Abstract
In order to investigate the relationship between hemostatic abnormalities and portal vein thrombosis (PVT) in hepatocellular carcinoma (HCC), platelets, prothrombin time (PT), activated partial thromboplastin time (aPTT), thrombin time, fibrinogen,
Keywords
Introduction
The liver plays several key roles in blood coagulation, being involved in both primary and secondary hemostasis. 1 The hemostatic system is in a delicate balance between prothrombotic and antithrombotic processes, aiming to prevent excessive blood loss from injured vessels and to prevent spontaneous thrombosis. Liver failure is accompanied by multiple changes in the hemostatic system, because of reduced plasma levels of procoagulant and anticoagulant clotting factors synthesized by hepatocytes and sinusoidal cells. 2 Vitamin K deficiency may coexist, so that abnormal clotting factors are produced due to lack of gamma carboxylation. Moreover during liver failure, there is a reduced capacity to clear activated hemostatic proteins and protein inhibitor complexes from the circulation. Thus, the global effect of liver disease with regard to hemostasis is complex, so that patients with advanced liver disease can experience severe bleeding or even thrombotic complications. Finally, when marked portal hypertension develops with collateral circulation and secondary splenomegaly, thrombocytopenia develops due to splenic sequestration. However, thrombocytopenia may also be due to decreased hepatic thrombopoietin synthesis. There is also impaired platelet function. 1–3
Over the last years, portal vein thrombosis (PVT) is increasingly frequently being diagnosed by a wide use of ultrasound-Doppler equipment. Recently, the lifetime risk of getting PVT in the general population is reported to be 1%. 4 Two thirds of PVT cases are related with cirrhosis or hepatocellular carcinoma (HCC). Portal vein thrombosis seems most often to develop in the presence of both systemic and local risk factors, 5,6 but the relative importance of these factors for the course and treatment strategy of the condition remains unclear. Thus, there are several unresolved issues regarding PVT that due to the rarity of the condition cannot be clarified by large trials. 7 Portal vein thrombosis is rare in intrahepatic cholangiocarcinoma and metastatic liver tumors but common in HCC. Although in some cases invasion of the portal veins by tumor cells was the reason of PVT in HCC, in a significant percentage there is no satisfying explanation for the increased incidence of PVT in HCC. In order to investigate the relationship between hemostatic abnormalities and PVT in HCC, hemostatic state of cirrhotic liver and primary and metastatic liver malignancies was studied.
Methods
Platelet counts, prothrombin time (PT), activated partial thromboplastin time (aPTT), thrombin time (TT), fibrinogen,
Demographic characteristics of the groups were shown in Table 1 . Statistical difference was not present between the groups. Patients were considered to have cirrhosis, if they had a liver biopsy, or a history and clinical presentation consistent with cirrhosis. Hepatocellular carcinoma was diagnosed by biopsy or abdominal ultrasound and spiral abdominal computed tomography together with high levels of α-fetoprotein. Only biopsy-proven cholangiocarcinoma and metastatic liver tumor patients were accepted for the study. The presence of thrombosis in portal system was investigated with Doppler ultrasound examination. Healthy control participants were selected from among the applicants of checkup and vaccination, otherwise healthy. All blood samples of patients with HCC were taken before any local (ethanol injection, radiofrequency ablation, etc) or systemic therapy. Also patients with detected portal vein tumor thrombosis were excluded from the study. All the patients with PVT had experienced thrombosis at least 3 months ago. Patients using anticoagulant or antiplatelet agents were excluded from the study. Also, during the sample collection, none of the patients with PVT was under the antithrombotic treatment. Previous endoscopic variceal treatment (band ligation or sclerotherapy) was not an exclusion criterion. The blood samples of cholangiocarcinoma group were taken before performing endoscopic retrograde cholangiopancreatography, and none of them had cholangitis during the sample collection. All patients and control participants gave their informed consent to participate in the study, which was approved by the local ethic committee.
Demographic Features of the Study Groups a
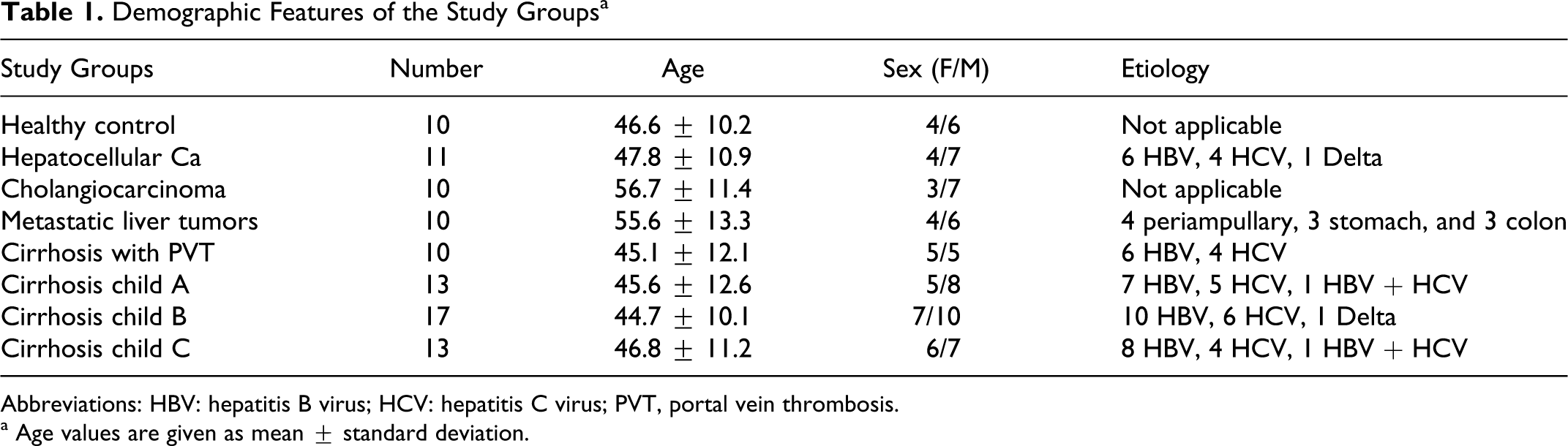
Abbreviations: HBV: hepatitis B virus; HCV: hepatitis C virus; PVT, portal vein thrombosis.
a Age values are given as mean ± standard deviation.
All blood samples were drawn fasting between 08:00 and 11:00
Platelet counts, aPTT, PT, TT, and fibrinogen were measured using conventional methods (Thromborel S, Pathromtin SL, BC thrombin Reagent, Multifibren U; BCT, Dade-Behring, Marburg, Germany). The activities of CFs V, VII, VIII, IX and XI, and protein C were assayed in stages using a parallel line bioassay based on aPTT, PT, or TT (coagulometric method; CFs V-, VII-, VIII-, IX-, and XI-deficient Plasma, BCT; Dade-Behring). The level of the
Statistical analysis
All statistical tests were done using SPSS 13 software. Data were presented as mean ± standard deviation. For nonparametric values, Kruskal-Wallis test and for parametric values 1-way analysis of variance and t test were used. Statistical significance was set as P values lower than .05.
Results
Results are given in Table 2
. When compared with the control group,
The Hemostatic Parameters of the Groups a
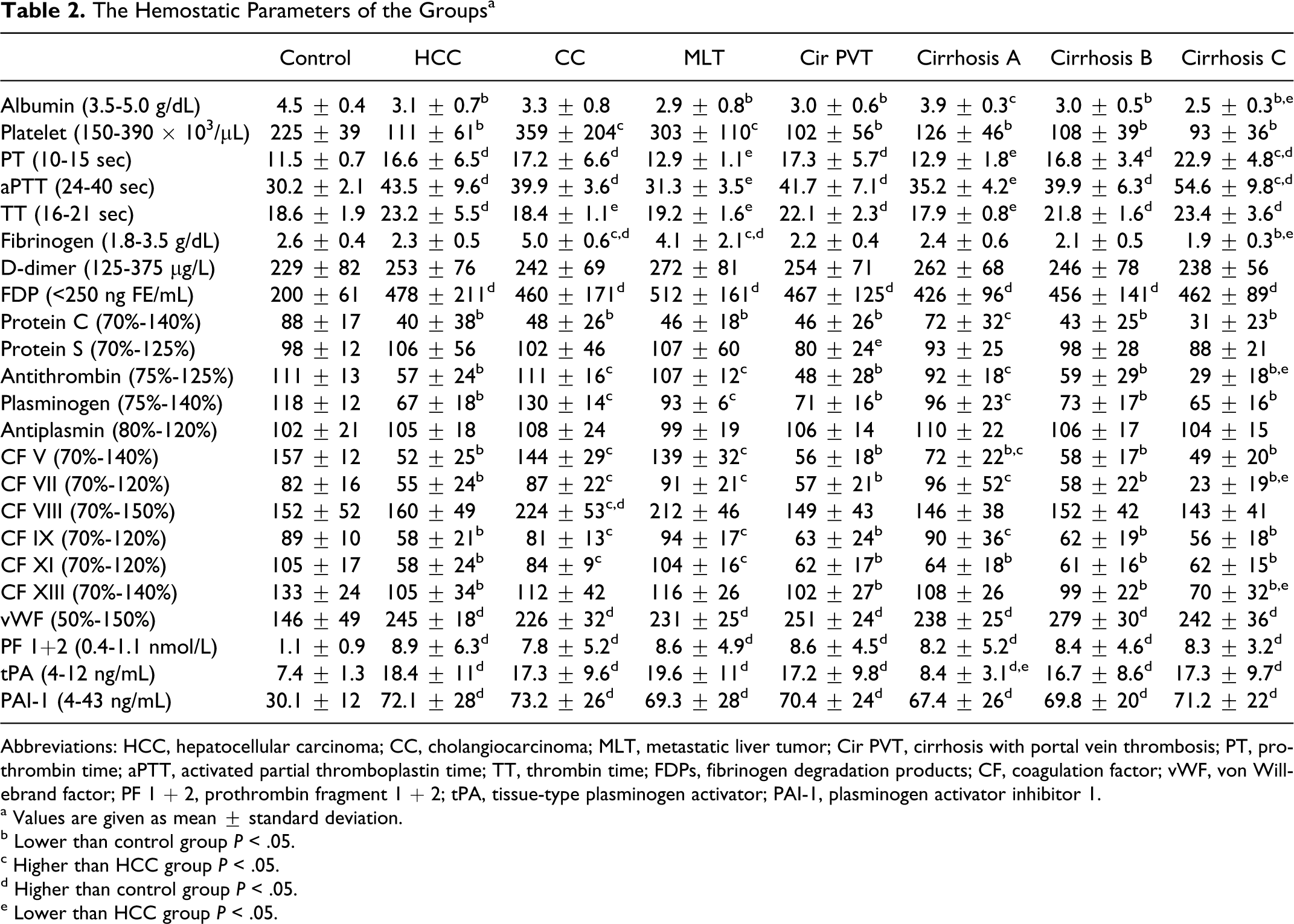
Abbreviations: HCC, hepatocellular carcinoma; CC, cholangiocarcinoma; MLT, metastatic liver tumor; Cir PVT, cirrhosis with portal vein thrombosis; PT, prothrombin time; aPTT, activated partial thromboplastin time; TT, thrombin time; FDPs, fibrinogen degradation products; CF, coagulation factor; vWF, von Willebrand factor; PF 1 + 2, prothrombin fragment 1 + 2; tPA, tissue-type plasminogen activator; PAI-1, plasminogen activator inhibitor 1.
a Values are given as mean ± standard deviation.
b Lower than control group P < .05.
c Higher than HCC group P < .05.
d Higher than control group P < .05.
e Lower than HCC group P < .05.
When compared with control, HCC, cirrhosis with PVT, Child-Pugh classes B and C cirrhosis groups showed similar findings. While
When we compare the other disease groups with HCC group, we saw that Child-Pugh class B cirrhosis group did not have any differences when compared to the HCC group. Also cirrhosis with PVT group did not show any differences from the HCC group with the exception of protein S level. The protein S level of cirrhosis with PVT group was found significantly lower than the HCC group. While albumin, protein C, antithrombin, plasminogen, and CFs V, VII, and IX levels of Child-Pugh class A cirrhosis group were found to be higher than HCC group, PT, aPTT, and TT levels were found to be lower than the HCC group. When we compare Child-Pugh class C cirrhosis and HCC groups, we saw that PT and aPTT levels of the Child-Pugh class C cirrhosis group were higher and albumin, fibrinogen, antithrombin and CFs VII and XIII levels of the Child-Pugh class C cirrhosis group were lower than the HCC group. Platelet, fibrinogen, antithrombin, plasminogen, and CFs V, VII, IX, and XI levels of cholangiocarcinoma and metastatic liver tumor groups were found to be higher than the HCC group. Also CF VIII level of the cholangiocarcinoma group was higher than the HCC group. While PT, aPTT, and TT levels were all lower in the metastatic liver tumor group when compared with HCC group, only TT level of the cholangiocarcinoma group was found to be lower than the HCC group. Findings of the metastatic liver tumor group were similar to the cholangiocarcinoma group, except that the levels of aPTT and plasminogen were found lower and the level of CF 11 was higher in metastatic liver tumor group when compared with cholangiocarcinoma group. So, similar differences were seen between the HCC and metastatic liver tumor groups, as present between the HCC and cholangiocarcinoma groups.
When we look at the results of the cirrhosis group, we detect important correlations between the Child-Pugh class and hemostatic parameters. Albumin, platelets, fibrinogen, protein C, antithrombin, plasminogen, and CFs V, VII, IX and XIII levels were all decreased significantly with the deterioration of the liver function from Child-Pugh class A to C. Also PT, aPTT, TT, and tPA levels were all increased significantly with the deterioration of the liver function from Child-Pugh class A to C. When we compare the cirrhosis with PVT group, with the PVT-negative cirrhosis groups we detect that the findings of the cirrhosis with PVT group was similar to the Child-Pugh class B cirrhosis group, with the exception of protein S level.
Discussion
Liver cirrhosis is the most frequent underlying disease in patients with nonneoplastic thrombosis occurring in the portal vein. In a large autopsy series of patients with PVT, neoplasia was present in 67%, cirrhosis in 28%, and in only 5% of patients PVT was considered primitive. 4 Hepatocellular carcinoma is the most frequent cause of PVT in cirrhosis, being present in up to 44% of cases, 8 and always it has to be searched for when a new diagnosis of PVT is made.
It is generally thought that liver cirrhosis leads to a progressive bleeding tendency because of the complex alteration of the clotting system induced by liver failure, but recent studies have demonstrated that liver cirrhosis, causing a reduced synthesis of either procoagulant or anticoagulant proteins is still associated with a balanced equilibrium of the clotting system set at a lower level. A series of acquired or inherited conditions can easily tilt this equilibrium toward either bleeding or thrombosis. Epidemiological studies show a prevalence of venous thrombosis of 0.5% to 1% in patients with liver cirrhosis is even greater than in the general population. 9,10 As in all venous thrombosis, the pathogenesis of PVT in noncirrhotic and in cirrhotic patients is multifactorial, even if not all known risk factors can be studied easily in this setting due to the impaired synthetic and metabolic ability of the liver. Congenital or acquired, local or systemic factors can interact causing reduction in blood flow velocity, endothelial damage and clotting unbalance. 5 So, in order to investigate the relationship between hemostatic abnormalities and PVT in HCC this study was done.
Platelets number and function are important in primary hemostasis. About one third of patients with chronic liver disease develop thrombocytopenia, which worsens in parallel with disease progression associated with increased platelet sequestration due to hypersplenism and/or impaired thrombopoietin synthesis. 11,12 von Willebrand factor is synthesized by the endothelium and mediates platelet-to-platelet interactions and platelet adhesion to the subendothelium. In chronic liver disease, endothelial shear stress related to portal hypertension may contribute to the high plasma levels of vWF via a nitric oxide stimulus. 13 A correlation between severity of liver disease and vWF plasma antigen levels has been documented. 14 A very recently published meta-analysis of primary hemostasis (platelets and its functions) in liver cirrhosis reported that primary hemostasis may not be defective in cirrhosis and a low platelet count should not be considered as an automatic index of an increased risk of bleeding. 15 In our study while cholangiocarcinoma and metastatic liver tumor groups’ platelet levels were not different from the control, the platelet levels of all the cirrhosis groups and HCC group were found significantly lower than the control group. Also vWF activities of all disease groups were found significantly higher than the control.
The liver is the site of synthesis of fibrinogen and CFs II, V, VII, IX, X, XI, XII, and XIII. 16 Besides the low levels of these CFs due to impaired synthesis capacity of the liver, dysfunctional proteins are also found in patients with liver failure. It has been shown that in liver disease part of the circulating prothrombin and protein C lack their c-carboxyglutamic acid residues, and this is presumably also true for the other vitamin-K-dependent proteins (CFs VII, IX, and X, and protein S). Coagulation factor VIII is synthesized mainly by the hepatic but also nonhepatic sinusoidal endothelial cells, 17 thus the plasma concentration of CF VIII is not decreased with liver disease and may be even increased, as many chronic liver diseases are associated with chronic inflammation. 18 Coagulation factor XIII are synthesized by liver, monocytes, and macrophages, so its level may remain normal. The CF activities of our study groups were consistent with the literature.
Plasma fibrinogen is an acute-phase reactant and remains normal or increased in patients with liver disease. Low concentrations due to decreased synthesis, yet above 100 mg/dL, are only seen with very severe liver disease. 19 However, the high fibrinogen concentrations found in patients with chronic hepatitis, cholestatic jaundice, and HCC do not result in increased clot formation as most is a nonfunctional fibrinogen present in 60% to 70%. Fibrinogen may be decreased in severe cirrhosis and TT may be prolonged in patients with hypofibrinogenemia, dysfibrinogenemia, or high levels of FDP. Also fibrinogen levels of metastatic tumors were found increased according to healthy controls and nonmetastatic tumors. 20 In our study, the fibrinogen levels of all cirrhosis groups were similar with the control, with the exception of Child-Pugh class C cirrhosis group, as expected. Also the TT all of the cirrhosis groups was found to be longer than the control, with the exception of Child-Pugh class A cirrhosis group, despite the normal level of fibrinogen. Cholangiocarcinoma and metastatic liver tumor groups’ fibrinogen levels were found statistically higher than the control group.
In liver diseases, the concentration of antithrombin falls due to reduced synthesis and/or increased consumption. Usually the antithrombin deficit is mild and thrombotic complications are very rare. 21 During acute or chronic liver disease, the concentrations of protein C and S decrease concomitantly with the other CFs but usually not below 20% of the normal. 22 Low levels of anticoagulant proteins might not only compensate for the defective thrombin-generating capacity due to low levels of procoagulant proteins in patients with liver failure, reduced thrombin inhibition might also facilitate thrombosis. Tripodi et al 23 recently reported that hypercoagulability of plasma from patients with cirrhosis appears to result from increased levels of factor VIII and decreased levels of protein C. In our study, while protein S activities of all disease groups were not different from the control, protein C activities of all disease groups (with the exception of Child-Pugh class A cirrhosis group) were found to be significantly lower than the control group. On the other hand antithrombin activities of cholangiocarcinoma, metastatic liver tumor, and Child-Pugh class A cirrhosis groups were found to be similar to the control.
Thrombin itself is impossible to quantitate, so the use of surrogate markers is necessary. Prothrombin fragment 1 + 2 is an index of in vivo thrombin generation; 1 molecule of PF 1 + 2 is released with the generation of each thrombin molecule. The measurement of PF 1 + 2 would be an excellent marker of thrombin generation. The appearance of elevated plasma levels of PF 1 + 2 has been used as evidence for in vivo clotting activation in cirrhosis.
Plasminogen is an essential component of the fibrinolytic system, which is converted to plasmin by tPA and urokinase plasminogen activator and inhibited by PAI-1, PAI-2, and antiplasmin. All the proteins involved in fibrinolysis, except for tPA and PAI-1, are synthesized in the liver. Reduced plasma levels of plasminogen and antiplasmin are found in cirrhosis. 24 Conversely, tPA levels are increased in liver disease due to decreased clearance, whereas its inhibitor PAI-1 is normal or only slightly increased in plasma. The inhibitor concentrations are insufficient to counteract the increase in tPA, accounting for increased fibrinolysis. 25 In our study while antiplasmin activities of all of the groups were found to be normal, both tPA and PAI-1 levels of all disease groups were found to be significantly higher than the control group. Also plasminogen activities of the cirrhosis groups were found to be significantly lower than the control.
The FDP assay measure amounts of the fibrin and fibrinogen split products in the blood and directly indicates the level of activity of the fibrinolytic system. Plasma
As reported by the Coagulation in Liver Disease Study Group, 29 a resetting of the coagulation and anticoagulation systems at a lower level was postulated, because during liver disease both procoagulant and anticoagulant pathways are affected in a parallel manner. The appearance of elevated plasma levels of PF 1 + 2 and thrombin–antithrombin complexes has been used as an evidence for in vivo clotting activation in cirrhosis. However, accumulation of indicators of both clotting activation and fibrinolysis could also be the consequence of a reduced clearance of these molecules by the diseased liver. Thrombotic complications can paradoxically occur in patients with cirrhosis even if an increased risk of hemorrhage is considered clinically. Despite prolonged coagulation tests, these patients cannot be viewed as being “anticoagulated.” The clinical consequence of the concomitant reduction in both pro- and anticoagulant proteins is unknown.
Hypercoagulability (deep vein thrombosis and pulmonary embolism) is a well-known condition in patients with cancer. But splanchnic or PVT is exceedingly rare in solid metastatic tumors. 30–32 When we compare cholangiocarcinoma and metastatic liver tumor groups with HCC and cirrhosis with PVT groups, we detect interesting findings. Despite the significantly higher levels of the procoagulants (platelets, fibrinogen, CFs V, VII, VIII, IX, and XI) in cholangiocarcinoma and metastatic liver tumor groups, we cannot detect any PVT in these 2 groups. Interestingly, antithrombin and plasminogen activities of cholangiocarcinoma and metastatic liver tumor groups were found to be normal and significantly higher than the HCC and cirrhosis with PVT groups. So we can say that anticoagulants (especially antithrombin) are more important in the occurrence of PVT related with HCC and cirrhosis. Also the low protein S activity of the cirrhosis with PVT group may be important for the occurrence of PVT in this group of patients. These findings suggested that the abnormalities of coagulation and fibrinolysis systems have some role in provoking thrombosis of portal veins in HCC, in addition to the invasion of portal veins by hepatoma cells.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
