Abstract
The approach to the patient with gastrointestinal bleeding (GIB) can be very complex. A multidisciplinary panel of physicians with expertise in Gastroenterology, Anesthesiology, and Transfusion Medicine worked together to provide the best knowledge and guide clinical practitioners in the real setting of health institutions, characterized by disparate availability of human and technical resources. The authors propose a global and personalized approach according to different clinical scenarios to improve the outcomes of patients with GIB, for whom the reduction of inappropriate transfusions is crucial. The goal of this document is to provide clear and objective guidance through interventional algorithms toward a goal-directed approach according to the clinical situation and supported by the latest available scientific data on GIB management in different settings.
Introduction
Specific guidelines for the management of gastrointestinal bleeding (GIB) are already published, but many unanswered questions and some controversies persist. This document reflects the opinion of a group of multidisciplinary experts with experience in the management of patients with GIB and presents their proposals based on current knowledge (Figure 1). This practical clinical approach must be adapted according to an individual clinical situation, available resources, medical expertise, and current scientific knowledge.

Gastrointestinal bleeding management algorithm.
These recommendations are not a substitute for clinician’s judgment and must be used considering other determinants that may contribute to the cause of bleeding. This last paragraph must be positionated before the Figure 1. Is not a legend of this figure.
Methods
On February 2019, a group of 14 advisors divided into 3 working groups according to their area of expertise—transfusion medicine (n = 5), anesthesiology (n = 4), and gastroenterology (n = 5)—gathered to issue a global and personalized approach statement to the management of patients with GIB according to different scenarios in the clinical practice. The aim was to develop a series of interventional algorithms that enabled an easy and practical control of GIB based on a multimodal and multidisciplinary approach. Each group of experts was assigned the consensus’ corresponding area of expertise. A comprehensive literature review about each topic in the manuscript, based on available published data, was conducted on PubMed database using the keywords “gastrointestinal bleeding,” “algorithm,” “coagulopathy,” “blood management,” “transfusion,” “goal-directed therapy,” “coagulation,” “variceal,” and “non-variceal.” Based on retrieved evidence, each working group independently developed the corresponding algorithms, which were finally assembled in 1 manuscript that was shared, reviewed, and approved by all advisors.
Initial Assessment and Resuscitation
Gastrointestinal bleeding is a common medical emergency worldwide with mortality rate of 7% at hospital admission, which increases to 26% if bleeding occurs during hospitalization for other causes. 1 Management of comorbidities ad initium has an impact on clinical outcomes (algorithm 1).
In unstable patients, it is of great value to have multidisciplinary coordination between Gastroenterology, Surgery, Anesthesiology, Transfusion Medicine, Interventional Radiology, and Intensive Care Medicine.
The initial approach to the patient presenting with GIB includes clinical evaluation and simultaneous hemodynamic and cardiorespiratory stabilization. 2,3 It is important to identify signs of liver disease (LD) and other comorbidities, patient’s medications such as oral anticoagulants (OACs), antiplatelet therapy (APT), and nonsteroidal anti-inflammatory drug (NSAID) (algorithm 2). Previous abdominal or vascular surgeries, such as abdominal aortic repair, are also important for differential diagnosis. Vital signs must be monitored according to clinical severity, speed of evolution, and hospitalization level. It is difficult to quantify blood loss in GIB. In an early phase, normal hemoglobin (Hb) and blood pressure (BP) do not exclude significant hemorrhage. In this stage, tachycardia is the best early sign of severity, except if the patient is under β-blocker therapy. 2,3
In a healthy individual, an estimated blood loss of 40% of the total blood volume (TBV) is associated with tachycardia, hypotension, tachypnea, and diaphoresis. Paleness, cool extremities, oliguria, and changes in mental status follow sustained hypotension (systolic BP <90 mm Hg or mean BP <65 mm Hg). Resuscitation measures must be according to the Airway, Breathing, Circulation, Disability, Exposure approach.
Recommended measures include considering oxygen supplementation and orotracheal intubation to protect the patient’s airway in cases of persistent hematemesis or change in consciousness level; inserting two 16-18 gauge peripheral venous accesses; collecting blood samples to perform complete blood count, coagulation, biochemistry, pretransfusional tests and, when available, viscoelastic tests (VET); administering crystalloids (polyelectrolytes); transfusion of packed red blood cells (PRBCs) in cases of severe bleeding; assuring normovolemia and normothermia; and nill by mouth. Nasogastric/orogastric intubation is not recommended on a routine basis. 3
A shock index (SI; heart rate/systolic BP ratio) ≥1 is a marker of active bleeding, allowing to identify unstable patients. 2,3 In a patient with hemodynamic instability and SI ≥1, the probability of identifying the source of bleeding is greater in angiographic examination, 2 and computerized tomography angiography (CTA) may be relevant as a first diagnostic approach in such patients. The key step is hemodynamic stabilization to assure subsequent diagnostic and therapeutic management.
Hemodynamic targets are described in Algorithm 1. In the absence of hemodynamic recovery after administration of crystalloids and PRBCs, 4,5 administration of noradrenaline should be considered in an initial dose of 0.1 to 0.2 μg/kg/min until a maximum of 2 μg/kg/min to ensure “permissive hypotension” with mean BP >65 mm Hg, until resolution of the bleeding cause.
Therapeutic targets are stop bleeding, maintaining Hb levels between 7 and 9 g/dL, 4 –6 and preventing coagulopathy while avoiding hemodilution, hypothermia, acidosis, and hypocalcemia. Transfusional support should be provided according to clinical evolution and laboratory results. Coagulopathy management should preferably be VET oriented. 7,8
The priority is to identify the semiology of active hemorrhage (hematemesis, melena, persisting/remaining hematochezia, or live/bright blood in the nasogastric probe) and LD existence. When hematochezia is associated with significant hemodynamic instability, upper GIB (UGIB) must be considered. 2 In patients with chronic LD (CLD), a portal hypertensive cause of GIB is the main hypothesis and specific measures should be initiated early (even before etiologic confirmation), including vasopressor therapy and prophylactic antibiotherapy.
Potentially Aggravating Factors in GIB
The aging of populations and associated comorbidities is one of the biggest challenges in the management of health conditions, including GIB (algorithm 2).
Liver Insufficiency
Liver disease (LD) is associated with decreased clearance of proteins involved in fibrinolysis, decreased production of coagulation factors (CF; vitamin K-dependent factors, fibrinogen, FV, FXIII), natural anticoagulants (proteins C and S, antithrombin), dysfibrinogenemia, low platelet count/impaired platelet function, and increased FVIII/von Willebrand factor (vWF). 9 This increased vWF levels can partially compensate the thrombocytopenia which frequently occurs in patients with cirrhosis; thus, primary hemostasis is preserved. 9 Patients with cirrhosis also have an increased risk of thrombosis. 9 In the context of variceal hemorrhage, over-resuscitation may aggravate bleeding by markedly increasing BP. The use of plasma, which may be associated with fluid overload and portal venous pressure increase, should be avoided. 7,9 Based on the latest data, hepatic coagulopathy should be managed with coagulation factor concentrates (CFC) instead of plasma, as they are more effective and have fewer side effects in volume overload and/or infections. 9
Renal Insufficiency
In patients with impaired kidney function, GIB is associated with high mortality risk. The increased levels of uremia and other toxins promote platelet dysfunction and reduce their ability to aggregate. Thrombocytopenia is usually mild.
In chronic kidney disease, anemia correction should target the hematocrit to 30% (grade 1C) 10 and low levels of fibrinogen should be corrected with fibrinogen concentrate (FC). Desmopressin (DDAVP; 0.3 μg/kg) induces the release of vWF to plasma, reducing bleeding time in 1 hour after infusion. The administration of tranexamic acid (TXA) is not indicated in cases of renal failure. 7
Antithrombotic Therapy
Antithrombotic drugs, namely OAC, heparins, and APT are widely used in the treatment and prophylaxis of thrombotic events. However, these therapies have risks, and bleeding is their main complication.
Patient approach depends on the type of antithrombotic therapy, GIB severity, and risk of thromboembolic events (algorithm 2). 11,12
The prescribed APT usually belongs to one of 2 groups: cyclo-oxygenase 1 inhibitors or adenosine diphosphate receptor inhibitors (P2Y12). For APT reversal, 11 suspend drug (except acetylsalicylic acid) and contact the Transfusion Medicine Department (TMD) to discuss platelet concentrate (PC) transfusion in cases of severe bleeding. 13
The anticoagulant effect of vitamin K antagonists (VKAs) is assessed by the international normalized ratio (INR). It is suggested that endoscopic therapy should not be delayed in patients with severe bleeding if INR ≤ 2.5. 14 For VKA reversal, 12,15 suspend drug (warfarin, acenocoumarol, phenindione), administrate vitamin K and prothrombin complex concentrate (PCC): 15 to 30 IU/kg, 15 if necessary (discuss with TMD).
Direct OACs (DOACs) dabigatran, apixaban, edoxaban, and rivaroxaban do not require routine monitoring. In most cases, qualitative coagulation tests are sufficient to demonstrate whether relevant anticoagulant activity is present or not.
For the direct thrombin inhibitor dabigatran, prothrombin time (PT) and INR are poorly sensitive. If activated partial thromboplastin time (aPTT) is within reference values, no relevant pharmacological activity is usually present. Thrombin time is very sensitive and, if normal, ensures no anticoagulant activity.
Regarding the remaining DOAC—the direct FXa inhibitors apixaban, edoxaban, and rivaroxaban—aPTT cannot be used due to poor sensitivity, PT has low sensitivity, and INR is inadequate for the evaluation of anti-Xa activity.
Reversal of anticoagulation with DOAC is facilitated by the short half-life of these drugs. In most situations, suspending the DOAC will stop its activity within less than 48 hours. For rapid reversal, the use of orally activated charcoal is recommended (if last dose has been taken less than 2 hours ago) 11,12 and it is suggested to promote DOAC excretion (increased diuresis, hemodialysis for dabigatran), 16 and use anticoagulation reversal agents 11,12 : (A) nonspecific—PCC 25 to 50 IU/kg; (B) specific—idarucizumab (Praxbind; “dabigatran antidote”) 2.5 g × 2 (total: 5 g) intravenous, bolus, or infusion within 10 minutes.
Unfractionated and low-molecular-weight heparins also present bleeding risk. In cases of major GIB requiring rapid reversal, 12 diluted intravenous protamine sulfate should be administered, not exceeding 5 mg/min until a maximum dose of 50 mg.
Cardiovascular Pathology/Coronary Stent
A significant number of patients require double APT (DAPT) consisting of the combination of acetylsalicylic acid and a P2Y12 platelet receptor inhibitor. Patients with myocardial infarction or acute coronary syndrome (ACS) and those undergoing percutaneous coronary intervention for balloon angioplasty and stenting have indication for APT (algorithm 2).
In patients with coronary stent, the decision to discontinue or maintain APT will depend on the likelihood of ischemia versus risk of prolonged/recurrent bleeding. 3,17 In patients with ACS experiencing major bleeding under DAPT, monotherapy with acetylsalicylic acid should be maintained. 3,17
Data regarding therapy restart are scarce. The consensus between the Societies of Cardiology and Gastroenterology recommends resuming therapy as soon as hemostasis is achieved. 3,11,14,17
Gastrointestinal Bleeding
Gastrointestinal bleeding is responsible for 350 hospitalizations/100 000 individuals annually in the United States. 18 It can be divided into 3 types: UGIB, middle GIB (MGIB), and lower GIB (LGIB). Upper GIB represents approximately 50% of all admissions by gastrointestinal (GI) hemorrhage, followed by LGIB with 40% and lastly MGIB with 10% of the episodes. A multidisciplinary approach is necessary for the optimal management of these patients. Pre-, peri-, and postprocedures are of utmost importance, as management of comorbidities ad initium can determine clinical outcomes.
Upper GIB
Acute UGIB is a frequent medical emergency. Despite advances in diagnosis and therapeutics, mortality following acute UGIB has remained relevant over the last 2 decades. Peptic ulcer bleeding-related 30-day mortality ranges from 5% to 12% and can be as high as 26% if occurring in patients previously hospitalized for other reasons. 19,20 There are no reliable statistics for the Portuguese population. 21
Portal hypertension (PHT)-related UGIB has a specific prognosis and management. Liver cirrhosis is the most frequent cause of PHT. Variceal bleeding is a dreaded decompensation of cirrhosis and mortality differs whether it is an isolated complication (20% 5-year mortality) or in association with other complications (over 80% 5-year mortality). Therefore, the cirrhosis stage, liver function scores (Child-Pugh score; model for end-stage LD [MELD] score), and presence of other (concomitant or previous) cirrhosis complications should be considered since higher scores are associated with worse prognosis. The immediate therapeutic goal in these patients is bleeding control, in order to prevent early recurrence (within 5 days) and reduce mortality. 22
The authors propose 2 distinct algorithms for the management of nonvariceal and variceal UGIB in pre-, peri-, and post-upper GI (UGI) endoscopy to identify the location and type of lesion, assess risk factors for rebleeding, and perform hemostatic endoscopic procedures (algorithms 3.1.1. and 3.1.2.).
Upper GIB associated with PHT (Algorithm 3.1.1.)
Variceal bleeding is a medical emergency commonly requiring intensive care. Data show that the use of vasoactive agents is associated with lower 7-day all-cause mortality 23 and lower transfusion requirements. Therefore, intravenous vasoactive drugs (terlipressin, octreotide, or somatostatin) should be started before diagnostic endoscopy as soon as possible, together with proton pump inhibitors (PPIs). Concomitant infections (urine, ascites, respiratory tract) should be excluded.
Patients with cirrhosis presenting with (variceal or nonvariceal) GIB are at high risk of bacterial infections. Short-term (maximum 7 days) antibiotic prophylaxis reduces this risk, recurrent bleeding, and death. Infection and death rates are lower in Child-Pugh Class A patients. Intravenous ceftriaxone is more effective than oral norfloxacin, mainly due to a high rate of quinolone-resistant organisms. The specific antibiotic recommended should be based on individual patient-risk characteristics and local antimicrobial susceptibility, with ceftriaxone (1 g/24 hours) being the first choice in patients with advanced cirrhosis, on quinolone prophylaxis, or living in areas with a high prevalence of quinolone resistance. 23
In these patients, UGI endoscopy should be performed in the first 12 hours after admission. For variceal bleeding, the most common endoscopic treatment is rubber band ligation. If massive bleeding is detected and endoscopic treatment cannot be implemented or is ineffective, a Sengstaken-Blakemore tube or a self-expandable covered stent (ELLA stent) has the potential to be lifesaving and gain time to try another endoscopic treatment or transjugular intrahepatic portosystemic shunt.
The use of lactulose or lactitol as soon as oral or enteric route is available following bleeding control prevents another complication: encephalopathy. Once acute bleeding is controlled, secondary prophylaxis of variceal bleeding must be implemented, usually with nonselective β-blockers and programmed band ligation.
Nonvariceal UGIB (Algorithm 3.1.2.)
Validated risk scores must be considered in the assessment of these patients. The Rockall score for mortality and the Glasgow Blatchford score (GBS) for the need for intervention and pre-endoscopy risk stratification are the most relevant. 3
Patients with GBS score 0 to 1 can be treated as outpatients if diagnosis and treatment strategies can be implemented in a timely manner. Patients should be informed about risk signs and maintain contact with the discharging hospital. Discharge from the emergency department without inpatient endoscopy should be considered for patients with systolic BP ≥110 mm Hg, pulse <100 beats/min, Hb ≥13.0 g/dL for men or ≥12.0 g/dL for women, blood urea nitrogen (BUN) <18.2 mg/dL, and absence of melena, syncope, LD, and cardiac failure.
For the remaining patients, intravenous PPI should be started as soon as possible by administering an 80-mg bolus followed by continuous infusion (8 mg/h), as long as this measure does not delay the endoscopy. Nasogastric or orogastric aspiration/lavage is not mandatory, but intravenous erythromycin (3 mg/kg) 30 to 120 minutes prior to endoscopy improves visualization, particularly in cases with signs of active bleeding.
Following hemodynamic resuscitation, UGI endoscopy should be performed early in inpatient setting (≤24 hours). Very early (<12 hours) UGI endoscopy can be deleterious and should be performed in patients with high-risk clinical features, namely hemodynamic instability (tachycardia, hypotension) that persists despite ongoing volume resuscitation attempts, inhospital bloody emesis/nasogastric aspirate, or contraindication to anticoagulant suspension. 24
During endoscopy, the local and type of lesion are investigated as well as its rebleeding risk, and endoscopic hemostasis can be performed. For peptic ulcer—the most frequent type of lesion—, hemostasis is performed in ulcers with spurting or oozing bleeding (Forrest classification Ia and Ib, respectively) or a nonbleeding visible vessel (Forrest classification IIa), as these lesions have a high risk of rebleeding. 3 For those with an adherent clot (Forrest classification IIb), careful endoscopic clot removal can be achieved; if an underlying active bleeding or nonbleeding vessel is visible, it should be submitted to endoscopic hemostasis. Those with a flat pigmented spot (Forrest classification IIc) or clean base (Forrest classification III) do not require local hemostasis.
High-dose PPI therapy should be warranted for patients undergoing endoscopic hemostasis and for Forrest IIc ulcers. The PPI infusion must be maintained for 72 hours.
Guidelines do not recommend routine second-look endoscopy. However, if there is clinical evidence of rebleeding following successful initial endoscopic hemostasis, repeating upper endoscopy and hemostasis are indicated. In cases of failure of the second hemostasis attempt, transcatheter angiographic embolization or surgery should be considered.
In patients with peptic ulcer bleeding, Helicobacter pylori infection must be excluded. Infected patients should be given specific treatment and its efficacy should be confirmed.
Lower GIB
The annual incidence of LGIB is 33 to 87 cases/100 000 individuals and most patients are older than 70 years. 25 Bleeding spontaneously stops in 80% to 85% of cases and mortality rate ranges between 2% and 4%, being higher in older patients and in those with comorbidities. 26
Considering etiology, diverticulosis is the most common cause of acute LGIB. Other potential causes include colonic polyps and cancer, colitis (ischemic, inflammatory bowel diseases, infectious, noninfectious as radiation), angiectasia, postpolypectomy, rectal ulcer, hemorrhoids, anal fissure, and others, including Dieulafoy lesions and colonic or rectal varices. 2,26
Initial patient assessment and hemodynamic resuscitation should be simultaneously performed (Algorithm 3.2). 26 Focused clinical history, physical examination, and laboratory assessment should be retrieved, and bleeding severity, possible location, and etiology assessed. 26
Risk factors for poor outcomes should be identified. In clinical practice, a simple score such as BLEED (Algorithms 3.2 and 3.2.1.) can be used, comprising 5 items: ongoing fresh bleeding, low systolic BP (<100 mm Hg), elevated PT (>1.2× the control), erratic/altered mental status, and unstable comorbid illness (any organ system abnormality that would ordinarily require intensive care unit [ICU] admission). 27 Patients with any of these criteria are classified as high risk and should be preferentially managed in an ICU.
Identifying the bleeding source after successful resuscitation is a clinical priority and not so straightforward in LGIB as in UGIB setting. 2 Colonoscopy should be the procedure of choice for almost all patients. 26 It should be performed after adequate colon cleansing with 4 to 6 liters of a polyethylene glycol-based solution or equivalent.
However, the timing of colonoscopy depends on patient’s hemodynamic status. The first approach is to calculate the SI. 2 A positive SI (≥1) is a marker of active bleeding and the patient is classified as unstable. 2 In cases of a strong suspicion of UGIB (presence of hematochezia and orthostasis), a UGI endoscopy must be performed. When UGI endoscopy is not feasible or deemed unnecessary, there is indication for CTA. 2 After a positive CTA, specific treatment should be implemented as soon as possible, either by intervention radiology (angiography with embolization), an endoscopic method (UGIB or LGIB), or even surgical intervention. 2 If CTA does not reveal the bleeding source, the patient should be admitted for lower GI endoscopy.
Patients with negative SI (<1) are classified as stable. In such cases, risk is assessed using the Oakland score. A score ≤8 indicates minor bleeding and the patient can be discharged for outpatient management with 95% confidence. 2 However, these patients must be submitted to an elective colonoscopy ideally in the first 2 weeks after discharge. 2
If Oakland score is >8, the patient should be admitted for observation and further studies. 2 Once again, if there is clinical suspicion of UGIB and/or BUN (mg/dL)/creatinine (mg/dL) is >30, a UGI endoscopy should be considered. If the patient has bright rectal bleeding, anorectal inspection by proctoscopy or flexible sigmoidoscopy should be considered, since benign anorectal conditions can account for more than 15% of cases. 28 For the remaining cases, colonoscopy is mandatory after adequate bowel cleansing. In patients with high-risk clinical features and/or signs/symptoms of ongoing bleeding, a colonoscopy should be performed within the first 24 hours. 26,29 In cases without high-risk clinical features or serious comorbidities and in those with high-risk clinical features without signs or symptoms of ongoing bleeding, a colonoscopy should be performed on the next available list. 26
Although most LGIB cases spontaneously stop, 3 main treatment options are available for these patients: endoscopy, transcatheter mesenteric embolization, or surgery. 2 Surgery is rarely required for LGIB and should be reserved for the minority of patients with persistent or refractory bleeding despite endoscopic and radiologic intervention. 2,26,29
Patients with cirrhosis represent a specific group. Data on LGIB in these patients are very limited, but this form of GI hemorrhage may be associated with life-threatening complications similar to UGIB. Endoscopic treatment of colonic or rectal varices can involve direct sclerosis, band ligation, or cyanoacrylate injection (Algorithms 3.2.1. and 3.2.2.).
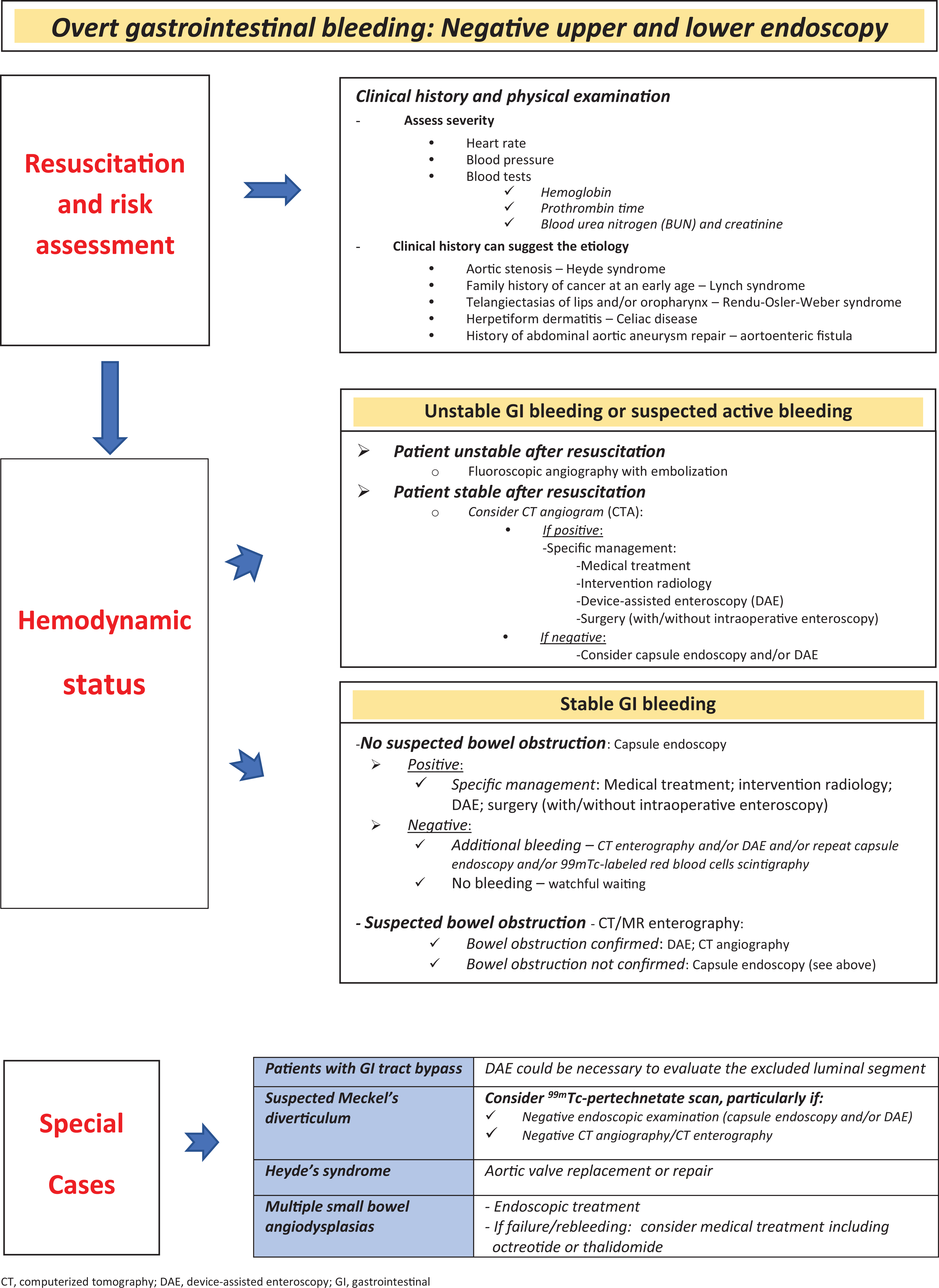
Middle GIB
Middle GIB refers to hemorrhage from the papilla of Vater to the ileocecal valve (small bowel bleeding). MGIB can have multiple etiologies: vascular lesions (angiodysplasia, Dieulafoy’s lesion), inflammatory bowel disease, Meckel’s diverticulum, small bowel neoplasms; NSAID-related erosions or ulcers; and aortoenteric fistula, radiation enteropathy, or small bowel varices. 30
For patients with suspected MGIB, a second-look upper endoscopy and/or colonoscopy should be considered (Algorithm 3.3). 30,31 After negative upper and lower endoscopy, a small bowel study must be conducted. As before, in these cases the hemodynamic status also determines the technique of choice. 32 More recent evidence suggests that CTA is a potential first-line tool for evaluation of all acute GIB episodes. 30,32,33 The main candidates for CTA are unstable patients who respond to resuscitation and stable patients with risk factors for active bleeding. 33 However, if the patient has a rapidly active hemorrhage and does not appropriately respond to resuscitation, direct fluoroscopic angiography is the best method, since it has the potential for immediate diagnosis and treatment.
For stable patients, capsule endoscopy (CE) is the best tool to examine the small bowel and should be performed as soon as possible, optimally within 14 days after the bleeding episode. 30 –32 It is interesting to notice that CE can be useful even in patients with acute severe bleeding. 34 A “watch-and-wait” approach with periodic clinical re-evaluation is generally acceptable after negative CE if the patient has no further bleeding and/or a significant drop in Hb levels. 31 Significant small bowel lesions, more frequently in the proximal segments (duodenum), can be missed during CE, and cross-section imaging or device-assisted enteroscopy should be offered to patients with persistent bleeding.
Scintigraphy with red blood cells (RBCs) tagged with 99mTc enables the detection of bleeding at rates as low as 0.1 to 0.5 mL/min. However, this procedure is not easily available, has a several-hour delay until final results are obtained, and the anatomical bleeding location is poor. 33
Additionally, in younger patients with ongoing bleeding and negative evaluation on CE, computerized tomography enterography, or other testing modalities, Meckel’s diverticulum diagnosis should be considered.
If a bleeding source is identified in any given examination, specific treatment should be implemented whenever feasible. Therapeutic alternatives include endoscopy, radiology, and surgery. Surgery is usually the last alternative except in specific cases, such as bleeding neoplasms. It must be guided by previous examinations, and intraoperative enteroscopy can be required during surgical intervention.
Coagulopathy Management in GIB
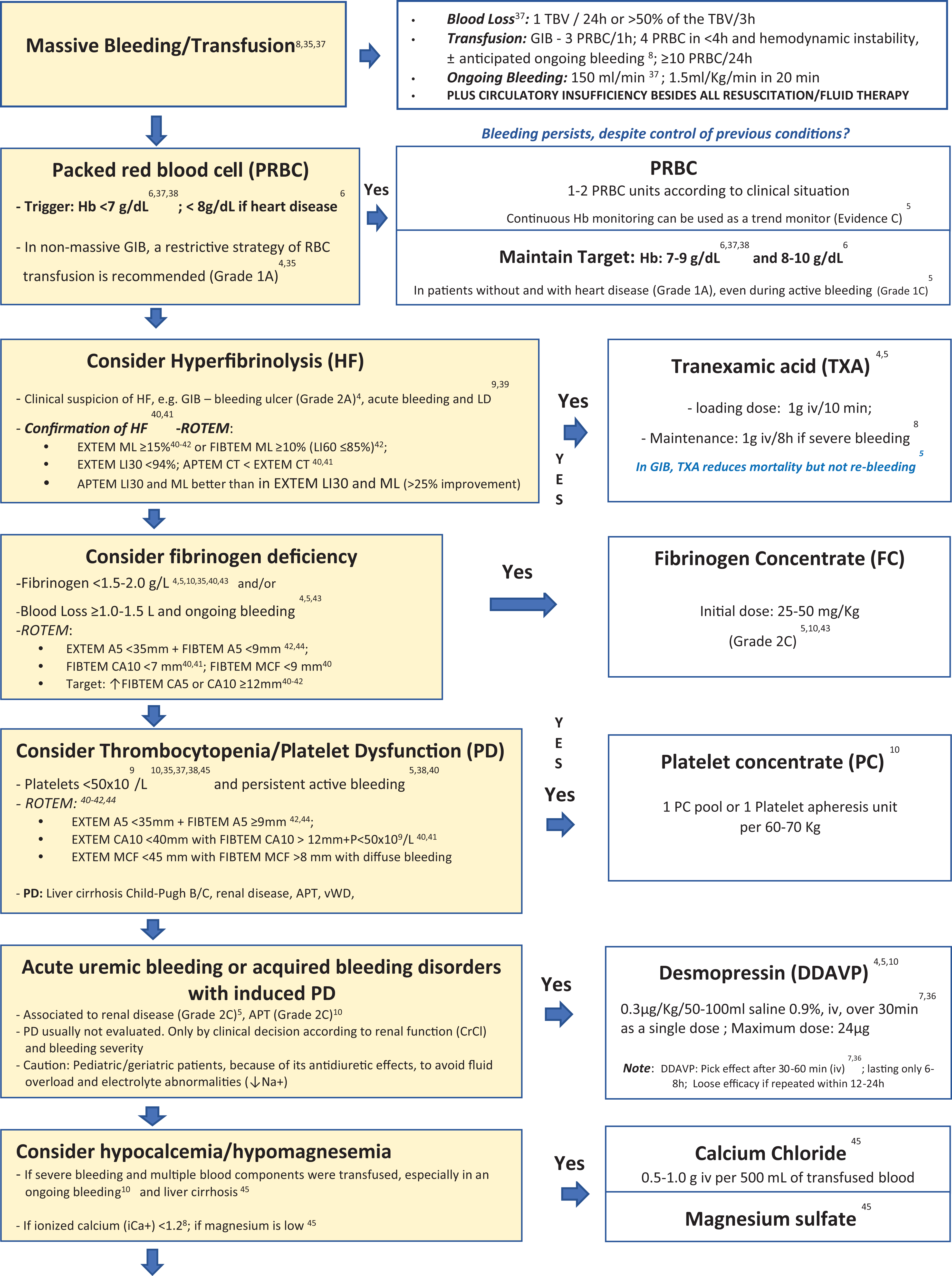
Restrictive transfusional therapy is recommended to avoid an increase in blood volume, especially in cases of pre-existing PHT 7 in patients with acute UGIB, unless there is massive life-threatening hemorrhage. 35 It is also important to follow a multimodal, preferably VET-guided goal-directed approach on massive bleeding management, ensuring hemostasis (Algorithm 4). 9,36
Massive bleeding/transfusion is defined according to the volume of blood loss, ongoing bleeding rhythm, and number of transfused blood units. 35,37 A pragmatic clinical-based definition is provided by the combination of bleeding with SI ≥1.0. 37
Several randomized controlled trials (RCTs) and observational studies support a restrictive RBC transfusion strategy with a general Hb threshold of 7 g/dL and 8 g/dL for RBC transfusion and a target Hb level of 7 to 9 g/dL and 8 to 10 g/dL after transfusion for patients without and with cardiac diseases (ischemic heart disease, including ACS, and after cardiac surgery), 6,37,38 respectively.
In UGIB, TXA reduces mortality with no identified risks, although its routine use is generally not recommended for UGIB or LGIB 39 due to important uncertainties about its safety and effectiveness in this indication. However, some experts state that TXA should be considered as adjuvant therapy in cases in which the standard of care for GIB fails. 39 The use of TXA is considered in nontraumatic major bleeding 35 and in presence of bleeding prompting transfusion. 39 Currently, TXA may be considered on a case-by-case basis, particularly if there is evidence of hyperfibrinolysis. 40 –42 In GIB, TXA reduces mortality but not rebleeding (evidence B). 5 The Sevilla Document 4 suggests TXA administration in patients with bleeding peptic ulcer (grade 2A), but its use in bleeding CLD patients remains unknown. However, when fibrinolysis in combination with excessive bleeding occurs in patients with LD, TXA (30 mg/kg) should be administered under VET control. 9 Ongoing RCT (HALT-IT-Tranexamic acid for the treatment of acute GIB: study protocol for a randomised controlled trial and EXARHOSE-Efficacy and tolerance of early administration of tranexamic acid in patients with cirrhosis presenting with acute upper GIB: a study protocol for a multicentre, ramdomised, double-blind, placebo controlled trials) are investigating the effective role of TXA in GIB.
Fibrinogen concentrate: Fibrinogen is a critical CF for clot formation. Fibrinogen promotes platelet aggregation via binding of glycoprotein IIb/IIIa receptors on platelet surfaces. 43 In GIB, when the patient has already lost 1 to 1.5 liters of blood and ongoing bleed is present and/or fibrinogen values are <1.5 to 2.0 g/L, fibrinogen replacement with FC should be considered 4,5,43 at doses of 25 to 50 mg/kg, depending on VET or Clauss results and on clinical situation. 42 –44
Platelet concentrate: Thrombocytopenia is common in LD and indirectly correlates with the degree of PHT and hepatic decompensation. Current guidelines and expert groups, including the present one, recommend considering the use of PC in patients with UGIB 38 and LD 7 with active bleeding plus platelet 5,35,37,38,45 count <50 × 109/L and VET analysis (if available). 40 –42,44 Non- or mildly thrombocytopenic patients with life-threatening bleeding who have been under APT represent specific cases regarding PC transfusion and should be the object of an individualized approach based on general clinical condition. 13
Desmopressin improves dysfunctional platelet activity by increasing vWF-FVIII levels from endothelial stores and decreases bleeding time within 1 hour after infusion. 7,36 In acquired bleeding disorders, DDAVP has been used mainly in patients with active bleeding and uremia plus impaired kidney function and also in those with APT, 5,10 as a single intravenous dose of 0.3 to 0.4 µg/kg. 7,36 No bleeding benefits from variceal hemorrhage or liver transplant were observed. 7
Calcium chloride and magnesium sulfate: Hypocalcemia and hypomagnesemia are often associated with massively transfused patients. Its evaluation and correction are imperative, 8,10,45 accompanied by close monitoring during its infusion. Administration of plasma and synthetic colloids promotes early hypocalcemia. As citrate in blood components is primarily metabolized by the liver, in patients with cirrhosis receiving massive transfusion, this effect may be more severe. 45
Prothrombin complex concentrate (PCC) contains nonactivated 4 CF (II, VII, IX, and X) and anticoagulants (proteins C and S). 37 Half-lives of each factor largely differ, with FII displaying the longest (60 to 72 hours) and FVII the shortest (6 hours). Fresh frozen plasma (FFP) contains several blood proteins, in particular all CF and natural anticoagulants. Prothrombin complex concentrate advantages over FFP mainly include smaller volume of higher 4 CF concentration, 37,46 with a lower risk of fluid overload 46 and faster and better correction of coagulation parameters. 46 The risk of thromboembolic complications with PCC is mainly attributed to the patient’s underlying thrombotic risk factors. The 4F-PCC is effective as adjuvant treatment, with an acceptable safety profile not only for emergent reversal of OACs (VKA, DOAC) but also for coagulopathy associated with acute major/life-threatening bleeding. 4 –6,37,46 In presence of active bleeding, prothrombin reaches a critical level (with reduced thrombin generation) when blood loss exceeds 150% to 200% (≥1.5 TBV), evidenced by VET and when PT or aPTT is >1.5 times the normal. 5,43 In cases of high bleeding tendency and prolonged clotting time despite adequate fibrinogen substitution, PCC administration (20-30 IU/kg) is suggested by the 2017 European guidelines (Grade 2C) 5 and others 10,36,42 –44 and/or FFP (12-15 mL/kg). 35,43 However, FFP single dose has been shown to have limited efficacy. 38,43 Additionally, patients with LD may not tolerate such FFP volume, 46 due to the risk of circulatory overload and PHT. 7,9 Consequently, the administration of FFP should be avoided in these patients. 9 An ex vivo study showed that PCC was more effective than FFP in increasing thrombin generation. 46 Also due to its safety profile, PCC has been used in patients with severe LD or hepatic failure, 46 –48 with emergent severe bleeding, and in preparation for urgent/elective surgery with bleeding 46 risk (20-30 IU/kg dose) 7 when immediate hemostasis is necessary. 7 Due to the frail hemostatic balance, it should be anticipated that these patients will develop a clinically significant dilutional coagulopathy and hemostatic failure with blood loss less than 1 blood volume (<1 TBV). 46 Prothrombin complex concentrate can be less effective in controlling hemorrhage in patients with Child-Pugh C cirrhosis and acute liver failure, 47 due to the deficit in CF as FV (<25%) 48,49 and fibrinogen, when they are not supplemented before PCC administration. 47 Prothrombin complex concentrate may be an option for selected patients with LD and excessive life-threatening GIB after carefully weighing risk versus benefit and after fibrinogen and FV supplementation 47 if needed, with FC and FFP (up to 12 mL/kg), respectively. 35,47 Based on the latest data, hepatic coagulopathy should be managed with CFC rather than FFP, as they are more effective and have fewer side effects. 9 Administration of PCC should be guided by VET to avoid overtreatment and thrombosis. 9
FXIII concentrate: FXIIIa is important for clot stabilization through cross-linking of fibrin monomers and increased clot resistance to fibrinolysis. 33,49 Fibrinogen, platelets, and FXIII are critical for clot stabilization. In acute hepatic failure and CLD, a decrease in FXIII synthesis is observed. 9 A significant decrease in fibrinogen, fibrin clot strength, and α angle is detectable when FXIII decreases below 60%. 50 Some massive bleeding algorithms recommend FXIII administration in cases of ongoing severe bleeding 36 and clot instability not related to hyperfibrinolysis. 40 In both cases, the administration should be guided by VET evaluation. 36,40 There are insufficient data regarding the amount of blood loss required to induce critical FXIII decrease (<60%). Special attention is necessary in cases of hemodilution, especially if colloids are used, because FXIII <60% is reached with only moderate blood loss (<1 TBV). Factor XIII supplementation is recommended by several updated guidelines 5,49 and also included in coagulation support algorithms for bleeding patients with functional FXIII deficiency, alongside with fibrinogen and PCC administration under VET analysis. 36,40,43 Recent guidelines state that “in cases of ongoing or diffuse bleeding and low clot strength despite adequate fibrinogen concentrate, it is likely that FXIII activity is critically reduced” and “in cases of significant FXIII deficiency (eg.<30 to <60% activity), FXIII concentrate (30IU/Kg) can be administered (Grade 2C). 5,49 ” If poor improvement is observed in α angle and fibrinogen/fibrin polymerization after fibrinogen administration, critical FXIII reduction should be considered together with correction with substituting FXIII. 50 In these cases, the authors suggest replacing FXIII with FFP (12-20 mL/kg) 43 if FXIII concentrate (30 IU/kg or 1.250 IU) 5,40,43,49 is not available.

Initial assessment and resuscitation.

Potentially aggravating factors in gastrointestinal bleeding.

Variceal upper gastrointestinal bleeding. 22

Non-variceal upper gastrointestinal bleeding. 3

Lower gastrointestinal bleeding. 2

Activated recombinant FVII (rFVIIa) has been used off-label in an array of life-threatening conditions, such as massive bleeding associated with trauma, surgery, and CLD. The 2017 European guidelines 5 and the Seville Document 4 suggest the off-label use of rFVIIa for refractory life-threatening bleeding and/or when comprehensive coagulation therapy fails (Grade 2C), 5 including intracranial, postpartum, and perioperative cardiac or hepatic bleeding (Grade 2C). 4 The authors suggest considering the administration of rFVIIa (90-120 μg/kg) as a rescue agent in cases of uncontrolled persistent or “life-threatening bleeding,” after standard treatment and correction of acidosis (Grade 1C), 5 hypocalcemia, hypothermia, fibrinogen levels, thrombocytopenia, and hyperfibrinolysis for clot formation improvement.
Coagulopathy management in gastrointestinal bleeding.
Footnotes
Acknowledgments
The authors gratefully acknowledge Rosa Leal and Joana Rodrigues from CSL Behring.
The authors further acknowledge the medical writing assistance provided by Joana Cavaco-Silva (
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Luciana Gonçalves received honorary from Boehringer Ingelheim, Daichii Sankio, Leo, Bayer, Pfizer, Bristol Myers Squibb, and CSL Behring. Manuela Gomes received honorary from Octapharma and CSL Behring. Nuno Almeida received honorary from Norgine and Allergan. Rosário Orfão received honorary from CSL Behring, Octapharma and 3M.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: CSL Behring provided financial support for this study but had no role in study design or development, entirely respecting the authors’ scientific independency.
