Abstract
Portal venous system thrombosis (PVST), a common complication of liver cirrhosis, is closely associated with thrombophilia. To explore the association of homocysteine (Hcy), anticardiolipin antibody (aCL), and anti-β2 glycoprotein I antibody (aβ2GPI), which are possible thrombophilic factors, with PVST in liver cirrhosis. Overall, 654 non-malignant patients (219 with and 435 without liver cirrhosis) admitted between January 2016 and June 2020 were retrospectively evaluated. Presence of PVST, degree of main portal vein (MPV) thrombosis, and clinically significant PVST were identified. Hcy level, hyperhomocysteinemia (HHcy), aCL positivity, and aβ2GPI positivity were compared according to the presence of liver cirrhosis and PVST. Positive aβ2GPI was significantly more frequent in patients with liver cirrhosis than those without, but Hcy level and proportions of HHcy and positive aCL were not significantly different between them. PVST could be evaluated in 136 cirrhotic patients. Hcy level [10.57 μmol/L (2.71-56.82) versus 9.97 μmol/L (2.05-53.44); P = 0.796] and proportions of HHcy [4/44 (9.1%) versus 13/81 (16.0%); P = 0.413] and positive aCL [1/23 (4.3%) versus 10/52 (19.2%); P = 0.185] and aβ2GPI [9/23 (39.1%) versus 21/52 (40.4%); P = 0.919] were not significantly different between cirrhotic patients with and without PVST. There was still no significant association of Hcy level, HHcy, aCL, or aβ2GPI with PVST based on Child-Pugh classification, MPV thrombosis >50%, and clinically significant PVST. Hcy, aCL, and aβ2GPI may not be associated with PVST in liver cirrhosis, suggesting that routine screening for Hcy, aCL, and aβ2GPI should be unnecessary in such patients.
Keywords
Introduction
Portal venous system thrombosis (PVST) is a common and severe complication of liver cirrhosis. 1 The reported prevalence of non-malignant PVST in liver cirrhosis ranges from 10% to 25%. 2 Failure to detect and treat PVST in a timely fashion may not only lead to mesenteric ischemia, but also cause cavernous transformation of portal vein and aggravate portal hypertension. 3,4
Thrombophilia has been considered as a major contributor for PVST. However, at present, the utility of thrombophilia work-up in cirrhotic patients with PVST is still controversial. The American College of Gastroenterology (ACG) guideline recommends thrombophilia work-up among patients with liver cirrhosis and PVST, if they have previous history of thrombosis, concomitant thrombosis at unusual sites such as hepatic veins, and family history of thrombosis. 5 But the ACG guideline does not specify which types of thrombophilic conditions are needed to be screened. The European Association for the Study of the Liver (EASL) guideline recommends screening for genetic thrombophilia alone, such as factor V Leiden and prothrombin gene G20210A mutation 6 and methylenetetrahydrofolate reductase C677T mutation, 7 in patients with liver cirrhosis and PVST, but not acquired thrombophilia. 8 But the EASL guideline does not point out which groups of cirrhotic patients with PVST should undergo such thrombophilia work-up.
Hyperhomocysteinemia (HHcy) 9,10 and antiphospholipid antibody syndrome (APS) 11 are 2 thrombophilic factors for venous and arterial thrombosis. However, their role in the pathogenesis of PVST in liver cirrhosis is obscure. First, our previous systematic review identified only 3 original studies regarding association of homocysteine (Hcy) level and HHcy with PVST in liver cirrhosis, and the findings were inconsistent among these studies. 7 Second, another previous systematic review suggested that the association between antiphospholipid antibodies and PVST in liver cirrhosis was unclear. 12 The findings are difficult to be explained that unclassified anticardiolipin antibody (aCL), rather than positive aCL immunoglobulin G (IgG) or IgM, was significantly associated with PVST in liver cirrhosis. Additionally, only 2 original studies explored the association of anti-β2 glycoprotein I antibody (aβ2GPI) with PVST in liver cirrhosis, and rarely identified positive aβ2GPI in such patients.
Herein, the present study aimed to clarify the association of Hcy level, HHcy, aCL, and aβ2GPI with PVST in non-malignant patients with liver cirrhosis.
Methods
Patients
A total of 911 adult (>18 years) patients, who were consecutively admitted to the Department of Gastroenterology of the General Hospital of Northern Theater Command between January 2016 and June 2020 and treated by an attending physician, were retrospectively screened. Finally, 654 patients (219 cirrhotic patients and 435 non-cirrhotic patients) were included in this study (Figure 1). The study protocol was approved by the medical ethical committee of our hospital with an approval number of Y (2020) 032. The requirement for informed consent was waived due to the retrospective nature of this study.

Flowchart of patient selection.
Diagnosis and Assessment
Liver cirrhosis was diagnosed by the history of chronic liver diseases, clinical symptoms and signs, laboratory tests, and abdominal images. If necessary, liver biopsy was performed. The severity of liver disease was estimated according to the Child-Pugh and Model for End-stage Liver Disease (MELD) scoring systems. 13
As previously described, 14,15 PVST was confirmed by evaluating the patency of portal venous system vessels using contrast-enhanced computed tomography (CT) or magnetic resonance imaging (MRI) scans or CT portal venography. Portal venous system included left portal vein, right portal vein, main portal vein (MPV), splenic vein, confluence of superior mesenteric vein (SMV) and splenic vein, and SMV. Degree of MPV thrombosis was divided into MPV thrombosis >50% and MPV thrombosis <50%. Clinically significant PVST would be considered, if any one of the following criteria was met: (1) MPV thrombosis >50% with SMV thrombosis and (2) complete MPV thrombosis with or without SMV thrombosis. 16
Measurements
Hcy level was quantitatively determined using Beckman Coulter AU5821 automated analyzer (Beckman Coulter, America). Reference range was 0 to 20 μmol/L. If Hcy level exceeded 20 μmol/L, HHcy would be diagnosed. aCL and aβ2GPI were qualitatively determined using kits based on enzyme-linked immune sorbent assay from EUROIMMUN Medical Diagnostics (EUROIMMUN, China). aCL and aβ2GPI activity were classified as negative and positive.
Statistical Analyses
Continuous variables were expressed as median (range) and compared by the non-parametric Mann-Whitney U test. Categorical variables were expressed as frequency (percentage) and compared by the Chi-square test or Fisher’s exact test. Hcy level, HHcy proportion, aCL positivity, and aβ2GPI positivity were compared according to the presence of liver cirrhosis (with versus without liver cirrhosis), Child-Pugh classification (Child-Pugh class A versus Child-Pugh class B/C), PVST (with versus without PVST), grade of PVST (MPV thrombosis >50% versus MPV thrombosis <50% and MPV patency), and severity of PVST (with versus without clinically significant PVST). Spearman correlation analysis was also performed to explore the correlation between Child-Pugh score and Hcy level, HHcy proportion, aCL positivity, and aβ2GPI positivity in liver cirrhosis. Coefficients, which are denoted as rs, were calculated. All statistical analyses were performed by using IBM SPSS Version 22.0 statistical software (IBM Corp., Armonk, NY). A 2 tailed P value of less than 0.05 was considered statistically significant.
Results
Liver Cirrhosis Versus No Liver Cirrhosis
Patients with liver cirrhosis were significantly older [56 years (22-90) versus 53 years (16-94); P = 0.001] than those without, but the proportion of male was statistically similar between the 2 groups [151/219 (68.9%) versus 276/435 (63.4%); P = 0.163]. The proportion of positive aβ2GPI was significantly higher in patients with liver cirrhosis than those without [44/108 (40.7%) versus 2/18 (11.1%); P = 0.016]. Median Hcy level [10.16 μmol/L (2.05-56.82) versus 10.25 μmol/L (3.04-96.25); P = 0.273] and proportions of HHcy [29/201 (14.4%) versus 45/395 (11.4%); P = 0.295] and positive aCL [21/108 (19.4%) versus 2/18 (11.1%); P = 0.605] were not significantly different between the 2 groups.
Child-Pugh Class A Versus Child-Pugh Class B/C in Liver Cirrhosis
Among the 219 cirrhotic patients, 88 and 131 had Child-Pugh class A and B/C, respectively. Cirrhotic patients with Child-Pugh class B/C were significantly older than those with Child-Pugh class A [60 years (33-80) versus 54 years (22-90); P = 0.033], but the proportion of male was statistically similar between the 2 groups [93/131 (71.0%) versus 58/88 (65.9%); P = 0.458]. The proportions of HHcy [22/119 (18.5%) versus 7/82 (8.5%); P = 0.065] and positive aβ2GPI [32/67 (47.8%) versus 12/41 (29.3%); P = 0.058] were higher in patients with Child-Pugh class B/C than those with Child-Pugh class A, but these differences were not statistically significant between them. Hcy level [10.54 μmol/L (2.05-56.82) versus 9.97 μmol/L (2.71-38.71); P = 0.393] and proportion of positive aCL [14/67 (20.9%) versus 7/41 (17.1%); P = 0.626] were not significantly different between the 2 groups. Similarly, Child-Pugh score significantly correlated with HHcy (rs = 0.168; P = 0.017) and positive aβ2GPI (rs = 0.191; P = 0.048) in liver cirrhosis, but not Hcy level (rs = 0.130; P = 0.066) or positive aCL (rs = 0.096; P = 0.324).
PVST Versus No PVST in Liver Cirrhosis
PVST was evaluable in 136 cirrhotic patients by contrast-enhanced CT (n = 122), and contrast-enhanced MRI (n = 13), and CT portal venography (n = 1). Among them, 47 patients were diagnosed with PVST and 89 without PVST. Age [56 years (39-83) versus 55 years (28-81); P = 0.439] and proportion of male [34/47 (72.3%) versus 60/89 (67.4%); P = 0.554] were statistically similar between cirrhotic patients with and without PVST. Median Hcy level [10.57 μmol/L (2.71-56.82) versus 9.97 μmol/L (2.05-53.44); P = 0.796] and proportions of HHcy [4/44 (9.1%) versus 13/81 (16.0%); P = 0.413] (Figure 2), positive aCL [1/23 (4.3%) versus 10/52 (19.2%); P = 0.185] (Figure 3), and positive aβ2GPI [9/23 (39.1%) versus 21/52 (40.4%); P = 0.919] (Figure 4) were not significantly different between the 2 groups.

No significant difference of HHcy proportion between patients with and without PVST in liver cirrhosis based on Child-Pugh class.
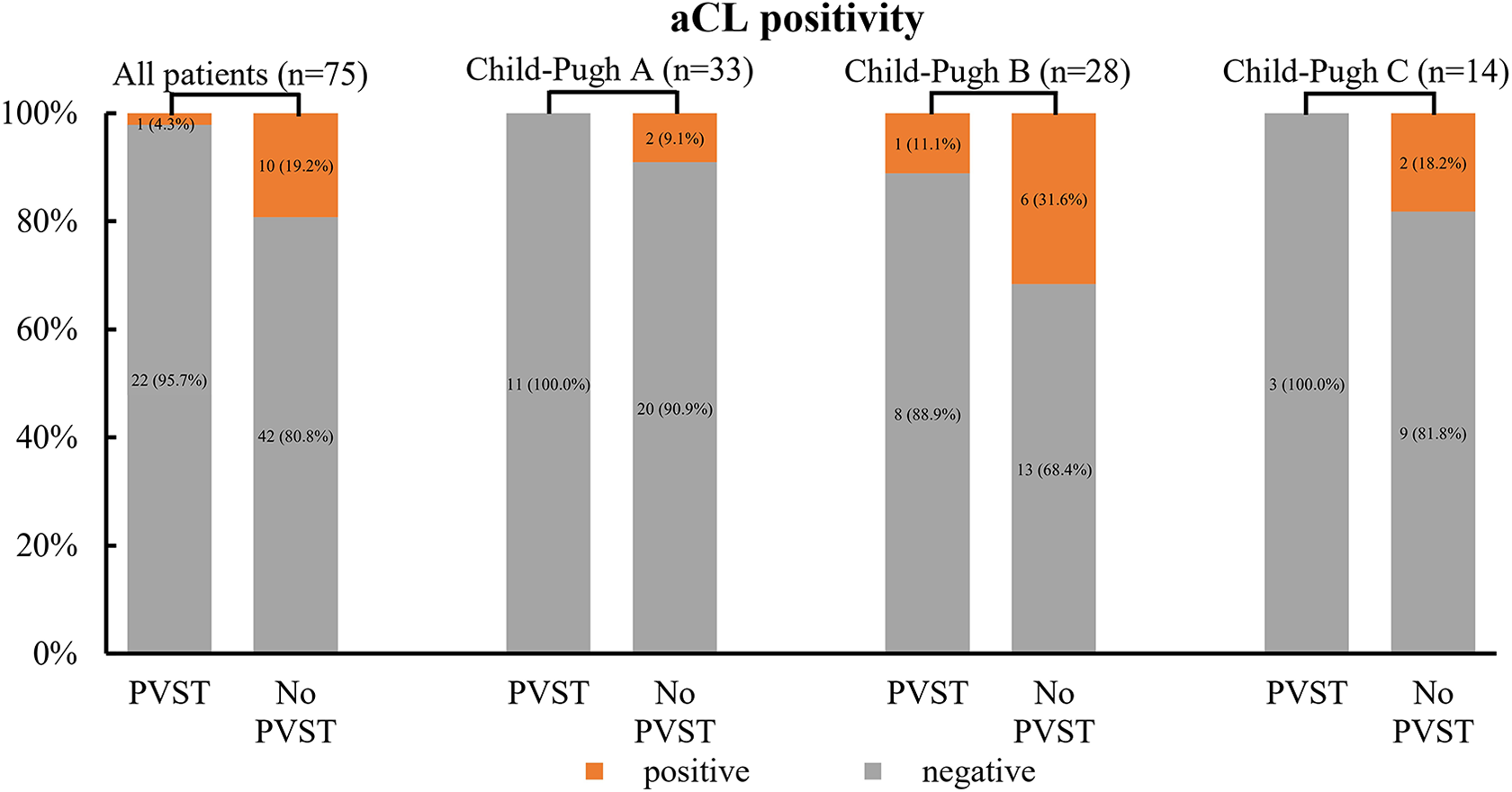
No significant difference of aCL positivity between patients with and without PVST in liver cirrhosis based on Child-Pugh class.

No significant difference of aβ2GPI positivity between patients with and without PVST in liver cirrhosis based on Child-Pugh class.
Among the 59 Child-Pugh class A patients, median Hcy level [10.71 μmol/L (2.71-38.71) versus 10.54 μmol/L (3.68-31.79); P = 0.803] and proportions of HHcy [1/22 (4.5%) versus 5/33 (15.2%); P = 0.384] (Figure 2), positive aCL [0/11 (0%) versus 2/22 (9.1%); P = 0.542] (Figure 3), and positive aβ2GPI [2/11 (18.2%) versus 6/22 (27.3%); P = 0.886] (Figure 4) remained not significantly different between patients with and without PVST.
Among the 53 Child-Pugh class B patients, median Hcy level [10.09 μmol/L (4.40-56.82) versus 9.54 μmol/L (3.87-53.44); P = 0.849] and proportions of HHcy [2/18 (11.1%) versus 4/31 (12.9%); P = 1.000] (Figure 2), positive aCL [1/9 (11.1%) versus 6/19 (31.6%); P = 1.000] (Figure 3), and positive aβ2GPI [5/9 (55.6%) versus 10/19 (52.6%); P = 1.000] (Figure 4) remained not significantly different between patients with and without PVST.
Among the 24 Child-Pugh class C patients, median Hcy level [10.75 μmol/L (8.28-27.94) versus 10.96 μmol/L (2.05-34.36); P = 0.720] and proportions of HHcy [1/4 (25%) versus 4/17 (23.5%); P = 1.000] (Figure 2), positive aCL [0/3 (0%) versus 2/11 (18.2%); P = 1.000] (Figure 3), and positive aβ2GPI [2/3 (66.7%) versus 5/11 (45.5%); P = 1.000] (Figure 4) remained not significantly different between patients with and without PVST.
MPV Thrombosis >50% Versus MPV Thrombosis <50% and MPV Patency in Liver Cirrhosis
Among the 136 cirrhotic patients with evaluable PVST, 21 were diagnosed with MPV thrombosis >50% and 115 with MPV thrombosis <50% and MPV patency. Median age [59 years (42-81) versus 55 years (28-83); P = 0.192] and proportion of male [14/21 (66.7%) versus 80/115 (69.6%); P = 0.791] were statistically similar between patients with MPV thrombosis >50% and those with MPV thrombosis <50% and MPV patency. Median Hcy level [9.70 μmol/L (2.71-56.82) versus 9.97 μmol/L (2.05-53.44); P = 0.944] and proportions of HHcy [3/20 (15%) versus 14/105 (13.3%); P = 0.735] (Supplementary Figure 1A), positive aCL [1/14 (7.1%) versus 10/61 (16.4%); P = 0.643] (Supplementary Figure 1B), and positive aβ2GPI [7/14 (50%) versus 23/61 (37.7%); P = 0.397] (Supplementary Figure 1C) were not significantly different between the 2 groups.
Clinically Significant PVST Versus No Clinically Significant PVST in Liver Cirrhosis
Among the 136 cirrhotic patients with evaluable PVST, 8 were diagnosed with clinically significant PVST and 128 without. Median age [61 years (45-73) versus 54 years (28-83); P = 0.123] and proportion of male [6/8 (75.0%) versus 88/128 (68.8%); P = 1.000] were statistically similar between patients with and without clinically significant PVST. Median Hcy level [11.73 μmol/L (8.55-27.94) versus 9.97 μmol/L (2.05-56.82); P = 0.149] and proportions of HHcy [1/7 (14.3%) versus 16/118 (13.6%); P = 1.000] (Supplementary Figure 2A), positive aCL [0/6 (0%) versus 11/69 (15.9%); P = 0.583] (Supplementary Figure 2B), and positive aβ2GPI [1/6 (16.7%) versus 29/69 (42.0%); P = 0.434] (Supplementary Figure 2C) were not significantly different between the 2 groups.
Discussion
The present study has explored the association of Hcy level, HHcy, aCL, and aβ2GPI with PVST in liver cirrhosis. As for the study design, a major advantage of the present study should be the use of contrast-enhanced CT or MRI or CT portal venography to comprehensively and accurately identify the presence of thrombosis within portal venous system; and another advantage should be that degree of MPV thrombosis and clinically significant PVST are analyzed in order to determine the potential impact of these possible thrombophilia factors on the severity of PVST. The major findings were as follows: (1) Hcy level and proportion of HHcy were not significantly higher in cirrhotic patients than in non-cirrhotic patients, but Child-Pugh score significantly correlated with HHcy in liver cirrhosis; (2) cirrhotic patients had a significantly higher proportion of positive aβ2GPI than non-cirrhotic patients, and Child-Pugh score significantly correlated with positive aβ2GPI in liver cirrhosis; and (3) Hcy level, HHcy, aCL, and aβ2GPI were not significantly associated with the risk of PVST in liver cirrhosis, regardless of severity of liver disease and PVST.
Patients with liver cirrhosis have significantly higher Hcy level and/or prevalence of HHcy than healthy subjects 17,18 and patients with chronic active hepatitis and fatty liver. 17 This is because liver cirrhosis has reduced levels of folate and vitamin B12 and B6 and decreased expression of genes, all of which are related to Hcy clearance. 19,20 Unfortunately, the present study did not confirm a significant association of Hcy level or HHcy with liver cirrhosis. This unexpected phenomenon might be ascribed to the fact that non-cirrhotic patients selected in the present study were a heterogeneous group of population and suffered from various types of digestive diseases other than liver cirrhosis and malignancy. In addition, among the patients with liver cirrhosis, the severity of liver disease is often in parallel with the prevalence of HHcy. 18,21 Similarly, the present study found that patients with Child-Pugh class B/C had a higher proportion of HHcy, and Child-Pugh score positively correlated with HHcy.
Hcy may cause a toxic effect on the vascular endothelium, which activates clotting cascade and induces the development of arterial and venous thrombosis. 22 Elevated Hcy levels significantly increase the risk of myocardial infarction and ischemic stroke, 23 but HHcy might not be associated with an increased risk of recurrent venous thrombosis. 24 The association of Hcy level and HHcy with PVST in patients with liver cirrhosis is also inconsistent. In a retrospective study, higher Hcy level and HHcy should be significant risk factors for PVST in liver cirrhosis even after correction for vitamin and methylenetetrahydrofolate reductase. 18 But it should be noted that more than half of PVST patients included in this study had a concomitant diagnosis of hepatocellular carcinoma, which may have an impact on Hcy level, coagulation system, and PVST. By comparison, in another study, which included non-malignant patients with liver cirrhosis, HHcy was not associated with an increased risk of PVST in both univariate and multivariate analyses. 25 A cross-sectional study also enrolled non-malignant patients with liver cirrhosis and found that Hcy level was not significantly different between patients with and without PVST. 26 In consistence with the latter 2 studies, the present study also demonstrated that Hcy level and prevalence of HHcy were statistically similar between cirrhotic patients with and without PVST, regardless of severity of liver disease. Furthermore, we found no impact of Hcy on the severity of PVST.
APS is characterized by arterial and venous thrombosis and/or pathological pregnancy in the presence of persistent antiphospholipid antibody positivity. 27 Antiphospholipid antibodies for a diagnosis of APS include aCL IgG and IgM antibodies, aβ2GPI IgG and IgM antibodies, and lupus anticoagulants. 28 Notably, we found that the positivity of antiphospholipid antibodies was more frequent in patients with liver cirrhosis, which was consistent with previous studies. 29,30 This phenomenon could be explained by the fact that hepatitis viral infections, especially hepatitis C virus infection, 31 alcohol abuse, 32,33 and autoimmune liver disease 34 disrupt hepatocytes membranes and then stimulate the production of neoantigens, which can be recognized by the immune system, thereby releasing antiphospholipid antibodies. On the other hand, liver necrosis and fibrosis can induce hepatocytes and endothelial cells to express adhesion molecules, thus leading to the production of autoantibodies. 35 Accordingly, the severity of liver disease might be associated with antiphospholipid antibodies. 30,32 The present study also found that Child-Pugh class B/C patients had a higher proportion of positive aβ2GPI, and Child-Pugh score positively correlated with positive aβ2GPI.
In a prospective study, only 1 of 10 patients with PVST and none of 51 patients without PVST had positive aCL. 36 Such a particularly low proportion of positive aCL was also observed in 2 other studies (0% and 0% in patients with and without PVST in the study by Chen et al, respectively; 2.6% and 3.2% in patients with and without PVST in the study by Ventura et al, respectively). 3,18 These data failed to establish the association of aCL with PVST in liver cirrhosis. Despite Amitrano et al found a relatively higher frequency of aCL positivity in patients with liver cirrhosis (aCL IgG 43.9% and 48.7%, aCL IgM 14% and 17.1% in patients with and without PVST, respectively), 25 they did not indicate any significant association of positive aCL IgM and IgG with PVST. Similarly, 2 studies, which quantitatively measured the titers of aCL, also found that aCL activity was not significantly different between patients with and without PVST. 26,37 In consistence with these previous studies, the present study found that positive aCL was relatively infrequent (4.3% and 19.2% in cirrhotic patients with and without PVST, respectively), and that aCL was not significantly associated with PVST, regardless of severity of liver disease.
In a study, none had positive aβ2GPI in cirrhotic PVST and non-PVST groups. 38 By comparison, the present study found a relatively high proportion of positive aβ2GPI (39.1% and 40.4% in patients with and without PVST, respectively), but the relationship between positive aβ2GPI and PVST could not be established. Besides, a prospective study demonstrated that the baseline aβ2GPI level could not predict the development of PVST during a 1-year follow-up. 39
Only one previous study had explored the influence of APS on the extension of non-cirrhotic and non-malignant PVST, and did not find any difference in the prevalence of APS between patients with thrombosis within MPV alone and those within MPV and SMV. 40 The present study further found that aCL and aβ2GPI positivity were not significantly associated with MPV thrombosis >50% or clinically significant PVST.
The present study has several major limitations. First, this was a cross-sectional study, so it could not explain the cause-effect association of Hcy, aCL, and aβ2GPI with PVST in liver cirrhosis. Second, multiple measurements of Hcy, aCL, and aβ2GPI were lacking. Third, the titers of aCL and aβ2GPI were not quantitatively measured. Fourth, IgM and IgG types of aCL and aβ2GPI were not clarified. Fifth, lupus anticoagulants, another marker of antiphospholipid antibody, were not measured. Sixth, Hcy level was a component of lipid profile laboratory tests at our hospital, so a majority of our patients underwent Hcy measurement. But only a minority of patients underwent aCL and aβ2GPI measurement.
Conclusion
Positive aβ2GPI may be associated with presence of liver cirrhosis and severity of liver disease, and HHcy may be positively associated with the severity of liver cirrhosis. However, we cannot establish any significant relationship of Hcy, aCL, and aβ2GPI with the presence of PVST, MPV thrombosis >50%, and clinically significant PVST. Therefore, screening for Hcy, aCL, and aβ2GPI should not be considered as routine thrombophilia testing for PVST in liver cirrhosis.
Supplemental Material
Supplemental Material, sj-jpg-1-cat-10.1177_10760296211010969 - No Association of Homocysteine, Anticardiolipin Antibody, and Anti-β2 Glycoprotein I Antibody With Portal Venous System Thrombosis in Liver Cirrhosis
Supplemental Material, sj-jpg-1-cat-10.1177_10760296211010969 for No Association of Homocysteine, Anticardiolipin Antibody, and Anti-β2 Glycoprotein I Antibody With Portal Venous System Thrombosis in Liver Cirrhosis by Le Wang, Xiaozhong Guo, Xiangbo Xu, Shixue Xu, Juqiang Han, Ran Wang, Zeqi Guo, Fangfang Yi and Xingshun Qi in Clinical and Applied Thrombosis/Hemostasis
Supplemental Material
Supplemental Material, sj-jpg-2-cat-10.1177_10760296211010969 - No Association of Homocysteine, Anticardiolipin Antibody, and Anti-β2 Glycoprotein I Antibody With Portal Venous System Thrombosis in Liver Cirrhosis
Supplemental Material, sj-jpg-2-cat-10.1177_10760296211010969 for No Association of Homocysteine, Anticardiolipin Antibody, and Anti-β2 Glycoprotein I Antibody With Portal Venous System Thrombosis in Liver Cirrhosis by Le Wang, Xiaozhong Guo, Xiangbo Xu, Shixue Xu, Juqiang Han, Ran Wang, Zeqi Guo, Fangfang Yi and Xingshun Qi in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Abbreviations
Authors’ Note
Le Wang, Xiaozhong Guo, Xiangbo Xu, Shixue Xu, and Juqiang Han are co-first authors. Conceptualization: Xingshun Qi; Methodology: Le Wang, Xiangbo Xu, Shixue Xu, and Xingshun Qi; Validation: Le Wang and Xingshun Qi; Formal analysis: Le Wang and Xingshun Qi; Investigation: Le Wang, Xiaozhong Guo, Xiangbo Xu, Juqiang Han, Zeqi Guo, and Xingshun Qi; Data curation: Le Wang, Xiangbo Xu, Shixue Xu, Ran Wang, Zeqi Guo, Fangfang Yi, and Xingshun Qi; Writing–original draft: Le Wang and Xingshun Qi; Writing–review and editing: all authors; Supervision: Xingshun Qi; Project administration: Xingshun Qi. The details of any images and tables can be published and all authors have made an intellectual contribution to the manuscript and approved the submission and publication. The data presented in this study are available on request from the corresponding author. The data are not publicly available due to privacy. The study protocol was approved by the medical ethical committee of our hospital with an approval number of Y (2020) 032. The requirement for informed consent was waived due to the retrospective nature of this study.
Acknowledgments
The authors would like to greatly appreciate the insightful comments from Prof. Dominique Valla and Prof. Ton Lisman to further improve this paper.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partially supported by the Science and Technology Project Foundation of Shenyang (19-112-4-005) and Science and Technology Plan Project of Liaoning Province (2020JH2/10300163).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
