Abstract
Low-molecular-weight heparin (LMWH) is often recommended as a bridging therapy during temporary interruptions in warfarin treatment, despite lack of evidence. The aim of this study was to see whether we could find benefit from LMWH bridging. We studied all planned interruptions of warfarin within the Swedish anticoagulation register Auricula during 2006 to 2011. Low-molecular-weight heparin bridging was compared to nonbridging (control) after propensity score matching. Complications were identified in national clinical registers for 30 days following warfarin cessation, and defined as all-cause mortality, bleeding (intracranial, gastrointestinal, or other), or thrombosis (ischemic stroke or systemic embolism, venous thromboembolism, or myocardial infarction) that was fatal or required hospital care. Of the 14 556 identified warfarin interruptions, 12 659 with a known medical background had a mean age of 69 years, 61% were males, mean CHADS2 (1 point for each of congestive heart failure, hypertension, age ≥75 years, diabetes, and 2 points for stroke or transient ischemic attack) score was 1.7, and CHA2DS2-VASc score was 3.4. The total number of LMWH bridgings was 7021. Major indications for anticoagulation were mechanical heart valve prostheses 4331, atrial fibrillation 1097, and venous thromboembolism 1331. Bridging patients had a higher rate of thrombotic events overall. Total risk of any complication did not differ significantly between bridging (1.5%) and nonbridging (1.2%). Regardless of indication for warfarin treatment, we found no benefit from bridging. The type of procedure prompting bridging was not known, and the likely reason for the observed higher risk of thrombosis with LMWH bridging is that low-risk procedures more often meant no bridging. Results from randomized trials are needed, especially for patients with mechanical heart valves.
Introduction
When oral anticoagulation with warfarin needs to be temporarily interrupted, low-molecular-weight heparin (LMWH) is commonly administered to prevent thromboembolic events. 1 Well-made studies of bridging showing positive benefit/risk ratio are scarce, with many studies comparing LMWH to unfractionated heparin instead of placebo or no treatment. 2 Guidelines are mainly based on meta-analyses. 3 Studies on recurrence of venous thromboembolism 4 and arterial thromboembolism from atrial fibrillation 5 were not in favor of LMWH bridging. Bridging is recommended in guidelines for patients with atrial fibrillation at high risk 6,7 and with mechanical heart valves, although evidence is scarce. 8,9
We have tested the hypotheses that LMWH bridging during temporary interruptions of warfarin therapy lacks positive effect on the risk of thromboembolism or death within 30 days of stopping warfarin and is associated with an increased risk of serious hemorrhage.
Methods
Design
For the study period from January 1, 2006, to December 31, 2011, all temporary interruptions of warfarin within the anticoagulation register and dosing system Auricula were extracted.
Every resident in Sweden has a unique civil registration number, making it possible to merge data on individuals from nationwide registers. We requested all diagnoses up until 30 days after the studied period within the National Patient Register, and date and cause of death from the Cause of Death Register, if any. We defined diagnoses constituting medical background and outcomes (given in the Online Supplementary Appendix). Prescriptions of drugs that could affect thrombocyte aggregation were extracted from the prescription register. Data were merged into an anonymized database, with every planned warfarin interruption given an identification number. An individual could have any number of interruptions.
Procedures
We categorized LMWH treatments (dalteparin, enoxaparin, or tinzaparin). Doses of LMWH are registered as free text in Auricula. Twenty-two doses were unclear and had to be registered as missing. Low dose was defined as a recommended dose for prophylaxis of venous thromboembolism: dalteparin 2500 to 5000 IU, enoxaparin 20 to 40 mg, or tinzaparin 3500 to 4500 IU, subcutaneously once daily. High dose was weight-adjusted full dose; intermediate dose was between low and high.
The propensity score, the probability of receiving bridging treatment, was estimated from a logistic regression model with risk factors: Factors associated with increased risk of cerebral or peripheral embolism: mechanical heart valve prosthesis, mitral valve prosthesis, and components of the risk scores CHADS2 and CHA2DS2-VaSc. Factor associated with increased risk of bleeding: previous bleeding defined in the Online Supplementary Appendix and factor associated with increased risk of complications: chronic obstructive pulmonary disease. Factors influencing the choice between LMWH bridging and no bridging (kidney failure), and drugs that inhibit thrombocyte function: acetylsalicylic acid/aspirin, other thrombocyte aggregation inhibitors, nonsteroidal anti-inflammatory drugs (NSAIDs), and selective serotonin reuptake inhibitors (SSRI).
10
Indication for anticoagulation: atrial fibrillation (AF), high-risk AF (AF and CHADS2 ≥ 4), venous thromboembolism, mechanical heart valve, and mechanical mitral valve.
By comparing propensity scores between cases and controls within strata, clusters of comparable individuals were created by a “full optimal match”, that is, every individual was assigned to (exactly) 1 cluster which was determined by minimizing the total average discrepancy between the scores within each cluster. 11 Within each cluster, weights were assigned with regard to the treated population, that is, each treated had a weight of one, and the sum of the control weights within each cluster equaled the sum of the treated weights within the same cluster. Thus, the weighted controls were a population having equal size and distribution of covariates as the treated population. These weights and clusters were used in the Cox regression analyses to obtain treatment effects for the treated as well as robust estimates of the standard errors. 12
We defined indication groups to be studied closer: mechanical heart valve prosthesis (the presence of a mechanical heart valve regardless of other indications for warfarin), atrial fibrillation, and venous thromboembolism. Atrial fibrillation and venous thromboembolism excluded the presence of mechanical heart valve. Low-molecular-weight heparin dose groups were also analyzed separately.
Data Sources
Auricula is a Swedish nationwide register and dosing support system for oral anticoagulation. 13 It was started in 2006 and now includes over 125 000 patients and 6 million international normalized ratio values. More than 200 centers, primary health-care centers as well as specialist clinics use Auricula, corresponding to approximately half of Swedish patients on oral anticoagulation. The most common indication is atrial fibrillation (70%), followed by venous thromboembolism (20%) and heart valve indications, mostly mechanical valves (10%). From Auricula, we extracted data on warfarin treatment indications, start and stop dates of temporary warfarin interruptions, bridging therapy with type and dose of LMWH, if any, as well as sex and age.
The Swedish National Patient Register (NPR) has registered diagnoses of patients discharged from hospital since 1987 and diagnoses at specialist clinics since 2001. International Classification of Diseases, tenth edition (ICD-10, World Health Organization) has been used since 1997. Primary care diagnoses are not registered. The register has a high validity and lacks information on primary diagnosis at discharge in only 0.5% to 0.9% of cases. 14 Medical background was extracted from the NPR. Renal failure was defined from diagnoses and not graded.
The Swedish prescription register was searched for prescriptions of drugs that inhibit thrombocyte aggregation, such as aspirin, other thrombocyte aggregation inhibitors, NSAIDs, and SSRIs.
Sweden’s Cause of Death Register has records on date and causes of death for all deaths in Sweden.
Outcomes and Follow-Up
Complications that were fatal or required hospital care within 30 days after the first day of warfarin interruption were defined according to ICD-10 codes given in supplemental Table S1. Bleeding was intracranial, gastrointestinal, or other. Thrombotic complications were venous thromboembolism, ischemic stroke or systemic embolism, or myocardial infarction. Only the first complication of every specific subtype was registered for every interruption, to avoid overregistering. Follow-up was until 30 days after the start of the last temporary warfarin interruption, if and when another warfarin interruption started, or death. Any warfarin interruption could theoretically result in any or all of the complications defined above, that is, 0 to 3 bleedings and 0 to 3 thrombotic complications.
Statistical Analysis
We present patient characteristics as numbers with percentages, or means and standard deviations, as appropriate. Categorical data were analyzed using χ2 tests. Cox regression analysis was performed to obtain hazard ratios. Significance was defined as P < .05.
Data were analyzed using SPSS Statistics (Version 22; SPSS Inc, IBM Corporation, New York), and R version 3.0.0 (R Foundation for Statistical Computing, Vienna, Austria. URL http://www.R-project.org/). Confidence intervals are at 95%.
Results
Study Population
We identified 14 578 planned temporary interruptions of warfarin treatment during the 6 study years, with data on LMWH missing for 22. In 12 659 cases, there was information in NPR prior to the bridging maneuver, meaning we could extract a medical history. Table 1 presents the background characteristics of patients, for all warfarin interruptions as well as according to indication for warfarin. Supplemental Figures S1 and S2 describe the success of matching.
Background Characteristics of the Studied Population.
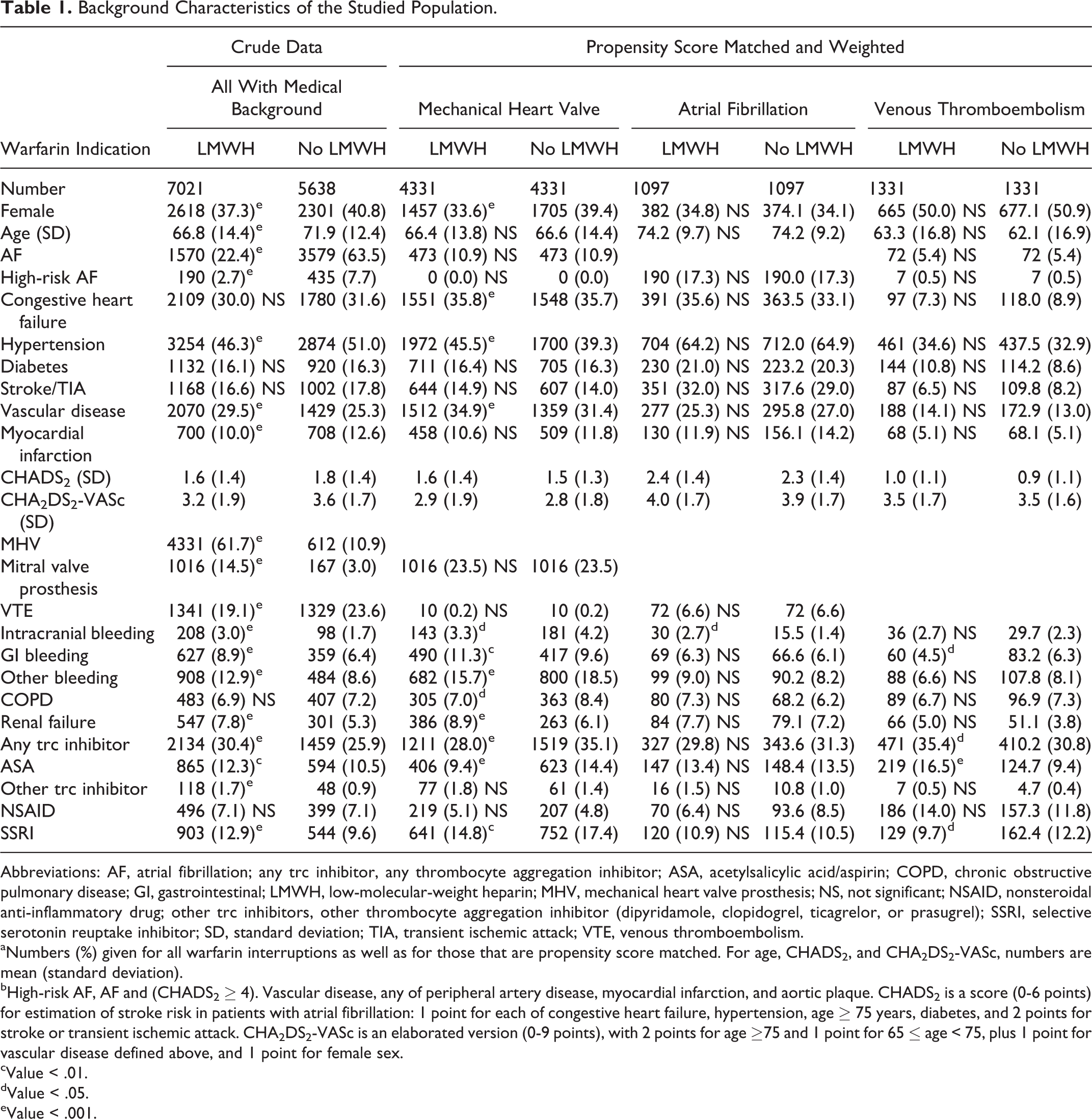
Abbreviations: AF, atrial fibrillation; any trc inhibitor, any thrombocyte aggregation inhibitor; ASA, acetylsalicylic acid/aspirin; COPD, chronic obstructive pulmonary disease; GI, gastrointestinal; LMWH, low-molecular-weight heparin; MHV, mechanical heart valve prosthesis; NS, not significant; NSAID, nonsteroidal anti-inflammatory drug; other trc inhibitors, other thrombocyte aggregation inhibitor (dipyridamole, clopidogrel, ticagrelor, or prasugrel); SSRI, selective serotonin reuptake inhibitor; SD, standard deviation; TIA, transient ischemic attack; VTE, venous thromboembolism.
aNumbers (%) given for all warfarin interruptions as well as for those that are propensity score matched. For age, CHADS2, and CHA2DS2-VASc, numbers are mean (standard deviation).
bHigh-risk AF, AF and (CHADS2 ≥ 4). Vascular disease, any of peripheral artery disease, myocardial infarction, and aortic plaque. CHADS2 is a score (0-6 points) for estimation of stroke risk in patients with atrial fibrillation: 1 point for each of congestive heart failure, hypertension, age ≥ 75 years, diabetes, and 2 points for stroke or transient ischemic attack. CHA2DS2-VASc is an elaborated version (0-9 points), with 2 points for age ≥75 and 1 point for 65 ≤ age < 75, plus 1 point for vascular disease defined above, and 1 point for female sex.
cValue < .01.
dValue < .05.
eValue < .001.
For all interruptions, regardless of the indication for anticoagulation, 64% of controls had atrial fibrillation, but only 22% of bridgings. On the other hand, mechanical heart valves were present in about 1 in 10 controls, but in over 6 in 10 bridgings. Bridging patients were more often male, younger, and had a lower prevalence of hypertension and myocardial infarction, but had higher prevalence of previous intracranial, gastrointestinal, and other bleedings. Duration of warfarin interruption was 5.4 (standard deviation 4.7) days for LMWH treated, and 7.5 (4.9) for non-LMWH treated.
Outcomes
We identified 134 complications in the NPR during the 30-day follow-up, from 12 659 warfarin interruptions (1.06%; Table 2). Death outside of hospital (without a diagnosis in the NPR) occurred in 51 cases. We included all deaths in our analyses. Kaplan-Meier curves of outcomes according to indication for warfarin are given in the online supplemental Figure S3.
Risks of Complications in the Respective Study Groups.a
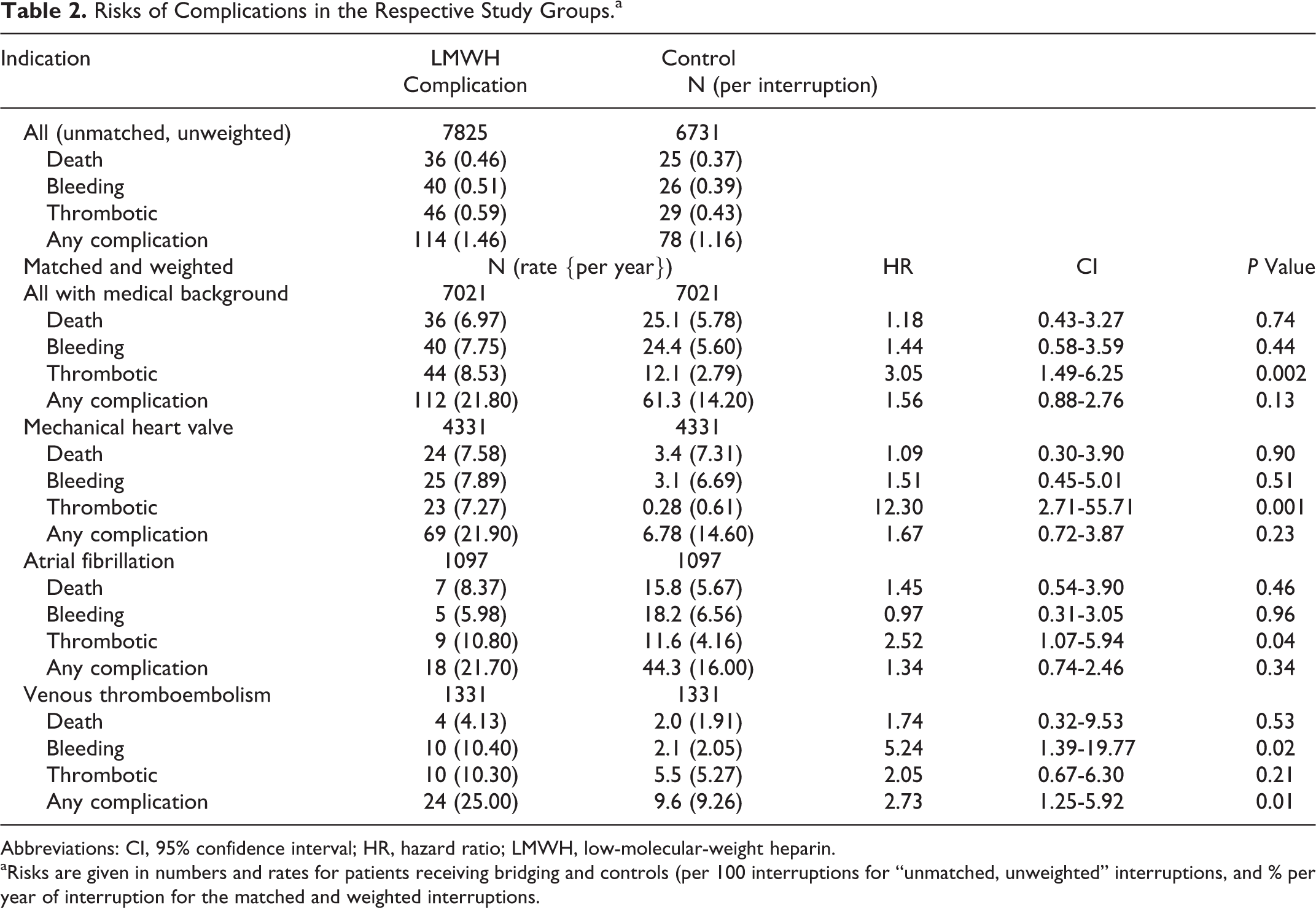
Abbreviations: CI, 95% confidence interval; HR, hazard ratio; LMWH, low-molecular-weight heparin.
aRisks are given in numbers and rates for patients receiving bridging and controls (per 100 interruptions for “unmatched, unweighted” interruptions, and % per year of interruption for the matched and weighted interruptions.
The use of thrombocyte aggregation inhibitors was associated with increased probability of thrombotic complications for the whole population and for patients with atrial fibrillation.
LMWH Dose and Risk of Complications
There was no significant relation between dose of LMWH and risk of complications (Table 3).
Risk of Complications According to Dose of LMWH Bridging.a
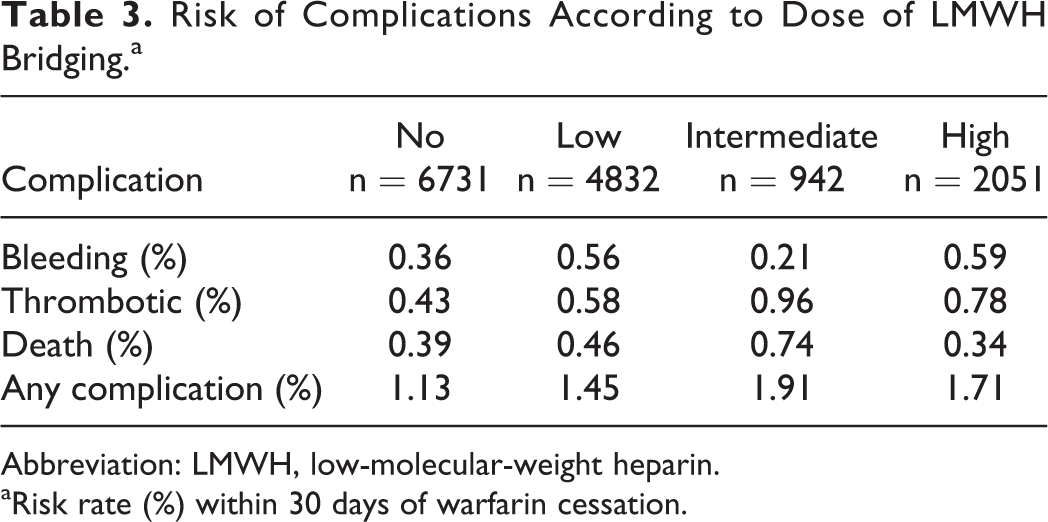
Abbreviation: LMWH, low-molecular-weight heparin.
aRisk rate (%) within 30 days of warfarin cessation.
Mechanical Heart Valve
For the 4331 propensity-matched bridgings in patients with mechanical valve, death, thrombotic complication, and any complication were statistically significant more common in the bridging group, whereas bleeding was not. We analyzed data on patients with mitral valve prosthesis (1091 with LMWH bridging, 178 without), but these results did not differ from those for mechanical heart valves in general (data not shown).
Atrial Fibrillation
There was a slightly but significantly higher rate of thrombotic complications in the bridging group (1097 bridgings vs 1097 matched and weighted controls). Patients with atrial fibrillation and CHADS2 ≥ 4 consistently had a higher risk of complications than other groups. This risk did not diminish with bridging (crude data, not shown).
Venous Thromboembolism
In the venous thromboembolism group (1331 bridgings vs 1331 matched and weighted controls), LMWH bridging was associated with increased risk of bleeding but with no difference in risk of thrombotic complications.
Discussion
In this large cohort of almost 15 000 bridging maneuvers, we found no benefit from LMWH bridging. About 1% of temporary interruptions of warfarin resulted in a complication requiring specialist care within 30 days. This might seem like a low risk, but means 30-day rates of bleedings and thrombotic events of 0.45% and 0.52%. Comparison of annual rates for warfarin-treated patients within Auricula during the same study period (2.24% and 2.65%) shows that warfarin interruption is associated with greatly increased risks. 15
The observed increased rate of complications is mainly due to thromboses in the group receiving LMWH, which would seem like a paradox and is contrary to some previous findings of increased risk of bleeding but without differences in risk of thrombotic complications. 4,5 Observational data from a US atrial fibrillation register showed that bridging was associated with increased risk of bleeding, but not thrombotic events. 16 However, another retrospective study of patients with atrial fibrillation reported higher rates of thrombotic events with LMWH bridging during interruption of warfarin, albeit with small numbers and possible confounding factors. Bleeding rates were shown to be higher with bridging for warfarin and dabigatran interruption. 17 Since it is unlikely that LMWH in itself would cause thrombosis, the likely reason would be differences between the patients receiving bridging and those who do not. We performed propensity score matching to reduce differences in risks between patient groups, leaving the probable reason for the higher rate of thrombotic complications to be different procedures prompting warfarin interruption. Several factors influence hemostasis when a patient stops warfarin, receives bridging, and undergoes surgery. 1 This could explain the counterintuitive relation between LMWH and thrombosis. Despite our best efforts, we could not get reliable data on what type of procedure was performed. It could be that low-risk procedures often meant no LMWH. It is possible that some patients benefit from bridging and maybe some patients should receive high dose of LMWH.
As for reliability of outcome data, death is a certain endpoint but cause of death is uncertain when out of hospital, without laboratory and radiological examinations, and with a very low autopsy rate. The number of deaths out of hospital was 51. Of these, only a small number were classified as a diagnosis that would qualify as a complication, with the majority being heart failure or infection. This is a common problem with register studies and makes it difficult to use cause of death as an endpoint, unless there is a diagnosis in the NPR. Lastly, it is possible that some patients were given unfractionated heparin as in-patients, which would not be registered in Auricula, but in our experience this is extremely uncommon in Sweden.
We have studied nonrandomized treatments in the absence of randomized data. This opens up for a possibility of bias as to which patients were offered LMWH bridging. However, there is no systematic exclusion of any patient groups regarding age, indication for anticoagulation, or comorbidities, meaning we have studied a real-life population. By comparison, our matched atrial fibrillation group was about 74 years old, slightly older than in a recent randomized trial, with a higher prevalence of stroke (30% vs 9%), similar rates of congestive heart failure (34% vs 32%), and lower rates of hypertension (65% vs 87%) and renal failure (7% vs 11%). 5 Comparing our data on bridgings in patients with venous thromboembolism to a North American register study, 4 our patients were slightly younger (63 vs 66), had a higher prevalence of congestive heart failure (8% vs 1%) but a lower prevalence of hypertension (34% vs 47%) and diabetes (10% vs 17%). Auricula covers all patients, both in primary care and at specialist centers within the county councils that have decided to join, and that means about half of Sweden’s population.
In the last few years, several bridging studies have been published. One prospective randomized trial showed no reduction in the risk of arterial thromboembolism, but increased risk of major bleeding. This study was restricted to 1884 patients with atrial fibrillation, with mean CHADS2 scores of 2.3 and 2.4 in control and treatment arms, respectively. Fewer than 15% (260 patients) were at high risk for stroke (CHADS2 ≥ 4). 5
We planned a propensity scoring of bridgings in atrial fibrillation patients with CHADS2 ≥ 4, but with only 187 bridgings possible to match, matching was deemed futile. Analysis of crude data (not shown, 625 cases) showed neither benefit nor harm for this group. Altogether, we found no support for LMWH bridging in atrial fibrillation.
Another recent, retrospective cohort study of 1178 patients with previous venous thromboembolism found that bridging was associated with a significantly increased risk of bleeding, but a nonsignificantly lower risk of recurrent venous thromboembolism. 4 With risk-matched data on a larger number of patients, we confirm increased risk and lack of efficacy from LMWH bridging for patients with venous thromboembolism.
For patients with mechanical heart valve prostheses, there is solid evidence for warfarin, but increased risk of thrombosis with the more short-acting dabigatran. 18 Low-molecular-weight heparin has a similarly short duration and would therefore not be expected to perform well. In our analysis with propensity score matching of risk factors for thromboembolism and bleeding, we found no support for LMWH bridging in patients with mechanical heart valves.
Since this is a retrospective register study, our findings need to be confirmed in randomized clinical trials. Such a trial in relevant patient groups is recruiting since 2006. 19
Conclusion
We found no benefit from LMWH prophylaxis in short-term warfarin interruptions irrespective of warfarin indication, in this large retrospective national register study.
Footnotes
Authors’ Note
V.S. and A.S. conceived the study. V.S. and H.R. extracted the data and did the statistical analyses. V.S. drafted the manuscript. All authors interpreted the data, critically reviewed the manuscript, and approved for publication. This study was approved by the regional ethics review board in Umeå, Sweden (EPN nr 2011–349-31M and 2012–277-32M) and conformed to the declaration of Helsinki.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This was an academic study supported by the Department of Public Health and Clinical Medicine, Umeå University and the Department of Research and Development, County Council of Västernorrland (grants LVNFOU385111 and LVNFOU481841).
Supplemental Material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
