Abstract
This study investigated the time trends and hospital factors affecting the use of drugs for infectious disease-associated disseminated intravascular coagulation (DIC) based on a national administrative database. A total of 14 324 patients with infectious disease-associated DIC were referred to 1041 hospitals from 2010 to 2012 in Japan. Patients’ data were collected from the administrative database to determine time trends and hospital factors affecting the use of drugs for DIC. Three study periods were established, namely, the fiscal years 2010 (n = 3308), 2011 (n = 5403), and 2012 (n = 5613). The use of antithrombin, heparin, protease inhibitors, and recombinant human soluble thrombomodulin (rhs-TM) for DIC was evaluated. The frequency of use of antithrombin, heparin, and protease inhibitors decreased while that of rhs-TM significantly increased from 2010 to 2012 in Japan (25.1% in 2010, 43.1% in 2011, and 56.8% in 2012; P < .001, respectively). Logistic regression showed that the study period was associated with the use of rhs-TM in patients with DIC. The odds ratio (OR) for 2011 was 2.34 (95% confidence interval [CI], 2.12-2.58; P < .001) whereas that for 2012 was 4.34 (95% CI, 3.94-4.79; P < .001). A large hospital size was the most significant factor associated with the use of rhs-TM in patients with DIC (OR, 3.14; 95% CI, 2.68-3.66; P < .001). The use of rhs-TM has dramatically increased. A large hospital size was significantly associated with the increased use of rhs-TM in patients with DIC from 2010 to 2012 in Japan.
Keywords
Introduction
Disseminated intravascular coagulation (DIC) is a serious clinical condition characterized by excess production of fibrin and consumption bleeding in small vessels with resultant activation of the coagulation system. 1,2 This critical condition usually requires accurate diagnosis and prompt treatment of both the DIC and its underlying cause. Such treatment is important to improve affected patients’ survival rates and prognoses. 1 –3 Some previous studies have found that patients with DIC have high mortality rates; thus, DIC has been recognized as a life-threatening condition worldwide. 4 –6
Although the current management of DIC is primarily focused on treating any associated underlying medical conditions, clinical practice guidelines have pointed out that anticoagulant therapy should also be required. 7 Historically, drugs such as antithrombin, heparin, and protease inhibitors have been frequently used as anticoagulant therapies for patients with DIC, particularly for DIC associated with infectious disease. 8,9 Recombinant human soluble thrombomodulin (rhs-TM), a new biological agent and promising treatment option for DIC, was approved in 2008 in Japan. 10 The use of rhs-TM is expected to replace traditional management for patients with DIC in Japan. 10
However, no epidemiological studies have evaluated changes in the use of old and new drugs for patients with DIC over time based on a national administrative database. In addition, no data are available on hospital factors that influence the choice of new drugs for patients with DIC with adjustment for time trend effects. Clarification of time trends or institutional attributes regarding the use of drugs for anticoagulant therapy could contribute to studies of quality management of patient medical care for DIC.
In this study, we investigated the recent time trends of the use of specific drugs as anticoagulant therapy for patients with infectious disease-associated DIC and hospital factors affecting the use of drugs for DIC among Japanese hospitals. This was achieved with the use of the national administrative database of the Diagnosis Procedure Combination (DPC), which is the Japanese case-mix system currently in use.
Material and Method
The health care system of Japan has severe financial problems because of the expenses of new medical technology, a rapidly aging society, and extended patient hospitalization periods. 11 –13 To address these issues, the Ministry of Health, Labour, and Welfare and its affiliated research institute have begun investigating whether the Japanese case-mix classification system, namely, the DPC, can be used to standardize medical profiling and payment. 11 –13 Japanese case-mix projects based on the DPC system were introduced to 82 academic hospitals (the National Cancer Center, the National Cardiovascular Center, and 80 university hospitals) in 2003. 11 –15 Reimbursement from health insurance using the DPC system is common practice in Japan. According to the administrative database of the DPC system, the number of acute-care hospitals has increased. Enormous amounts of inpatient data are collected annually, covering approximately 90% of the total acute-care inpatient hospitalizations. 11 –15
This system also collects important data during hospitalization in addition to the characteristics of the unique reimbursement system. Each patient’s financial data, claim information, and discharge summary (including the principal diagnosis, complications, and comorbidities during hospitalization) are thoroughly recorded in the administrative database of the DPC system. These data are coded using the International Classification of Diseases and Injuries, Tenth Revision (ICD-10) code. Additionally, this administrative database contains comprehensive medical information, including all interventional or surgical procedures, medications, and devices that have been indexed in the original Japanese code. The Ministry of Health, Labour, and Welfare of Japan assigns these codes. 11 –15 The date and amount of daily care delivered are also recorded in the DPC administrative database. 11 –15
Study Setting
From the DPC administrative database, we selected 14 324 Japanese patients with infectious disease-associated DIC in 1041 hospitals participating in the DPC (83 academic and 958 community hospitals) between 2010 and 2012. These hospitals are dispersed throughout Japan and play leading roles in providing acute-care medicine, advancing medical research, and educating students and medical residents. 11 –15 For the present analysis, 3 study periods were established according to the following fiscal years: 2010 (n = 3308), 2011 (n = 5403), and 2012 (n = 5613).
The survey of DPC-participating hospitals has been conducted by the DPC research group. The DPC-participating hospitals send all the anonymized data to the DPC research group, which in turn sends it to the server in our department. 11 –15 The use of DPC data was permitted by all institutions and hospitals that provided detailed data. The research protocol of the study was approved by the ethics committee of medical care and research of the University of Occupational and Environmental Health, Kitakyushu, Japan.
Study Variables
Study variables included age; sex; chronic comorbid conditions; use of ambulance transportation and the intensive care unit (ICU); hospital type, size, region, and volume; proportion of hospitals with emergency centers; in-hospital mortality; length of stay (LOS); medical cost incurred during hospitalization; and drugs used as anticoagulant therapies for patients with DIC.
Age was stratified as follows: <60, 60 to 79, and ≥80 years. The severity of chronic comorbid conditions was assessed using the Charlson Comorbidity Index (CCI), which is widely used to record comorbidities and has been validated in various studies. 11 –15 The CCI was calculated for each patient as in previous studies that have demonstrated the association between CCI and ICD-10 code. 11 –15 The CCI was expressed as the score of all comorbid conditions and was initially evaluated as a continuous variable. However, categorical variables defining 4 categories of the severity of chronic comorbid conditions were created to simplify the presentation of the results: 0, none; 1, mild; 2, moderate; and ≥3, severe. 13 –15 Hospital type was classified as academic or community. 11 –15 Hospital size was categorized into 3 groups according to the number of hospital beds, namely, small (<200 beds), medium (200-600 beds), and large (>600 beds). 13 –15 Hospital region was classified as urban or rural. 16 Hospital volume was expressed as the number of patients treated during the study period and was initially evaluated as a continuous variable. However, categorical variables defining 3 categories of hospital volume were created to simplify the presentation of the results, namely, low-volume hospitals (<10 patients per year), medium-volume hospitals (10-20 patients per year), and high-volume hospitals (>20 patients per year). The analysis of medical costs incurred during hospitalization was based on an exchange rate of approximately 100 yen = 1 US dollar (October 2014). Antithrombin, heparin, protease inhibitors, and rhs-TM were selected as the anticoagulant drugs evaluated in this study as described in the Japanese clinical practice guidelines for DIC. 7
Statistical Analysis
The chi-square test was used for categorical data, and 1-way factorial analysis of variance was used for continuous variables. We used a multiple logistic regression model to estimate the odds ratios (ORs) and their 95% confidence intervals (CIs) for the time trend of the use of drugs as anticoagulant therapy for patients with DIC. The 2010 study group served as the reference group. Age; sex; chronic comorbid conditions; use of ambulance transportation and the ICU; hospital type, size, region, and volume; and hospitals with emergency centers were considered to be potential confounders and were included in the logistic regression model. These analyses were performed for each drug in this study. The independent hospital factors influencing the use of rhs-TM in patients with infectious disease-associated DIC were investigated in additional analyses using a logistic regression model.
All statistical analyses were performed using the STATA statistical software package, version 11.0 (Stata Corporation, College Station, Texas). A P value of <.05 was considered to be statistically significant.
Results
In total, 14 324 patients with infectious disease-associated DIC were included in this study (3308 patients in 2010, 5403 patients in 2011, and 5613 patients in 2012). The total in-hospital mortality rate was 43.6% from 2010 to 2012. The total mean LOS of all patients was 38.1 days, and the mean medical cost during hospitalization was US$25 029.70.
The clinical characteristics and presentations of patients and hospitals are shown in Table 1. No significant differences were observed in a number of independent variables from 2010 to 2012 with regard to the patients’ characteristics. In addition, the frequency of use of the ICU was similar between 2010 and 2012 (18.0% in 2010, 17.6% in 2011, and 17.6% in 2012; P = .874). The rate of patients treated in large hospitals decreased in accordance with the fiscal year (30.7% in 2010, 30.1% in 2011, and 26.6% in 2012; P < .001). Similarly, the rate of patients treated in hospitals with emergency centers decreased from 2010 to 2012 (39.4% in 2010, 38.1% in 2011, and 34.3% in 2012; P < .001). With respect to treatment outcomes, lower mortality was observed in 2012 than in 2010 or 2011 (46.8% in 2010, 44.4% in 2011, and 40.8% in 2012; P < .001). However, there were no significant differences in LOS from 2009 to 2011 (P = .126). Significant variations in the mean medical cost incurred during hospitalization were observed between 2010 and 2012 (P < .001).
Characteristics of Patients and Hospitals by Fiscal Year According to a National Administrative Database.
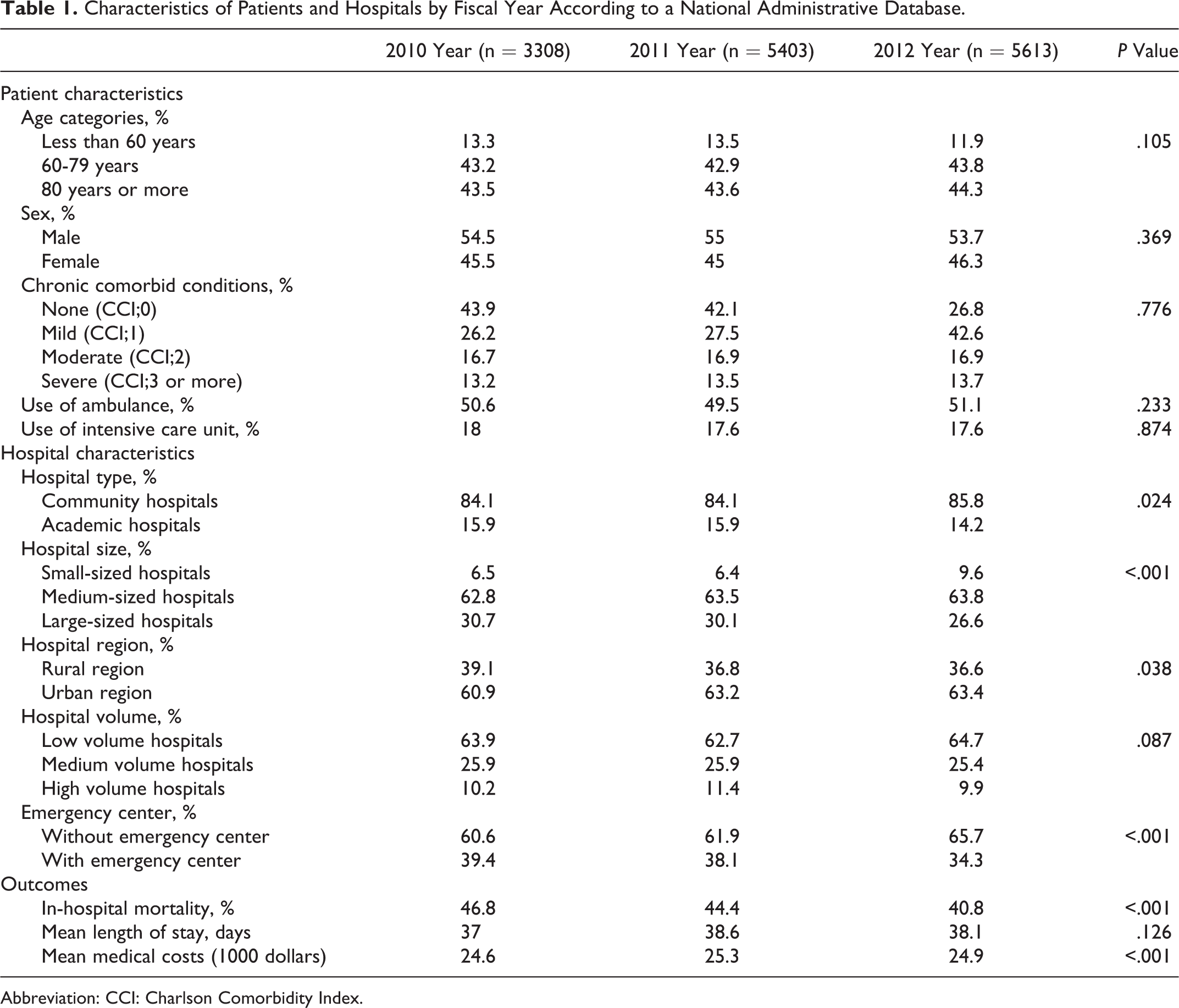
Abbreviation: CCI: Charlson Comorbidity Index.
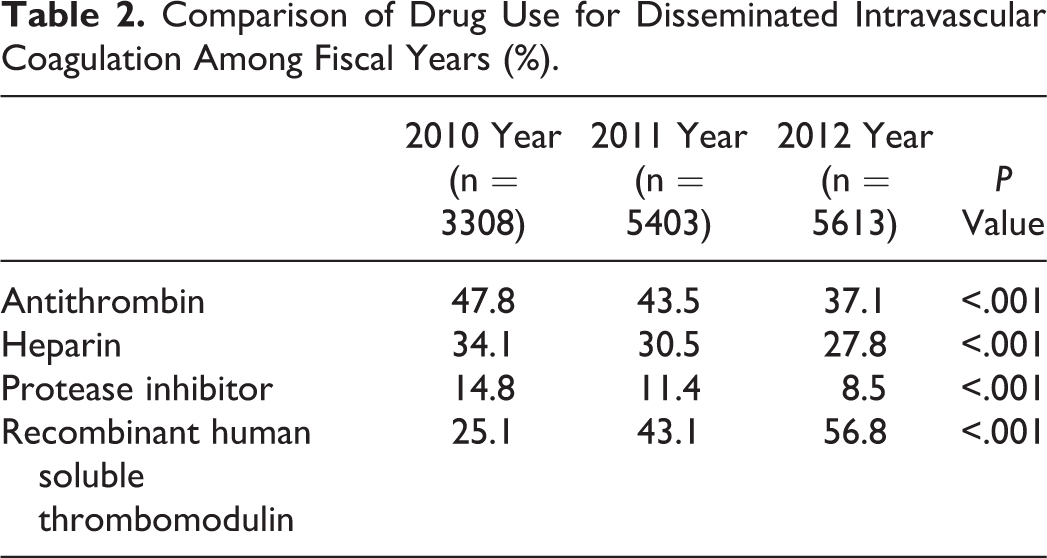
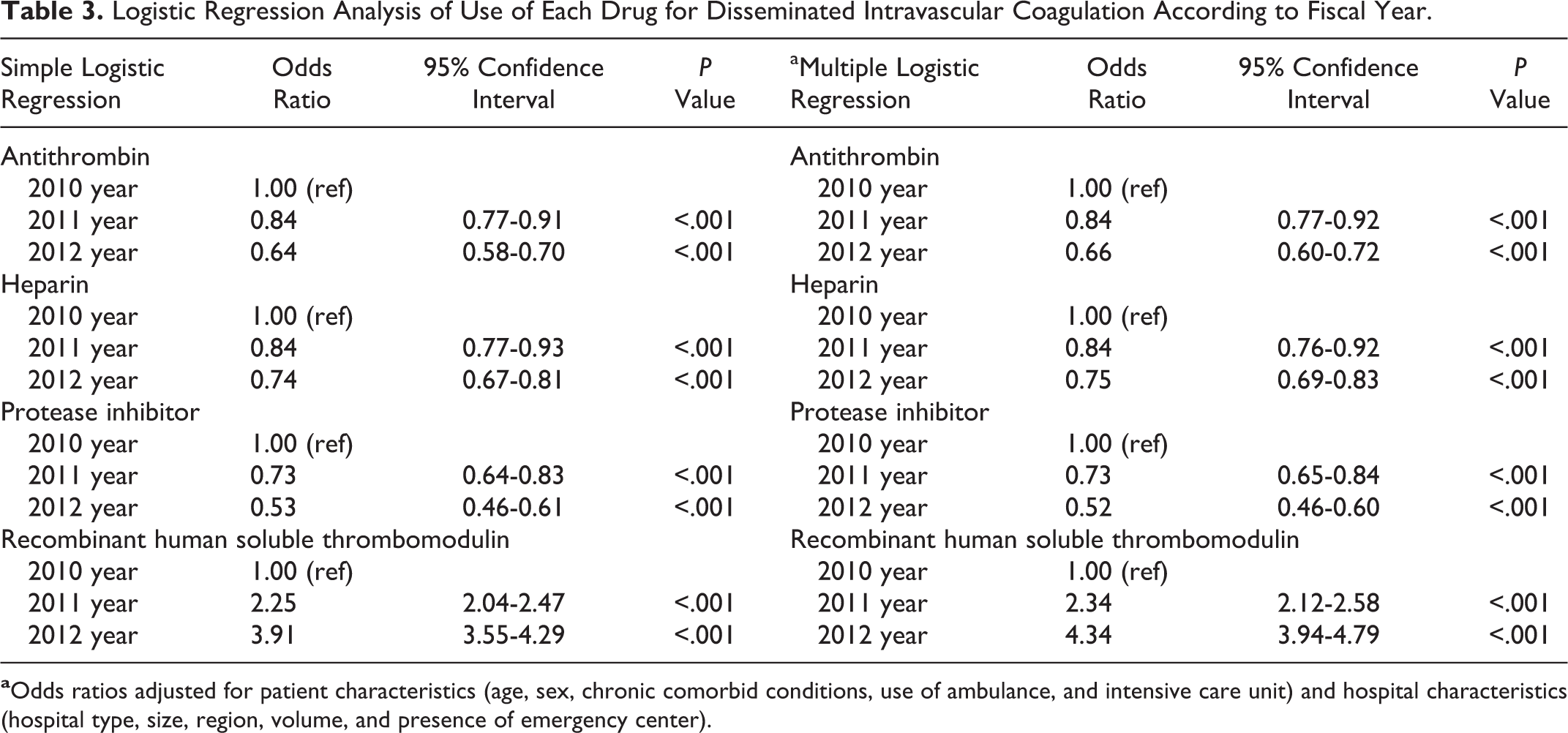
A comparison of the drug use in patients with DIC from 2010 to 2012 is presented in Table 2. The use of antithrombin, heparin, and protease inhibitors significantly decreased with each fiscal year (P < .001). In contrast, rhs-TM was used significantly more frequently in 2012 than in 2010 or 2011 (25.1% in 2010, 43.1% in 2011, and 56.8% in 2012; P < .001). Multiple logistic regression analysis also revealed a declining use of antithrombin, heparin, and protease inhibitors from 2010 to 2012. However, the fiscal year significantly affected the increasing use of rhs-TM in patients with infectious disease-associated DIC. The OR for 2011 was 2.34 (95% CI, 2.12-2.58; P < .001) while that for 2012 was 4.34 (95% CI, 3.94-4.79; P < .001; Table 3).
Comparison of Drug Use for Disseminated Intravascular Coagulation Among Fiscal Years (%).
Logistic Regression Analysis of Use of Each Drug for Disseminated Intravascular Coagulation According to Fiscal Year.
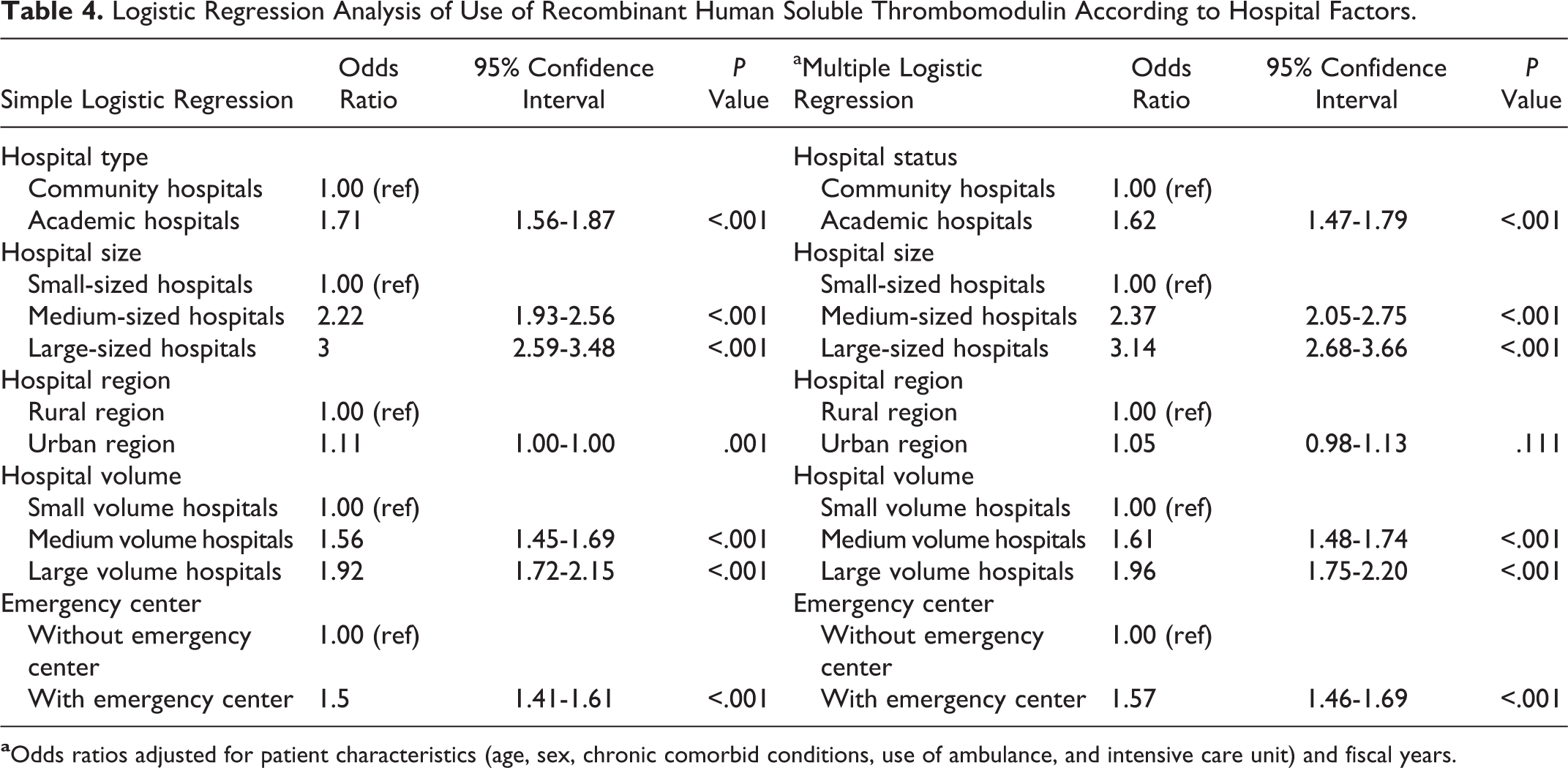
The results of logistic regression analyses of the use of rhs-TM according to hospital factors are shown in Table 4. With the exception of hospital region, each hospital factor influenced the increasing frequency of use of rhs-TM in patients with infectious disease-associated DIC. However, simple logistic regression analysis showed that a large hospital size was the most significant factor influencing the use of rhs-TM (OR, 3.00; 95% CI, 2.59-3.48; P < .001). After adjustment for patients’ characteristics and fiscal years, a significant association between the increasing use of rhs-TM and a large hospital size was observed in this study (OR, 3.14; 95% CI, 2.68-3.66; P < .001).
Logistic Regression Analysis of Use of Recombinant Human Soluble Thrombomodulin According to Hospital Factors.
Discussion
Using a national administrative database, we investigated the recent time trends and hospital factors associated with the use of drugs for infectious disease-associated DIC. This study demonstrated that from 2010 to 2012 in Japan, the use of rhs-TM in these patients dramatically increased and that a large hospital size was a significant factor associated with this increased use.
Many studies have focused on the efficacy or safety of drugs for DIC. However, very few have evaluated time trends of the use of drugs for DIC. Confirmation of chronological changes in the use of drugs using a large patient database clearly demonstrates changes in the circumstances surrounding the treatment of patients with DIC, and such information could be useful to improve the quality of care for these patients. The present study is the first to demonstrate a rapid change in the use of specific drugs for infectious disease-associated DIC.
The use of rhs-TM has been rapidly increasing since its launch in 2008, and its frequency of use reached 56% in 2012 according to the present study. Several recent studies have reported that rhs-TM has contributed to improved prognoses for patients with DIC associated with various infectious diseases, including sepsis. 17 –19 Saito et al 17 reported that rhs-TM more significantly improved the outcomes of DIC and more effectively alleviated bleeding symptoms in patients with DIC than did heparin therapy. Yamakawa et al 18 also showed that administration of rhs-TM reduced the 28-day mortality rate among patients with severe sepsis-associated DIC. In addition, the Japanese clinical practice guidelines for the diagnosis or treatment of DIC, published in 2009, are the first to introduce rhs-TM as a promising treatment for patients with DIC. 7 Such studies and reports can provide important information for clinicians regarding the selection of rhs-TM for patients with DIC. Furthermore, the early experience of using rhs-TM in individual hospitals, particularly large-sized or higher volume hospitals, is an important factor for clinicians to recommend using rhs-TM in successive patients with infectious disease-associated DIC. Jones et al 20 demonstrated that early experience with the use of a new drug can strongly influence its future use. Thus, many clinicians have favorably evaluated the efficacy of rhs-TM based on previous reports or their own experience in the treatment of DIC, which may help to explain the rapid increase in its use from 2010 to 2012 in Japan.
In this study, a large hospital size was the most influential factor among the hospital characteristics affecting the use of rhs-TM for patients with infectious disease-associated DIC. Although the reasons for this finding are unclear, several factors may be associated with the increased use of rhs-TM in large hospitals. First, the pharmaceutical industry usually targets large hospitals to promote new drugs. 21 Information from the pharmaceutical industry may contribute greatly to the awareness of new drugs among clinicians engaged in the treatment of DIC. Second, Garjón et al 22 reported that specialists more rapidly adopt new drugs than do generalists. It is plausible that larger hospitals have more specialists than do smaller hospitals. Therefore, the effect of advertisement from the pharmaceutical industry or the presence of greater numbers of specialists may contribute to the increased use of rhs-TM for patients with infectious disease-associated DIC in larger hospitals.
The data chosen for analysis represent a major strength of the current study. Enormous amounts of inpatient data are collected annually, covering almost all of Japan’s acute-care inpatient hospitalizations. 11 –15 One of the benefits of this national database is that it enables evaluation of a large number of Japanese hospitals in an unbiased manner; our investigation involved a nationally representative sample of patients with DIC in a community setting. 11 –15 In addition, detailed medical data including patient and hospital characteristics are recorded yearly. 11 –15 Therefore, this administrative database enables interested parties to evaluate the various outcomes of DIC treatment using detailed medical data of individual patients.
Some potential limitations of this study warrant mention. First, the data were obtained from DPC-participating hospitals. Data from non-DPC-participating hospitals should also be analyzed to confirm the circumstances for use of rhs-TM in the future. Second, we could not include clinician-associated characteristics in our analysis because our administrative database does not include data such as clinicians’ level of skill, type of specialty, or years in practice. These background factors may affect the choice of old or new drugs for the treatment of DIC. Therefore, further clinical studies that consider how these clinician-related factors affect the choice of drugs for patients with DIC may be required.
In spite of these limitations, this study confirmed the occurrence of rapid changes in the treatment of patients with infectious disease-associated DIC. The current study has important implications for future research in this area. First, the results obtained from our study are not necessarily applicable to other countries. For example, Italian or Japanese clinical practice guidelines for DIC treatment have recommended the use of rhs-TM, while British clinical practice guidelines have not mentioned the use of rhs-TM. 23 –26 In addition, developing nations tend to have lower diffusion speeds and lower maximum penetration levels of new drugs than do developed countries. 27 Therefore, further investigation of the use of rhs-TM in other countries is vital to confirm the impact of the increasing use of rhs-TM in patients with infectious disease-associated DIC. Second, although previous studies of rhs-TM have focused on the efficacy or safety of this drug in patients with DIC, no reports have evaluated the financial implications of its use in terms of parameters such as the medical cost of treatment with rhs-TM. Unlike heparin and protease inhibitors, rhs-TM is a very expensive drug. 28 The medical cost incurred during hospitalization did not dramatically increase with the frequency of use of rhs-TM in this study. However, the use of rhs-TM can undoubtedly increase the medical costs incurred during hospitalization. Therefore, a survey on the medical-related economic outcomes of patients who have received rhs-TM would be very informative. Successive monitoring of the medical costs incurred during hospitalization of patients receiving rhs-TM as well as the rate of use of rhs-TM will help to elucidate the real influence of the use of rhs-TM for patients with DIC, particularly in large hospitals where rhs-TM is being used more frequently. Such monitoring could provide useful information that will help to increase the quality of medical care for patients with DIC in Japan.
In conclusion, this study has demonstrated that from 2010 to 2012 in Japan, the use of rhs-TM for the treatment of infectious disease-associated DIC rapidly increased and that a large hospital size was a significant factor affecting this increase. Further investigation of the use of rhs-TM in other countries as well as successive monitoring of medical costs incurred during hospitalization of patients receiving rhs-TM will help to confirm this change in the treatment of DIC.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Grants-in-Aid for Research on Policy Planning and Evaluation from the Ministry of Health, Labour, and Welfare, Japan.
