Abstract
A variety of antithrombotic drugs are used during percutaneous coronary interventions (PCIs). We aimed to investigate the practicability of the use of bivalirudin and GPIs in patients receiving PCI. We searched 7 of 629 relevant records from PubMed, the Cochrane Library, EMBASE, and Web of Science for randomised controlled trials. There were no significant differences in the rates of major adverse cardiac events (MACE) between bivalirudin plus GPI and heparin (all P > .05). Bivalirudin plus planned GPI was similar to bivalirudin monotherapy in terms of the risk of MACE (risk ratio [RR] = 1.07; 95% confidence interval [CI] = .91 − 1.27; P = .55). Bivalirudin plus provisional GPI was associated with lower bleeding risk (RR = .57; 95% CI = .47 − .69; P < .01) compared to using heparin plus GPI. Compared to bivalirudin alone, bivalirudin plus planned GPI evidently increased bleeding risk (RR = 2.20; 95% CI = 1.73 − 2.79; P < .01). Patients receiving bivalirudin or heparin therapy had semblable efficacy endpoints, but those receiving bivalirudin had a significantly lower bleeding risk. For high-risk bleeding patients, bivalirudin plus provisional GPI can have a better antithrombotic effect than heparin, without increasing the bleeding risk.
Keywords
Introduction
Bivalirudin and glycoprotein IIb/IIIa inhibitors (GPIs) are powerful antithrombotic that play a significant role in percutaneous coronary interventions (PCI). Some studies have shown that bivalirudin with or without GPI is superior to heparin in terms of anticoagulant efficacy; furthermore, heparin causes an increase in the rate of massive bleeding and thrombocytopenia.1,2 However, other studies have shown no difference in the occurrence of major adverse cardiovascular events (MACE) between these agents and have suggested that a combination therapy of bivalirudin and GPI increases the risk of bleeding.3,4
Although the efficacy of P2Y12 inhibitors is improving considerably, patients undergoing primary PCI require the best possible anticoagulation strategy due to the short duration of receiving the medications and the high risk of thrombosis. In comparison with heparin, bivalirudin is advantageous to a certain extent as it is non-inferior or even superior in terms of the occurrence of MACE.5,6 Previous trials had many treatment options and complex grouping, and most meta-analyses have compared the outcomes of bivalirudin and that of heparin.7–9 However, the difference between the use of bivalirudin plus planned GPI and the provisional use of GPI remains unclear.10,11
The study investigated the practicability of bivalirudin plus a GPI use in PCI. We considered that the administration of heparin alone was not sufficient as a control intervention, we planned to compare bivalirudin plus a GPI and bivalirudin monotherapy or heparin with GPI.
Materials and Methods
This review followed the recommendations of the Preferred Reporting Items for Systematic Reviews and Meta-analysis (PRISMA) statement. 12 The protocol was registered in PROSPERO as CRD42021251303.
Data Sources
Two researchers independently searched for articles published in the PubMed, Cochrane Library, EMBASE, and Web of Science databases between January 2000 and May 2021 using the following search terms: “randomised controlled trial‘, “bivalirudin‘, “glycoprotein IIb/IIIa‘, “percutaneous coronary intervention‘, “ST-elevation myocardial infarction‘, and “non-ST-elevation myocardial infarction‘. Additionally, researchers searched for other relevant trials that were not contained in the above-mentioned databases.
Selection Criteria
The following selection criteria were used: (1) studies involving patients undergoing PCI in a hospital setting; (2) randomised controlled trial (RCT) of bivalirudin plus planned or provisional GPI use; (3) studies that reported clinical outcomes and had follow-up durations of at least 30 days; and (4) articles published as systematic reviews, meta-analyses, and comments along with studies that had a small sample size (n < 50) were excluded.
Data Assessment
Two authors (S.L. and D.L.) independently extracted relevant information. Disagreements were discussed by the two authors or a third author (C.L.) assessed the study. The following specific data were retrieved from the articles: personal characteristics, efficacy endpoints, safety endpoints, and adverse events. Major bleeding was regarded as a primary safety endpoint within 30 days. The primary efficacy endpoint was the occurrence of a compound event of MACE within 30 days (including death, myocardial infarction, revascularisation, and stent thrombosis), Secondary efficacy endpoints were individual events, such as death, myocardial infarction, revascularisation, and stent thrombosis including acute and subacute).
Two authors independently reviewed the eligibility and quality of the studies obtained. Differences were resolved by two authors, or a senior researcher performed the assessment. According to the Cochrane Handbook, researchers assessed the risk of bias, which is classified as low quality, high quality, or unclear, and the evaluation criteria included the following items: random sequence generation, allocation concealment, blinding of participants and personnel, bleeding outcome assessment, incomplete outcome data, selective reporting, and other issues. 13
Data Analysis
The researchers used Forest Plot to present the data. P values <.05 were considered statistically significant (two-sided). The pooled estimated outcome was calculated by the fixed-effect or random-effect model, which depended on whether the heterogeneity was evident using the Cochran's Q test and the I2 test. A Cochran’s P < .10 and I2 > 50% were regarded as obvious heterogeneity. All data were calculated and analysed by RevMan 5.4 software (The Cochrane Collaboration, Copenhagen, Denmark).
Results
Baseline Characteristics
The process of article selection is presented in Figure 1. The database search identified 629 studies. After removing duplicates, 391 of the initial 476 studies were excluded upon reviewing titles and abstracts, and the remaining 85 potentially suitable studies were scrutinised further by reading the full text. Finally, seven RCTs were eligible for analysis.(Supplementary Table S1 provides detailed information on each trial, and Supplementary Figures S1 and S2 provide the risk of bias for each study)

Process of article selection included in the meta-analysis.
Overall, studies involving a total of 17 490 subjects were included in this review. Specifically, 3169 patients received bivalirudin plus planned GPI, 5569 received bivalirudin plus provisional GPI, and 2779 were treated with bivalirudin alone. Furthermore, 4886 patients were diagnosed with ST-elevation myocardial infarction (STEMI) and were enrolled for further analysis. Three studies14–16 compared bivalirudin plus planned GPI versus heparin plus GPI; although an additional trial 17 had met the inclusion criteria during screening, it was finally excluded owing to its low quality. Four studies compared bivalirudin plus provisional GPI versus heparin plus GPI2,18–20 and two studies14,16 compared bivalirudin plus planned GPI versus bivalirudin monotherapy. Regarding outcomes, all trials included death, myocardial infarction, revascularisation, and major bleeding, either as composite or individual endpoints. All study protocols recommended a loading dose of P2Y12 inhibitors and aspirin before PCI. Clopidogrel dose was 75 mg once daily and aspirin was 100 mg once daily after the PCI. Dual antiplatelet therapy was implemented for at least 30 days.
Clinical Results
The incidence of MACE was similar between the bivalirudin plus GPI and the heparin plus GPI groups (RR = 1.06; 95% CI = .95 − 1.18; P = .27). Similarly, no differences were observed in the myocardial infarction and revascularisation rates between interventions. Although the overall mortality analysis showed no significant differences between the groups, a subgroup analysis of the data revealed a lower death rate in the bivalirudin plus provisional GPI group than in the heparin plus GPI group (RR = .69; 95% CI = .49 − .96; P = .03; Figures 2, 3, 4, and 5).

Individual and summary risk ratios with 95% confidence intervals (Cls) of major adverse cardiac events. GPI, glycoprotein IIb/IIIa inhibitor.

Individual and summary risk ratios with 95% confidence intervals (Cls) of death. GPI, glycoprotein IIb/IIIa inhibitor.
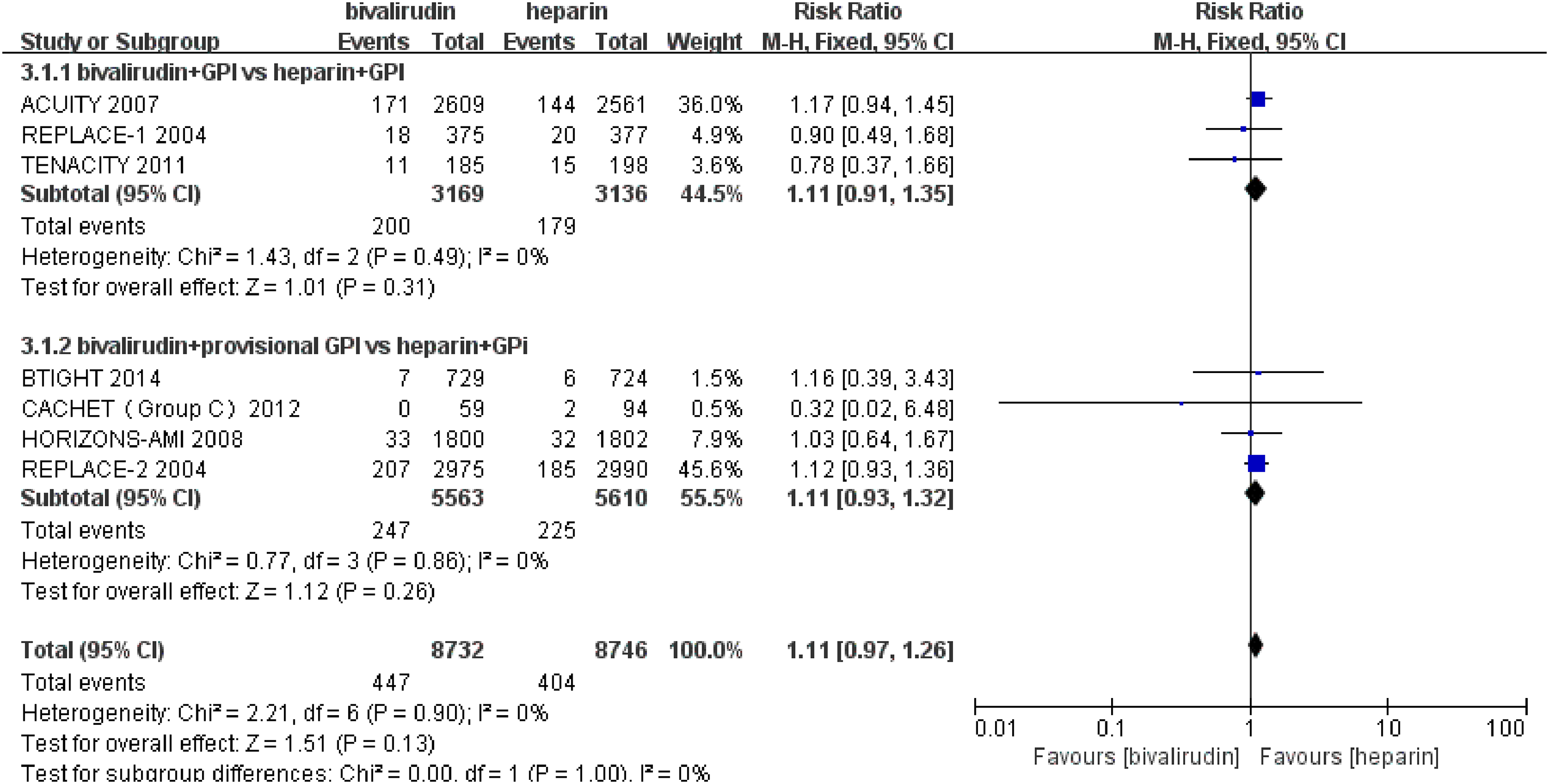
Individual and summary risk ratios with 95% confidence intervals (Cls) of myocardial infarction. GPI, glycoprotein IIb/IIIa inhibitor.
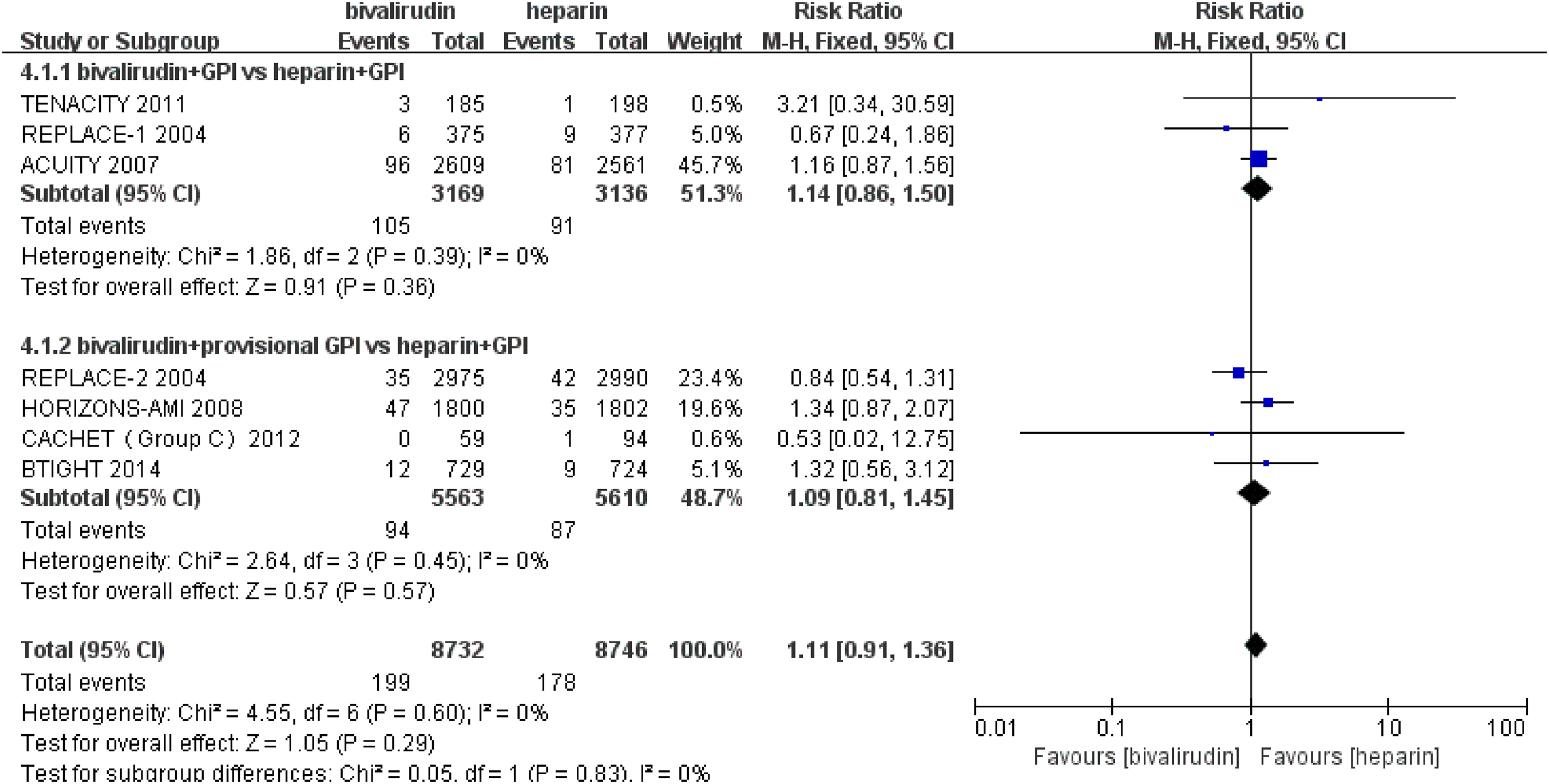
Individual and summary risk ratios with 95% confidence intervals (Cls) of revascularisation. GPI, glycoprotein IIb/IIIa inhibitor.
The incidence of major bleeding within 30 days was similar between the bivalirudin plus planned GPI and the heparin plus GPI groups (RR = 1.05; 95% CI = .76 − 1.46; P = .77). However, the incidence was lower in bivalirudin plus provisional GPI use than in heparin plus GPI (RR = .57; 95% CI = .48 − .69; P < .01). In the overall analysis, bivalirudin plus GPI was safer to use than heparin plus GPI in this regard (RR = .66; 95% CI = .45 − .98; P = .04; Figure 6).

Individual and summary risk ratios with 95% confidence intervals (Cls) of major bleeding within 30 days. GPI, glycoprotein IIb/IIIa inhibitor.
Subsequently, we compared the outcomes between bivalirudin plus planned GPI and bivalirudin alone. Both groups showed a similar risk of MACE (RR = 1.07; 95% CI = .91 − 1.27; P = .41). With respect to safety, bivalirudin alone showed a lower bleeding risk than bivalirudin plus GPI (RR = 2.20; 95% CI = 1.73 − 2.79; P < .01; Supplementary Figures S3 and S4).
Regarding STEMI, the incidence of MACE and stent thrombosis was similar between the two groups. Regarding the bleeding risk, bivalirudin plus provisional GPI was significantly safer than heparin with GPI (RR = .46; 95% CI = .27 − .80; P = .006; Supplementary Figures S5, S6, and S7).
Discussion
In this meta-analysis, we included seven RCTs that investigated the efficacy of bivalirudin combined with GPI. There was no difference in the primary and secondary endpoints between the bivalirudin plus provisional GPI and heparin plus GPI groups. Furthermore, the safety endpoint of the bivalirudin group was superior to that of the heparin plus GPI; and available results showed that although bivalirudin plus planned GPI or provisional GPI were similar in the rates of efficacy endpoints, bivalirudin plus provisional GPI use showed a lower bleeding rate at 30 days. Therefore, GPIs should be used according to the individual bleeding risk of each patient because major bleeding is a powerful predictor of death. 21
Unlike in previous meta-analyses, in this study, we carefully selected the inclusion criteria and limited the doses of bivalirudin used. 22 Some trials had relatively specific inclusion criteria, whereas others excluded STEMI and non-STEMI (NSTEMI), or patients with diabetes, which we also excluded.23,24 Previous meta-analyses focused on comparing bivalirudin with heparin and did not categorise the use of GPI explicitly;8,9 thus, we classified the use of GPI into planned and provisional.
Bivalirudin monotherapy was significantly safer than bivalirudin plus GPI in preventing major bleeding. Although bivalirudin alone possesses relatively lower rates of bleeding, this does not imply that it can replace GPIs, especially in patients with medium-high risk ACS and those with a severe thrombus load.25,26 Additionally, the ACUITY study 16 included patients without STEMI, with a severe thrombus load, and a high risk of ischaemia; therefore, these patients were more likely to benefit from GPI treatment. This may have influenced the results observed between the two groups.
Previous record demonstrated that STEMI and NSTEMI groups are semblable in the efficacy. 27 With regards to stent thrombosis, we found no significant difference in the STEMI group. However, we cannot draw definitive conclusions because only a small number of trials have studied stent thrombosis. Some records suggest that using bivalirudin for patients would bring a certain amount of stent thrombosis risk. 5 This may be explained by the fact that bivalirudin requires several infusions to achieve the maximum effect, while the infusion time and dose are shorter and smaller, respectively, for bivalirudin. 28 In the BRIGHT study, the risk of stent thrombosis was equivalent between these therapies because the bivalirudin infusion time was extended.
The recent VALIDATE-SWEDEHEART trial 29 compared bivalirudin with heparin monotherapy in patients with STEMI or those with NSTEMI who underwent PCI. The trial showed that two groups had similar major bleeding rates; therefore, it contradicts the results of some previous studies.2,30 GPIs can reduce slow flow or no-reflow, and their use is routinely considered in patients with high-risk ACS. However, this trial selected heparin monotherapy as a control group; this deserves further analysis.
This meta-analysis had some limitations. First, owing to the strict inclusion criteria, a relatively small number of comparisons were possible. Second, some trial data were insufficient. For example, the CACHET trial had a short period of observation for the safety endpoint and the REPLACE-1 trial did not provide complete endpoint details. Finally, we did not evaluate the use of P2Y12 inhibitors and the choice of surgical access and the femoral or radial artery; further studies are needed to assess these particular issues.
In conclusion, bivalirudin was not superior to heparin in terms of composite or individual MACE endpoints. Regarding safety, bivalirudin demonstrates a relatively lower risk of major bleeding compared to that of heparin. Therefore, for most patients, there is no significant difference between the outcomes of using heparin and of using bivalirudin. However, for high-risk bleeding patients, bivalirudin plus provisional GPI can have a better effect than heparin; furthermore, the timing of the use of a GPI is important.
Footnotes
Acknowledgments
We thank Professor Jia for guidance on the content of the article and Doctor Liu for her support in data analysis.
Declarations of Interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
None
