Abstract
Purpose
Coagulation abnormalities are one of the most important complications of severe COVID-19, which might lead to venous thromboembolism (VTE). Hypercoagulability with hyperfibrinogenemia causes large vessel thrombosis and major thromboembolic sequelae. Statins are potentially a potent adjuvant therapy in COVID-19 infection due to their pleiotropic effect. This study aims to evaluate the effectiveness of statins in reducing the risk of thrombosis among hospitalized critically ill patients with COVID-19.
Methods
A multicenter, retrospective cohort study of all critically ill adult patients with confirmed COVID-19 admitted to Intensive Care Units (ICUs) between March 1, 2020, and March 31, 2021. Eligible patients were categorized based on their usage of statins throughout their ICU stay and were matched with a propensity score. The primary endpoint was the odds of all cases of thrombosis; other outcomes were considered secondary.
Results
A total of 1039 patients were eligible; following propensity score matching, 396 patients were included (1:1 ratio). The odds of all thrombosis cases and VTE events did not differ significantly between the two groups (OR 0.84 (95% CI 0.43, 1.66), P = 0.62 and OR 1.13 (95% CI 0.43, 2.98), P = 0.81, respectively. On multivariable Cox proportional hazards regression analysis, patients who received statin therapy had lower 30-day (HR 0.72 (95 % CI 0.54, 0.97), P = 0.03) and in-hospital mortality (HR 0.67 (95 % CI 0.51, 0.89), P = 0.007). Other secondary outcomes were not statistically significant between the two groups except for D-dimer levels (peak) during ICU stay.
Conclusion
The use of statin therapy during ICU stay was not associated with thrombosis reduction in critically ill patients with COVID-19; however, it has been associated with survival benefits.
Keywords
Introduction
Since the outbreak of Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) globally, 230 million and more than 4.7 million deaths as of 25 September 2021 were reported. 1 The severity of Corona Virus Infection Disease 2019 (COVID-19) symptoms varies from one person to another, as if they may be either asymptomatic or severe in some cases requiring intensive unit (ICU) admission. The progression of clinical symptoms can be related to several mechanisms. However, one previous study concluded that endothelial dysfunction, inflammatory, and immunological response can play a crucial role in the pathogenesis of viral pneumonia, including COVID-19 patients. This effect can trigger a wide array of severe adverse complications that further increase mortality, such as arterial and venous thrombotic events. White blood cells (WBC), neutrophils, lymphocytes, neutrophil/lymphocyte ratio (NLR), and platelets/lymphocyte ratio (PLR) are all easy and obtainable markers of systemic inflammation that aids patient’s prognosis and follow up. Higher levels of NLR in COVID19 patients suggested a poor outcome in COVID19 patients. Additionally, PLR was also higher in COVID-19 patients, which correlated with poor prognosis and longer length of hospital stay.2–8
Patients with cardiovascular risk factors or a history of cardiovascular disease are associated with poor prognosis and increased mortality when infected with COVID-19.9,10 In critically ill patients with COVID-19, the risk of in-hospital and ICU mortality in patients with heart disease failure was significantly higher. 11 Most of the mortality in COVID-19 patients was attributed to respiratory-related complications. 12 However, a shred of growing evidence finds that increased mortality and morbidity related to cardiac complications, including new or worsening heart failure, new or worsening arrhythmia, myocardial infarction.13,14
Initial studies concluded that the incidence of venous thromboembolism (VTE) was between 15-25% in hospitalized patients with COVID-19. Additionalstudies have shown that the risk of VTE may be as high as 85% despite all antithrombotic prophylaxis administered to COVID-19 patients.15,16 However, recently a multiplatform, randomized trial found that therapeutic anticoagulation use in moderately ill patients with COVID-19 was associated with an increased probability of survival and reduced use of ICU-level organ support compared with usual thromboprophylaxis. 17 Unfortunately, this trial noticed no positive outcome among critically ill patients with COVID-19 who received therapeutic anticoagulation. 18
Statin therapy are known for their lipid-lowering properties, are commonly used in clinical practice, and recommended by many guidelines for primary and secondary prevention of atherosclerotic cardiovascular disease (ASCVD).19–21 Additionally, statins have pleiotropic effects such as improving endothelial function, anti-inflammatory, immunomodulatory and antithrombotic effects. 19 These pleiotropy effects were the basis for exploring the role of statins in patients with COVID-19.. Previous published studies about the effect of statins on mortality in COVID-19 patients showed different conclusions. More recently, an updated meta-analysis, including 25 cohort studies, found that statin use is associated with lower mortality risk. 20
To date, little attention has been paid to the role of statins on venous thromboembolism (VTE) among COVID-19 patients. In non-COVID-19 patients, Joseph et al conducted a pooled analysis of (HOPE-3) and (JUPITER) trials to investigate the association between rosuvastatin and VTE risk. This trial showed a lower VTE risk among patients who received rosuvastatin as compared to placebo (HR 0.53; 95% CI, 0.37-075). 22 Also, a meta-analysis from nine randomized control trials (n = 1165) showed that statin could significantly decrease plasma D dimer levels. 23 Despite many studies on statin benefits in COVID-19 patients, there is a lack of evidence about the effectiveness of statins in reducing the risk of VTE in critically ill patients with COVID-19 infection. Therefore, the aim of this study is to evaluate the role of statins in reducing the risk of thrombosis among hospitalized critically ill patients with COVID-19.
Methods
Study Design
A multicenter, retrospective cohort study including critically ill adult patients admitted to the Intensive care unit (ICU) from March 1, 2020, until March 31, 2021, with confirmed COVID-19. The study was approved by King Abdullah International Medical Research Center (KAIMRC) (Ref.# RC20/683/R). All patients were followed until they were discharged from the hospital or died during the in-hospital stay. Informed consent was waived due to the retrospective observational nature of the study.
Study Participants
We included adults patients (age ≥ 18 years) admitted to the ICUs with confirmed COVID-19. Patients were diagnosed with COVID-19 using Reverse Transcriptase-Polymerase Chain Reaction (RT-PCR) nasopharyngeal or throat swabs. Patients were excluded if the ICU length of stay (LOS) ≤ one day, died within 24 h of ICU admission, were labeled as "Do-Not-Resuscitate", had a history of VTE, Systemic lupus erythematosus (SLE), or Antiphospholipid antibody syndrome (APLS) (Figure 1). Eligible patients were then categorized into two groups based on the statin therapy used during ICU stay (control vs statin). Statins were prescribed in the ICU as part of the medication reconciliation process if they had been prescribed in the pre-ICU period or were newly initiated in the ICU for various indications at the discretion of the ICU treating team.

Critically ill patients with COVID-19 flowchart.
Study Setting
The study was conducted at four hospitals in Saudi Arabia: King Abdulaziz Medical City (Riyadh), King Abdulaziz University Hospital (Jeddah), King Abdullah bin Abdulaziz University Hospital (Riyadh), and King Salman Specialist Hospital (Hail). The primary site for this multicenter retrospective study was King Abdulaziz Medical City (Riyadh).
Data Collection
Each patient’s data were collected and managed using Research Electronic Data Capture (REDCap®) software hosted by King Abdullah International Medical Research Center (KAIMRC). We collected patients’ demographic data, comorbidities, vital signs, and laboratory tests such as renal profile (ie, estimated glomerular filtration rate (eGFR)), liver function tests (LFTs) (ie, total bilirubin), coagulation profile (ie, INR, aPTT, platelets count), and surrogate markers (ferritin, D-dimer, and C-reactive protein (CRP)) within 24 h of ICU admission. Moreover, severity score baseline (ie, Acute Physiology and Chronic Health Evaluation II (APACHE II) and Sequential Organ Failure Assessment (SOFA)), Glasgow Coma Score (GCS), acute kidney injury, use of prone positioning, the needs for mechanical ventilation (MV) and MV parameters (eg, PaO2/FiO2 ratio (lowest), FiO2 requirement (highest)) within 24 h of ICU admission were recorded. Surrogate biomarkers of the COVID-19-associated inflammation (ferritin, D-dimer, and C-reactive protein (CRP)) were collected and reported with their baseline and peak values during the ICU stay. In addition, statin therapy, tocilizumab, corticosteroids, and pharmacological VTE prophylaxis use were documented for the eligible patients (Additional file 1).
Baseline Severity Score Definitions
Acute Physiology and Chronic Health Evaluation II (
Sequential Organ Failure Assessment (
Glasgow Coma Score (
Sample Size Calculation
A total of 310 subjects (155 in each group) required to detect the minimal difference in the incidence of all thrombosis cases of 10% between statin and non-statin group, with 5% level of significance and 80% power of test. The sample size was calculated using power analysis and sample size (PASS) software (PASS 15 Power analysis and sample size software (2017). NCSS, LLC. KAYSVILLE, Utah, USA, ncss.com/software/pass).
Outcomes
The primary endpoint was all thrombosis cases. The secondary endpoints were VTE, 30-day/in-hospital mortality, ICU-acquired complication
Outcome Definition (s)
Arterial/venous thrombosis defined using the International Statistical Classification of Diseases (ICD)10-CM code (ie, Myocardial infarction (MI), ischemic stroke, pulmonary embolism, deep vein thrombosis). 23
The 30-day mortality was defined as a death occurring for any cause within 30 days of the admission date during hospital stay; patients who were discharged from the hospital alive were presumed to be survived.
New-onset atrial fibrillation (A fib.) was defined as new onset or a first detectable episode of atrial fibrillation (any type), whether symptomatic or not.
Statistical Analysis
We presented continuous variables as mean and standard deviation (SD), or median with lower quartile (Q1) and upper quartile (Q3), as appropriate and categorical variables as number (percentage). The normality assumptions were assessed for all numerical variables using a statistical test (ie, Shapiro–Wilk test\Kolmogorov Smirnov test) and graphical representation (ie, histograms and Q-Q plots). Model fit assessed using the Hosmer-Lemeshow goodness-of-fit test.
Baseline characteristics and outcome variables were compared between the two study groups. For categorical variables, we used the Chi-square or Fisher’s exact test. We compared the normally distributed continuous variables using student t-test and other non-normally distributed continuous variables with the Mann-Whitney U test. Multivariable Cox proportional hazards regression analyses were performed for the 30-day and in-hospital mortality. Multivariable regression analysis and negative binomial regression were used for the other outcomes considered in this study. The odds ratios (OR), hazard ratio (HR), or estimates with the 95% confidence intervals (CI) were reported as appropriate. No imputation was made for missing data as the cohort of patients in our study was not derived from random selection. We considered a P value of < 0.05 statistically significant and used SAS version 9.4 for all statistical analyses.
Propensity score matching procedure (Proc PS match) (SAS, Cary, NC) was used to match patients who received statin therapy (active group) to patients who did not (control group) based on patient’s age, BMI, APACHE 2 score, D-dimer, aPTT, MV status, systemic corticosteroids use, pharmacological DVT prophylaxis use, stroke and atrial fibrillation as coexisting illness. A greedy nearest neighbor matching method was used in which one patient who received statin therapy (active) group matched with one patient who did not (control), which eventually produced the smallest within-pair difference among all available pairs with treated patients. Patients were matched only if the difference in the logits of the propensity scores for pairs of patients from the two groups was less than or equal to 0.5 times the pooled estimate of the standard deviation.
Results
A total of 1039 patients were eligible based on the selected inclusion/exclusion criteria. Of these patients, 279 (26.8%) received statin during ICU stay. After propensity score matching (1:1 ratio), 396 patients were included based on predefined criteria. Most of the patients who received statin were using it for chronic conditions (eg, DLP, IHD). Statins were discontinued on only fifteen patients (7.8%) during ICU stay; liver injury was the most common cause (6 patients), followed by rhabdomyolysis (5 patients). Atorvastatin (81.8%) with a median dose of 20 mg was the most often used statin, followed by Rosuvastatin (16%) with a median dose of 20 mg.
Demographic and Clinical Characteristics
The majority of the patients included in both arms (68.9%) were male, with a mean age of 61.6 (SD + 14.8). Diabetes mellitus (59.0%), hypertension (55.3%), and dyslipidemia (19.3%) were the most prevalent underlying comorbidities in our patients. There were some notable differences between the two groups before propensity score matching; patients who did not receive statin therapy (control group) were younger, placed more in a prone position, had higher total WBC count, aPTT, D-dimer, ferritin, and creatine phosphokinase (CPK) levels baseline within 24 h of ICU admission. On the other hand, patients who received statins had significantly higher MV needs within 24 h of ICU admission, C-reactive protein (CRP) baseline, systemic corticosteroids, and pharmacological DVT prophylaxis use compared with the control group. Following the propensity score matching based on the selected criteria, most of these baseline and demographic characteristics were similar between the two groups. A Summary of the patients’ baseline characteristics is available in
All Thrombosis Cases/ Venous Thromboembolism (VTE)
There was no significant difference in all thrombosis events in crude analysis between the statin and control group (9.1% vs. 10.1; p-value = 0.75, respectively). Patients who received statin showed a lower thrombosis events by 16% in compared to the control group, but it failed to reach the statistical significance at multivariable logistic regression analysis (OR (95%CI): 0.84 (0.43, 1.66), p-value = 0.62)
Regression Analysis for the Outcomes After PS.

Δ^ Denominator of the percentage is non-atrial fibrillation as comorbidity.
Δ Denominator of the percentage is the total number of patients.
$ Propensity score matched used based patient’s age, BMI, APACHE 2 score, D-dimer, aPTT, MV status, systemic corticosteroids use, pharmacological DVT prophylaxis use, stroke and atrial fibrillation as co-existing illness.
In crude analysis, the prevalence of VTE rate was not statistically significant among patients who received statin therapy compared to patients who did not (5.0% vs. 4.0%; p-value = 0.63), respectively. Moreover, there was no statistically significant difference between the two groups after using multivariable logistic regression analysis (OR (95%CI): 1.13 (0.43, 2.98), p-value = 0.81) (Table 1).
30-day and in-Hospital Mortality
There was no significant difference in the 30-day mortality (42.9% vs 50.0%, p-value = 0.16) nor in-hospital mortality (47.5% vs 54.1%, p-value = 0.19) between the statin and the control group at crude analysis, respectively (Table 1). However, according to the cox proportional hazards regression analysis, patients who received statin had a lower 30-day mortality (HR 0.72; 95% CI 0.54, 0.97, P = 0.03) and in-hospital mortality (HR 0.67; 95% CI 0.51, 0.89, P = 0.007) compared with the control group (Table 1).
Complications During ICU Stay
Interestingly, patients who received statin therapy had a lower odds of liver injury, but it was not statistically significant (OR (95%CI): 0.53 (0.26, 1.07), p-value = 0.08). In terms of major bleeding, there was no significant difference in crude analysis in the patient who received statin compared to patients who did not (7.6% vs. 5.1%; p-value = 0.30). Although, results from the multivariable logistic regression analysis; demonstrated a higher odds of major and minor bleeding in the statin group ; however, it did not reach the statistical significance (OR (95%CI): 1.55 (0.68, 3.53), p-value = 0.30 and OR (95%CI): 1.14 (0.52, 2.50), p-value = 0.75, respectively). In addition, patients who received statin have a higher odds of new-onset atrial fibrillation; but, it was not statistically significant (OR (95%CI): 1.7 (0.67, 2.41), p-value = 0.47) (Table 1).
Follow-up Surrogate Markers During ICU Stay
Ferritin, and CRP as follow-up inflammatory markers during were the same between the two groups, while D-dimer was significantly lower in patients who received statins in comparison to those who did not with a beta coefficient (95%CI): −2.64 (−3.16, −2.12), p-value <0.0001 (Table 2).
Follow-Up Surrogate Markers During ICU Stay (Peak Levels)
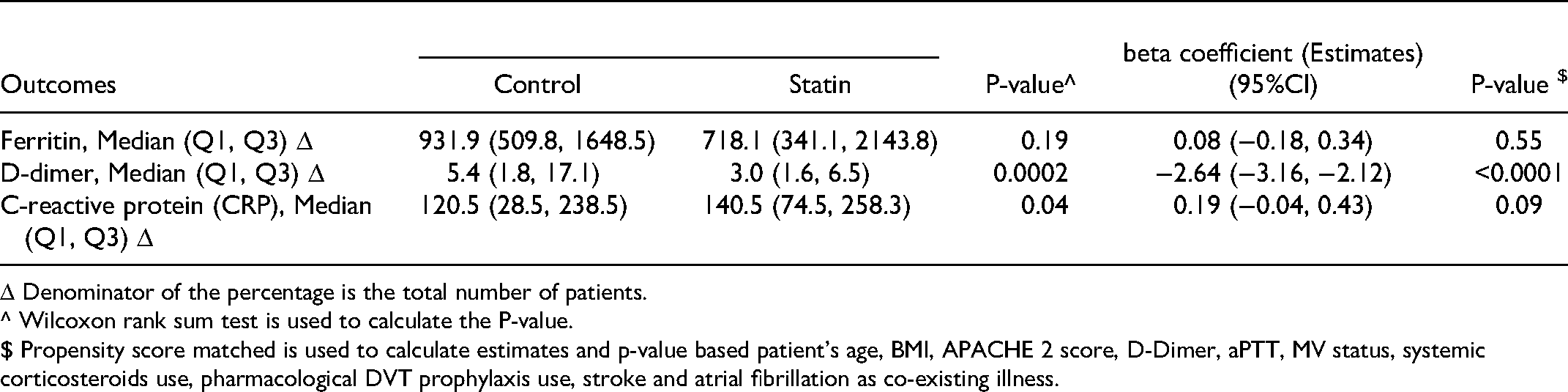
Δ Denominator of the percentage is the total number of patients.
^ Wilcoxon rank sum test is used to calculate the P-value.
$ Propensity score matched is used to calculate estimates and p-value based patient’s age, BMI, APACHE 2 score, D-Dimer, aPTT, MV status, systemic corticosteroids use, pharmacological DVT prophylaxis use, stroke and atrial fibrillation as co-existing illness.
Discussion
Our cohort study examined the association between statins use and VTE incidence in critically ill patients with COVID −19. Our data showed no statistically significant reduction in all thrombosis cases and VTE events in COVID-19 patients who received statins compared with patients who did not. On the other hand, patients who received statins during their ICU stay had lower 30-day and in-hospital mortality.
Even though our findings showed no statistically significant difference in VTE prevalence and all cases of thrombosis, critically ill patients with COVID-19 are known to have hypercoagulable states and a high incidence of VTE.27–29 A meta-analysis of 19 studies with 1599 patients showed that the prevalence of VTE, DVT, and PE in critically ill patients with COVID −19 was 28.4%, 25.6%, and 16.4%, respectively. 29 Similar to our finding, a randomized controlled trial (INSPIRATION-S) compared atorvastatin 20 mg daily with placebo in critically ill patients with COVID-19 found that starting the statin did not shows thrombosis benefits. 30 We propose two potential explanations for our observed findings. First, all patients in this study and previous studies were taking some form of VTE pharmacological prophylaxis.. Therefore, identifying patients who cannot receive thromboprophylaxis during the AC-free period will help assess statin's pleiotropic and anti-inflammatory effects to reduce VTE incidence. Additionally, our study had a small sample size of patients (n = 396); this potentially could have limited the ability to detect any difference between the groups.
Our study showed that the use of statins was associated with a significant reduction in 30-day mortality and in-hospital mortality in critically ill patients with COVID-19 compared with the control group. The effect of statins on reducing mortality in critically ill patients with COVID-19 has been previously investigated in several studies. A meta-analysis of 19 observational studies showed that statins use prior to COVID-19 diagnosis was associated with lower mortality. 31 This aligns with findings observed by Lori et al in which prior use of statins reduced the risk of death in patients with CVD and HTN. 32 Similarly, a multicenter observational study in Italy conducted on 842 patients with COVID-19 failed to demonstrate a statistically significant reduction in mortality among statin users but highlighted that statin use was associated with more severe disease. 33 Conversely, our cohort showed a significant reduction in D-dimer levels in the statin group than the control group during the stay, which may be translated to a reduction in the severity of disease progression that translated to the observed mortality benefit.
Despite these findings, statins did not improve mortality in a systematic review by Timotius et al as well in the recent INSPIRATION-S trial.30,34 In this RCT, only newly initiated statin therapy was included. It is imperative to note that our study was conducted on critically ill patients; most of them who are suffering from ARDS and severe inflammatory responses.Most of the participants in our study were long-term users of statins, which would suggest that continuous statins during the peak of the inflammatory response might be beneficial compared to mild or moderate disease patients who may not have ARDS or severe inflammation. 30
Most of our patients in this study (81.8%) received atorvastatin 20 mg, which is considered moderate intensity. Although the LDL lowering activity of statins is dose-dependent, some of the other pleiotropic activities could happen at even lower drug concentrations. 35 Therefore, statins intensity (ie, high-intensity) requires further studies to prove its effect on reducing VTE incidence or mortality in COVID-19. Moreover, our study did not report the type and dose of pharmacological VTE prophylaxis. During the early COVID-19 era, the Saudi Ministry of Health (MOH) COVID-19 protocol recommended the use of high dose VTE prophylaxis (enoxaparin 40 mg BID) in patients with elevated D-dimer (above 1) and body weight (above 100 kg). This may have affected the inability to detect any differences in VTE incidence and all thrombosis as most of our patients had an elevated BMI (mean above 30) and D-dimer (mean above 1); therefore, they may have been candidates for high dose VTE prophylaxis.
We believe that our multicenter cohort study is one of few studies that evaluated statins’ clinical and safety outcomes in critically ill patients with COVID-19 using propensity score matching and multiple regression analysis to minimize the bias. Nevertheless, we also determined some limitations in our study, such as the retrospective nature, which cannot prove causality and may have been affected by missing documentation that could be translated to unmeasured confounders. Another limitation is the lack of data on the duration of statin use before ICU admission and the timing of statin initiation (New Vs. Continuation). Even though we observed mortality benefits, we did not assess the long-term benefit of statin use after COVID-19 survival. Arterial thrombosis was numerically less in statin recipients who were already at risk for arterial thrombosis due to their underlying condition in addition to the pathogenesis of COVID19. This study could lead to a larger, well-designed, randomized controlled trial in critically ill patients with COVID −19 to investigate the efficacy and safety of statins on thrombosis risk and mortality.
Conclusion
The use of statin therapy in critically ill patients with COVID-19 did not result in VTE reduction but was associated with lower mortality. Additionally, patients who received statin therapy were more likely to have lower D-dimer levels, indicating that continuing statin therapy might be safe and beneficial. However, the dose and timing of statin therapy, along with other modes of prophylaxis are crucial to lowering the inflammatory process and progression of disease that could improve survival rates since inflammatory markers play a key role in complications, pathogenesis, and mortality rate. Further randomized clinical studies are required to evaluate and confirm these findings.
Summary of Demography and Baseline characteristics.
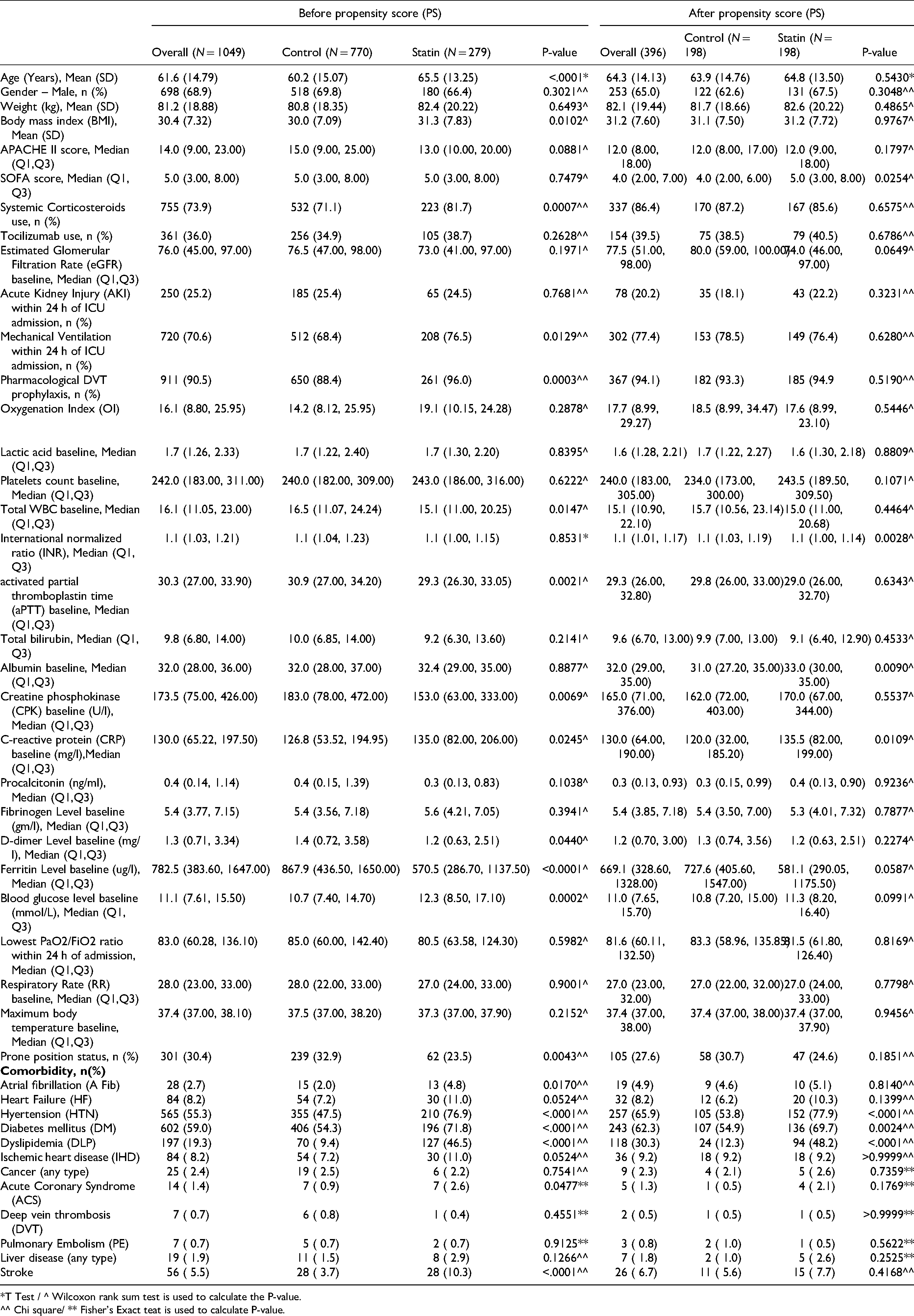
*T Test / ^ Wilcoxon rank sum test is used to calculate the P-value.
^^ Chi square/ ** Fisher’s Exact teat is used to calculate P-value.
Footnotes
Acknowledgements
We would like to acknowledge the investigators in the Saudi critical care pharmacy research (SCAPE) platform who participated in this project.
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Availability of Data and Material
The datasets used and/or analyzed during the current study are available from corresponding author on reasonable request.
Ethics Approval and Consent to Participate
The study was approved in January 2021 by King Abdullah International Medical Research Center Institutional Review Board, Riyadh, Saudi Arabia (Ref.# RC20/683/R). Participants’ confidentiality was strictly observed throughout the study by using anonymous unique serial numbers for each subject and restricting data only to the investigators. Informed consent was not required due to the research's method as per the policy of the governmental and local research center.
Patients and Public Involvement
Patients or the public were not involved in the design, or conduct, or reporting, or dissemination plans of our research.
Consent for Publication
Not applicable.
Competing Interests
No author has a conflict of interest in this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
