Abstract
Defibrotide is approved to treat hepatic veno-occlusive disease/sinusoidal obstruction syndrome (VOD/SOS) with renal/pulmonary dysfunction following hematopoietic cell transplantation (HCT) in adult and pediatric patients in the United States, and to treat severe hepatic VOD/SOS post-HCT in adult and pediatric patients aged >1 month in the European Union. The defibrotide prescribing information warns that defibrotide may increase bleeding risk in VOD/SOS patients. To broaden our understanding of the incidence of bleeding with defibrotide, we performed a meta-analysis of the published literature of defibrotide use outside of the post-HCT VOD/SOS setting. Of 1857 records identified, 125 reported on defibrotide; 23 contained data on bleeding events. The estimated overall incidence of bleeding events was 1% (95% confidence interval [CI]: 0%-2%) and 8% (95% CI: 3%-14%) in studies using intravenous defibrotide and studies with controls, respectively. The risk ratio for bleeding events with intravenous defibrotide versus controls was 0.36 (95% CI: 0.24-0.52; P < .00001) among studies with data on intravenous defibrotide and controls. This meta-analysis of defibrotide use outside of the post-HCT VOD/SOS setting suggests that the incidence of bleeding with defibrotide is lower than controls.
Introduction
Defibrotide (Defitelio) is a polydisperse mixture of predominately single-stranded polydeoxyribonucleotide sodium salts that has been shown in vitro to protect the endothelium from cytotoxic and inflammatory damage by reducing activation of endothelial cells. 1 –3 Preclinical data suggest that defibrotide reduces endothelial cell dysfunction by mechanisms that are antithrombotic, profibrinolytic, antiadhesive, and anti-inflammatory, thereby restoring the thrombotic-fibrinolytic balance and preserving endothelial homeostasis. 1,4 –6 Additionally, defibrotide has been shown to exhibit antithrombotic properties without significant effects on systemic coagulation. 1,6
In 1986, defibrotide was approved in Italy for the prophylaxis of deep vein thrombosis (DVT) and treatment of thrombophlebitis because of its profibrinolytic, anti-inflammatory, and antithrombotic effects. 7 Defibrotide is currently approved to treat adult and pediatric patients with hepatic veno-occlusive disease (VOD), also known as sinusoidal obstruction syndrome (SOS), with renal or pulmonary dysfunction following hematopoietic cell transplantation (HCT) in the United States, 8 and to treat adult and pediatric patients aged >1 month with severe hepatic VOD/SOS post-HCT in the European Union. 9 The recommended dose for treatment of VOD/SOS is 6.25 mg/kg every 6 hours (25 mg/kg/d) given as a 2-hour intravenous (IV) infusion.
Hepatic VOD/SOS is a potentially fatal complication post-HCT and may also result from chemotherapy alone. 10,11 Endothelial cell injury caused by HCT conditioning regimens or chemotherapy alone causes endothelial activation and an inflammatory response that leads to sinusoidal narrowing and the clinical manifestation of VOD/SOS. 12 Patients who have undergone HCT, including those with VOD/SOS, have a high risk of bleeding events due to low platelet levels and other comorbidities associated with the post-HCT period. 12 –14 The prescribing information for defibrotide contains a warning that defibrotide may increase the risk of bleeding in post-HCT VOD/SOS patients. Additionally, for treatment of VOD/SOS, concurrent administration of defibrotide with systemic anticoagulants, such as heparin, or fibrinolytic therapies is contraindicated. 8,9 It is of clinical interest to understand any potential association between defibrotide treatment and the underlying risk of bleeding events.
In a phase 3 study (NCT00358501) of patients with VOD/SOS and advanced multi-organ dysfunction post-HCT, rates of bleeding events with defibrotide (n = 102) were generally similar to or lower than matched historical controls (n = 32). 15 The incidence of common hemorrhagic events was 64% in the defibrotide group compared with 75% in the historical control group. The incidence of coagulopathy was 2% in the defibrotide group versus 16% in the historical control group; hypotension was observed in 39% of defibrotide-treated patients and 50% of historical controls. Real-world evidence of the incidence of bleeding associated with defibrotide was assessed in an expanded access study (T-IND; NCT00628498) of defibrotide in patients with post-HCT VOD/SOS, with and without multi-organ dysfunction (n = 1000); overall, 29% of patients experienced ≥1 hemorrhagic event. 16 Similarly, among adults who received defibrotide for treatment of severe VOD/SOS post-HCT in a postmarketing study conducted in France (n = 66), the rate of bleeding events was 21%. 17
To broaden our understanding of the incidence of bleeding events reported with defibrotide treatment, a meta-analysis was conducted using published literature. The analysis was limited to studies of defibrotide outside of the post-HCT VOD/SOS setting due to the inherent risk of bleeding associated with HCT. The overall incidence of bleeding events reported during IV defibrotide treatment was assessed, as well as the relative risk for bleeding when IV defibrotide treatments were compared to control treatments.
Methods
Search Criteria
A systematic search of PubMed (MEDLINE), the Database of Abstracts and Reviews of Effects, the Cochrane Central Register of Controlled Trials, ClinicalTrials.gov, and Embase, which was used to search for abstracts (eg, European Society for Bone and Marrow Transplantation, Blood and Marrow Transplantation, American Society for Hematology, and European Hematology Association) until July 24, 2018, was performed per a prespecified and clearly defined protocol based on Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. The search term for all databases was “defibrotide” in all fields. Duplicate results from these searches were removed.
Criteria for Study Selection
Defibrotide studies of adult or pediatric patients, including controlled trials assessing an intervention of interest, observational/retrospective studies, retrospective or post hoc analyses, and case series with ≥10 patients were identified. Studies of patients with VOD/SOS or HCT, case reports with <10 patients, and reviews were excluded. VOD/SOS and HCT studies were excluded because these results have been previously published. 15,16,18
Data Collection
The full text of the selected studies and conference abstracts were assessed for study design, sample size, dose, route of administration, treatment duration, and control comparators. Publications were evaluated for the presence of data on end points of interest, which included any bleeding events, site of bleeding (if reported), hemorrhage, other adverse events (AEs), and overall safety and efficacy summaries.
Statistical Analyses
All studies with relevant data were included in the meta-analysis. Overall incidence of bleeding was estimated for defibrotide and controls using the Freeman-Tukey double arcsine transformation and random-effects modeling (Stata software). The risk ratio of bleeding was calculated using the Mantel-Haenszel method and random-effects modeling (RevMan 5.3 software). Overall bleeding rate and risk ratio of bleeding were calculated from studies that used IV defibrotide administration only. Studies using only oral or intramuscular formulations were assessed but, due to the current defibrotide indication being for IV administration, these studies were not included in the final analysis.
Results
Literature Search Results
A total of 1857 publications were identified in the search, and 1349 publications were screened after the elimination of duplicates (Figure 1). The full text of 209 articles and conference abstracts were examined, of which 125 studies reported on defibrotide outside of the post-HCT VOD/SOS setting. The most common indications were DVT (23%) and peripheral arterial occlusive disease (17%). Trial size ranged from 10 to 4810 patients (median, 40 patients), and 4 studies had >1000 patients. The majority of studies were from Italy (119 studies), where defibrotide was initially approved for the prophylaxis of DVT and the treatment of thrombophlebitis in 1986. 7 Of the 125 studies identified in the literature search, 23 contained data related to bleeding events and were included in the meta-analysis. 19 –34
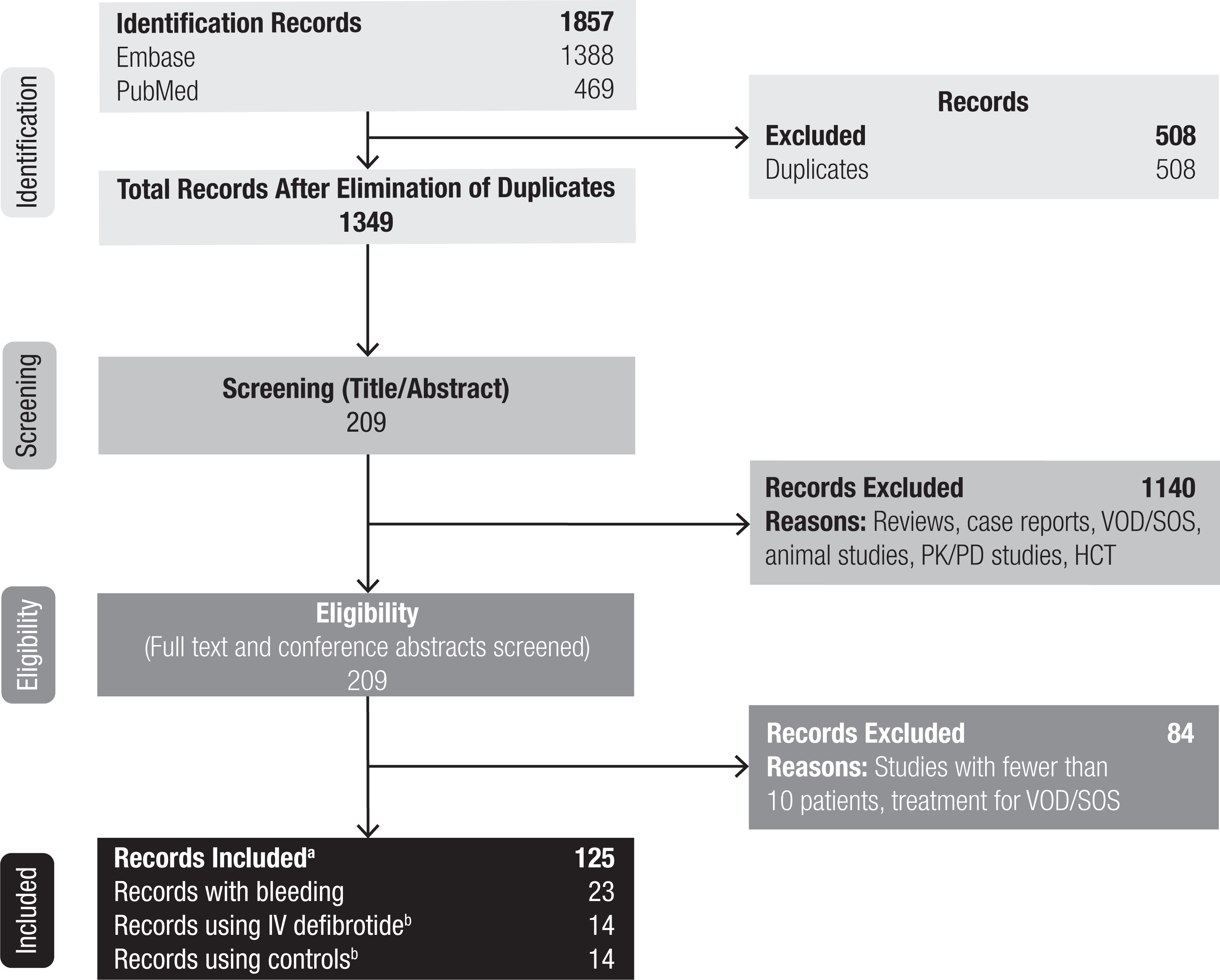
PRISMA diagram. HCT indicates hematopoietic cell transplantation; IV, intravenous; PK/PD, pharmacokinetic/pharmacodynamic; VOD/SOS, veno-occlusive disease/sinusoidal obstruction syndrome. aControlled studies assessing an intervention of interest, observational/retrospective studies, retrospective or post hoc analyses, and case series ≥10 patients were included. Studies of patients with VOD/SOS post-HCT, case reports with <10 patients, and reviews were excluded. bThese groups were not composed of the same 14 studies, although some studies were included in both groups.
Characteristics of Studies Included in the Meta-Analysis
Of the 23 studies included in the meta-analysis, 2 studies enrolled >1000 patients (Table 1). An IV formulation was used in 14 studies, and the other 9 studies used intramuscular or oral delivery, used more than one method of defibrotide administration, or the method of administration was not defined. The most common indications were prevention of DVT (13 of 23 studies) and treatment of thrombosis (4 of 23 studies). Seventeen of the studies had 2 treatment arms; among these, heparin was the most common comparator (14 of 17 studies). Of the other 6 studies, 4 were single-arm and 2 had 3 treatment arms (different defibrotide doses). The majority of the studies were conducted in adults (20 of 23), and the other 3 studies did not specify patient age. All the studies were conducted in Italy.
Summary of Studies Included in the Meta-Analysis.

Abbreviations: AMI, acute myocardial infarction; bid, 2 times a day; DF, defibrotide; DVT, deep vein thrombosis; IM, intramuscular; IV, intravenous; NR, not reported; PO, oral; qid, 4 times a day.
Bleeding Incidence
Table 2 provides a summary of the defibrotide dosing and bleeding events from the 23 studies included in the meta-analysis. Among these studies, 14 reported on use of IV defibrotide and 14 studies included a control arm; some studies were included in both groups. The incidence and risk ratio of bleeding were calculated based upon data from 14 studies that used IV defibrotide. The majority of studies that used IV defibrotide used a dose of 800 mg/d (12 of 14). The Milazzotto et al study assessed 2 dose levels of defibrotide (group 1: 2.8 g/d; group 2: 5.6 g/d), 39 and Tubaro et al used a dose that tapered from 3.6 to 1.2 g/d. 24 The most common reported cause of bleeding was intra- and/or postoperative bleeding (Table 2). Incidence of bleeding events reported in the 14 studies using only IV formulations of defibrotide ranged from 0% to 10% in individual studies, and the estimated overall bleeding rate was 1% (95% confidence interval [CI]: 0%-2%; Figure 2A). In 14 studies with control treatments (12 heparin, 1 urokinase, 1 not reported), incidence of bleeding events with controls ranged from 0% to 37%, with 6 studies reporting ≥10%. Across the 14 studies with available data for controls, the estimated overall bleeding incidence was 8% (95% CI: 3%-14%; Figure 2B).

Bleeding rates for IV defibrotide (A) and controls regardless of route of administration (B). CI indicates confidence interval; ES, effect size; IV, intravenous.
Defibrotide Dosing and Reported Bleeding Events.

Abbreviations: bid, 2 times a day; DF, defibrotide; IM, intramuscular; IV, intravenous; N/A, not available; NR, not reported; PO, oral; qid, 4 times a day.
aBleeding events included intraoperative blood loss >300 mL.
Among the 10 studies that reported data on both IV defibrotide and active controls (8 heparin, 1 urokinase, 1 conventional supportive treatment), the risk ratio for bleeding favored defibrotide over controls (0.36 [95% CI: 0.24-0.52; P < .00001]; Figure 3).
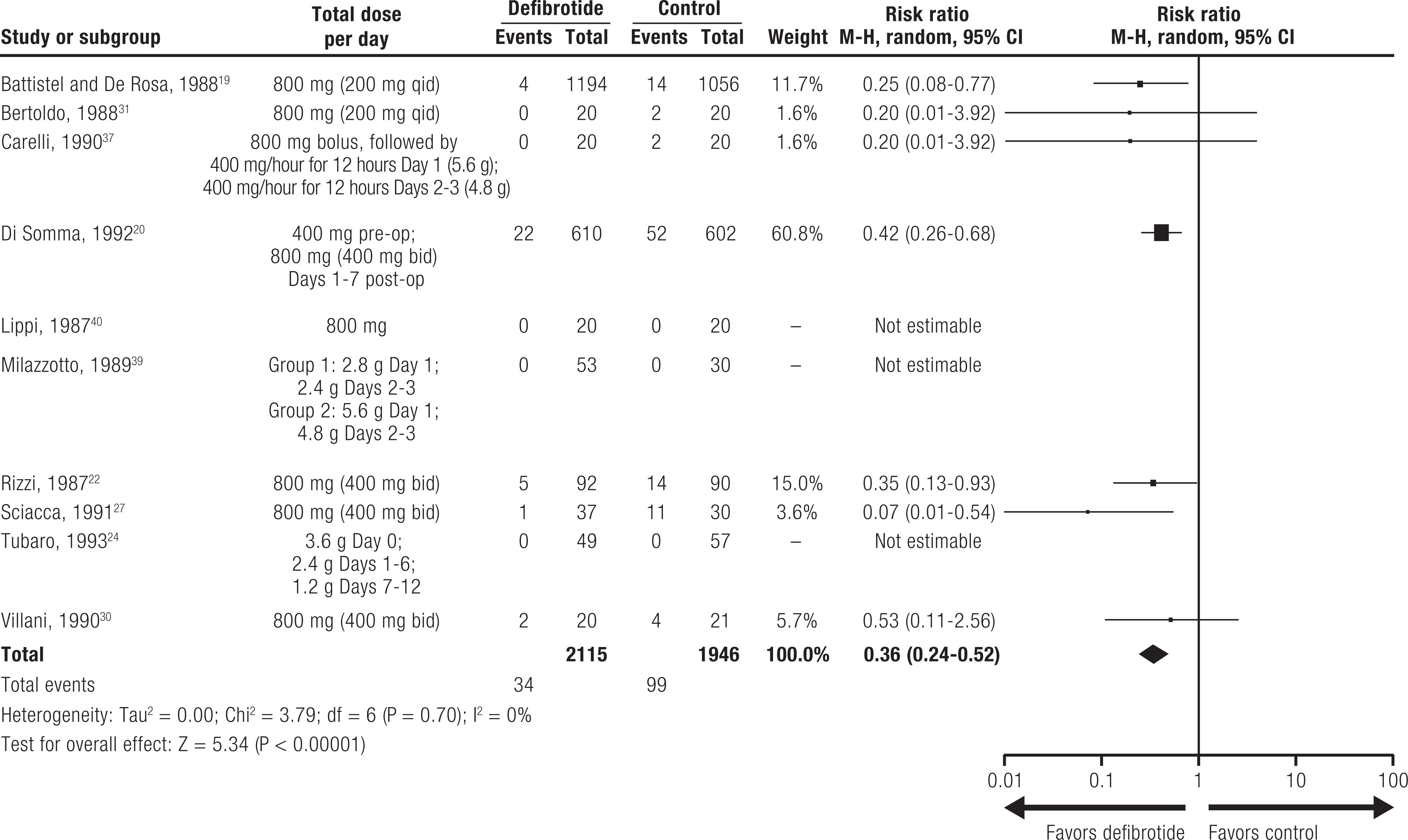
Risk ratio of bleeding for IV formulation of defibrotide versus controls. bid indicates 2 times a day; CI, confidence interval; df, degrees of freedom; IV, intravenous; M-H, Mantel-Haenszel; post-op, postoperative; pre-op, preoperative; qid, 4 times a day.
Discussion
In this meta-analysis of patients treated with defibrotide outside of the post-HCT VOD/SOS setting, the overall incidence of bleeding with defibrotide was low (at 1%), and there was a lower incidence of bleeding with defibrotide versus controls such as heparin or urokinase. In studies with active controls reporting bleeding incidence in patients treated with IV defibrotide, the risk of bleeding was lower with defibrotide compared to controls.
Defibrotide is thought to act by stabilizing endothelial cells and restoring thrombo-fibrinolytic balance. The mechanism of action is multifaceted, with demonstrated antithrombotic, fibrinolytic, antiadhesive, and anti-inflammatory properties. 1 Available data suggest that the mechanisms of defibrotide and heparin are distinct, as defibrotide may not have systemic anticoagulant effects. 1,6 In addition, it has been shown that defibrotide does not alter activated partial thromboplastin time, prothrombin time, or thrombin time. 42 Defibrotide has been shown to increase levels of tissue plasminogen activator, thrombomodulin, and tissue factor pathway inhibitor and reduce levels of plasminogen activator inhibitor-1, tissue factor, and von Willebrand factor. 1,3,6 In addition, it has been reported that the antithrombotic actions of defibrotide may be limited to the microvasculature, with less activity on macrovascular endothelial cells. 6,43
There are several important limitations to this analysis. First, there were variations in AE reporting across the studies. In addition, all of the studies in the analysis were conducted in Italy and, with the exception of one, 33 all occurred before the year 2000, so the results may not be generalizable to other countries and may not reflect the current standard of care or AE management practices. Furthermore, older abstracts (prior to 1986) may not have been available for some congresses, and therefore may be missing from this analysis. Congress abstracts were identified using Embase; however, a more extensive search of local congresses in local languages may have yielded more studies. The results of the meta-analysis are limited to adults, as no pediatric studies were included and thus, conclusions should not be generalized to the pediatric patient population. Finally, the 800 mg/d dose (∼11 mg/kg/d for a 70-75 kg patient) used in most of the studies included in the analysis is lower than the recommended dose for VOD/SOS treatment (25 mg/kg/d). 8 It is noteworthy that one of the studies included in the analysis (Tubaro et al, 1993) 24 used an initial dose of 50 mg/kg/d and reported no major bleeding events (n = 106 patients treated with defibrotide). In addition, the study by Milazzotto et al 39 treated patients with 2 defibrotide doses, 2.8 g/d in one group (n = 30) and 5.6 g/d in the other (n = 23), and also reported no bleeding events. The duration of defibrotide treatment in these studies ranged from 7 to 40 days.
Conclusion
This meta-analysis of defibrotide use outside of the post-HCT VOD/SOS setting suggests that bleeding risk with defibrotide is low (at 1%), and there is a lower incidence of bleeding with defibrotide treatment versus controls such as heparin or urokinase. Important limitations of this analysis include variations in AE reporting across studies and the use of a defibrotide dose in most included studies that is lower than the currently approved recommended dose.
Footnotes
Authors’ Note
All relevant data are provided within the manuscript and supporting files. William Tappe was affiliated with Jazz Pharmaceuticals, Palo Alto, CA, USA, at the time the meta-analysis was conducted.
Acknowledgments
Medical writing and editorial assistance were provided by Erica Chevalier-Larsen, PhD, of SciFluent Communications, Inc, and were financially supported by Jazz Pharmaceuticals.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: W.T. was an employee of Jazz Pharmaceuticals at the time of the analysis and holds stock and/or stock options in Jazz Pharmaceuticals. S.A., O.T., and M.I. have consulted for Jazz Pharmaceuticals.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Jazz Pharmaceuticals.
