Abstract
Lung cancer is the leading cause of death from cancer in Japan. Studies in other countries have reported a venous thromboembolism (VTE) rate of 4%–20% in cancer patients. In this study, we aimed to determine the incidence of VTE in lung cancer patients in Japan and compared the characteristics of patients with and without VTE. In this retrospective cohort study, the clinicopathological characteristics of study patients with and without concomitant VTE were compared. Patients with lung cancer treated at Fukui Prefectural Hospital, Japan from 2008 to 2017. Of the 1471 patients with lung cancer studied, 28 developed VTE. Five patients developed pulmonary thromboembolism (PTE) alone, 9 PTE with concomitant deep vein thrombosis, and 14 deep vein thrombosis alone. Compared with patients in the non-VTE group, the VTE group was significantly younger (mean value ± SD 66.3 ± 10.1 vs. 73.0 ± 10.6 years, p = 0.001), contained significantly more patients with stage IIIb–IV disease (p = 0.002), and had a significantly higher rate of chemotherapy (p < 0.001) and radiation therapy (p = 0.007). There was no significant difference in median survival time from lung cancer diagnosis between the VTE and non-VTE groups. The 1-year mortality rate after VTE diagnosis was 60.7%. Lung cancer was the most frequent cause of death, followed by infection and VTE. Several baseline characteristics differed between patients with and without VTE. The prognosis may worsen after development of VTE, suggesting that lung cancer patients should be carefully monitored for it.
Introduction
The Japanese Circulation Society published guidelines for the Diagnosis, Treatment, and Prevention of Pulmonary Thromboembolism (PTE) and Deep Vein Thrombosis (DVT) in 2004. Since then, venous thromboembolism (VTE) has been diagnosed more frequently in Japan, partly as a result of improvements in recognition and diagnosis of the disease. 1 Increasing attention is being paid to the importance of thrombosis in cancer patients. Studies in countries other than Japan have reported a VTE incidence rate of 4%–13% in lung cancer patients. 2 -5 Khorana et al. reported that lung cancer is associated with a particularly high risk of VTE. 6 In Japan, lung cancer is the leading cause of death among cancer patients, 7 suggesting that VTE may be an especially important problem in this patient cohort. However, few studies on this topic have been conducted in Japan. In the present study, we sought to identify some of the characteristics associated with VTE in Japanese lung cancer patients.
Methods
All procedures were conducted in accordance with the Declaration of Helsinki. This was a retrospective cohort study of patients with lung cancer who were treated at the Fukui Prefectural Hospital from 2008 to 2017. The follow-up period was 3 years or more after diagnosis of lung cancer. Data for patients with a recorded diagnosis of VTE were extracted in accordance with the following criteria. VTE was defined as proven VTE by imaging (e.g., contrast-enhanced computed tomography [CT], 8 or echography of the lower limb veins, or mismatch of lung ventilation/perfusion scintigraphy) in patients with a high pretest clinical probability of VTE in the physician’s opinion (e.g., because of medical history, symptoms, physical findings, or Wells score), or proven VTE detected coincidentally by contrast-enhanced CT performed for follow-up of cancer or investigation of other diseases. Contrast-enhanced CT anywhere from the chest to lower limbs had been performed on 654 patients (44.5%) after diagnosis of lung cancer. All diagnoses made on the basis of radiology reports were checked. Wells scores of ≥3 for DVT and ≥2 for PTE were considered to indicate high risk. 9,10 Clinical characteristics (sex, age, body mass index, smoking status, performance status, comorbidities, lung cancer stage, biomarkers, histological type, and treatments for lung cancer) and long-term outcomes were compared between the VTE group and non-VTE group. Disease stage was grouped into stages I–IIIa, which were treated mainly with surgery, and stages IIIb–IV, which were treated mainly with radiation therapy or chemotherapy. 11,12 Data collected in the VTE group included symptoms, severity of PTE, 13 treatments for lung cancer and VTE, and outcomes (including survival after VTE onset, cause of death, and recurrence rate of symptomatic VTE within 1 year, 14 anticoagulation interruption rate, and hemorrhagic complications). Hemorrhagic complications were assessed according to the Bleeding Academic Research Consortium [BARC] definitions, 15 which clearly delineate the degree of bleeding, including minor bleeding. Patient data were analyzed for fatal or clinically severe bleeding events requiring blood transfusion or surgical intervention, or intracranial hemorrhage. Differences between the VTE and non-VTE groups were compared with Fisher’s exact test, unpaired t-test, Mann–Whitney U test, or log-rank test. Cumulative incidence was assessed using Gray’s test.
Results
During the 10-year study period, 1746 patients with lung cancer were enrolled at the Fukui Prefectural Hospital, 1471 (84.2%) of whom had been followed for more than 3 years or until the patient died. The 3-year survival rate of all lung cancer patients was 34.5%. Twenty-eight patients developed VTE, its cumulative incidence being 2.5% at 3 years. The characteristics of the VTE and non-VTE groups are summarized in Table 1. The VTE group, 57% of whom were men, were significantly younger than the non-VTE group at diagnosis of lung cancer (mean ± SD 66.3 ± 10.1 vs. 73.0 ± 10.6 years, p = 0.001). More patients in the VTE than the non-VTE group had IIIb–IV disease and had undergone chemotherapy or radiation therapy. The incidence of VTE in patients with small cell lung cancer was the highest (5.56%), however, the significant difference in the incidence of VTE in patients with any histologic subtypes was not confirmed by multivariate logistic regression independently of stage, PS, chemotherapy and radiation therapy (Adenocarcinoma: p = 0.830, Squamous cell carcinoma: p = 0.167, Large cell carcinoma: p = 0.990, SCLC: p = 0.233).
Baseline Characteristics of Patients With Lung Cancer.

Data are presented as the number (%) except as indicated.
Stage according to the Tumor, Node, Metastasis classification system for lung cancer, 7th edition (11).
VTE, venous thromboembolism.
NSCLC, non-small cell lung cancer; SCLC, small cell lung cancer.
There were no significant associations with the following common risk factors: body mass index, diabetes, hypertension, dyslipidemia, stroke, chronic heart failure, chronic respiratory disease, atrial fibrillation, smoking, and performance status. However, baseline leukocyte and platelet counts were found to have a significant association with VTE. The median survival time from cancer diagnosis was shorter in patients with VTE (420 vs. 531 days, p = 0.348); this difference was not significant. Five of the 28 patients in the VTE group (18%) developed PTE alone, 9 (32%) PTE with concomitant DVT, and 14 (50%) DVT alone, which involved the superior vena cava, jugular, subclavian, or great saphenous vein. Twenty-one patients had symptomatic VTE, the commonest symptoms being dyspnea in 12 patients and edema in 9; 7 patients had asymptomatic VTE. Of the 23 patients with symptomatic VTE, 15 (11 with PTE, 4 with DVT) had a high pretest clinical probability of VTE according to their Wells scores. The 7 patients with asymptomatic VTE were diagnosed by contrast-enhanced CT performed as part of the preoperative examination or for assessment of the therapeutic effect of chemotherapy or of metastasis. PTE severity was non-massive in 8 patients, sub-massive in 4, massive in one, and one patient had a cardiac arrest. Of the 23 patients who developed DVT, 10 had central type thrombosis, 6 had peripheral type lower limb vein thrombosis, 1 had great saphenous vein thrombosis (with concomitant PTE), 3 had superior vena cava thrombosis, 2 had subclavian vein thrombosis, and 1 had jugular vein thrombosis. There were no patients in whom stage progressed between the diagnosis of lung cancer and onset of VTE; however, in assessment of the effect of anticancer therapy, 20 patients were judged to have progressive disease at the onset of VTE.
The most frequent means of diagnosis was contrast-enhanced CT (26 patients). Of the 28 VTE patients, 21 have since died, most frequently from lung cancer itself (66.7%), followed by infection (14.2%) and VTE (9.5%). The median time from diagnosis of lung cancer to VTE onset was 227 days; the median survival time after VTE onset was 135 days; and the 1-year mortality rate after VTE onset was 60.7%. Fourteen of the 28 patients with VTE had received platinum-based chemotherapy for lung cancer, the median time to VTE from the start of platinum-based chemotherapy being 37 days (range, 13–721 days). Twenty-six of the 28 patients had received anticoagulant therapy. Eleven patients received warfarin (PT-INR from 1.5 not exceed to 2.5), 15 received oral anticoagulants (14 edoxaban 30 mg or 60 mg once daily (60 mg in patients with body weight (BW) ≥ 60 kg and 30 mg in those with BW < 60 kg), one rivaroxaban 15 mg twice daily), and 1 received only unfractionated heparin. All patients had received the recommended dose of oral anticoagulants considering BW and creatinine clearance. Fondaparinux sodium 2.5 mg once daily was co-administered to 3 patients. No symptomatic VTE recurrence was identified. Anticoagulation therapy was discontinued within 90 days 38.5% of patients and within 1 year in 69.2% (Figure 1). Apart from death, the most frequent cause of anticoagulant termination was bleeding (airway, gastrointestinal tract, and brain metastases bleeding) (Figure 2). The anticoagulants associated with bleeding events were warfarin (3 patients), edoxaban (two), and unfractionated heparin (one). All bleeding events were classified as BARC type 2. No patients experienced BARC type 3 or higher bleeding with fatal or clinically severe bleeding.
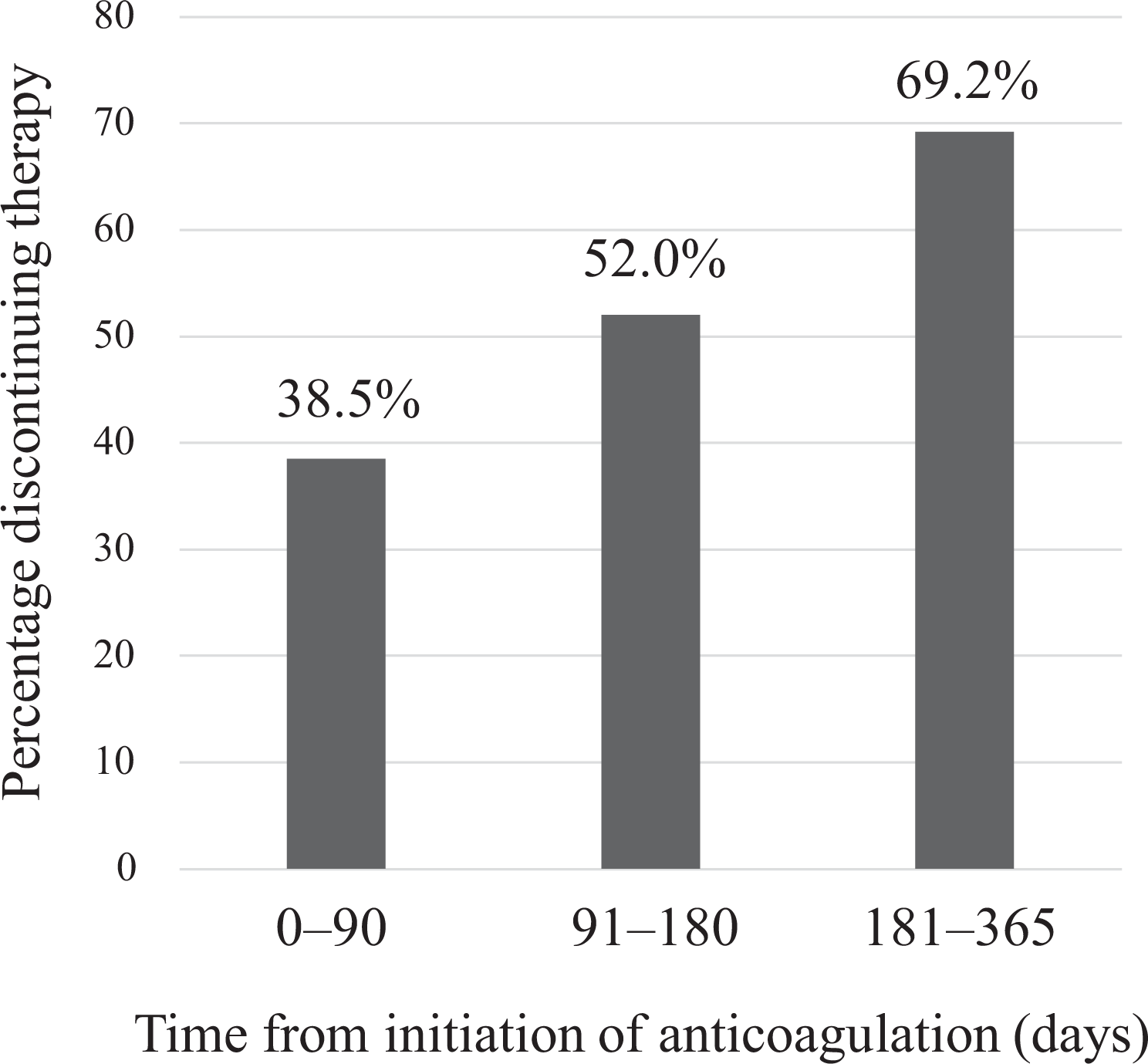
The proportion of discontinuation of anticoagulation therapy during the first year after VTE diagnosis.

Reasons for discontinuation of anticoagulation therapy.
Discussion
The overall incidence of lung cancer-associated VTE in the present study of Japanese patients was lower than the incidences reported in studies in other countries. The COMMAND VTE Registry is a multicenter registry of Japanese patients with cancer who have symptomatic VTE. Yamashita et al. reported that the 1-year mortality rate for all patients in the registry after a diagnosis of VTE was 49.6%, and Sakamoto et al. reported that advanced cancer and chemotherapy were found to be high risk factors for VTE. 16,17 In the present study focusing on patients with lung cancer, we found that a significantly higher proportion of patients in the VTE group (n = 28) than the non-VTE group (n = 1443) had advanced stage disease and were treated with chemotherapy. Chemotherapy and radiation therapy were confirmed to be risk factors for VTE by multivariate logistic regression independently of stage and PS (Chemotherapy: p = 0.011, radiation therapy: p = 0.013). It may be the increased use of chemotherapy or radiation therapy in patients with advanced stage of disease that leads to VTE. The 1-year mortality rate in our study was higher than that in the COMMAND VTE Registry. We observed no cases of cancer stage progression between the diagnosis of lung cancer and the onset of VTE in this study, which may not be only because many patients already had advanced lung cancer at VTE onset, but also suggests that VTE can occur even at the early stages of cancer. In the Khorana risk score, platelet count of 35 × 10(4)/µL or more and leukocyte count more than 11 × 10(3)/µL are predictors of thrombosis. In the present study, we found significant associations between baseline leukocyte and platelet counts and VTE; this finding may partially support the predictive ability of the Khorana risk score. 6 The significant difference in median leukocyte and platelet counts in VTE patients and non-VTE patients was also confirmed by Mann–Whitney U test (hemoglobin: 13.5 vs 12.8 g/dl, p = 0.112, leukocyte: 7.4 vs 6.4 ×10(3) /µL, p = 0.030, platelet: 26.3 vs 21.7 ×10 (4) /µL, p = 0.003).
Figure 3 shows survival curves according to VTE status during the observation period. After diagnosis of lung cancer, the survival rate was initially better in the VTE than non-VTE group; however, this pattern was reversed after about 1 year of follow-up. Patients who are relatively young are more likely to be treated with chemotherapy or radiation therapy after diagnosis of lung cancer, and development of VTE early after anticancer therapy may result in a worse prognosis. Kanaji et al. reported an incidence of thrombosis, including arterial thrombosis, of 2.2% (16/716) in patients with lung cancer and the thrombosis group had a shorter overall survival time than the non-thrombosis group (280 vs. 538 days, p = 0.0454). 18 In the present study focusing on venous thrombosis, the overall survival time of the non-thrombosis group was comparable, however, the difference in median survival time between the VTE and non-VTE groups was not statistically significant. This suggests that venous thrombosis with anticoagulant therapy does not have a worse prognosis than arterial thrombosis. It has been reported that most VTE events occur within the first 100 days after starting platinum chemotherapy. 5 Thus, the above data indicate that special care should be taken, not only to prevent VTE, but also to detect and treat it early in younger patients and those with advanced lung cancer and/or receiving anticancer therapy.

Comparison of survival of lung cancer patients with and without VTE.
Additionally, special attention should be paid to the possibility of bleeding during anticoagulant therapy. Current guidelines recommend that patients who have VTE because of reversible factors, such as lower limb fixation, pregnancy, and prolonged sitting, receive anticoagulant therapy for only 3 months to diminish the risk of bleeding and development of side effects. However, it is recommended that cancer patients receive longer term anticoagulant therapy (until the cancer is cured) to minimize recurrence of VTE. 19 In the present study, the most frequent reason for discontinuing anticoagulant therapy, apart from terminal phase of life or death, was bleeding. Many patients did not continue this treatment beyond 1 year. In particular, we need to consider bleeding from airways or brain metastases in lung cancer patients undergoing anticoagulant therapy. Additionally, cancer patients may have cytopenia associated with anticancer drug therapy, and dysfunction of other organs. Therefore, the danger of continuous anticoagulation in lung cancer patients, even those at high risk of VTE, is not trivial.
Our study has some limitations. It was a retrospective study using data from our electronic medical record system and is specific to patients who have been diagnosed with, and treated for, both lung cancer and VTE at our hospital. Our findings are not necessarily applicable to other patient cohorts. Additionally, because only 44.5% of patients underwent contrast-enhanced CT, the rate of asymptomatic VTE may have been underestimated. A prospective study of VTE in lung cancer patients is necessary to verify our findings.
Conclusions
We report here the results of a retrospective study of lung cancer patients who developed VTE. Several baseline characteristics differed between patients in the VTE and non-VTE groups. The 1-year survival rate after VTE onset was 60.7%, and the prognosis may worsen after development of VTE. Guidelines (19) recommend long-term anticoagulant therapy to prevent recurrence in cancer patients with VTE; however, bleeding events were the biggest impediment to continued therapy in our patient cohort. Because development of VTE is not rare in lung cancer patients, special attention should be paid to this risk in this patient group.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
