Abstract
Objective
Pancreatic ductal adenocarcinoma (PDAC) is the fourth leading cause of cancer-related deaths in Japan. Previous studies from other countries have reported venous thromboembolism prevalence rates of 12 to 36% in patients with pancreatic cancer. In this study, we aimed to determine the incidence of VTE in patients with PDAC in Japan and compare the characteristics of patients with and without VTE.
Methods
In this retrospective cohort study, clinicopathological characteristics of patients with and without concomitant VTE were compared.
Patients
Patients with PDAC treated at Fukui Prefectural Hospital, Japan from 2010 to 2019.
Results
The 1-year survival rate of all patients with pancreatic cancer was 40.7%. Among 432 patients with PDAC, 31 developed VTE. Seventeen (55%) patients received anticoagulant therapy. Compared with the non-VTE group, the VTE group had significantly more patients whose body mass index was >25 kg/m² (p = .035) and had a significantly higher rate of chemotherapy (p = .024). There was no significant difference in median survival time from PDAC diagnosis between the VTE and non-VTE groups. The 6-month mortality rate after VTE diagnosis was 54.8%. PDAC-related death was the most frequent cause of death, and thrombus-related death was not observed.
Conclusion
Several baseline characteristics differed between patients with and without VTE. The incidence of VTE in patients with PDAC is high. However, because the prognosis of PDAC itself remains quite poor, VTE may not have a significant effect on prognosis.
Introduction
In 2004, the Japanese Circulation Society published guidelines for the diagnosis, treatment, and prevention of pulmonary thromboembolism (PE) and deep vein thrombosis (DVT). Since then, venous thromboembolism (VTE) has been diagnosed more frequently in Japan, partly because of the improvements in the recognition and diagnosis of the disease. 1 Increasing attention is being paid to the importance of thrombosis in patients with cancer. In recent years, it has been reported that cancer is associated with a high risk of VTE and VTE is the second leading cause of death in cancer patients. 2 An intricate relationship between cancer and VTE has been demonstrated. Khorana et al. reported that pancreatic cancer is associated with a particularly high risk of VTE. 3 Pancreatic cancer is the fourth leading cause of cancer-related death in Japan, and its prognosis remains poor, with only 8.5% of patients being alive at 5 years after diagnosis. 4 Previous studies have reported VTE prevalence rates of 12 to 36% in patients with pancreatic cancer5-9. Since 2013, primary thromboprophylaxis with a Grade 1B level of evidence has been recommended for patients with locally advanced pancreatic cancer receiving chemotherapy according to the International Initiative on Cancer and Thrombosis clinical practice guidelines. In Japan, patients with pancreatic cancer have poor prognosis, suggesting that VTE is an especially important problem in this patient cohort. However, only a few studies on this topic have been conducted in Japan. In the present study, we aimed to identify characteristics associated with VTE in our data of Japanese patients with pancreatic cancer.
Methods
All procedures were conducted according to the Declaration of Helsinki. This was a retrospective cohort study of patients with pancreatic ductal adenocarcinoma (PDAC) who were diagnosed and treated at the Fukui Prefectural Hospital from 2010 to 2019 and followed up until the terminal phase of life or death. Data of patients with a recorded diagnosis of VTE were extracted according to the following criteria. The VTE group was defined as proven VTE on imaging (eg, contrast-enhanced computed tomography [CT] 10 or echography of the lower limb veins) in patients with a high pretest clinical probability of VTE according to the physician's opinion (eg, because of medical history, symptoms, physical findings) or proven VTE detected coincidentally on contrast-enhanced CT performed for follow-up of cancer or investigation of other diseases. Contrast-enhanced CT was performed for all patients after diagnosing PDAC. All diagnoses that were made based on radiology reports were checked. Clinical characteristics (sex, age, body mass index, smoking status, performance status, comorbidities, cancer stage, biomarkers, types of chemotherapy for PDAC, and cause of death) and long-term outcomes were compared between the VTE and non-VTE groups. Data collected for the VTE group included symptoms, treatment for VTE, and outcomes. Differences between the VTE and non-VTE groups were compared using Fisher's exact test, unpaired t-test, or log-rank test. Cumulative incidence was assessed using Gray's test. All p-values were two sided, and p-values ≤.05 were considered statistically significant. All statistical analyses were performed using EZR (Saitama Medical Center, Jichi Medical University, Saitama, Japan), which is a graphical user interface for R (The R Foundation for Statistical Computing, Vienna, Austria). 11
Results
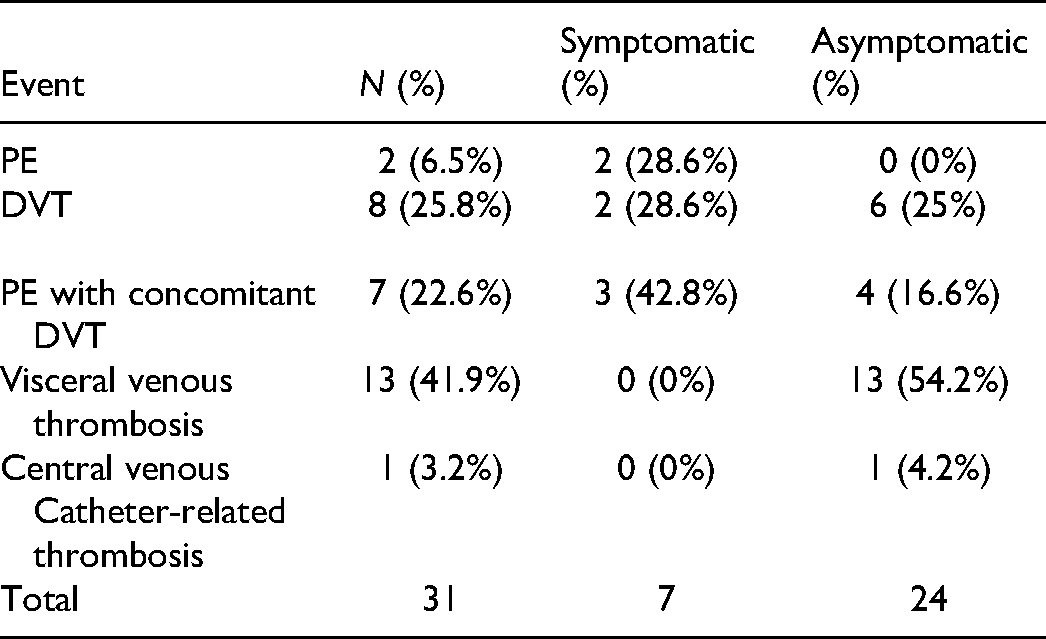
During the 10-year study period, 564 patients with pancreatic cancer were registered at the Fukui Prefectural Hospital; 432 (76.6%) patients with PDAC were followed until the terminal phase of life or death (excluding patients with endocrine tumors or those who visited only for a second opinion or palliative care or were receiving only proton therapy in our proton therapy center). More than half of the patients had metastasis (stage 0, 5 cases [1%]; stage I, 33 cases [8%]; stage II, 113 cases [26%]; stage III, 51 cases [12%]; stage IV, 230 cases [53%]). The 1-year survival rate of all patients with pancreatic cancer was 40.7%. Thirty-one (7.2%) patients developed VTE, with a cumulative incidence of 14.3% at 3 years. Two patients were simultaneously diagnosed with VTE and PDAC. The characteristics of the VTE and non-VTE groups are summarized in Table 1. The VTE group had significantly more patients whose body mass index (BMI) was >25 kg/m² (p = .035) and had a significantly higher rate of chemotherapy (especially with gemcitabine or paclitaxel) (p = .024) than the non-VTE group. There were no significant associations with the following common risk factors: sex, age, smoking, hypertension, stroke, performance status, diabetes, dyslipidemia, congestive heart failure, chronic pulmonary diseases, atrial fibrillation/flutter, liver diseases, metastasis, and biomarkers. Twenty-three patients developed cerebral infarction after PDAC diagnosis. Seventeen (55%) of the 31 patients in the VTE group developed PE or DVT, 13 (42%) developed visceral venous thromboembolism (VVT), and one developed catheter-related thrombosis (Table 2). The median survival time from cancer diagnosis was similar in both the groups (VTE vs non-VTE: 249 vs 249 days, p = .973) (Figure 1). Three hundred eighty-eight (90%) patients died, most frequently from pancreatic cancer itself (including cholecystitis, liver abscess, duodenal hemorrhage associated with pancreatic cancer) (92%), followed by cerebrovascular events (2.8%) and infections not associated with pancreatic cancer (1.5%) (Figure 2). Seven (23%) patients had symptomatic VTE, and two patients had a cardiac arrest. Twenty-four (77%) patients had asymptomatic VTE, all of whom were proven to have VTE detected coincidentally on contrast-enhanced CT performed as part of a preoperative examination or for assessing metastasis or the therapeutic effect of chemotherapy. Amon 15 patients who developed DVT, 10 had central type thrombosis, 4 had peripheral type lower limb vein thrombosis, and 1 had brachiocephalic vein thrombosis. Among 31 patients with VTE, 29 died (all of them died from pancreatic cancer). The median time from PDAC diagnosis to VTE onset was 106 days, the median survival time after VTE onset was 139 days, and the 6-month mortality rate after VTE onset was 54.8%. Table 3 shows treatment for VTE. Seventeen of the 31 patients received anticoagulant therapy. Three patients received warfarin (PT-INR 1.5-2.5), 12 received oral anticoagulants (edoxaban 30 mg or 60 mg once daily [60 mg in patients with body weight {BW} ≥60 kg and 30 mg in those with BW <60 kg]), 2 received only unfractionated heparin, and 5 received an inferior vena cava filter. All patients received the recommended dose of oral anticoagulants, considering BW and creatinine clearance. No symptomatic recurrence of VTE was observed. Anticoagulation therapy was discontinued within 90 days in 47.1% of patients and within 1 year in 88.2% (Figure 3). Besides the terminal phase of life or death, the most frequent cause of anticoagulant termination was bleeding (26%: bleeding associated with direct infiltration of PDAC, including intraperitoneal and gastrointestinal bleeding, or unknown sources of bleeding) (Figure 4). The anticoagulants associated with bleeding events were edoxaban (three cases) and unfractionated heparin (one case).
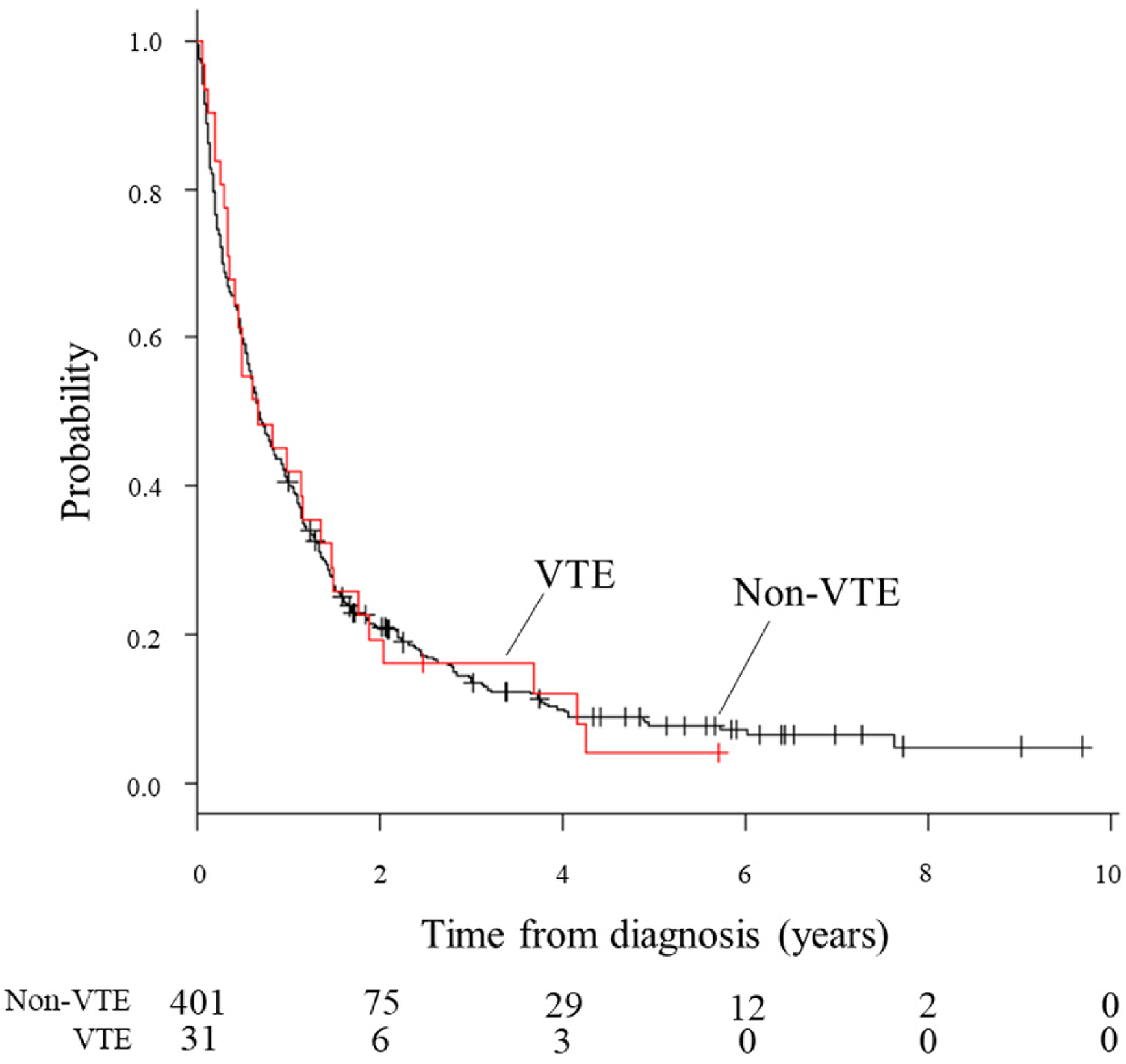
Comparison of survival of pancreatic cancer patients with and without VTE.
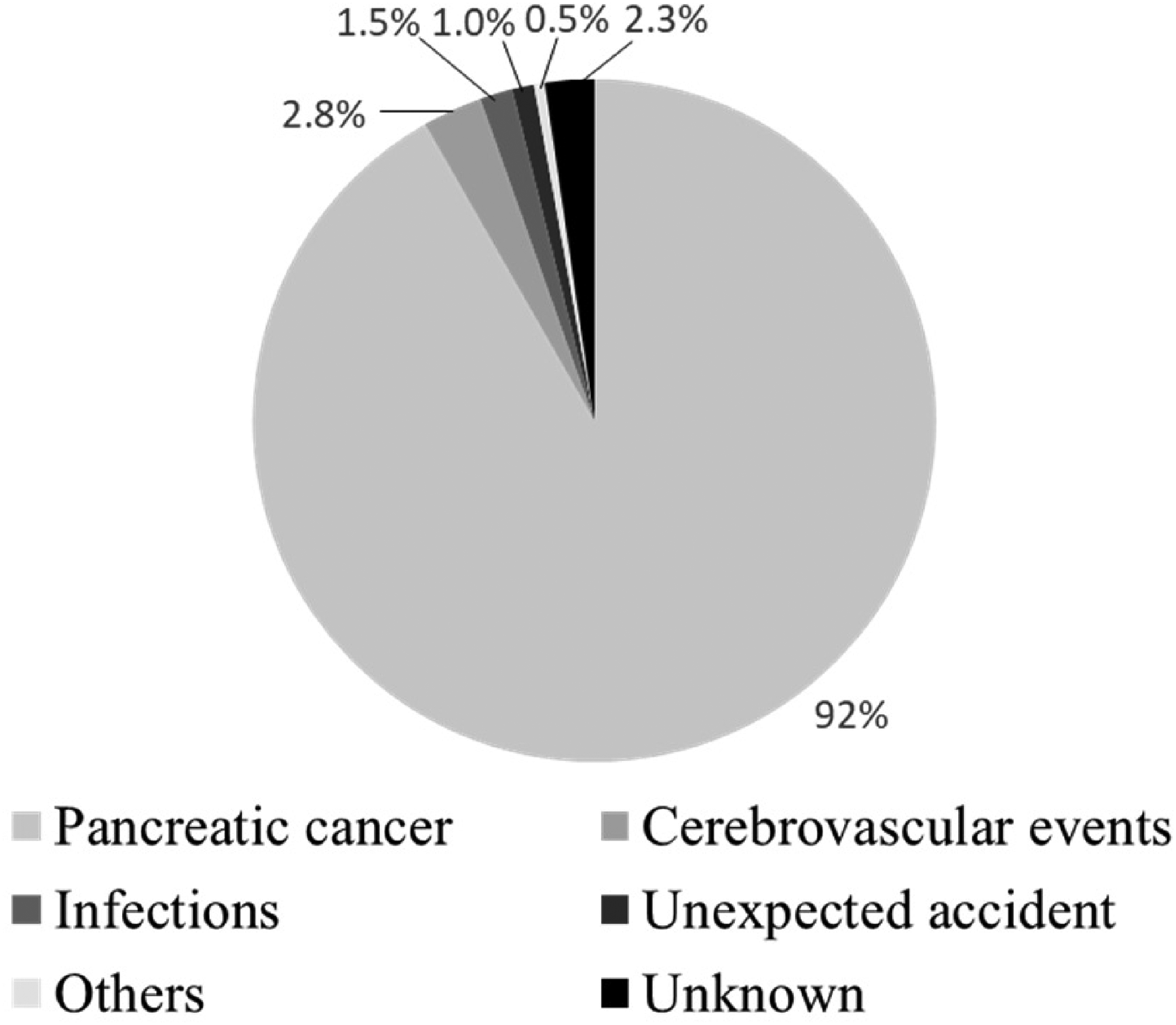
Cause of death of patients with pancreatic cancer Pancreatic cancer includes cholecystitis, liver abscess, and duodenal hemorrhage associated with pancreatic cancer. Infections means not associated with pancreatic cancer.

The proportion of discontinuation of anticoagulation therapy during the first year after VTE diagnosis.

Reasons for discontinuation of anticoagulation therapy.
Baseline characteristics of patients with pancreatic cancer.

VTE: venous thromboembolism.
S-1: Tegafur, Gimeracil, Oteracil Potassium.
Types of venous thromboembolism events in this study.
Treatment for VTE.

*¹ One of the 18 patients was received only IVC filter placement, and 4 patients received IVC filter placement and anticoagulation therapy.
Discussion
In a report from the United States, 20.79% of patients with PDAC developed VTE, and the cumulative incidence of VTE was 8.07% at 3 months and 19.21% at 12 months. The median time from PDAC diagnosis to VTE onset was 4.49 months, and patients who developed VTE during the follow-up had shorter progression-free survival (PFS) and overall survival (OS) (median: PFS, 6.66 months vs 9.56 months; OS, 9.13 months vs 14.55 months). 12 Additionally, in an Asian study from Korea, the overall incidence of pancreatic cancer-associated VTE was similar to that reported in studies from Western countries. Compared with patients in the non-VTE group, patients who were simultaneously diagnosed with VTE and PDAC had poorer prognosis (median OS: 2.1 months vs 10.7 months, p < .001). 13 In contrast, two monocentric cohorts of patients with pancreatic cancer found no difference in OS between patients who developed VTE and those who did not.14,15 In this study, the incidence of VTE in Japan was relatively lower when compared to Western countries. Khorana et al. 3 reported that obese individuals had a significantly higher incidence of VTE. Therefore, the lower incidence of VTE may be attributed to the relatively lower prevalence of obesity in Japan. The COMMAND VTE Registry is a multicenter registry of Japanese patients with cancer who have symptomatic VTE. Yamashita et al. reported that the 1-year mortality rate for all patients in the registry after a diagnosis of VTE was 49.6%. 16 The 6-month mortality rate in our study was higher than the 1-year mortality in the COMMAND VTE Registry. Sakamoto et al. reported that advanced cancer and chemotherapy were found to be high-risk factors for VTE. 17 In the present study focusing on patients with PDAC, we found that a significantly higher proportion of patients in the VTE group (n = 31) than in the non-VTE group (n = 401) had a BMI over 25 kg/m² and were treated with chemotherapy. Obesity and chemotherapy were confirmed as risk factors for VTE. Gemcitabine has been reported to show increased tissue factor and decreased tissue factor pathway inhibitor activity. Moreover, patients with pancreatic cancer receiving gemcitabine-based palliative chemotherapy developed VTE in 10.6% of cases and had significantly shorter OS (3.7 months vs 6.4 months, p = .005). 18 In this study, OS was shorter (301 vs 398 days, p = .557), but there was no significant difference. This may also mean that gemcitabine prolongs the prognosis of Japanese patients, even if they develop VTE. Folfirinox has also been reported to result in a risk of thrombosis; however, our study could not prove it because of the small number of patients being treated with folfirinox. In the Khorana risk score, platelet count of 35 × 104/µL or more and leukocyte count of more than 11 × 103/µL or hemoglobin level less than 10 g/dL were predictors of thrombosis. In the present study, we did not find significant associations between baseline biomarkers and VTE. This finding may not support the predictive ability of the Khorana risk score. 3
Several studies have found an association between VTE onset and poorer prognosis. In contrast, Figure 3 shows survival curves according to the VTE status during our observation period. After PDAC diagnosis, the survival rate in the VTE group was similar to that in the non-VTE group. Additionally, special attention should be paid to the possibility of upper gastrointestinal bleeding or intra-abdominal bleeding owing to direct infiltration during anticoagulant therapy. Current guidelines recommend that patients with VTE, because of reversible factors, such as lower limb fixation, pregnancy, and prolonged sitting, should receive anticoagulant therapy for only 3 months to diminish the risk of bleeding and development of side effects. However, it is recommended that patients with cancer receive long-term anticoagulant therapy (until the cancer is cured) to minimize VTE recurrence. 19 In the present study, the most frequent reason for discontinuing anticoagulant therapy, besides the terminal phase of life or death, was bleeding. Most patients did not continue anticoagulant therapy for more than 1 year. In particular, bleeding from the digestive tract should be considered in patients with PDAC undergoing anticoagulant therapy. Therefore, the danger of continuous anticoagulation in patients with PDAC, even those at a very high risk of VTE, is not trivial. In this study, about half of the VTE cases developed VVT, such as portal vein thrombosis. There is no consensus regarding the necessity of anticoagulant therapy for VVT. However, some retrospective studies focusing on the association between VVT and survival also found an association between the onset of VVT and increased mortality.20,21 In this study, 9 patients with VVT did not receive anticoagulant therapy because of bleeding or terminal stage, and there was no significant difference in the OS (222 vs 249 days, p = .390). This retrospective study of patients with PDAC found no difference in OS between patients who developed VTE (PE, DVT, or VVT) and those who did not. This lack of difference in survival between both the groups might be explained by the patient's short prognosis because the stage of the disease in more than half of the patients with PDAC is almost already advanced at the time of diagnosis. This may also explain why there was no significant difference in the number of patients with metastasis. Although, as with other types of cancer, if the prognosis of pancreatic cancer patients improves with the improvement of diagnostic ability and treatment for pancreatic cancer in the future, the effect of VTE on the prognosis will be more important, and it may be necessary to discuss anticoagulant prophylaxis. However, at the time of this study, it was noted that VTE had little effect on the prognosis of patients with pancreatic cancer.
Our study has some limitations. This was a single-center retrospective study that was performed using data from our electronic medical record system and was specific to patients diagnosed with and treated for both PDAC and VTE at our hospital. The mortality of pancreatic cancer was close to what had already been reported in the literature. The vast majority of VTE events were incidental or asymptomatic. Although all patients in this study were diagnosed or judged for therapeutic effect using contrast CT, the incidence of VTE may have been underestimated because regular screening for VTE was not performed.
Conclusions
We report the results of this retrospective study of patients with PDAC who developed VTE. Several baseline characteristics differed between patients in the VTE and non-VTE groups. The 6-month mortality rate after VTE onset was high; however, this retrospective study of patients with PDAC found no difference in OS between patients who developed VTE and those who did not. This lack of difference in survival between both the groups might be explained by the patient's short prognosis because the stage of the disease in more than half of the patients with PDAC is almost already advanced at the time of diagnosis. In this situation, VTE may not be a prognostic factor for Japanese patients with PDAC.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
