Abstract
Although a bioabsorbable bone hemostatic agent (BBHA) was developed approximately 20 years ago to overcome the shortcomings of conventional bone wax, its bleeding control capacity has not yet been studied. This study was aimed at investigating the efficacy and safety of BBHA in total knee arthroplasty (TKA). Sixty-two patients who underwent unilateral primary TKA for knee osteoarthritis were included and randomized to the control or BBHA group. Before releasing the tourniquet, BBHA was applied on the bone-cut surface that was not covered by implants. The primary variable was the drainage volume during the postoperative period. The secondary outcomes were total estimated blood loss (EBL), hemoglobin level, hematocrit level, erythrocyte sedimentation rate (ESR), C-reactive protein (CRP) level, range of motion (ROM), pain visual analog scale (VAS) score, and rate of complications. There were no significant differences in drainage volume or EBL between the 2 groups. Hemoglobin and hematocrit levels were higher in the BBHA group during the 4-week postoperative period; however, the intergroup differences were not significant. The ESR, CRP, ROM, and pain VAS scores in the BBHA group were not significantly different from the corresponding values in the control group. No specific complications were observed. Although BBHA was found to be safe without complications, it did not decrease bleeding after TKA in general cases. Further studies are necessary to evaluate the efficacy of BBHA in patients with coagulation problems.
Introduction
Bleeding control in surgery remains a significant challenge in modern medicine. In particular, orthopedic surgery is associated with the challenge of managing bone bleeding from the exposed or cut surface of the cancellous bone. The disastrous situation of uncontrollable bleeding from the cancellous bone has been reported in several studies. 1 –3 Among all the fields of orthopedic surgery, total knee arthroplasty (TKA) is generally known to induce significant blood loss due to extensive bone cutting and exposure of a large surface of the cancellous bone. Specifically, bleeding due to an unavoidable mismatch between the implant size and exposed cancellous bone after cutting is one of the remaining important issues in TKA (Figure 1A). Various techniques have been attempted to directly prevent bleeding from the cancellous bone, such as application of fibrin or thrombin sealant, hydrogen peroxide washing, and sealing with bone wax. 4 –8

Intraoperative photograph showing the exposed bone surface and procedure of applying BBHA. A, The bone cut surface that was not covered by implants was a result of a mismatch between the implant size and exposed cancellous bone (asterisk). B, BBHA (circle) was applied to the exposed bone cut surface (dotted circle). BBHA indicates bioabsorbable bone hemostatic agent.
Bone wax is a topical bone hemostatic agent composed of a mixture of beeswax, paraffin, and isopropyl palmitate and serves as a barricade and tamponade on the bleeding site. 7,8 Owing to its advantages such as ease of manipulation and cost-effectiveness, bone wax has been widely used in the field of orthopedic surgery. However, it is not absorbed by the body, hinders osteogenesis, and therefore impairs bone healing. 7,8 There is also much evidence about granuloma formation, allergic reaction, and infection, which results in more serious complications, such as complete paralysis after spinal surgery and sigmoid sinus obstruction following mastoid surgery. 8 –10
Recently, an alternative agent to bone wax, a biodegradable bone hemostatic agent (BBHA), has been developed to overcome these limitations. BBHA does not seem to inhibit bone growth, is not biochemically altered by the human body, and is eliminated without inducing an inflammatory reaction. 7,8,11 –15 Furthermore, Wellisz et al reported a decreased rate of infection associated with BBHA in an experimental animal model compared to the infection rate associated with conventional bone wax. 16 However, no study has evaluated the bleeding control capacity of BBHA. Therefore, this study was aimed at investigating the efficacy and safety of BBHA through TKA.
Materials and Methods
This prospective randomized, single-blinded, placebo-controlled comparative study was approved by the institutional review board (1705-027-851) and registered at cris.nih.go.kr (KCT0004298). From November 2017 to August 2019, patients between 50 and 80 years of age who were scheduled to undergo unilateral primary TKA for knee osteoarthritis were enrolled. The exclusion criteria were as follows: current anticoagulation treatment; current or a history of bleeding disorder (platelet count of <150,000/mm, 3 international normalized ratio of >1.4, or prolonged partial thromboplastin time [>1.4 times normal]); a history of major comorbidities (including previous myocardial infarction, severe heart failure [New York Heart Association Class III or IV], a severe pulmonary disease such as asthma or chronic obstructive or restrictive pulmonary lung disease, renal failure [GFR >60 mL/min/1.73 m2], or hepatic failure [Child class >B]); a history of thromboembolic disease such as a cerebrovascular attack, DVT, or PE; severe deformity (mechanical axis or flexion contracture of >20°); severe bone defect considering metal augment, bone graft, or stem fixation; a previous operation of the ipsilateral knee; a history of peripheral nerve disorder; and neuromuscular comorbidities such as cerebral infarct and parkinsonism cerebral infarct, which could have an effect on postoperative rehabilitation.
A priori power analysis was conducted based on the results of a previous study. 17 It was estimated that 28 patients were needed in each group to potentially find an effect at a compensated alpha level of 0.05 and a power of 80% by using a 2-sided test. To allow for 10% exclusions and dropouts, 62 patients (31 in each group) were enrolled in the present study. These 62 patients were randomly assigned to the control or BBHA group by using a computer-generated randomization table. During follow-up, one patient in the control group and 2 patients in the BBHA group dropped out. Finally, 59 patients (30 in the control group and 29 in the BBHA group) were analyzed (Figure 2).

CONSORT diagram of the randomized controlled trial. BBHA indicates bioabsorbable bone hemostatic agent.
BBHA (Noboseal, CGBio, Seoul, Korea)
The BBHA used in the present study is a biodegradable and soluble bone hemostatic agent that mainly consists of an alkylene oxide block copolymer. 11,12 The poloxamer changes its form depending on the temperature and reduces the strength of bone wax at body temperature. At 25°C (room temperature), it has a strength of more than 11 N, and its strength changes to approximately 4 N when applied to the human body, making it easy to attach to irregular bone surfaces.
Procedures of BBHA Application and Surgical Technique
A single surgeon performed all the surgeries using the same procedure. After tourniquet inflation, an incision was made along the anterior midline followed by a standard mid-vastus arthrotomy. A posterior-stabilized knee prosthesis with fixed bearing was implanted, and routine patella resurfacing was performed with cement fixation. Before deflating the tourniquet, BBHA was applied to the bone-cut surface that was not covered by the implant (Figure 1B). The tourniquet was thereafter deflated, and meticulous bleeding control was performed. A suction drain was inserted, and the joint capsule and subcutaneous layer were closed. Tranexamic acid 1.0 g was administered through the drain line with clamping for 4 h; the drain was removed on postoperative day 2.
Postoperative Management and Rehabilitation
All patients received the same multimodal perioperative pain management, including intravenous patient-controlled analgesia, oral analgesics, and rescue injection. Standardized rehabilitation programs were used postoperatively. All patients were encouraged to have continuous passive motion and begin isometric quadriceps strengthening and straight-leg raise exercises on the first postoperative day. Walking with the aid of a walker was started on the second postoperative day. Discharge was commonly recommended on postoperative day 7.
Outcome Assessment
Drainage volume during 2 days after the operation was measured as the primary outcome variable. The drained fluid in the suction drain was collected at operation day, postoperative day 1, and postoperative day 2, and the volume was measured. The secondary outcomes were total estimated blood loss (EBL), hemoglobin level, hematocrit level, erythrocyte sedimentation rate (ESR), C-reactive protein (CRP), range of motion (ROM), and the degree of pain and complications. EBL was evaluated at postoperative day 2 and 6 by calculating the difference between preoperative hemoglobin and the lowest postoperative hemoglobin level. Based on hemoglobin balance, EBL was calculated using the method described by Good et al 18 Hemoglobin and hematocrit levels were assessed on postoperative day 1, 2, and 6, and week 4. ESR, CRP level, and ROM were assessed at postoperative day 6 and week 4. The angle of the maximal extension and the further flexion angle were measured with a goniometer by an independent trained researcher blinded to the trial. The degree of pain was measured using a visual analog scale (VAS, range 0-10) at postoperative days 1, 2, and 6 and week 4. 19 A clinical investigator blinded to participants' treatment recorded all values. Complications related to the use of intra-articular materials, such as infection, hemarthrosis, and allergic reactions were recorded.
Statistical Analysis
Data are described as mean and standard deviation values. Categorical variables (sex, side, and complication) were analyzed using Pearson’s chi-squared test or Fisher’s exact test with relative ratio while continuous variables (age, BMI, hemoglobin and hematocrit levels, ESR and CRP, ROM, VAS, drainage volume, and EBL) were analyzed with Student’s t-test. Statistical analyses were performed using the Statistical Package for the Social Sciences (SPSS) version 22 (IBM Corp.). A P-value of less than 0.05 was considered statistically significant.
Results
There were no significant differences in demographic characteristics between the 2 groups (Table 1). The mean drainage volumes during the 2-day postoperative period in the control and BBHA groups were 384.9 ± 132 mL and 408.0 ± 146 ml, respectively. There were no significant differences between the 2 groups (Table 2). The mean EBL of the control and BBHA groups during the 6-day postoperative period was 756.0 ± 433 mL and 747.1 ± 321 mL, respectively. Intergroup differences were not significant.
Summary of Patients’ Demographic and Preoperative Data.a

Abbreviations: BBHA, bioabsorbable bone hemostatic agent; BMI, body max index; ESR, erythrocyte sedimentation rate; CRP, C-reactive protein; ROM, range of motion; VAS, visual analog scale.
a The values are presented as the mean and the standard deviations.
Comparison of Postoperative Clinical Data Between 2 Groups.a
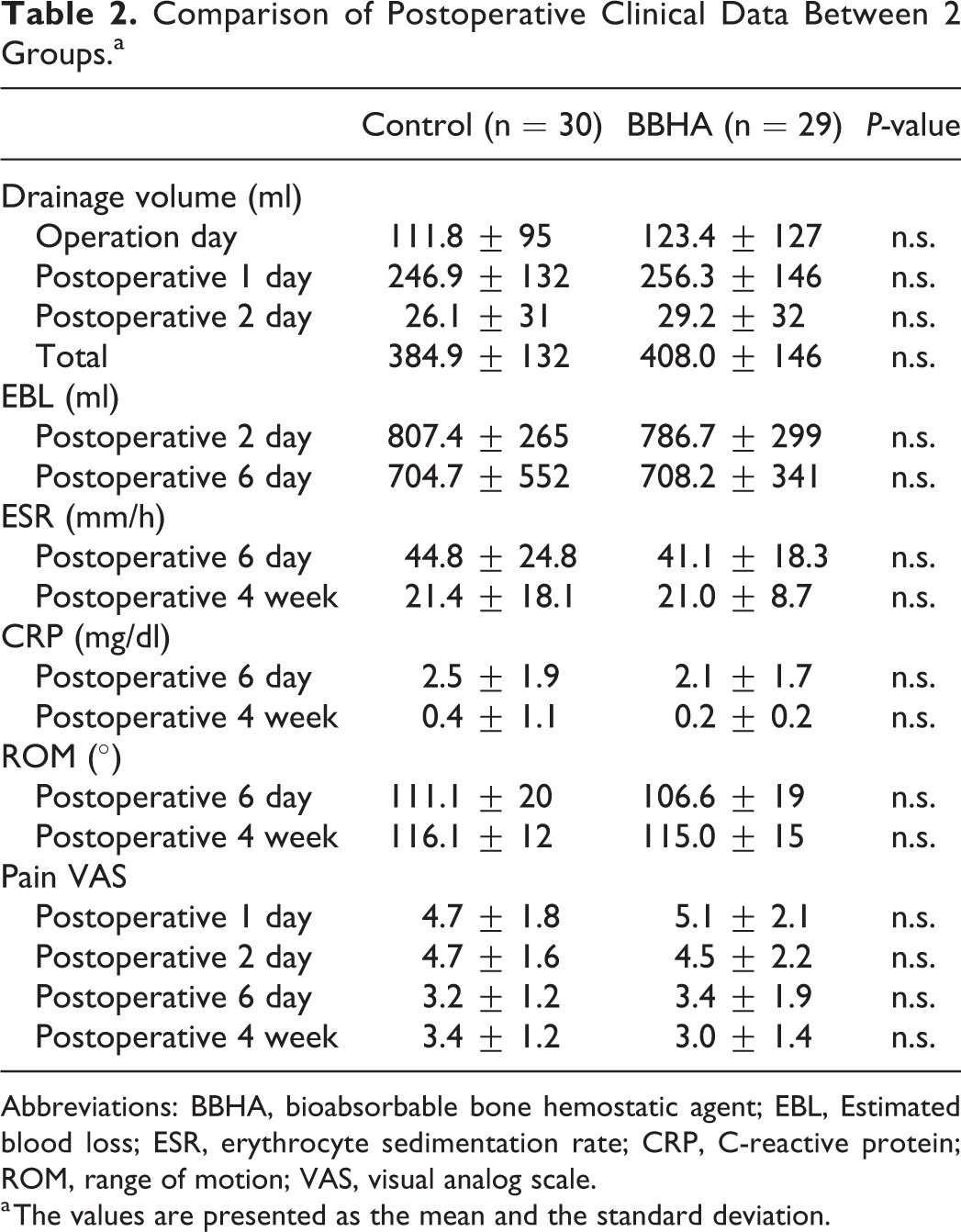
Abbreviations: BBHA, bioabsorbable bone hemostatic agent; EBL, Estimated blood loss; ESR, erythrocyte sedimentation rate; CRP, C-reactive protein; ROM, range of motion; VAS, visual analog scale.
a The values are presented as the mean and the standard deviation.
The mean hemoglobin level during the observed postoperative period in the BBHA group maintained higher than that in the control group (Figure 3A). In particular, the mean hemoglobin levels on the first postoperative day in the control and BBHA groups were 10.9 ± 1.2 g/dl and 11.2 ± 1.0 g/dl, respectively. The corresponding values for the second postoperative day were 10.01 ± 1.1 g/dl and 10.5 ± 0.87 g/dl. However, there were no significant differences between the 2 groups.
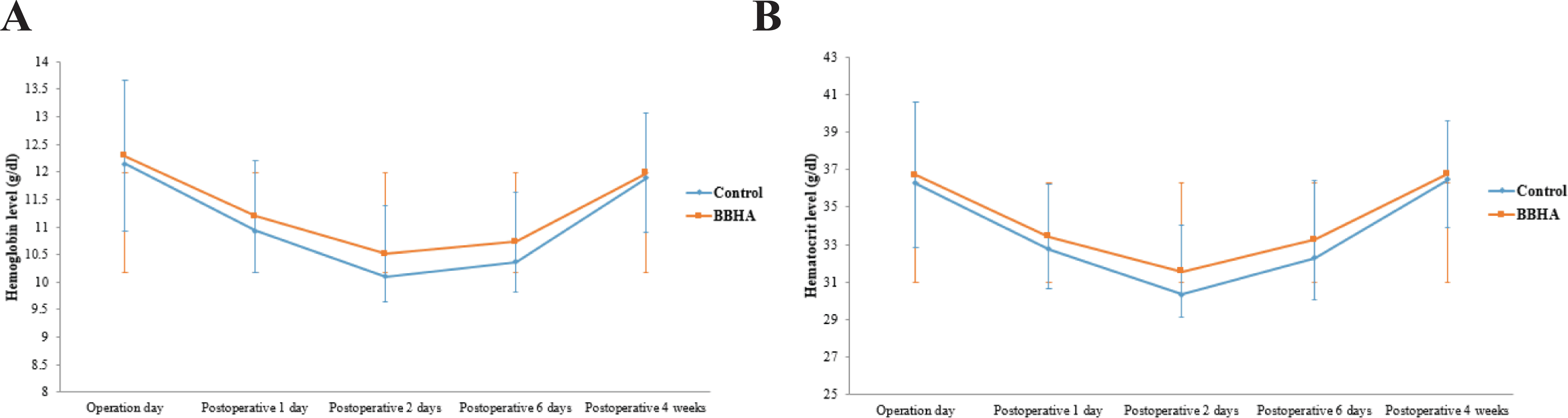
Comparison of hemoglobin (A) and hematocrit levels (B). Mean hemoglobin and hematocrit levels of the BBHA group remained higher than those of the control group during the observed postoperative period. However, there were no significant differences between the 2 groups. BBHA indicates bioabsorbable bone hemostatic agent.
The mean hematocrit level during the observed postoperative period in the BBHA group also maintained higher than that in the control group (Figure 3B). In particular, the mean hematocrit levels on the first postoperative day in the control and BBHA groups were 32.8 ± 3.1 g/dl and 33.1 ± 2.8 g/dl, respectively. The corresponding values for the second postoperative day were 30.3 ± 1.1 g/dl and 31.6 ± 2.5 g/dl. However, there were no significant differences between the 2 groups.
The mean ESR and CRP levels at postoperative day 6 and postoperative 4 weeks in the BBHA group were lower than those in the control group (Table 2). However, there were no significant differences between the 2 groups. ROM and pain VAS scores of the BBHA group were not significantly different from those of the control group. No specific complications were observed in either group.
Discussion
The most important finding of this study was that there were no significant differences in the values pertaining to bleeding control between the control and BBHA groups. The hemoglobin and hematocrit levels during the 4-week postoperative period were higher in the BBHA group than in the control group, but the intergroup differences were not significant. BBHA group did not show significant differences in other variables to evaluate the safety such as ESR, CRP, ROM and pain VAS compared to the control group. No specific complications related to the use of this intra-articular material were observed.
To the best of our knowledge, this is the first clinical study to investigate the bleeding control capacity of BBHA. Since the development of BBHA in 2001, no in vitro or in vivo study to determine its efficacy in bleeding control has been published. The in vivo studies published were conducted to evaluate safety, including bone healing, inflammation, and infection, in animal or human models. 7,8,11 –16 However, this first study on bleeding control showed no significant effect of BBHA.
BBHA is composed of a water-soluble alkylene oxide block copolymer without beeswax, which is one of the major components of conventional bone wax. 7,8 It dissolves within 24-48 hours and is excreted in the urine. 20,21 Although this hydrophilic property seems to provide many benefits compared to bone wax, a proper implanted period apparently generating its role of bleeding control has not been established. This issue becomes more significant in a specific environment such as the knee joint and should be considered as such. The knee joint is affected by dynamic factors due to the turbulence force from the joint fluid and contact force from the surrounding soft tissue, which are increased by joint movements characterized as flexion and extension ROM. Because of these factors, BBHA could be more easily diffuse or get washed out before its effects become apparent. According to a similar randomized control study on the effect of conventional bone wax on the remaining exposed cancellous bone in TKA, bone wax effectively reduced blood loss without complications. 6 Therefore, when biomaterials are applied in the knee joint, stable and long-term implantation that could sustain in the dynamic environment are more important.
Although the intergroup differences were not significant, hemoglobin and hematocrit levels were higher in the BBHA group. Many parameters, such as drainage volume in the suction drain, EBL calculated by Good’s method, serum hemoglobin or hematocrit level, and transfusion rate, have been used in several previously published studies. 6,8,22 However, there is still no consensus on which parameter is ideal for evaluating the bleeding control capacity. The present study was powered to detect differences between the control and BBHA groups based on drainage volume. Considering that hemoglobin and hematocrit levels were higher in the BBHA group during the observation period, if the values were determined as primary variables and powered, the result could be significant. Therefore, further studies are necessary to determine the most significant parameter to evaluate the effect of bleeding control.
The safety of BBHA was revealed in the present study as well as in many previously reported studies with similar results. 7,8,11 –13,15,16,22 Wellisz et al conducted an in vivo study on the spine in an animal model and investigated the degree of inheritance with bone healing and soft-tissue response. 12 As a result, the BBHA group showed no negative effect on bone healing without a specific inflammatory reaction at the host site, compared to the bone wax group. In another study, Wellisz et al reported a decreased rate of infection in an experimental animal model compared to that in the conventional bone wax group. 16 There was only one published clinical study on BBHA, which solely dealt with bone healing. 14 The study reported less interference of BBHA with bone healing compared with conventional bone wax in a histologic examination of the sternal bone after cardiac surgery. Although a histologic examination was not conducted in the present study, clinically easily presented and detected values were used to investigate safety. Inflammation and infection were evaluated by checking laboratory results (ESR and CRP). By measuring active movement (ROM) and subjective feeling (pain VAS), impairment of functional restoration was evaluated. Lastly, the complications were evaluated by recording gross visible changes such as effusion and discharge.
This study has several limitations. First, the present study excluded patients with a potential bleeding tendency, such as those with a bleeding disorder, those undergoing anticoagulation therapy, and immune-compromised patients with major comorbidities. Therefore, the generalizability of this study to patients with these medical conditions is limited and the efficacy of BBHA remains unproven in patients at high risk of bleeding. In addition, this point could be the reason why the results were equivalent with the control. Further study including patients with bleeding disorder or anticoagulation therapy could give a clearer picture. Second, the present study used tranexamic acid as a routine bleeding control protocol. This combined use of hemostatic agents could result in the low efficacy of BBHA. In other words, if only the BBHA was used in TKA, its efficacy could be proven. However, many institutions use tranexamic acid, and this antifibrinolytic agent is considered a routine peri- and postoperative medication by being applied intra-articularly, intravenously, or orally. 22 Therefore, the present study could have more clinical value by investigating the efficacy of BBHA in the setting of actual management in TKA.
Conclusions
Although the use of BBHA was safe without complications, it did not decrease bleeding after TKA in general. To evaluate the efficacy of BBHA in patients with coagulation problems, further studies are necessary.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: This study was funded by CG Bio Inc. The funding source had no role in the study design, collection, analysis or interpretation of the data, writing of the manuscript, or in the decision to submit the manuscript for publication.
