Abstract
To evaluate associations among coagulation-related variables, resolution of disseminated intravascular coagulation (DIC) and mortality, we retrospectively investigated 123 patients with sepsis-induced DIC treated with recombinant human soluble thrombomodulin (rTM). Changes in coagulation-related variables before and after treatment with rTM were examined. Further, associations between coagulation-related variables and DIC resolution were evaluated. The platelet count, prothrombin international normalized ratio (PT-INR), and fibrin/fibrinogen degradation products (FDP) significantly (p < .001) improved after rTM administration in survivors (n = 98), but not in nonsurvivors (n = 25). However, the DIC score significantly (p < .001) reduced in survivors and in nonsurvivors. Among coagulation-related variables examined before rTM, only PT-INR was significantly (p = .0395) lower in survivors than in nonsurvivors, and PT-INR before rTM was significantly (p = .0029) lower in patients attaining than not attaining DIC resolution (n = 87 and 36, respectively). The 28-day mortality was significantly lower in patients attaining than not attaining DIC resolution (11.5% vs 41.7%, p = .0001). In conclusion, the initiation of rTM administration before marked PT-INR elevation may be important to induce DIC resolution and thus to decrease mortality in patients with sepsis-induced DIC. Conversely, the treatment with rTM in patients with marked PT-INR elevation may be not so effective in achieving such goals.
Keywords
Introduction
Sepsis is the most common cause of disseminated intravascular coagulation (DIC), which leads to organ dysfunction through intravascular thrombosis.1,2 DIC is characterized by systemic activation of coagulation, which induces the intravascular fibrin deposition and microvascular thrombosis. Subsequently, the ongoing activation of coagulation results in severe depletion of coagulation factors and platelets, which induces diffuse and ongoing bleeding.1,2 There are two phases in DIC which are the early phase – hypercoagulation, and the late phase – diffuse bleeding from various sites. Inflammation deregulates the coagulation cascade and leads to various coagulopathies including DIC in septic patients.2–4 The essential therapy in patients with sepsis is treatment of underlying infection using appropriate antibiotics and source controls. However, DIC can persist or develop even after the initiation of appropriate therapy. In such situations, supportive therapy to manage DIC and to modulate cross-talk between inflammation and coagulation may reduce morbidity and mortality in patients with sepsis. 4
Thrombomodulin (TM), which is localized in the vascular endothelium, acts as an anticoagulant. After it binds to thrombin, the thrombin-TM complex activates protein C to produce activated protein C, which inactivates factors Va and VIIIa, thereby inhibiting formation of thrombin (IIa).5,6 Furthermore, TM attenuates inflammation through various mechanisms.7,8
A phase III randomized controlled trial (RCT) comparing efficacies of recombinant human soluble TM (rTM) and heparin in patients with DIC showed that rTM improved DIC and relieved bleeding symptoms more effectively than heparin. 9 Accordingly, rTM was approved as an agent for treatment of DIC in 2008 in Japan, and has since been used clinically. A multicenter propensity score-matched analysis of sepsis-induced DIC reported that rTM reduced in-hospital mortality in patients requiring mechanical ventilation. 10 In addition, a multicenter retrospective study comparing 452 propensity score-matched pairs of septic patients with DIC revealed that rTM significantly reduced in-hospital mortality. 11 Although the Sepsis Coagulopathy Asahi Recombinant LE Thrombomodulin (SCARLET) trial performed as a multicenter RCT found that rTM did not significantly reduce 28-day mortality compared with a placebo in patients with sepsis-associated coagulopathy, 12 a post-hoc analysis of this trial revealed that the subgroup of rTM-treated patients with higher baseline levels of prothrombin fragment 1.2 and thrombin-antithrombin complex had lower mortality than placebo-treated patients. 13 Therefore, rTM may play an important role in improving coagulopathy and mortality in septic patients with DIC.
The aim of the present study was to evaluate associations among inflammation- or coagulation-related variables, resolution of DIC, and mortality in patients with sepsis-induced DIC treated with rTM.
Methods
Study Design and Subjects
Prior to this retrospective observational study, the study protocol was approved by the Ethics Committee of Juntendo University Hospital (approval number H19-045), with a waiver of patients’ written informed consent.
We retrospectively studied consecutive patients who were diagnosed with sepsis-induced DIC and treated with rTM in the intensive care unit at Juntendo University Hospital between April, 2015 and December, 2019. Sepsis was defined according to the Sepsis-3 criteria. 14 Sepsis is defined as life-threatening organ dysfunction caused by a dysregulated host response to infection. For clinical operationalization, organ dysfunction can be identified as an acute change in total Sequential Organ Failure Assessment (SOFA) score 15 ≥ 2 points due to the infection. 14 Septic shock is a subset of sepsis, and it can be identified by requiring vasopressors to maintain a mean arterial pressure ≥ 65 mm Hg and serum lactate level > 2 mmol/L in the absence of hypovolemia. 14 DIC was diagnosed based on the DIC scoring system by the Japanese Association for Acute Medicine (JAAM). 16 JAAM's diagnostic criteria for DIC requires two-step checking before scoring. The first step is to prove the presence of diseases that can cause DIC. The target patients of this study were patients with sepsis. The second step is to rule out any coagulopathy similar to but different from DIC. Clinical conditions that we carefully ruled out included A) thrombocytopenia due to 1) dilution and abnormal distribution, massive blood loss and transfusion, massive infusion, 2) increased platelet destruction (idiopathic thrombocytopenic purpura, thrombotic thrombocytopenic purpura/hemolytic uremic syndrome, heparin-induced thrombocytopenia, drugs, viral infection, alloimmune destruction, antiphospholipid syndrome, HELLP syndrome, extracorporeal circulation), 3) decreased platelet production (viral infection, drugs, radiation, nutritional deficiency such as vitamin B12 and folic acid, disorders of hematopoiesis, liver disease, hemophagocytic syndrome), 4) spurious decrease (EDTA-dependent agglutinins, insufficient anticoagulation of blood samples, and 5) others (hypothermia, artificial devices in the vessel); B) prolonged prothrombin time due to anticoagulation therapy, anticoagulant in blood samples, vitamin K deficiency, liver cirrhosis, massive blood loss and transfusion; C) elevated FDP due to thrombosis, hemostasis and wound healing, hematoma, pleural effusion, ascites, anticoagulant in blood samples, and antifibrinolytic therapy; and D) others. After coagulopathy as stated above were excluded, DIC was diagnosed based on a DIC score ≥ 4 points on the JAAM's DIC score, 16 which is the sum of the points (graded 0 to 3) for the following four components: A) systemic inflammatory response syndrome (SIRS) criteria ≥ 3 is scored as 1; B) a platelet count ≥ 80 and < 120 × 109/L or a > 30% decrease within 24 h is scored as 1, a platelet count < 80 × 109/L or a > 50% decrease within 24 h is scored as 3; C) a prothrombin international normalized ratio (PT-INR) ≥ 1.2 is scored as 1; and D) a fibrin/fibrinogen degradation products (FDP) level ≥ 10 and < 25 µg/mL is scored as 1, and a FDP level ≥ 25 µg/mL is scored as 3.
rTM at a dose of 380 U/kg was intravenously infused for 30 min once a day, which was usually continued for 6 days or until improvement of DIC was achieved. In patients with severe renal dysfunction (estimated glomerular filtration rate < 30 mL/min/1.73 m2), the dose was reduced to 130 U/kg, considering its elimination by the kidneys.
Data Collection
Data on patients’ clinical characteristics and laboratory blood tests were collected from medical records, including age, sex, the outcome (28-day mortality), the Acute Physiology and Chronic Health Evaluation II (APACHE II) score, 17 the SOFA score, 15 the incidence of septic shock, requirements of mechanical ventilation and/or continuous renal replacement therapy, the DIC score, 16 the platelet count, PT-INR values, levels of FDP and C-reactive protein (CRP), requirements of surgical interventions, sites of infection, infecting microorganisms, the presence of documented bacteremia, and the dose and duration of rTM administration.
Resolution of DIC was defined as the DIC score ≤ 3 on the day after the last treatment with rTM. 18 To assess the efficacy of rTM, coagulation- and inflammation-related variables before and after treatment with rTM were examined and compared between survivors and nonsurvivors. In addition, these variables before rTM were compared between patients attaining and not attaining DIC resolution.
Statistical Analysis
Qualitative data are expressed as numbers (percentages) and were analyzed using the chi-square test. Quantitative data are expressed as medians [interquartile ranges: IQRs]. Changes in variables over time were analyzed with the Wilcoxon signed-rank test. Variables were compared between dichotomized groups with the Mann-Whitney U test. A p value less than .05 was considered statistically significant.
Results
Patient Characteristics
A total of 123 patients were diagnosed with sepsis-induced DIC and treated with rTM. Baseline clinical characteristics in these patients are shown in Table 1. The 28-day mortality rate in the total cohort was 20.3% (25/123). The APACHE II score, the SOFA score, and requirements of renal replacement therapy were significantly (p < .05) lower in survivors than in nonsurvivors. However, other variables, including age, sex, the incidence of septic shock, requirements of mechanical ventilation, requirements of surgery, sites of infection, infecting microorganisms including antibiotic-resistant bacteria, and documented bacteremia, were not different between survivors and nonsurvivors. There was no patient receiving antithrombotic agents, such as warfarin, for co-existing disease. Heparin was not used except for arterial line to monitor arterial pressure in which heparin was continuously infused at a rate of 6 U/h. Because of the extremely low dose, the systemic anticoagulant effect of heparin could be negligible.
Baseline characteristics in patients with sepsis-induced DIC.
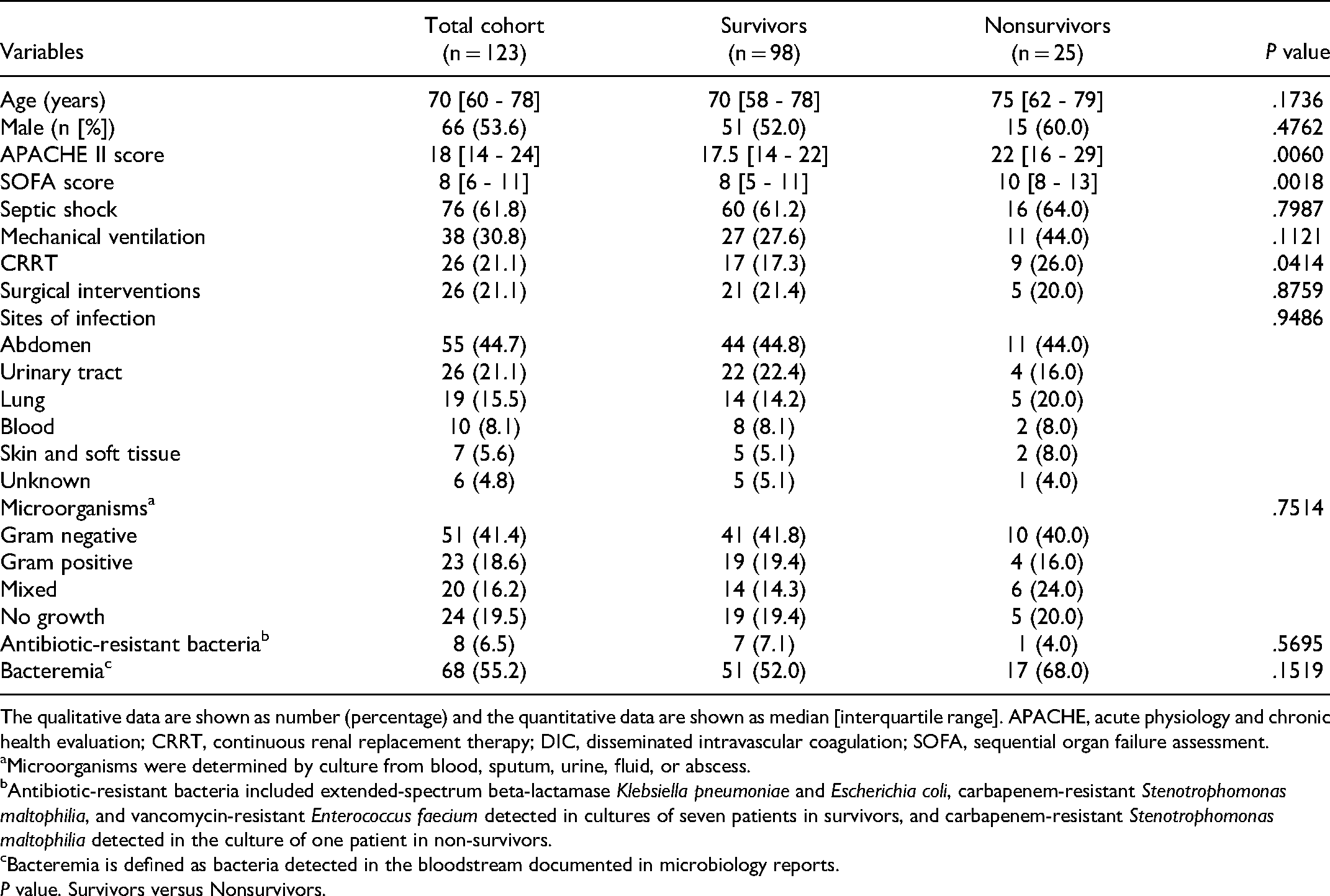
The qualitative data are shown as number (percentage) and the quantitative data are shown as median [interquartile range]. APACHE, acute physiology and chronic health evaluation; CRRT, continuous renal replacement therapy; DIC, disseminated intravascular coagulation; SOFA, sequential organ failure assessment.
Microorganisms were determined by culture from blood, sputum, urine, fluid, or abscess.
Antibiotic-resistant bacteria included extended-spectrum beta-lactamase Klebsiella pneumoniae and Escherichia coli, carbapenem-resistant Stenotrophomonas maltophilia, and vancomycin-resistant Enterococcus faecium detected in cultures of seven patients in survivors, and carbapenem-resistant Stenotrophomonas maltophilia detected in the culture of one patient in non-survivors.
Bacteremia is defined as bacteria detected in the bloodstream documented in microbiology reports.
P value. Survivors versus Nonsurvivors.
Effects of rTM on Coagulation- and Inflammation-Related Variables
In the total cohort, the dose and duration of rTM administration were 380 [130 - 380] U/kg/day and 5 [3 - 6] days, respectively. These were 380 [192.5 - 380] U/kg/day and 5 [3.25 - 6] days in survivors, and 380 [130 - 380] U/kg/day and 6 [3 – 7] days in nonsurvivors.
Coagulation- and inflammation-related variables before and after rTM administration are shown in Table 2. Before treatment with rTM, the DIC score, the platelet count, FDP, and CRP were not significantly different between survivors and nonsurvivors. Among coagulation- and inflammation-related variables, only PT-INR was significantly (p = .0395) lower in survivors than in nonsurvivors, although medians [IQRs] of PT-INR were much higher than normal (1.0 ± .1) in both survivors and nonsurvivors (1.42 [1.27 - 1.62] and 1.70 [1.31- 2.07], respectively) (Table 2).
DIC score, platelet count, coagulation biomarkers, and CRP level before and after rTM administration in patients with sepsis-induced DIC.

Data are shown as median [interquartile range]. CRP, C-reactive protein; DIC, disseminated intravascular coagulation; FDP, fibrin/fibrinogen degradation products; PT-INR, prothrombin international normalized ratio; rTM, recombinant human soluble thrombomodulin.
**p < .001, ***p < .0001 versus before rTM, P value, Survivors versus Nonsurvivors.
After treatment with rTM, the platelet count increased significantly (p < .0001) while the DIC score, PT-INR, FDP, and CRP decreased significantly (p < .0001) in the total cohort and also in survivors. On the other hand, the platelet count, PT-INR, or FDP did not change significantly in nonsurvivors. However, the DIC score and CRP decreased significantly (p < .001) not only in survivors but also in non-survivors, albeit to lesser extents in nonsurvivors compared with survivors (Table 2). The DIC score decreased significantly after rTM even in nonsurvivors primarily because one component of the DIC score – the SIRS criteria score decreased in most (22/25) of nonsurvivers, despite that other three components, including the platelet count, PT-INR and FDP, did not change significantly. Other than above-mentioned coagulation- or inflammation-related variables, the SOFA score decreased significantly (p < .001) after rTM not only in survivors (from 8 [5-11] to 2 [0-3]) but also in nonsurvivors (from 10 [8-13] to 6 [5-9]), albeit to a lesser extent in nonsurvivors.
After treatment with rTM, the platelet count was significantly higher while PT-INR, FDP, and CRP, and the DIC score were significantly lower in survivors than in nonsurvivors (p = .0038 for FDP, and p < .0001 for others) (Table 2). The SOFA score also was significantly (p < .01) lower in survivors than in nonsurvivors. The DIC resolution rate was significantly higher in survivors than in nonsurvivors (77/98 [78.6%] vs 10/25 [40.0%], p < .001).
Comparisons of variables before treatment with rTM between patients attaining DIC resolution (n = 87) and those not attaining DIC resolution (n = 36) are shown in Table 3. The APACHE II, SOFA, and DIC scores, and PT-INR were significantly lower while the platelet count was significantly higher in patients attaining than not attaining DIC resolution (p < .01, respectively). Interestingly, PT-INR before rTM was significantly (p = .0029) lower in patients attaining than not attaining DIC resolution, although medians [IQRs] of PT-INR were much higher than normal in both patients attaining and not attaining DIC resolution (1.42 [1.26 −1.59] and 1.71 [1.38 - 2.27], respectively) (Table 3). Other variables, including demographic data, FDP, or CRP were not different between these patient groups. The 28-day mortality was significantly lower in patients attaining than not attaining DIC resolution (10/87 [11.5%] vs 15/36 [41.7%], p = .0001).
Comparison of various variables before rTM administration between patients attaining DIC resolution and those not attaining DIC resolution.
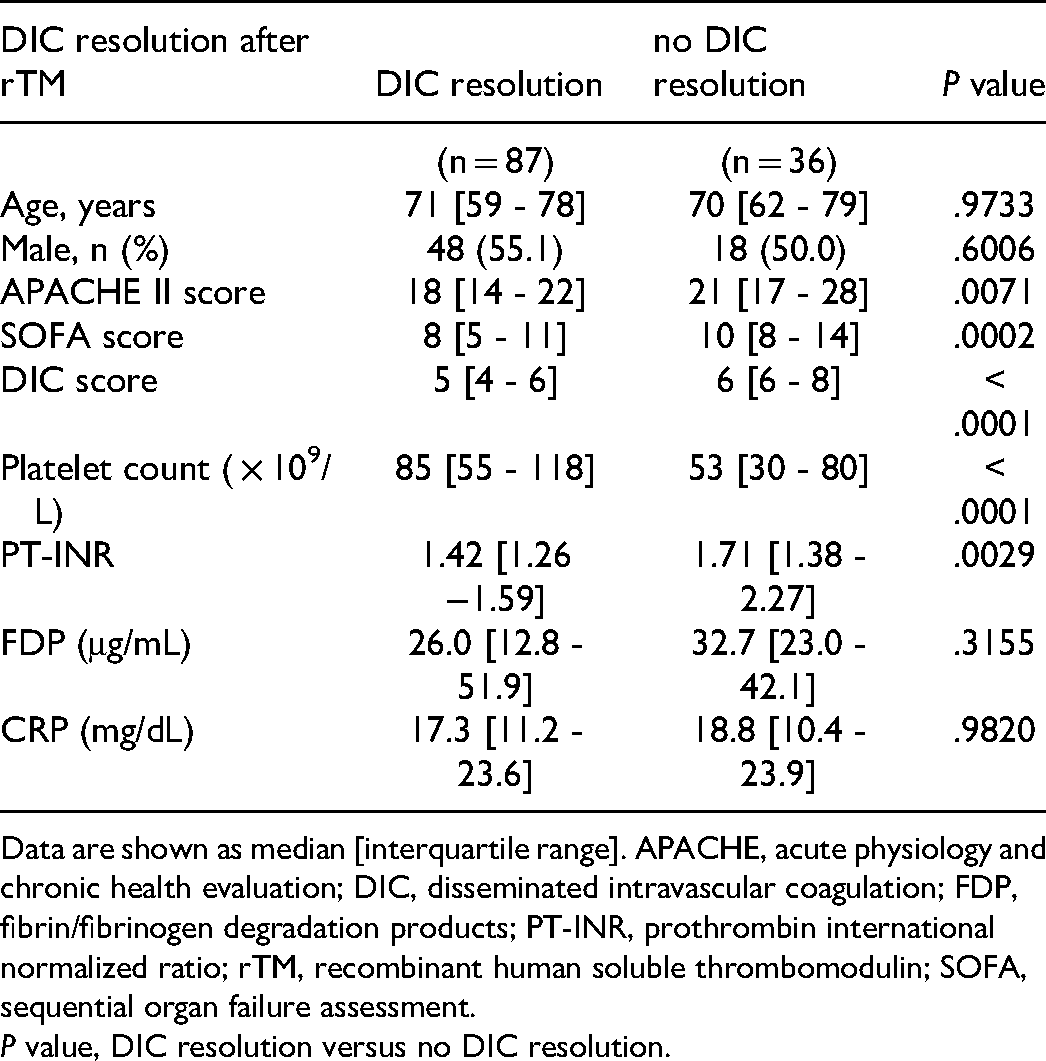
Data are shown as median [interquartile range]. APACHE, acute physiology and chronic health evaluation; DIC, disseminated intravascular coagulation; FDP, fibrin/fibrinogen degradation products; PT-INR, prothrombin international normalized ratio; rTM, recombinant human soluble thrombomodulin; SOFA, sequential organ failure assessment.
P value, DIC resolution versus no DIC resolution.
There were no major rTM-related significant adverse events, including bleeding, in any patients.
Discussion
In the present study, we found that the DIC score, CRP, and the SOFA score significantly decreased after rTM administration in the total cohort, and not only in survivors but also in nonsurvivors, suggesting that rTM was helpful in improving DIC, systemic inflammation, and organ function. We also found that among coagulation- and inflammation-related variables before rTM, only PT-INR was significantly associated with mortality, and PT-INR was also significantly associated with DIC resolution, suggesting that PT-INR was associated with the patient's outcome after treatment with rTM.
In the present study, the DIC resolution rate was significantly higher in survivors than in nonsurvivors (78.6% vs 40.0%), and mortality was significantly lower in patients attaining than not attaining DIC resolution (11.5% vs 41.7%). Comparisons between survivors and nonsurvivors showed that mortality was significantly associated with APACH II and SOFA scores and PT-INR, but not with the DIC score, platelet count, FDP, or CRP. Comparisons between patients attaining and not attaining DIC resolution showed that DIC resolution was significantly associated with APACH II, SOFA, and DIC scores, the platelet count and PT-INR before treatment with rTM, but not with FDP or CRP. After treatment with rTM, however, coagulation-related variables, including the platelet count, PT-INR, and FDP, improved significantly only in survivors, but not in nonsurvivors. These results suggested that not only favorable responses of sepsis to therapies with antibiotics and rTM, but also favorable responses of coagulopathy to therapies were crucial in improving the patient's outcome.
PT-INR evaluates how well the coagulation factors in the extrinsic and common coagulation pathways, including I (Fibrinogen), II (Prothrombin), V, VII, and X, work in coordination. Elevated PT-INR suggests bleeding tendency secondary to reduced coagulation activity resulting from DIC.1–4 In the present study, we showed that among coagulation- and inflammation-related variables before rTM, only PT-INR was significantly associated with mortality, and PT-INR was also significantly associated with DIC resolution. Reportedly, rTM can effectively suppress hypercoagulability indicated by increased levels of prothrombin fragment 1.2 and thrombin-antithrombin complex, compared with a placebo, by downregulating the thrombin generation mediator in sepsis-induced DIC. 19 Further, rTM-treated patients who had higher baseline levels of prothrombin fragment 1.2 and thrombin-antithrombin complex had lower mortality than placebo-treated patients, 13 suggesting that rTM is effective in reducing mortality in patients with hypercoagulability. A previous study classified bacterial sepsis into three subtypes – “inflammopathic”, “adaptive”, and “coagulopathic” subtypes, among which the “coagulopathic” subtype with profound coagulopathy was associated with higher mortality. 20 Previous studies mentioned above may suggest that rTM can be effective in the treatment of “coagulopathic” subtype of DIC.13,19
On the other hand, because the anticoagulant effect of rTM is dependent on thrombin activity in circulation, the ability of rTM to activate protein C and thus, the anticoagulant effect of rTM are expected to be reduced after thrombin depletion.5,6 Actually, the present study showed that among coagulation- and inflammation related variables before rTM, only PT-INR was significantly lower in survivors than in nonsurvivors, although median PT-INR values were much higher than normal in both patient groups. Further, PT-INR was also significantly lower in patients attaining than not attaining DIC resolution, although median PT-INR values were much higher than normal in both patient groups. These results suggested that marked PT-INR elevation before rTM treatment could be associated with reduced DIC resolution and higher mortality. In this regard, diagnosing DIC in early phases of sepsis and starting treatment of DIC with rTM before marked PT-INR elevation indicating depletion of coagulation factors seemed crucial in inducing DIC resolution and reducing mortality in patients with sepsis-induced DIC.
Endothelial expressions of TM and protein C receptors were significantly reduced in patients with sepsis. 21 Accordingly, replacement with rTM is expected to compensate for such downregulations of the endothelial thrombomodulin-protein C receptor pathway in septic patients. Although the primary anticoagulant effect of rTM is mediated by activated protein C, plasma activated protein C levels did not increase after administration of rTM in septic patients with DIC. 22 These results suggest that activated protein C acts only locally and does not circulate systemically in response to rTM. Therefore, the risk of bleeding associated with rTM could be significantly lower, compared with heparin. 9 Indeed, we did not experience any significant adverse events related to rTM, including bleeding, in the present study.
TM, which is localized in the vascular endothelium, integrates biological pathways related to coagulation, innate immunity, and inflammation, and TM thus has anti-inflammatory as well as anticoagulation effects.5–8 The recombinant lectin-like domain of rTM binds to Gram-negative bacteria and lipopolysaccharides, enhances their elimination from the body, and protects the host by suppressing inflammatory responses. 23 In the present study, CRP, which is the most commonly used biomarker of infection and inflammation, 24 significantly decreased after treatment with rTM not only in survivors but also in nonsurvivors. Further, the SIRS criteria score that constitute one component of the DIC score decreased significantly after rTM even in nonsurvivers. These results suggested that rTM, in addition to antibiotics, played an important role in ameliorating systemic inflammation. Also in the present study, the DIC score and the SOFA score decreased significantly after treatment with rTM not only in survivors but also in nonsurvivors, also suggesting that rTM, in addition to antibiotics, played an important role in improving organ functions by reducing systemic inflammation and by regulating coagulation function.
This study had several limitations. First, only a small number of patients were included because of the study conducted at a single institute. Second, the data were retrospectively collected. Third, there was no control group. Fourth, antibiotics also affect CRP. Because rTM therapy has been recognized as a standard treatment for sepsis-induced DIC in Japan, ethics consideration prevented us from allocating patients to a group without rTM. Alternatively, we compared coagulation- and inflammation-related variables before and after treatment with rTM. Further, we compared these variables between survivors and nonsurvivors, and between patients attaining and not attaining DIC resolution. Clearly, further studies are required to verify the efficacy of rTM in patients with sepsis-induced DIC.
Conclusion
In the present study, the DIC score, the SOFA score, and CRP significantly reduced after rTM administration in the total cohort, and not only in survivors but also in nonsurvivors, suggesting that rTM was helpful in improving DIC, systemic inflammation, and organ function. Before rTM administration, PT-INR was significantly lower in survivor than in nonsurvivors and it was significantly lower in patients attaining than not attaining DIC resolution, although median PT-INR values were much higher than normal even in survivors and in patients attaining DIC resolution. Therefore, the initiation of rTM administration before marked PT-INR elevation may be important to induce DIC resolution and thus to decrease mortality in patients with sepsis-induced DIC. Conversely, the treatment with rTM in patients with marked PT-INR elevation may be not so effective in achieving such goals.
Footnotes
Author Contributions
CM designed the protocol and drafted the manuscript. CM and IK performed data collection. CM and DS analyzed the data and performed the statistical analysis. MH edited the manuscript and supervised the study. All authors read and approved the final manuscript.
Ethics and Patient Consent
Ethical approval to report this study was obtained from the Ethics Committee of Juntendo University Hospital (approval number H19-045).
The need for patient approval and informed consent was waived due to the retrospective nature of the study. Instead, the protocol summary was publicized on the university website clearly informing of the patients’ right to refuse participation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
