,22,26,35 Similarly, hyperfibrinolysis, usually defined as EXTEM ML > 15% of the MCF, can be identified by ROTEM, however, in our study, only two patients demonstrated an ongoing hyperfibrinolytic process. 10 Overall, these data could support the clinical usefulness of ROTEM in identifying patients who may be candidates for antifibrinolytic treatment, however, further studies are needed to further elucidate the extent of this potential interaction.
This study has several limitations. Due to the low incidence of APL, number of patients observed in defined period of time was consequently low. This low sample size limits the applicability and generalizability of the results. Furthermore, we could not perform a multivariate analysis and so far, couldn't define ROTEM predictors for HED. Even though, it would be interesting and useful to define cut-off values of ROTEM parameters for prophylactic transfusion in APL patients, this was not feasible with such a small sample size and this study design. Despite limitations, we believe that our results may help in understanding APL coagulopathy, and given the limited number of previously published studies, we truly believe that our data may help guide further studies involving larger sample sizes for the elucidation of ROTEM's role in detecting patients with increased risk of HED and severe bleeding.
Conclusion
We found that patients with APL have abnormal ROTEM parameters that are reflective of abnormal coagulation in regard to platelet count and function, fibrinogen level and function, and fibrinolytic activity. Based on its values in patients with severe bleeding and HED, as well as its correlations with WBC and DIC score, EXTEM CT has a potential value for aiding in the identification of patients at increased risk for fatal hemorrhage at initial diagnosis of APL and prior to initiation of APL treatment. More importantly, ROTEM could be used for timely evaluation of hemostasis and management of APL-specific coagulopathy, both of which may allow for an increased survival rate throughout the critical initiation period of APL therapy. Nonetheless, future prospective studies with a larger number of patients are needed to validate and verify the utilization of ROTEM in patients with APL, determine cut off values for APL-specific coagulopathy management with blood products, and confirm the predictive correlations between ROTEM parameters and HED.
Footnotes
Acknowledgments
We would like to acknowledge Nada Miscevic (NMScientific Inc., Toronto, Canada) for helping in the preparation of this manuscript and for providing critical review and helping in manuscript drafting. We also express our thanks to all personnel at Clinic for Vascular Surgery, Clinical Centre of Serbia, for performing ROTEM analysis. Finally, we wish to acknowledge the support of the personnel of the hematology clinic during this study.
Authors’ Approval
All authors read and approved the final version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
All procedures performed in this study involving human participants were approved by Ethical committee of Clinic of Hematology, Clinical Center of Serbia (protocol number III 41004).
Consent to Participate
Informed consent was obtained from all individual participants included in the study.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
