Abstract
Given that the presence of antiphospholipid (aPL) antibodies has been proposed to be associated with thrombosis in newly diagnosed patients with lymphoma, we conducted a prospective cohort study on these patients. In all, 154 patients were enrolled. More than half were advanced-stage diffuse large B-cell lymphoma. Approximately one-third (35.7%) of the patients had the presence of aPLs, with single-, double-, and triple-aPL positivities of 29.9%, 5.2%, and 0.6%, respectively. Of the 154 patients, 8 (5.19%) developed symptomatic thrombosis during follow-up. There were no significant differences in the incidences of thrombosis for the aPL-positive and aPL-negative groups (5.5% vs 5.1%;
Introduction
Patients with cancer are more at risk of thrombosis than the general population, having a 5-fold higher risk than those without cancer. In fact, the incidence of thrombosis is as high as 10% to 15% during the course of their cancer. 1,2 This applies to both hematologic malignancies and solid cancers. 3 –5 The incidence in large groups of patients with malignant lymphomas has varied between 1.5% and 59.5%, depending on the patients’ lymphoma subtype, disease stage, chemotherapy regimen, and the intensity of the chemotherapy protocol. 3 –5 The highest incidences have been found among patients with major risk factors for thrombosis, namely, high-grade non-Hodgkin lymphoma, a high international prognostic index score, and the presence of a mediastinal mass and/or central nervous system lymphoma. Minor thrombotic risk factors have been reported in those with old age, a higher stage of disease, immobilization, and/or the presence of a central venous catheter. Most thromboses occur upon the diagnosis of the cancer or early in the course of the cancer treatment (particularly during the first 3 months). 3 –9
Currently, the pathogenesis of thrombosis in patients with lymphoma is still not well understood. Venous stasis from tumor compression or immobilization is well-known risk factors that contribute to thrombosis, particularly in patients with lymphoma with a huge mass or with immobilization due to a neurological deficit found in central nervous system lymphoma (CNSL). 10 Nevertheless, thrombosis may occur in lymphomas that are not large or do not have any CNS involvement. This suggests that there might be other causes, such as a hypercoagulable state, that lead to thrombosis. Given that lymphomas have a tendency to produce antibodies to many proteins, 11 immune-mediated thrombophilia is a possible cause of thrombosis in some patients with lymphoma.
Recent research has revealed a correlation between a thrombophilic state and a higher incidence of thrombosis in many kinds of malignancy, including lymphoma. Thus, 27% of newly diagnosed patients with lymphoma were found by one study to have lupus anticoagulants (LAs) and/or anticardiolipin (aCL) antibodies, compared with the incidence of 8% for the general population. 12 Interestingly, during the follow-up period of that research, the rate of thrombosis in the series was significantly higher among the patients with antiphospholipid (aPL) antibodies than without aPLs (5.1% vs 0.75% patients per year, respectively). 12 Those data have since been corroborated by another study of 66 cases of immune-mediated thrombophilia in lymphoma: Thrombosis occurred in 22 of 48 evaluable cases. Given that, after completion of the chemotherapy course, the LA became negative in more than one-third of the patients, its presence was suggested to be associated with the malignant disorder. 13 Although a number of studies also supported this proposed hypothesis of correlation between the presence of aPLs and thrombosis in patients with lymphoma, 14,15 others have not. 16 The pathogenesis of thrombosis of lymphoma and the presence of aPLs, therefore, remains unclear. 17
In addition, we are not aware of any study having been conducted on an Asian population. We conducted this study to determine the incidence of aPL positivity in newly diagnosed patients with lymphoma with the goal of developing a predictive score for the presence of aPLs in those patients.
Methods
Patients and Procedures
This prospective, cohort study enrolled newly diagnosed patients with lymphoma. The patients were aged 15 years or older, and they were followed up at Siriraj Hospital, Mahidol University, Bangkok, Thailand, October 16, 2017, to February 28, 2018. We excluded patients with a history of any of the following: (1) previous venous/arterial thrombosis, (2) an unexplained recurrent abortion, (3) familial thrombophilia, (4) an autoimmune disease, (5) anticoagulant use, and (6) a liver disease or elevated liver enzyme (> 3 times the upper normal limit). Before the patients received lymphoma treatment, blood samples were collected for aPLs, comprising LA, aCL-immunoglobulin M (IgM), aCL-IgG, anti-β2 glycoprotein I (aβ2GPI)-IgM, and aβ2GPI-IgG. Lupus anticoagulant was detected using the dilute Russell’s viper venom time and activated partial thromboplastin time (aPTT) tests (actin FSL reagent, Siemens) which is different from aPTT testing in routine coagulogram workup (actin FS). The enzyme-linked immunosorbent assay method was used to measure aCL-IgM, aCL-IgG, aβ2GPI-IgM, and aβ2GPI-IgG. We utilized commercially available kits (Euroimmun) for all testing. Following the guidelines outlined in the Declaration of Helsinki and all of its subsequent amendments, the institutional review board of the Siriraj approved our study. It was supported by the Faculty of Medicine Siriraj Hospital, Mahidol University.
Clinical manifestations were collected; these consisted of the symptoms and physical signs of all of the patients at their initial presentation. A hospital-based chart review was performed for every patient until the end of the first chemotherapy regimen. Details of the thrombotic events that occurred throughout the first course of chemotherapy were also collected.
Statistical Analysis
The clinical characteristics were presented as median with interquartile range and as mean with standard deviation or percentage. The paired
Results
Baseline Characteristics
A total of 154 newly diagnosed patients with lymphoma were enrolled. Their mean age was 58.7 ± 16.5 years, with equal proportions of males and females. The most common lymphoma subtype was diffuse large B-cell (DLBC) lymphoma (52.6%), followed by marginal zone lymphoma (MZL; 9.7%), follicular lymphoma (FL; 6.5%), and primary CNSL (5.2%). Nearly half of the patients had lymphoma stage IV. The initial complete blood counts demonstrated a mean hemoglobin of 11.0 ± 2.3 g/dL, a median white blood cell count of 8.0 × 109/L (range: 1.5-44.0 × 109/L), and a median platelet count of 268 × 109/L (range: 21-764 × 109/L). The coagulogram results were a mean prothrombin time of 11.5 ± 1.6 seconds, a mean aPPT of 24.7 ± 3.3 seconds, and a median fibrinogen level of 442.1 mg/dL (range: 56.2-1, 396.9 mg/dL). Table 1 illustrates the baseline patient characteristics and initial laboratory findings.
Baseline Patient Characteristics and Initial Laboratory Findings.

Abbreviations: aPTT, activated partial thromboplastin time; Hb, hemoglobin; NK, natural killer; PT, prothrombin time; WBC, white blood cell.
Incidence of aPL Positivity and Thrombotic Events
Approximately one-third (35.7%) of the patients had the presence of aPLs, with single-, double-, and triple-positivities of 29.9%, 5.2%, and 0.6%, respectively. The following positive results were determined: LA, 17.5% which were positive by aPTT-based method using actin FSL reagent (14.3%), dilute russell viper venom time method (2.6%) and both methods (0.6%); aβ2GPI-IgM, 17.3%; aβ2GPI-IgG, 0.6%; aCL-IgM, 3.2%; and aCL-IgG, 3.9%. Overall, aPL positivity was found for 39.5% of the DLBC lymphoma cases, 33.3% of the Hodgkin lymphoma, 40% of the FL, and 40% of the MZL. Moreover, aPL positivity was found in 25% of angioimmunoblastic T-cell lymphoma cases, 25% of peripheral T-cell lymphoma, and 50% of natural killer/T-cell lymphoma. Of the 154 patients, 8 developed thrombotic events, with venous thrombosis in 7 and an arterial thrombosis in the 1. Interestingly, only 3 thrombotic cases were aPL positive. One patient had triple positivity for aPLs without developing clinical thrombosis. Table 2 details the thrombotic events.
Details of Thrombotic Events in 8 Patients.

Abbreviations: aPL, antiphospholipid; CNS, central nervous system; DLBCL, diffuse large B-cell lymphoma; PCNSL, primary central nervous system lymphoma.
Comparisons of Clinical Characteristics, Laboratory Results, and Outcomes
The clinical characteristics of the aPL-positive and aPL-negative patients (age, body mass index, comorbidities, and initial laboratory findings) were not significantly different. However, 2 factors—gender and lymphoma stage IV—were statistically significantly different. The incidences of thrombosis in the aPL-positive and aPL-negative groups was identical (5.5% vs 5.1%;
In the univariate and multivariate analyses, male gender and lymphoma stage IV had higher likelihoods of aPL positivity in patients with lymphoma, as presented in Table 3.
Univariate and Multivariate Analysis for Risk Factors of aPL Positivity.

Abbreviations: aPL, antiphospholipid; BMI, body mass index; Hb, hemoglobin; OR, odds ratio; PT, prothrombin time; WBC, white blood cell.
Development of a Predictive Score for aPL Positivity
The aPL predictive score of aPL positivity was assessed using the following multiple logistic regression model:
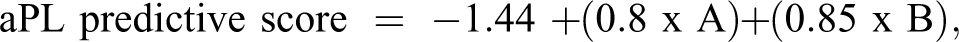
where
We used the receiver operating characteristic curve analysis of the aPL predictive score. The most suitable cutoff level for aPL positivity was obtained when the area under the curve was 0.64 (95% CI: 0.55-0.73). The sensitivity of the score was 83.5%, whereas the specificity was 34.3% (Table 4).
Sensitivity, Specificity, Positive, and Negative Predictive Values of aPL Predictive Score.

Abbreviations: aPL, antiphospholipid; AUC, area under the curve; PPV, positive predictive value; NPV, negative predictive value.
Discussion
The prevalence of aPLs in the normal population has been reported by several studies to range from 1% to 5.6%. 18 –21 In the present study, we analyzed the prevalence in newly diagnosed patients with lymphoma; we noted a considerably higher level (37.5%) in this group of patients, consistent with reports on other patients. 12,13 Notably, LA and aβ2GPI-IgM were the first and second most common aPLs in the aPL-positive group of our cohort. This finding differs from an earlier study, in which aCL antibodies were predominant, 12 while another study by Bairey et al 22 reported that aβ2GPI-IgM was the most common aPL found in lymphoma, with a prevalence of 40%. The current research is thus the first to report on Asian patients with lymphoma; furthermore, it used a prospective cohort design and enrolled a large number of patients. Although a number of case reports have demonstrated an association between aPLs and lymphoma, 23 –26 most of the lymphoma subtypes hitherto published have been MZL. This could be explained theoretically by its unique, plasmacytic-differentiation characteristic. Nonetheless, our data showed that aPLs can also be found not only in indolent lymphomas (such as FL or MZL) but also in DLBC and T-cell lymphomas. These findings support 2 aPL production mechanisms: direct synthesis by lymphoma cells (found in plasmacytic-differentiated lymphomas) 27 and immune system production of the antibodies against the protein expressed on lymphoma cells. 11 The explanation why advanced-stage lymphoma tends to have more aPL positivity might be that a higher tumor load can produce more antibodies. For primary antiphospholipid syndrome, female gender is well known to be predominant due to a close correlation with autoimmune diseases. This is similar to other immune-mediated cytopenia (eg, immune thrombocytopenia or autoimmune hemolytic anemia) in which a majority of patients are young female. Nonetheless, the proportion of males nearly equate to females in the elderly groups. 28 This might explain why our results demonstrate a higher possibility of aPL positivity for the male gender due to our high mean age of 58.4 years. In addition, it has been proposed that HIV infection might contribute to aPL positivity. 29 There were only 4 HIV patients included in this study. Two of them had DLBC lymphoma and the aPL results were both positive for LA. While the other 2 patients had Burkitt lymphoma with negative aPL results. However, due to limited number of HIV patients in this study, we cannot clearly define the association between HIV infection and aPLs.
A previous aPL score to predict diagnosis of aPL syndrome and thrombosis has been proposed to be utilized in systemic autoimmune patients. 30 The parameters used in this score mainly depend on aPL testing. However, we developed a predictive score with high sensitivity for aPL screening in newly diagnosed patients with lymphoma. Patients who score ≤−1 might not benefit from aPL screening. In other words, if a patient is either male or stage IV lymphoma, it is worthwhile performing an aPL test. Nevertheless, the high incidence of aPL positivity did not affect the frequency of thrombotic events in this patient group. Our results revealed that the incidence of symptomatic thrombosis after lymphoma diagnosis was approximately 5% for both the aPL-positive and aPL-negative groups. This contrasts with a previous report which found a significant relationship between aPL positivity and thrombosis. 12 The reasons behind this might be explained by certain limitations to our study. First, 60% of our thrombosis cases accounted for the compression of the venous system by the large lymphoma mass at presentation. As such, the hypercoagulable state did not contribute to thrombosis in those patients. In addition, the number of patients with thrombosis in the current research might not have been high enough to demonstrate a correlation between aPLs and the thrombotic rate. Thus, it would be beneficial to undertake further studies that include more patients in order to detect a greater number of thrombosis cases. Furthermore, although the sensitivity of aPL predictive score was good, the specificity was 34.3%. The current study recommends that high specificity tests were required for the aPL syndrome diagnosis. 31 We, therefore, have ongoing research on validating this score. Of note, routine aPTT coagulogram workup (actin FS) failed to show significant difference between aPLs-negative and aPLs-positive patients in univariate analysis to identify predictive factors of APL positivity for developing aPL predictive score, as shown in Table 3, that can be explained by its lower sensitivity to detect LA. Actin FSL reagent (aPTT-based method for LA screening) is different from actin FS reagent because it is designed to have lower phospholipid content, which is why it provides increased sensitivity to detect LA. 32 However, actin FSL reagent is not recommended to be used as a routine aPTT coagulogram workup to avoid false prolongation especially in elderly population. 32,33 Lastly, false positives of aβ2GPI and aCL can be found with several conditions, such as infections and inflammation; hence, this may have led to a high incidence of aPL positivity in our study. Repeated aPL profiles are needed to confirm the persistence of those findings. Although our results are unable to demonstrate a correlation between aPL positivity and the development of thrombosis, the positivity of aPLs in indolent lymphoma will alter the management plan for selected patients from watchful waiting to specific treatment.
Conclusions
There was a high incidence of aPL positivity in newly diagnosed patients with lymphoma. Nevertheless, there was no clear association between aPL positivity and the incidence of symptomatic thrombosis. An aPL predictive score was proposed to assume APLA positivity in newly diagnosed patients with lymphoma. We recommend testing for aPLs in patients with an aPL score higher than −1 because the positivity of aPLs may alter the plan of management, especially in cases of indolent lymphoma.
Footnotes
Authors’ Note
All procedures were conducted in accordance with the ethical standards of the responsible committee on human experimentation, and in accordance with the 1964 Declaration of Helsinki and all of its subsequent revisions. The local ethical commission approved the study. Written informed consent was included. All authors designed the study. SK collected and drafted the manuscript. WO and TR supervised the project and made critical revisions to the manuscript. YN performed all laboratory results. All authors read and approved the final manuscript. The data sets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Acknowledgments
The authors gratefully appreciate Prof Dr Med Peter Hokland for his helpful suggestions to the manuscript. The authors also thank Ms Khemajira Karaketklang for her assistance with the statistical analyses.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by grants from the Faculty of Medicine Siriraj Hospital, Mahidol University, Thailand.
