Abstract
Chronic thromboembolic pulmonary hypertension (CTEPH) is a serious complication of acute pulmonary embolism (PE) which remains underdiagnosed. A better understanding of risk factors for CTEPH would improve our ability to predict which PE survivors are at risk. Several medical conditions—including malignancy, splenectomy, thyroid hormone supplementation, the presence of an intravascular device, inflammatory bowel disease, osteomyelitis, and non-O blood group—have been associated with increased risk of CTEPH, primarily in studies comparing patients with CTEPH to individuals with non-thrombotic conditions. Because many of these conditions increase thrombosis risk, it remains unclear whether their association with CTEPH reflects a general effect on thrombosis risk, or a specific effect on the risk of developing CTEPH as an outcome of thrombosis. We performed a case-control study comparing the frequencies of these conditions in patients with CTEPH versus patients with acute PE who did not develop CTEPH. The conditions studied were equally frequent in the CTEPH and PE cohorts, although there was a trend towards an increased frequency of splenectomy and non-O blood group among the CTEPH cohort. Thus, other than the possible exceptions of splenectomy and non-O blood group, the investigated medical conditions do not appear likely to increase the risk of CTEPH as an outcome of acute PE, and thus are unlikely to be useful in predicting CTEPH risk among PE survivors.
Introduction
Chronic thromboembolic pulmonary hypertension (CTEPH) results from chronic obstruction of the pulmonary arteries with thromboembolic material, and is the most serious long term sequela of acute pulmonary embolism (PE). 1 CTEPH occurs in about 3% of PE survivors, 2 but approximately 25% of patients diagnosed with CTEPH have not had a previously recognized acute PE. 3 The pathophysiology of CTEPH is not well understood, and this hinders our ability to predict who is at risk for the disease.
Existing data suggest that CTEPH is significantly underdiagnosed, 4 but at the same time the low frequency of PE survivors who develop CTEPH makes screening unselected PE survivors for CTEPH low yield. One way to improve CTEPH case recognition is to identify risk factors that differentiate patients with acute PE who do not develop CTEPH from patients with acute PE who are more likely to develop CTEPH. That way, acute PE survivors who are predicted to be at the highest risk of developing CTEPH could be targeted for higher intensity follow up. The identification of risk factors differentiating those in whom acute PE resolves from those in whom acute PE leads to CTEPH could also provide significant insight into why thrombus resolution is impaired in acute PE survivors who develop CTEPH.
Several medical conditions have been identified as potential risk factors for CTEPH. These include cancer, 5 splenectomy,5–7 thyroid hormone supplementation, 5 non-O blood group,3,5 inflammatory bowel disease,6,8 osteomyelitis,6,8 and the presence of intravascular devices including totally implantable central venous access catheters (TICVACs), pacemakers, and ventriculo-atrial (VA) shunts, particularly when there is a history of device infection.5,6,8–10 Many of these conditions are known risk factors for venous thromboembolism (VTE) more generally.11–22 Because a prior history of VTE is one of the most important risk factors for CTEPH, these conditions may increase CTEPH risk indirectly through their effects on VTE risk. Alternatively, the presence of one or more of these conditions may exert a specific effect on the development of CTEPH as an outcome of acute PE.
If medical conditions that have been associated with CTEPH have a direct effect on the development of CTEPH as an outcome of PE, rather than just a general effect on the incidence of PE, then one would expect that these conditions would be more frequent in a cohort of patients with CTEPH than in a cohort of patients with otherwise uncomplicated acute PE. Unfortunately, most of these conditions have been identified as potential CTEPH risk factors in studies that did not compare patients with CTEPH to patients with acute PE. For example, cancer, thyroid hormone supplementation, non-O blood group, and the presence of TICVACs have been identified as potential CTEPH risk factors based on case-control studies comparing patients with CTEPH to patients with pulmonary arterial hypertension (PAH).5,9 The data linking infected intravascular devices to CTEPH comes predominantly from uncontrolled retrospective case series.10,23 Only one retrospective case-control study has compared the frequencies of medical conditions in patients with CTEPH versus those with otherwise uncomplicated acute PE. 6 In this study, splenectomy, the presence of a VA shunt for treatment of hydrocephalus, inflammatory bowel disease (IBD), and osteomyelitis were significantly more frequent in patients with CTEPH compared to those with otherwise uncomplicated acute PE, suggesting that these conditions may specifically predispose to CTEPH. 6 However in a follow up case-control study from the same group comparing a larger cohort of CTEPH patients to patients with PAH, the associations of IBD and osteomyelitis with CTEPH were not confirmed. 5
Thus, based on existing data, it is not possible to assess whether many of the medical conditions that have been associated with CTEPH are risk factors for CTEPH specifically, or just associate with CTEPH due to their known influence on VTE risk. Therefore, we assessed the frequency of medical conditions that have previously been associated with CTEPH in a cohort of patients with CTEPH, and in a matched cohort of patients with acute PE who did not develop CTEPH.
Methods
Study Populations
The cohorts included in this study have been described previously. 24 Briefly, we enrolled consecutive patients with a diagnosis of CTEPH who were seen in the pulmonary hypertension clinic at Intermountain Medical Center between 7/1/2015 and 6/30/2019. CTEPH was defined as the presence of pre-capillary pulmonary hypertension confirmed by right heart catheterization (mean pulmonary artery pressure [mPAP] ≥ 25 mm Hg and pulmonary capillary wedge pressure [PCWP] ≤ 15 mm Hg and/or pulmonary vascular resistance ≥ 3 Wood units by right heart catheterization), with evidence of chronic thromboembolic disease by both ventilation/perfusion scintigraphy (V/Q scan) and either or both of computed tomography pulmonary angiography (CTPA) or conventional pulmonary arteriography, after completing at least three months of anticoagulation from the time of the last acute PE.
The control cohort consisted of consecutive patients with a history of acute PE who were not suspected of having CTEPH who were enrolled during scheduled visits at either the thrombosis or pulmonary hypertension clinics at Intermountain Medical Center. To enroll in the study, PE patients had to meet the following inclusion criteria: 1) prior history of objectively documented acute PE (by CTPA or high probability V/Q scan), 2) age ≥ 18 years at the time of enrollment, 3) no clinical signs/symptoms of CTEPH at the time of enrollment (based on a targeted history and physical exam), and 4) no imaging evidence of chronic pulmonary embolism (ie potential subjects were excluded if they had a single CTPA study that was interpreted by the radiologist as showing chronic or acute-on-chronic PE, or serial CTPAs or V/Q scans showing unresolved chronic thromboembolic disease). Because the original study was designed to compare the frequencies of inherited thrombophilias between patients with CTEPH and acute PE, 24 PE subjects were also required to meet individual matching criteria to one or more CTEPH subjects at the time of enrollment. These matching criteria were: 1) age at first diagnosed VTE event (± 5 years), 2) lifetime history of DVT, and 3) any history of unprovoked VTE. This matching process was designed to ensure that the CTEPH and PE cohorts were balanced on criteria known to influence the likelihood of an individual with VTE carrying an inherited thrombophilia.25–27 The following conditions were considered environmental risk factors for VTE, and the absence of all of these defined unprovoked VTE: malignancy that was either active at the time of diagnosis of VTE or diagnosed within 6 months after the diagnosis of VTE, estrogen or testosterone therapy, pregnancy up to 3 months post-partum, surgery requiring general anesthesia in the 3 months prior to diagnosis of VTE, or trauma or medical illness requiring immobilization for ≥3 days in the 3 months prior to diagnosis of the VTE. All CTEPH subjects were matched to at least one, and no more than two, VTE subjects.
Data Collection
At the time of enrollment, all subjects completed a brief survey regarding their histories of relevant medical conditions. This was then corroborated through a detailed review of the subjects’ electronic health records. Data captured included any history of malignancy (excluding non-invasive non-melanoma skin cancers) diagnosed prior to the individual's diagnosis of CTEPH or PE, any history of active malignancy at the time of diagnosis of CTEPH or PE, active therapy with thyroid hormone supplementation at the time of diagnosis with CTEPH or PE, history of splenectomy prior to diagnosis with CTEPH or PE, presence of an intravascular device (TICVAC, tunneled central venous catheter, pacemaker, or VA shunt) at the time of diagnosis of CTEPH or PE, or any history of inflammatory bowel disease or osteomyelitis. Where available, a reported history of splenectomy or intravascular device implantation was corroborated by review of relevant prior imaging studies. Blood group data was obtained from laboratory results in the electronic health record, where available.
Statistical Analyses
The pre-specified analysis in this study was a comparison of the frequency of CTEPH-associated medical conditions between subjects with CTEPH and matched controls with PE. Crude odds ratios were calculated for each comparison, and unadjusted p-values calculated using the Chi-square test for proportions, and then corrected for multiple comparisons. For calculation of adjusted odds ratios, we employed conditional logistic regression stratified by matched case-control blocks to estimate the effect of the medical condition of interest on CTEPH occurrence after adjusting for confounding variables. We used the least absolute shrinkage and selection operator (LASSO) approach to identify confounding variables not used for matching, and their important two-way interaction terms to control. Sex, race/ethnicity, elapsed time since first VTE, history of VTE, recurrent VTE events, family history of CTEPH and BMI were identified as the important confounding variables to control. Although not a part of the primary analysis for this study, the results of genetic testing for the Factor V Leiden and Prothrombin G20210A variants were also included in this dataset, and have been published previously. 24 In that report, the Prothrombin G20210A variant was numerically more frequent in the CTEPH group, but this did not reach statistical significance. 24 In the LASSO analysis for this study, the Prothrombin G20210A variant arose as an additional confounding variable. These selected confounders and their interactions were controlled in the regression using the Covariate Balance Propensity Score approach.
Ethics
The study protocol was approved by the institutional review board at Intermountain Medical Center (IRB_1024723).
Results
We enrolled 91 patients with CTEPH and 157 matched acute PE controls. CTEPH patients had a median age at the time of CTEPH diagnosis of 63 years (Table 1), and 78.0% had been diagnosed with acute PE prior to their diagnosis with CTEPH. At diagnosis, the median WHO functional class among CTEPH patients was 3, and median 6-min walk distance at diagnosis was 411 meters. At the time of publication, 55 (60.4%) CTEPH subjects had undergone PTE surgery, and 3 (3.3%) had undergone balloon pulmonary angioplasty.
Hemodynamic and functional characterization of the CTEPH cohort (n = 91).

Abbreviations: CTEPH: chronic thromboembolic pulmonary hypertension, PE: pulmonary embolism, IQR: interquartile range, WHO: World Health Organization, mPAP: mean pulmonary artery pressure, mm Hg: millimeters of mercury, PCWP: pulmonary capillary wedge pressure, L/min: liters per minute, PVR: pulmonary vascular resistance, PTE: pulmonary thromboendarterectomy, BPA: balloon pulmonary angioplasty.
The demographics of the acute PE cohort closely matched those of the CTEPH cohort (Table 2). Both populations were primarily Caucasian—reflecting the demographics of the state of Utah—and nearly evenly split between sexes. The CTEPH and PE cohorts were closely matched in terms of age at first VTE, history of DVT, and history of unprovoked VTE. Among CTEPH patients who had acute VTE diagnosed prior to their CTEPH diagnosis, the median duration from first VTE to CTEPH diagnosis was 2.0 years (interquartile range 1.3-8.7 years). Among PE subjects, median follow up from first VTE to study enrollment was 2.7 years (interquartile range 0.9-7.5 years).
Demographic and clinical features of the CTEPH and PE cohorts.

Abbreviations: CTEPH: chronic thromboembolic pulmonary hypertension, PE: pulmonary embolism, BMI: body mass index, IQR: interquartile range, VTE: venous thromboembolism, DVT: deep vein thrombosis. P-values reported are for comparison between the CTEPH and PE cohorts, and use the two-tailed Student's t-test for continuous variables and Chi-square test for proportions for binary variables, and were corrected for multiple hypothesis testing.
For each of the conditions analyzed, crude odds ratios for CTEPH and PE were not significantly different (Table 3; the PE cohort was assigned an odds ratio of 1 for reference). After adjusting for confounding variables, the presence of an intravascular device was associated with a significantly decreased odds ratio of CTEPH, while for all other conditions analyzed, adjusted odds ratios remained not statistically different between the two cohorts. Inflammatory bowel disease and osteomyelitis were not included in the multivariable analysis as there were no subjects in the CTEPH cohort with either of these conditions. Among the variables analyzed, only splenectomy and non-O blood group showed a trend towards an increased odds ratio in patients with CTEPH in both the crude and multivariable analyses.
Comparison of the frequency of relevant medical conditions between patients in the CTEPH and PE cohorts.
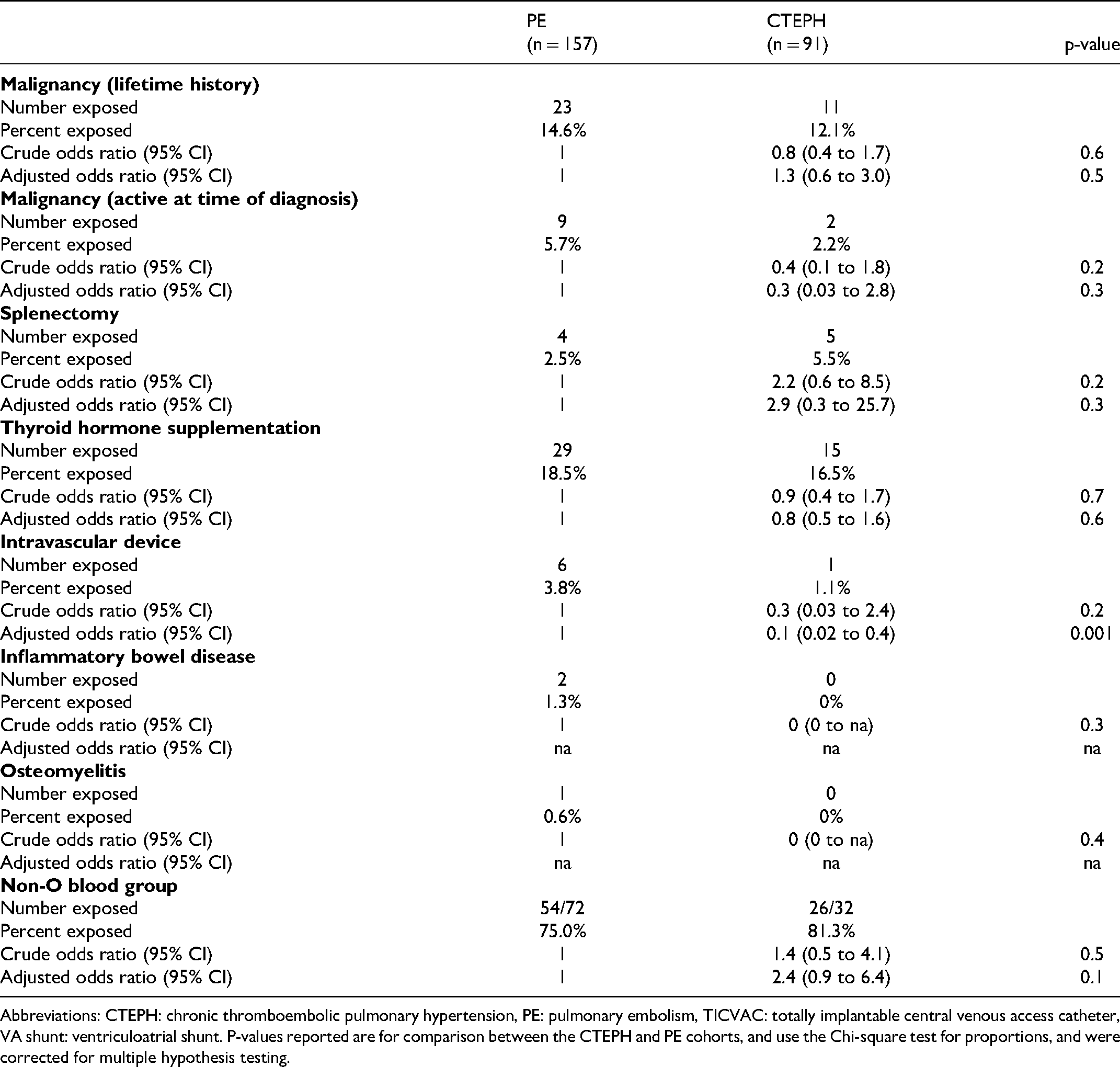
Abbreviations: CTEPH: chronic thromboembolic pulmonary hypertension, PE: pulmonary embolism, TICVAC: totally implantable central venous access catheter, VA shunt: ventriculoatrial shunt. P-values reported are for comparison between the CTEPH and PE cohorts, and use the Chi-square test for proportions, and were corrected for multiple hypothesis testing.
If the presence of one or more of the medical conditions previously associated with CTEPH truly alters risk of CTEPH, then patients with CTEPH who have one of these risk factors may differ from patients with CTEPH who do not have one of these risk factors. Thus, we performed an exploratory subgroup analysis examining clinical features in patients with CTEPH who had a history of one or more of the medical conditions analyzed in this study, compared with CTEPH patients who had none of these conditions. After correction for multiple hypothesis testing, we found no differences between these two cohorts in terms of age at diagnosis with CTEPH, VTE history, PTE status, nor hemodynamics (Table 4). There was a trend toward an increased frequency of prior diagnosis with acute PE among CTEPH patients who had one or more of these conditions, but this was not statistically significant.
Comparison of clinical features between CTEPH patients with and without medical conditions previously associated with CTEPH risk.

Abbreviations: CTEPH: chronic thromboembolic pulmonary hypertension, DVT: deep venous thrombosis, PE: pulmonary embolism, PTE: pulmonary thromboendarterectomy, mPAP: mean pulmonary artery pressure, PVR: pulmonary vascular resistance, WHO: World Health Organization. Conditions counted as “relevant medical conditions' were cancer, splenectomy, hypothyroidism, history of intravascular device, inflammatory bowel disease, osteomyelitis, and non-O blood group. P-values reported are for comparison between the population of CTEPH patients with and without a relevant medical condition, and use the two-tailed Student's t-test for continuous variables and Chi-square test for proportions for binary variables, and were corrected for multiple hypothesis testing.
Discussion
We report the frequencies of medical conditions that have been previously associated with CTEPH in a cohort of patients with CTEPH and in a matched comparison cohort of patients with a history of acute PE who did not go on to develop CTEPH. We find that both the crude and adjusted odds ratios for CTEPH relative to acute PE are no different when considering malignancy (either lifetime history of malignancy or active malignancy at the time of CTEPH diagnosis), splenectomy, thyroid hormone supplementation, and non-O blood group as potential risk factors for CTEPH. Similarly, crude odds ratios were no different for inflammatory bowel disease or osteomyelitis, however these conditions could not be included in the multivariable analysis as no patients in the CTEPH cohort had either condition. Surprisingly, the presence of an intravascular device was associated with a significantly reduced odds ratio for CTEPH in the multivariable analysis. These data suggest that malignancy, thyroid hormone supplementation, intravascular devices, inflammatory bowel disease, and osteomyelitis are unlikely to be risk factors for CTEPH specifically, and more likely have been associated with CTEPH in prior studies due to their effects on increasing VTE risk more generally. While our data did not find a statistically significant increase in either the crude or adjusted odds ratio for CTEPH among patients with splenectomy or non-O blood group, we caution against concluding that these conditions are not specific risk factors for CTEPH as an outcome of acute PE because the relatively small sample sizes in this study limits the statistical power. However, because splenectomy is uncommon and non-O blood group is common in both cohorts, it is unlikely that the presence of either condition—even if demonstrated to be specific CTEPH risk factors in larger cohorts—would have significant predictive power regarding an individual's risk of CTEPH after acute PE.
Although the medical conditions studied have previously been associated with CTEPH, these associations mostly come from studies that have compared patients with CTEPH to controls without a history of VTE.5,9 Because CTEPH is a thrombotic disease, it is not surprising that conditions like malignancy, splenectomy, inflammatory bowel disease, intravascular device placement, and non-O blood group would be identified more commonly in patients with CTEPH than in controls without VTE, as these conditions are known to be VTE risk factors.11–15,19–22 For example, patients who have undergone splenectomy have a risk of VTE that is increased 2- to 3-fold compared to non-splenectomized controls, with the risk of DVT and PE increased equally.11–13 Individuals with non-O blood group have a 1.5- to 1.8-fold higher risk of PE than those with blood group O,14,15 with the relative anti-thrombotic effect of blood group O likely mediated by lower circulating levels of von Willebrand Factor (vWF) and Factor VIII.28,29 Malignancy is a well-established risk factor for VTE, with specific tumor types associated with differential effects on VTE risk.19,20 Inflammatory bowel disease 21 and the presence of intravascular devices, 22 particularly when placed for cancer treatment, are both well-established risk factors for VTE.
The story is more complicated when it comes to thyroid hormone supplementation. In this study, we report a comparable frequency of thyroid hormone supplementation in patients with CTEPH (16.5%) as was reported in European CTEPH patients by Bonderman, et al. (19.9%). 5 In the Bonderman, et al. study, the comparison group was individuals with PAH, among whom 3.5% were on thyroid hormone supplementation, 5 a percentage that closely matches the estimated prevalence of overt hypothyroidism in European adults. 30 The higher than expected frequency of thyroid hormone supplementation in patients with CTEPH in the Bonderman, et al. study suggested that thyroid hormone supplementation is a risk factor for CTEPH, however to our knowledge, no other studies to date have reported the frequency of thyroid hormone supplementation in patients with acute PE. In the present study, in an acute PE population that was closely matched to the CTEPH population on such relevant clinical parameters as age at VTE and whether the VTE was associated with a provoking environmental risk factor, we identified thyroid hormone supplementation in a comparably high percentage of PE patients (18.5%). This unexpected result suggests that, much like many of the other potential CTEPH risk factors evaluated in this study, thyroid hormone supplementation is likely associated with CTEPH because it is associated with increased PE risk. While this result is somewhat unexpected, it is biologically plausible, as disorders of thyroid function have been strongly associated with hemostasis and thrombosis. Hyperthyroidism, for example, is strongly associated with increased VTE risk,31,32 and even levels of free T4 (fT4) that are at the higher end of the normal range seem to increase the risk of thrombosis.17,18 Increased fT4 levels tend to cause increases in vWF and Factor VIII levels, possibly driving the thrombotic risk associated with hyperthyroidism. 33 Hypothyroidism, on the other hand, is associated with reduced vWF and an increased bleeding tendency, 34 although at least one study has reported increased VTE in hypothyroid patients. 16 There is a surprising lack of data regarding thrombosis risk in patients with treated hypothyroidism, but our data suggests that thrombosis risk is likely to be increased in this patient population. Future work will be required to better understand this process.
The main limitation of our study is that it is a single center study with a relatively small sample size, due in large part to the fact that CTEPH is a rare disease. The frequencies of many of the conditions that we analyzed, including splenectomy, intravascular devices, osteomyelitis, and inflammatory bowel disease, were low in both the CTEPH and PE cohorts. Therefore, we cannot rule out that one or more of these conditions may in fact be more common in patients with CTEPH than those with otherwise uncomplicated acute PE if they were to be analyzed in larger cohorts. We postulate that splenectomy is the strongest such candidate. Splenectomy has been previously identified as a CTEPH risk factor both in studies comparing patients with CTEPH to PE controls, 6 and in two independent case control studies comparing patients with CTEPH to patients with PAH.5,7 There is also evidence that splenectomy modifies the clinical features of CTEPH, as prior studies have reported an increased frequency of splenectomy in patients with non-operable versus operable CTEPH,3,35 suggesting that splenectomy may be associated with an increased likelihood of distal relative to proximal chronic thromboembolic disease. Thus, splenectomy may both increase the risk of VTE,11–13 and independently increase the likelihood of developing CTEPH as an outcome of VTE. However, our results make clear that even if splenectomy increases CTEPH risk, it is a risk factor that is present in only a small minority of patients with CTEPH.
In conclusion, the present study suggests that medical conditions including malignancy, thyroid hormone supplementation, intravascular devices, inflammatory bowel disease, and osteomyelitis do not increase the risk of CTEPH as an outcome of acute PE, but rather associate with CTEPH due to their association with increased VTE risk. Thus, it is unlikely that these conditions would be useful to predict development of CTEPH after acute PE.
Footnotes
Funding
This work was supported by the Intermountain Research and Medical Foundation (grant number 20100033).
Author Contributions
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Intermountain Research and Medical Foundation (grant number 20100033).
