Abstract
We developed a computerized clinical decision support system (CCDSS) for venous thromboembolism (VTE) risk assessment. We aimed to demonstrate its relevance and evaluate associations between risk level and VTE incidence in patients undergoing total hip/knee arthroplasty. In this case-control study, VTE was confirmed using ultrasonography/computed tomography angiography in 1098 adults at a tertiary care hospital over five years (2013-2018). Postoperative VTE incidence was classified into three risk levels (moderate, high, and highest). The overall VTE incidence was 11.7%, which increased with a risk level of 0%, 5.8%, and 12.8% in moderate-risk, high-risk, and highest-risk patients, respectively. Highest-risk patients were significantly more likely to develop VTE than high-risk patients (odds ratio [OR] 2.4; 95% confidence interval [CI] 1.2-5.5; p = 0.01). VTE development was more likely in patients with risk scores ≥4 relative to those with risk scores of 2–3 (OR 1.8; 95% CI 1.2-2.7; p = 0.003) and −1 to 1 (OR 3.3; 95% CI 1.6-7.7; p < 0.001). This study indicates that risk level and VTE incidence are associated; our scoring system appears useful for patients undergoing total hip/knee arthroplasty.
Introduction
Venous thromboembolism (VTE), including deep vein thrombosis (DVT) and pulmonary embolism (PE), causes morbidity and mortality among patients undergoing total hip arthroplasty (THA) or total knee arthroplasty (TKA) worldwide. 1 The demand for THA and TKA is estimated to increase by 174% (572 000 patients) and 673% (3.48 million patients), respectively, from 2005 to 2030. 2 A systematic review integrating 38 studies (n = 11 769) in Asians reported a total incidence of DVT or PE of 14.2% and 0.73%, respectively, when anticoagulants were administered to prevent VTE after THA or TKA. 3 Additionally, the incidence of fatal pulmonary embolism has increased by 0.2%–6.0% over the last 30 years in Asia.4,5 VTE is associated with the risk of post-thrombotic syndrome and chronic thromboembolic pulmonary hypertension, both of which are linked to morbidity and increased management costs. 5 Studies have associated VTE management with increased total hospital costs.6–11
The American College of Chest Physicians guidelines recommend VTE risk assessment for THA or TKA patients; optimal strategies for thromboprophylaxis include pharmacologic and mechanical interventions. 1 However, while adherence to the recommendations of these guidelines among orthopedic inpatients appears to be increasing, adherence after discharge is decreasing. 12 Furthermore, only 11.8% of postoperative patients are covered by appropriate prophylactic measures in Asia. 13 Risk determination of VTE by physicians has been shown to be inaccurate. 14 The incidence of VTE is underestimated in Asian patients with thrombi due to less symptomatic outcomes relative to Western patients. Additionally, physicians’ lack of awareness of VTE and underestimation of VTE risk have led to a suboptimal implementation of guidelines and inadequate preventive management of VTE in Asia.1,15–18 A validated VTE risk assessment model may help physicians identify patients at risk. The Caprini risk assessment model includes more than 40 risk factors (1-5 scores) and stratifies patients into low, moderate to high, and highest VTE risk groups, 19 followed by VTE prophylaxis determination. The Caprini risk score is significantly and positively correlated with the number of risk factors and VTE occurrence.19–23 Several systematic reviews and meta-analyses have shown that computerized clinical decision support systems (CCDSS) were associated with an increased rate of appropriate prophylaxis for VTE.14,24 Furthermore, a retrospective study reported the usefulness of an artificial intelligence-based Clinical Decision Support System to improve the incidence of VTE detection. 25 However, the Caprini risk score has not been created for Asians, and few studies have investigated THA or TKA patients. Furthermore, since it is desirable to vary the type and degree of VTE prophylaxis for patients at risk of bleeding before surgery, it is necessary to develop a CCDSS that allows simple screening of Asian patients who are at a high risk of developing VTE but cannot use anticoagulants before drug use and make it an institution-wide policy. We developed a CCDSS for VTE risk assessment in 2008 for use not only in Asia but also globally, based on national consensus guidelines and previous studies.26,27 We evaluated the efficacy of a portable intermittent pneumatic compression device for VTE prophylaxis and wearing compliance in 38 lower extremity orthopedic patients whose VTE risk was assessed using CCDSS. We reported that the portable intermittent pneumatic compression device has the potential for safe VTE prophylaxis in patients at high risk for both venous thrombosis and bleeding. 28 Despite these efforts, the association between the VTE level, total risk score (TRS) in the CCDSS, and VTE incidence remains unknown. Evaluating this association may help physicians determine VTE risk level and optimal thromboprophylaxis strategies. 28
The primary objective of this study was to demonstrate the association between CCDSS VTE risk level and VTE incidence among THA or TKA patients who were assessed for VTE risk using CCDSS during hospitalization. The secondary objective was to examine the association between TRS and VTE incidence.
Methods
Study Design and Patients
This case-control study (level of evidence: III) was approved by the Institutional Review Board for Observation and Epidemiological Study at the University of Kitasato (KMEO B20-324) and conducted in accordance with the Declaration of Helsinki and Good Clinical Practice. Informed consent was obtained in the form of an opt-out option on the Kitasato University School of Medicine website. All collected data were anonymized. All participants underwent THA or TKA at the Kitasato University Hospital in Kanagawa Prefecture, Japan, between April 1, 2013, and August 31, 2018. The hospital is a university hospital with a 1185-bed tertiary care center. Perioperative patient data were collected from electronic medical records after patients were discharged. Two researchers independently reviewed the data.
Eligibility Criteria
The inclusion criteria were age ≥16 years, hospitalization ≥4 days, the use of the CCDSS during hospitalization, and THA or TKA history. The exclusion criteria were occurrence of VTE before surgery and no record of preoperative and postoperative ultrasonography.
VTE Definition
DVT was recorded after confirmation by a clinical laboratory technician within 7 days post-surgery with vascular ultrasonography (AplioTM Platinum Series; Canon, Tochigi, Japan).
PE was recorded when patients presented with early symptoms of PE following confirmation by a clinical radiologist using computed tomography angiography (SOMATOM Definition Flash; Siemens Healthineers, Forchheim, Germany). In this study, considering the incidence of DVT, vascular ultrasonography was performed on all patients on the day of the visit one month before admission and, in principle, within 7 days after surgery. Additional vascular ultrasonography was performed at the physician's discretion if symptomatic DVT was suspected. Computed tomography angiography was performed as needed at the physician's discretion for patients with initial symptoms of PE or confirmed DVT. All records were evaluated by a physician. The incidence of DVT or PE, either symptomatic or non-symptomatic, previously defined as a postoperative complication, was defined as VTE. Thereafter, patients were divided into the VTE and non-VTE groups.
VTE Risk Scoring System
The development of VTE was then evaluated weekly (Table 1). In risk assessment, the dose of VTE pharmacological prophylaxis or the severity of the disease was not considered a risk factor for VTE. Data for VTE risk factors were collected by nurses from the electronic medical records and double-checked by a physician. The presence of disease related to risk factors for VTE was determined by the presence of a physician's entry in the electronic medical records. The risk assessment for VTE was evaluated only during the hospitalization period, and the second and subsequent assessments were perfromed by nurses within one week of the previous assessment before being confirmed by a physician. We also classified patients into four groups according to TRS scores: group 1 (−2); group 2 (−1 to 1); group 3 (2 to 3); and group 4 (≥4).
CCDSS risk assessment model.

The criteria for thrombophilia included antiphospholipid antibody syndrome, hyperhomocysteinemia, increased prothrombin levels, factor V Leiden, increased factor VIII levels, hyperfibrinogenemia, antithrombin deficiency, protein C deficiency, and protein S deficiency. Abbreviations: BMI, body mass index; CCDSS, computerized clinical decision support system; VTE, venous thromboembolism
CCDSS
The CCDSS was devised as a convenient tool for clinicians to improve VTE prophylaxis for medicine and surgery patients. The CCDSS, shown to clinicians via an electronic medical chart, was available in all hospital wards and was used throughout hospitalization. The assessment of VTE risk level using the CCDSS consisted of three steps. First, the postoperative VTE risk level for each surgery was classified (low, moderate, and high); THA and TKA were categorized as high risk because of the high occurrence of postoperative VTE. Second, to determine the final VTE risk level for an individual patient, the corresponding 16 VTE risk factors were selected. Each risk factor had a risk score, which was automatically added if more than one risk factor was selected. Each risk factor was listed with weights of −2 to 8 (eTable 1). TRS was defined as the total score added to the selected risk factor. The final VTE risk level was determined by the risk of VTE due to the surgical procedure and the TRS score (eg, if the VTE risk due to the surgical procedure was high and the TRS was −2, the final VTE risk level was middle risk). The final VTE risk level (none, low, moderate, high, highest) and recommended steps toward VTE prophylaxis were displayed in the electronic medical record (eTable 2). All patients with or without symptomatic or asymptomatic VTE were assessed for the risk level of VTE with CCDSS.
VTE Prophylaxis Protocol
Pharmacological prophylaxis was defined as any anticoagulant administration at a prophylactic dose during hospitalization. All patients received 20 mg enoxaparin (Clexane® Sanofi-Aventis, Paris, France) subcutaneous injection once or twice daily, or 15 or 30 mg edoxaban (Lixiana® Daiichi-Sankyo, Tokyo, Japan) oral administration once daily. The dosages of each drug were determined according to the package insert in Japan. Pharmacological prophylaxis was started 24 h after surgery for enoxaparin and after 12 h for edoxaban. Pharmacological prophylaxis was administered for a maximum of 14 days. The duration of administration of each drug differed as per the discretion of physicians. Enoxaparin was administered to patients ≥65 years of age, and edoxaban to those <65 years of age. Pharmacological prophylaxis was combined with the optimal use of a mechanical method (eg, compression stocking and intermittent pneumatic compression). During hospitalization, the VTE occurrence in all participants was assessed. All patients admitted to the Department of Orthopedic Surgery, Kitasato University Hospital, and for whom the contraindications in the package insert for anticoagulants used for VTE prophylaxis did not apply, were included in the study.
Sample Size
Our study was designed to have a power of 80% to detect a 2-fold difference between the highest and high VTE groups in the primary endpoint. Therefore, 1380 patients had to be enrolled in this study, assuming an expected VTE incidence of 14% for the highest group, a sample size ratio of 5:1 for the highest and high groups, 125 dropout patients (10%), and α value of 0.05. A sample size calculation was performed for the primary outcome.
Statistical Analyses
We used Fisher's exact test and the chi-square test (with the Yates correction) to compare categorical variables between the VTE and the non-VTE groups. We analyzed continuous variables using the Mann–Whitney U test. If the VTE risk level was assessed multiple times during hospitalization in the CCDSS, the first TRS and VTE risk level assessed after admission were used for statistical analysis. To evaluate TRS characteristics, we defined TRS as a continuous variable. Normally distributed quantitative data were expressed as means, whereas non-normally distributed quantitative data were expressed as medians. Categorical data were expressed as absolute values and percentages of the population. Patients with missing values were excluded from the analyses. Univariate regression investigated the odds and rates for VTE among patients at different VTE risk levels and TRS groups. We excluded moderate-risk patients, or group 1, from the outcome analysis because of the assumed small number of patients. If significant differences were identified among the groups, we assessed trends in VTE occurrence in the CCDSS VTE risk level and TRS groups using the Cochran–Armitage trend test in the secondary analysis. 29 The distribution of the incidence rate of VTE according to the risk level and TRS, and the significance of the differences, were reported using the Fisher's exact test. Subgroup analyses were performed to assess the robustness of the primary endpoint in THA or TKA patients.
All statistical analyses were performed using Easy R (EZR version 1.36) software (Saitama Medical Center, Jichi Medical University, Saitama, Japan) and a graphical user interface for R (The R Foundation for Statistical Computing, Vienna, Austria). It is a modified version of R commander designed to add statistical functions frequently used in biostatistics. 30 All tests were two-tailed. If the appropriate number of patients could not be enrolled during the study, we presented nominal p values for our analysis, with a p value ≤0.05 considered statistically significant.
Results
Patient Characteristics
Clinical data were collected from 1198 patients within the study period. After exclusion due to the discovery of VTE before surgery (n = 83; 6.9%) or no record of preoperative or postoperative ultrasonography (n = 17; 1.4%), 1098 patients were enrolled instead of the assumed 1380. Incidence rates of DVT and PE were 11.5% (n = 126) and 0.18% (n = 2), respectively; 11.7% (n = 128) of patients developed VTE during postoperative hospitalization. The THA or TKA rates were 71.9% (n = 789) and 28.1% (n = 309), respectively. The median age of the VTE group was higher than that of the non-VTE group (74 vs 67 years). The median TRS for the VTE and non-VTE groups were 4 and 2, respectively. Age ≥60 years as a CCDSS risk factor was significantly associated with VTE occurrence. Risk factors included age, body mass index, cancer, severe infection, personal history of VTE, and thrombophilia (Table 2).
Individual risk factors, CCDSS score, and VTE rate in THA or TKA patients (n = 1098).

p values were calculated using the Fisher's exact test.
number (percentage)
Other than surgery, respiratory surgery, urology, and gynecology.
Criteria included antiphospholipid antibody syndrome, hyperhomocysteinemia, increased prothrombin levels, factor V Leiden, increased factor VIII levels, hyperfibrinogenemia, antithrombin deficiency, protein C deficiency, and protein S deficiency.
Abbreviations: BMI, body mass index; CCDSS, computerized clinical decision support system; THA, total hip arthroplasty; TKA, total knee arthroplasty; VTE, venous thromboembolism.
Primary Outcome
In this study, 13 (1.2%) and 156 (14.2%) patients were classified as moderate and high risk, respectively, while 929 patients (84.6%) were classified as highest risk (Figure 1). The incidence of VTE was associated with a risk level increase. The incidence of VTE during postoperative hospitalization was 12.8% (n = 119) in highest-risk, 5.8% (n = 9) in high-risk, and 0% (n = 0) in moderate-risk patients. Highest-risk patients were significantly more likely to develop VTE than were high-risk patients (OR 2.4; 95% CI 1.2-5.5; p = 0.01).
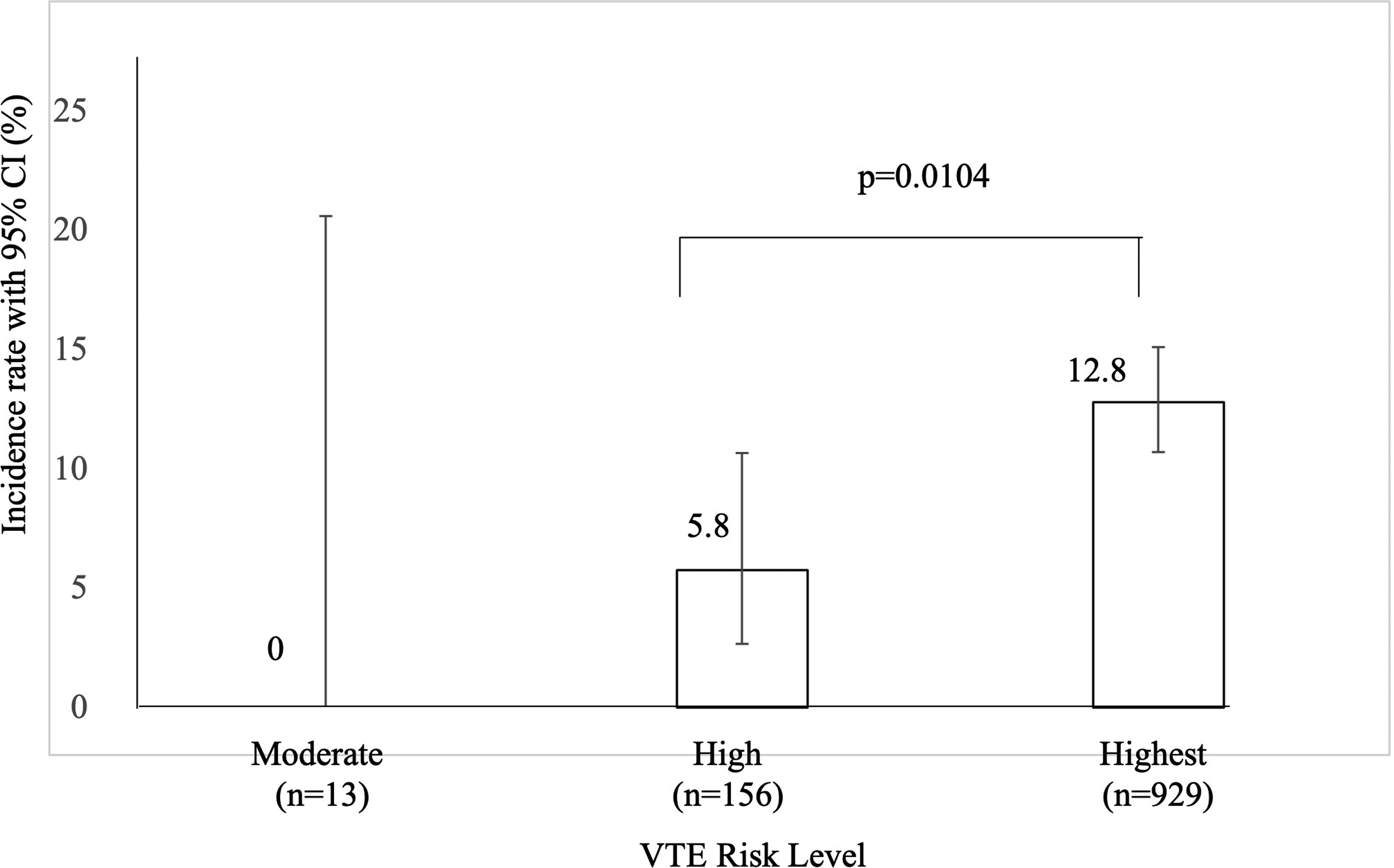
Incidence rate of VTE according to risk level (n = 1098). The incidence rates of VTE at the highest and high risk were 12.8% (n = 119) and 5.8% (n = 9), respectively, with a significant difference (p = 0.0104). The error bars indicate 95% CIs. Abbreviations: CI, confidence interval; VTE, venous thromboembolism.
Secondary Outcome
Most patients (84.6%) demonstrated a TRS of 2 to 3 (542 patients [49.4%]) or ≥4; VTE incidence increased in a linear fashion with increasing TRS (Figure 2). Patients with a TRS ≥4 were more likely to develop VTE than patients with a TRS of 2 to 3 (OR 1.8; 95% CI 1.2-2.7; p = 0.003) and −1 to 1 (OR 3.3; 95% CI 1.6-7.7; p < 0.001), while those with a TRS of 2 to 3 and −1 to 1 did not differ significantly and had a low VTE incidence (OR 1.8; 95% CI 0.86-4.3; p = 0.12) (Table 3). The Cochran–Armitage trend test demonstrated that VTE incidence increased with increasing risk level or TRS (p = 0.0043 and p < 0.001, respectively).

Incidence rate of VTE according to the TRS (n = 1098). As TRS increased, VTE incidence increased linearly. The error bars indicate 95% CIs. Abbreviations: CI, confidence interval; VTE, venous thromboembolism; TRS, total risk score.
Odds ratio for VTE in THA or TKA patients stratified according to the TRS.

The p values and odds ratios were calculated using the Fisher's exact test.
Data are shown as odds ratios with 95% confidence interval.
Abbreviations: THA, total hip arthroplasty; TKA, total knee arthroplasty; TRS, total risk score; VTE, venous thromboembolism.
Subgroup Analysis
eTables 3 and 4 show the ORs of VTE stratified according to CCDSS VTE risk level in THA and TKA patients. No significant difference was recorded in the likelihood of VTE occurrence between highest- and high-risk THA and TKA patients.
Discussion
We investigated the relationship among risk level, TRS in CCDSS, and occurrence of VTE in THA or TKA patients. VTE incidence in patients who underwent THA or TKA was 11.7%, and the OR for VTE incidence in highest-risk patients was 2.4-fold that of high-risk patients. Additionally, VTE incidence was significantly higher in patients with a TRS ≥4 than in those with a score of 2 to 3 or −1 to 1. VTE incidence in Asian patients after THA or TKA was associated with VTE risk level and TRS in the CCDSS. To our knowledge, this is the first study to show such an institution-specific risk assessment model and VTE incidence relationship in Asian patients. We found that the CCDSS may be a useful and efficacious tool to stratify VTE risk in THA or TKA patients, confirming the findings of previous studies using Caprini risk assessment models.21,23
Several studies have shown that Asian patients generally have a significantly lower incidence of VTE than Western patients; however, under certain conditions, such as postoperatively, Asian patients have been shown to have similar VTE incidences as Western patients.3,31 When appropriate prophylaxis is provided, symptomatic VTE can be reduced by 55%–60% for patients undergoing orthopedic surgery. 1 In fact, guidelines for VTE prevention are suboptimally implemented in Asian patients. 13 To the best of our knowledge, there is no VTE risk assessment tool specific to Asians; therefore, it is necessary to create a tool that can assess VTE risk accurately and concisely.
The follow-up duration varied among studies. This study assessed inpatient VTE only, with a mean hospitalization duration of 19 days. In contrast, the mean hospitalization duration for THA or TKA patients in the US was 2–5 days. 32 This is because the Japanese insurance system differs from that of other countries; moreover, Japan has adopted the Diagnosis Procedure Combination-based Payment System. 33 Therefore, the hospitalization period may be longer than that of the US.
Consistent with other validation studies, VTE incidence was higher in THA and TKA patients when the VTE risk level in CCDSS increased. However, the reliability of VTE risk assessment models in Asian patients after THA or TKA has not been fully evaluated. The observation of an increased rate of VTE during hospitalization in highest-risk versus high-risk patients is useful for identifying patients who may benefit from short-term prevention. However, further research is required to determine the relationship between VTE risk level and the period that patients remain at risk. 34
Both our study design and a previous retrospective database study by Bahl et al stratified surgical patients according to VTE risk level and examined VTE incidence according to the stratified risk score. 21 Our findings are consistent with Bahl's results reporting that the 30-day VTE occurrence increased with the increasing risk level. 21 Despite this, VTE incidence according to stratified risk level differed between the two studies. Here, VTE occurrence among highest-risk patients approached approximately 6.6-fold the occurrence recorded by Bahl (12.8% vs 1.9%). These differences may be explained by the differences in study methods. First, we included only THA or TKA patients, whereas the Bahl study included general, vascular, and urology surgical patients. The occurrence of VTE after THA or TKA was higher than that after general and urological surgery. Second, in contrast to Bahl's study, we checked all patients for VTE occurrence. By confirming the VTE of all THA or TKA patients, a more accurate VTE incidence could be calculated. The Caprini score, accepted in the 2012 American College of Clinical Pharmacology VTE guidelines, can also identify VTE risk among the surgical or inpatient population.21,23 However, data regarding the efficacy of a VTE risk scoring system in THA or TKA patients are limited, and Bateman et al showed that the Caprini model does not provide clinically useful risk stratification for THA and TKA. 35 Nevertheless, Bateman et al stated that this result may be partly due to the fact that the incidence of VTE was so low that they could not enroll enough patients.
Consistent with previous studies, we found that the VTE risk increase was associated with a TRS increase in THA or TKA patients.20,21,23 Finally, the 16 risk factors of the CCDSS, compared with >40 risk factors of the Caprini model, may facilitate the risk assessment of VTE occurrence in THA or TKA patients. In this study, we found that patients with a TRS ≥4 may have a higher incidence of VTE than those with a TRS of 2 to 3. Therefore, even at the highest risk level, patients with a TRS ≥4 should be carefully monitored for the development of early symptoms of VTE. The results of this study suggest the importance of evaluating TRS in patients at risk for VTE; further refinement of the CCDSS to improve its predictive accuracy for VTE and its routine incorporation into clinical practice would be highly beneficial.
No statistically significant differences were found between the highest- and high-risk THA and TKA patients. The calculated posterior power of the highest risk and high-risk patients for THA and TKA was 41.4% for THA and impossible to calculate for TKA. The sample size calculation for the analysis based on the highest and highest-risk VTE incidence required 1326 THA patients. This may be attributed to the small sample size of the high-risk group compared to the highest-risk group in THA and TKA, and the small difference in the incidence of VTE in each group in TKA. Moreover, the statistical multiplicity was unaccounted for, resulting in a lack of statistical significance. In this study, the high-risk and highest-risk patients in the THA or TKA group were 14.2% (n = 156) and 84.6% (n = 929), respectively. The reason for this was that THA or TKA was the procedure with a high risk of VTE in the CCDSS. Moreover, the presence of individual patient risk factors for VTE was likely to result in the highest risk. In fact, 72.2% of the patients were aged ≥60 years, and all patients aged ≥60 years were determined to be at the highest risk in the THA or TKA patient groups in the CCDSS.
Reduced medical costs and hospitalization are among the VTE prevention benefits. A previous study with THA and TKA patients recorded significantly higher total healthcare costs between patients with and without VTE. 36 Moreover, several systematic reviews have reported a significant difference in total hospital costs when VTE occurred during hospitalization compared with when it did not.7,37 Therefore, VTE prophylaxis should be implemented during early hospitalization in all hospitalized patients, depending on their individual VTE risk level.
CCDSS may be a useful tool to stratify patients into different VTE risk levels. Furthermore, we believe that repeated risk assessments for VTE and the implementation of appropriate preventive measures are necessary to reduce the incidence of VTE, ensure patient safety, and promote early recovery.5,38 Zhai et al also recommend that all patients need to undergo repeated risk assessments. 39 Repeated assessments can be burdensome for healthcare providers; thus, tools such as the CCDSS are needed to assess VTE accurately and concisely.
Several studies have reported that CCDSS has some shortcomings, but it is one of the most effective tools for improving the quality of healthcare. 40 In order to increase the effectiveness and convenience of CCDSS, several studies have been reported concerning the creation of smartphone applications and the integration of artificial intelligence into CCDSS.25,41 However, there was limited guidance in the literature on the design and reporting of CCDSS. Kawamoto et al provided 13 recommendations for the design, conduct, and reporting of CCDSS studies. 42 Moreover, the quality of the guideline recommendations needs to be checked carefully, as some guidelines contain a significant proportion of low-quality or very low-quality evidence or the opinions of experts in working groups. 43 We are planning to improve CCDSS based on these recommendations to improve its application and implementation in clinic settings.
This study had several limitations. First, as it was a single-center study, our results are not generalizable. Further studies regarding the association of VTE risk level with outcomes at other centers worldwide are warranted to validate the CCDSS efficacy. Furthermore, examining the CCDSS efficacy in different racial groups, with reliable data sources to apply the VTE risk level and scoring method, improves its applicability. However, the increase in VTE incidence, CCDSS VTE risk level, and score has important practical implications. Second, we only report VTE incidence during hospitalization, which may lead to underestimating VTE occurrence in THA or TKA patients. A previous meta-analysis demonstrated the 90-day postoperative rates of symptomatic, non-fatal VTE (3.0%) and fatal pulmonary embolism (0.003%) following THA or TKA. 44 A prospective study with a minimum 90-day follow-up of those patients would overcome this limitation. Third, our sample size was insufficient to investigate the association of the CCDSS VTE risk level with VTE occurrence. The statistical power was low, ie, 70.1%. In addition, the number of THA patients exceeded the number of TKA patients, probably because our orthopedic department specializes in the treatment of hip joints rather than knee joints. In addition, the distribution of patients with high- and highest-risk levels in the CCDSS was too abundant in this study, and more studies including low- and intermediate-risk patients are needed to evaluate the risk classification of the CCDSS. Therefore, it was not possible to include each patient population equally in this study. Further research with adequate sample size is required to detect any significant group differences. The major strengths of this study were that we created a CCDSS (which allows for more concise input than the Caprini score) and used this tool to examine the relationship between VTE incidence and the CCDSS VTE risk level among a large sample of Asian patients (with an average hospital stay of 19 days). There are three improvements from the Caprini risk score in developing the CCDSS for Asian patients.
First, the number of risk factors selected to simplify the evaluation for VTE is 16; second, the time of surgery has not been considered so that the risk of VTE can be evaluated even before surgery; and third, because Asians have a lower BMI than Americans, the BMI stratification has been changed from three (BMI>30, >40, >50) to two (BMI≥25-<30, ≥30). 45
In conclusion, the CCDSS may be an effective method for identifying patients at risk for VTE during hospitalization after THA or TKA. In the Japanese population, VTE incidence in THA or TKA patients using CCDSS was found to be lower than that in patients using enoxaparin for VTE prophylaxis. 46 We suggest assessing individual patient VTE risk levels to determine the appropriate thromboprophylaxis. Appropriate VTE prophylaxis should be adhered to according to guidelines developed based on high-quality evidence and expert opinion. Although this study included only THA or TKA patients, the VTE risk classification of the CCDSS was similar to that of other scoring systems.21–23 The CCDSS contains fewer VTE risk assessment factors than other existing scoring systems, reducing the burden on physicians and enabling appropriate VTE prophylaxis, hence potentially preventing prolonged hospitalization due to VTE. Finally, the CCDSS could be used to stratify THA or TKA patients according to the risk of VTE and provide the basis for a practical tool to improve adherence to VTE prophylaxis guidelines.
Supplemental Material
sj-docx-1-cat-10.1177_10760296221103868 - Supplemental material for Internal Validation of a Risk Scoring System for Venous Thromboembolism After Total hip or Knee Arthroplasty
Supplemental material, sj-docx-1-cat-10.1177_10760296221103868 for Internal Validation of a Risk Scoring System for Venous Thromboembolism After Total hip or Knee Arthroplasty by Akihiko Akamine, Naonobu Takahira, Masayuki Kuroiwa, Atsushi Tomizawa and Koichiro Atsuda in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Acknowledgements
We thank the working group of DVT prevention committee members at Kitasato University Hospital for their clinical assistance with this research. We also thank Shun Kunisawa for his work on the data collection. We thank the orthopedists who performed orthopedic surgery at Kitasato University Hospital. We thank Editage (![]() ) for English language editing.
) for English language editing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Statement
Ethics Approval
Ethical approval to report this case was obtained from Institutional Review Board for Observation and Epidemiological Study at the University of Kitasato (KMEO B20-324).
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Informed consent for patient information to be published in this article was not obtained because due to the retrospective nature of the study, the Institutional Review Board waived the need to obtain informed consent from the participants.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
