Abstract
Antiphospholipid syndrome (APS) is an autoimmune disorder characterized by antiphospholipid antibodies associated with thrombosis and pregnancy complications. Catastrophic APS is a severe form involving multiple organ systems with a high mortality rate. The pathogenesis involves antiphospholipid antibodies which target phospholipid-binding proteins and damage endothelial cells thus activating coagulation, triggering a pro-thrombotic state. Laboratory tests for antiphospholipid antibody detection include lupus anticoagulant testing in the coagulation laboratory and serological detection of anticardiolipin and anti-beta 2 glycoprotein I antibodies. Despite recent updates in the diagnostic criteria for APS the recent decades and our improved knowledge of this disease, there remain several key issues pertaining to diagnosis and testing with potential implications to patient management. Here we briefly review APS pathophysiology, strengths and weaknesses of classification criteria, laboratory challenges leading to test interpretation, and clinical management of this complex condition.
Introduction
Antiphospholipid Syndrome (APS) is rare autoimmune disorder characterized by antiphospholipid antibodies (aPL), prolonged phospholipid-dependent coagulation tests and an increased risk of thrombosis and/or pregnancy-related complications. 1 APS is often associated with autoimmune diseases particularly systemic lupus erythematosus (SLE) and is more common in females. 2 The incidence is rare, varying from 1 to 50 cases per 100 000 per year and is associated with significantly increased mortality compared to the general population. 3 Since its discovery and recognition in the twentieth century, APS has been known to present clinically with arterial or venous thrombosis and/or obstetric complications such as pregnancy losses and pre-eclampsia.4,5 Other clinical features may include livedo reticularis, thrombocytopenia, hemolytic anemia, valvular heart disease, and nephropathy. 6 Catastrophic APS (CAPS) is a particularly rare and severe form of APS characterized by acute clotting of multiple vascular beds with multiorgan failure and a high mortality. 7 The diagnosis and management of APS may be challenging due to disease heterogeneity and due to that fact that transient aPL may occur in a number of common settings particularly infection. In addition, aPL are found in a proportion of healthy people.4,8 aPLs can be demonstrated by lupus anticoagulant (LAC) testing in the coagulation laboratory or by immunological assays detecting antibodies to various proteins, most commonly cardiolipin (aCL) and beta-2-glycoprotein I (a-β2GPI).
While there has been significant progress in the development of diagnostic and classification criteria for APS, 5 several clinical and laboratory challenges persist that will be discussed in this narrative review. In view of improvements in our understanding of APS pathophysiology and treatment, we aim to review and compare recent updates to the clinical and laboratory diagnostic guidelines for APS and summarize current best practices to aid clinicians and pathologists.
This review will also approach aspects that are not covered by the guidelines, such as the classification of CAPS and complexities of APS testing in patients on anticoagulation therapy. Furthermore, we review challenges in LAC testing such as the optimal method to determine of normalized ratios, caveats of the mixing study, seronegative APS, and false negative aPLs. Furthermore, we suggest possible avenues for improvement in APS laboratory testing and potential areas of research.
Pathophysiology of Antiphospholipid Syndrome
The presence and persistence of aPL is central to the pathogenesis of APS. The primary targets of APL are phospholipid-binding proteins, typically cardiolipin with β2GPI acting as a co-factor. Physiologically, β2GPI is known to regulate hemostasis and complement.9,10 Figure 1 summarizes the pathophysiology of APS at the cellular level. Endothelial cells are proposed to play a central role in pathogenesis, with endothelial cell dysfunction and circulating aPL being essential initiating factors. 11 When β2GPI is exposed to anionic phospholipids due to platelet activation and inflammatory responses, it adopts an open conformation and exposes antigenic sites, including a cryptic antigen in domain I which binds autoantibodies. Another mechanism is that oxidative stress can unfold the β2GPI protein on endothelial cells, exposing these antigenic sites. Autoantibodies lock β2GPI in an open conformation and facilitate the binding of β2GPI:aPL complexes to receptors on endothelial cells, placental trophoblasts, monocytes, platelets, and neutrophils, thereby activating them. 11 Complement activation then occurs with C3a and C5a generation and the formation of the membrane attack complex which results in endothelial cell injury and further activation of neutrophils, monocytes, platelets, and endothelial cells, as well as activation of coagulation.8,9,12
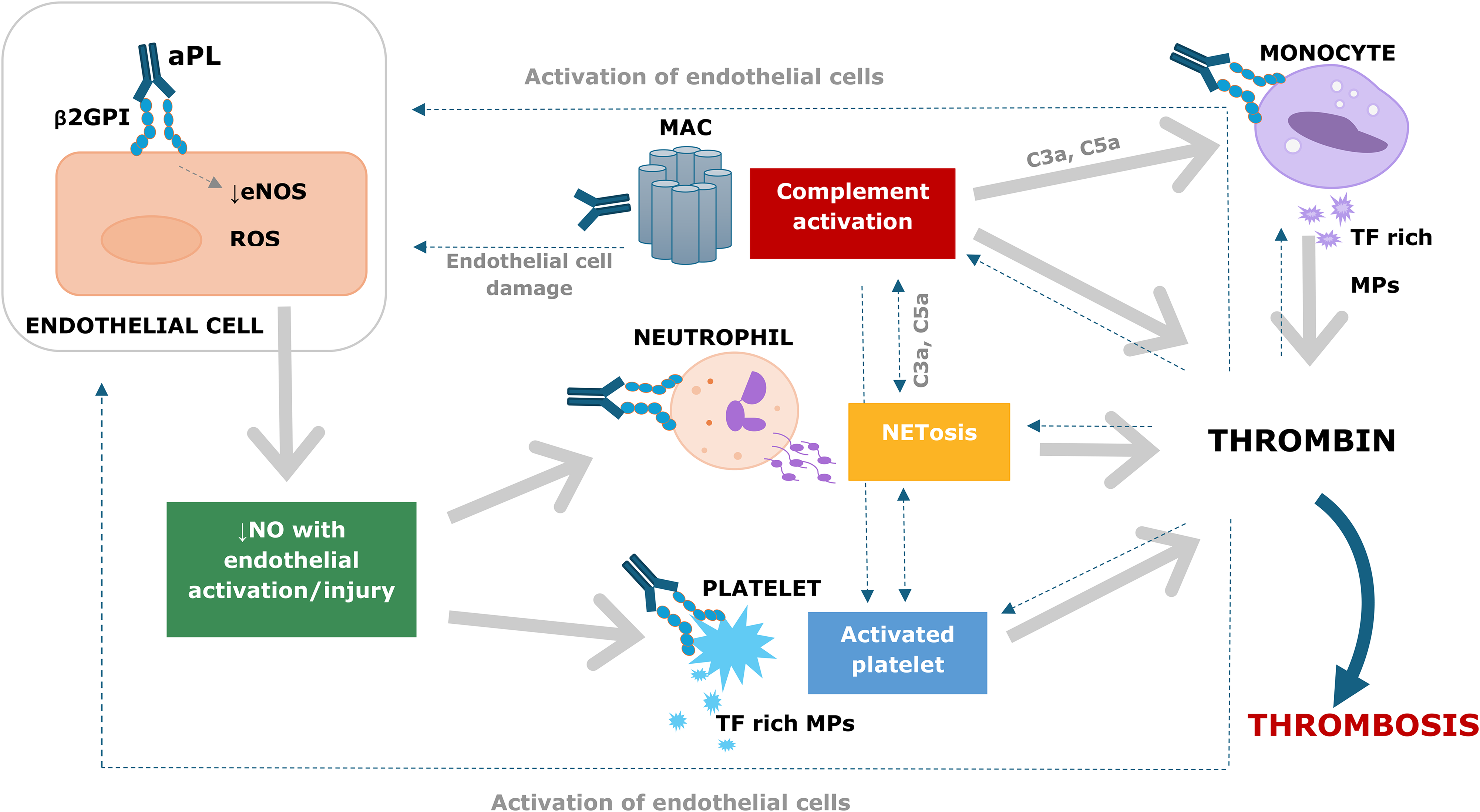
Endothelial-derived nitric oxide (NO) produced by endothelial NO synthase is required for normal endothelial function. Binding of β2GPI:aPL on endothelial cells disrupts NO production, leading to increased oxidative stress, and activation of endothelial cells, neutrophils and platelets with formation of neutrophil extracellular traps (NETS) and tissue factor rich microparticles. 11 Furthermore, platelet-monocyte aggregates form, which express tissue factor and activate coagulation.8,9,12 Other postulated mechanisms for APS include aPL-induced resistance to activated protein C and downregulation of tissue factor pathway inhibitor. 9
Evolution of Antiphospholipid Syndrome Diagnostic Criteria
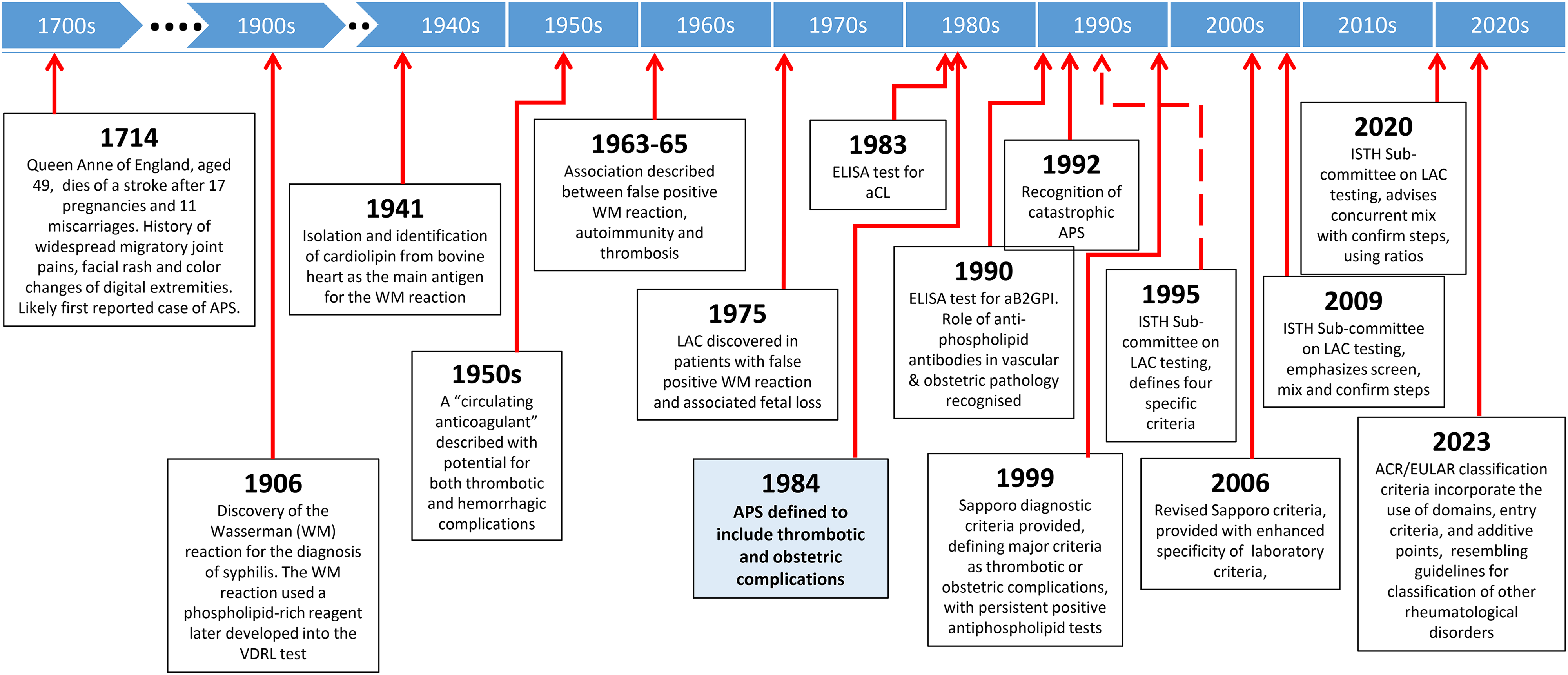
Figure 2 provides a timeline overview denoting historical milestones in the recognition and diagnosis of antiphospholipid syndrome. Table 1 compares and summarizes the evolution of diagnostic and classification criteria for APS over the last 25 years. The first diagnostic criteria for definite APS, the Sapporo Criteria, were developed in 1999, and included thrombotic and obstetric complications in the setting of persistent positive laboratory tests at least 6 weeks apart. 13 In 2006, through an international consensus, an update of the 1999 Sapporo Criteria was issued. 14 The revised criteria included the addition of a-β2GPI to the antibody test criteria, specified high titers of antibodies, and extended the requirement for persistent test positivity from 6 to 12 weeks to improve specificity. Features associated with APS but not included in the 2006 criteria included valvular heart disease, livedo reticularis, thrombocytopenia, nephropathy and neurological manifestations, including visual complications. In 2023, the American College of Rheumatology/European Alliance of Associations for Rheumatology (ACR/EULAR) provided major revisions to the Sapporo Criteria and algorithms. 15 ACR/EULAR proposed a new point- and domain-based scoring system which included weighting criteria and a minimum requirement of at least 3 points from the clinical domains, and at least 3 points from the laboratory domains. At least one documented clinical criterion and one qualifying laboratory test performed within the previous 3 years were required as “entry criteria” for APS diagnosis. The recent ACR/EULAR criteria enhanced diagnostic sensitivity at the expense of diagnostic specificity, risking over-diagnosis and potential for “indeterminate” diagnoses.14,15 Nonetheless, the use of “entry criteria” encourages clinicians to consider the APS diagnosis in the absence of specified Sapporo Criteria. 13
Abbreviations: APS, antiphospholipid syndrome; ACR, American College of Rheumatology; EULAR, European Alliance of Associations for Rheumatology; aCL, anti-cardiolipin antibodies; a-B2GPI, Anti-Beta 2 Glycoprotein I Antibodies; pt/pts, point(s).
*The 1999 and 2006 criteria13,14 recommended risk-stratifying by pre-existing thrombophilia or cardiovascular disease risks, which are addressed in the 2023 criteria 15 by separate weighted points (pts). **Pregnancy morbidity and obstetric manifestations include fetal loss with normal morphology or karyotype beyond the 10th week, one or more premature births before 34th week gestation, or three or more unexplained consecutive spontaneous abortions before 10th week.
Laboratory Testing
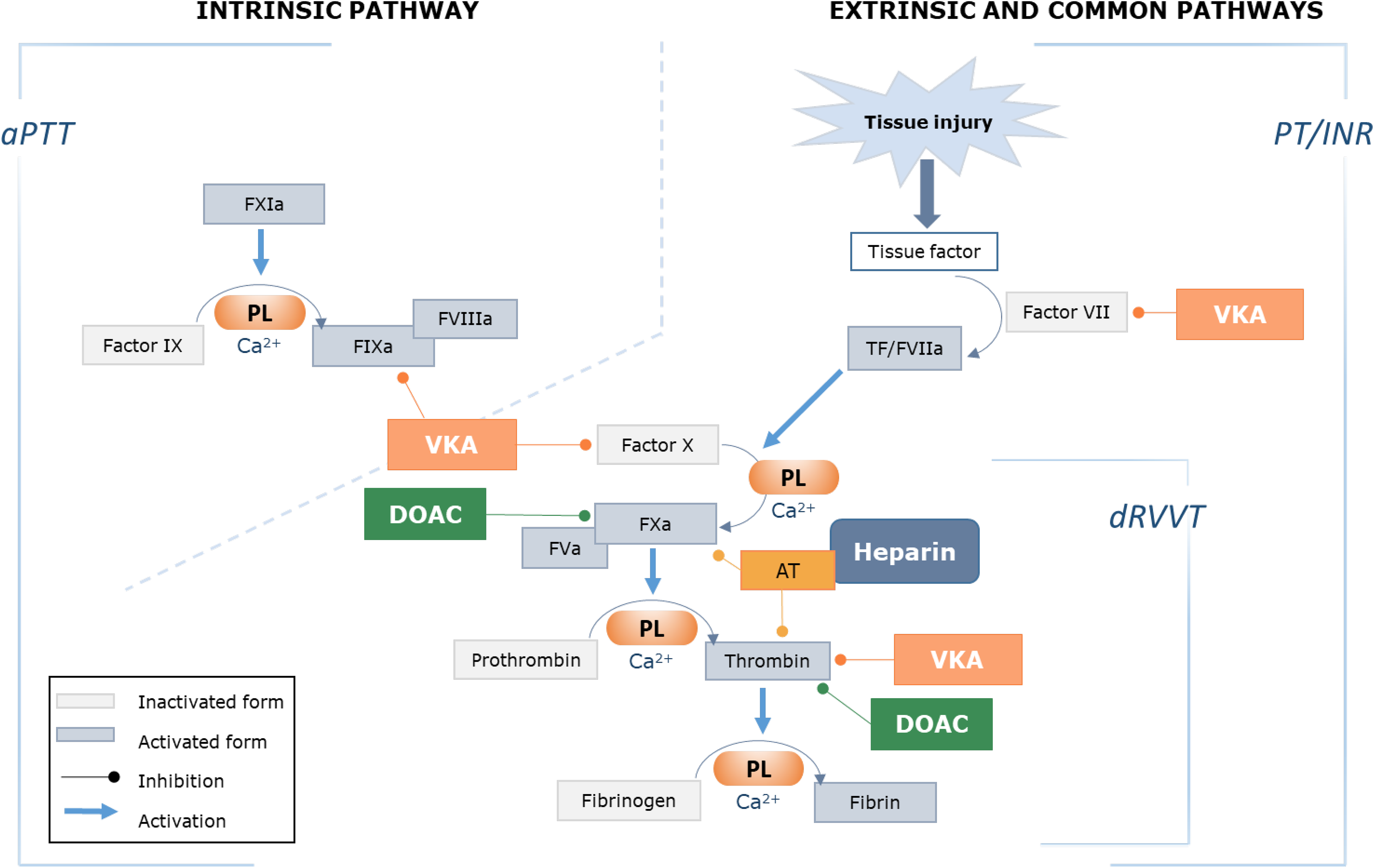
APLs may manifest as LACs using functional clot-based coagulation assays, or be detected immunologically as aCL and/or aB2GPI antibodies in the serum. According to Sapporo Criteria, in the setting of one or more positive clinical criteria, only one laboratory test (LAC, aCL and/or aB2GPI) needs to be persistently positive at least 12 weeks apart for an APS diagnosis (Table 1). 16 LACs are diagnosed by three basic principles: prolongation of a phospholipid-dependent clotting test, evidence of inhibition demonstrated by mixing studies, and evidence of phospholipid dependence (shortening of the prolonged clotting time with the addition of excess phospholipid).16,19 To improve sensitivity and specificity, two clot-based assays using different principles must be used. The dilute Russell viper venom time (dRVVT) and a sensitive activated partial thromboplastin time (aPTT) are the two recommended LAC screening tests. 17 The dRVVT is based on factor X activation and the aPTT on contact factor activation (Figure 3). There are variations in LAC testing between laboratories and in the use of mixing studies, instrumentation, reagents and calculations. Additional coagulation tests may be used where there is a strong suspicion of LAC with inconclusive DRVVT and aPTT results, or possible assay interference from oral anticoagulants. These additional tests include (but not limited to) the dilute prothrombin time and the kaolin clotting time with platelet neutralization. 20 LAC testing should be avoided during anticoagulant therapy, particularly with direct oral anticoagulants (DOACs) and vitamin K inhibitors, as well as during the acute thrombotic phase or during pregnancy due to elevated coagulation proteins. 17
The Sapporo Criteria include detection of aCL IgG/IgM antibodies in medium to high titer, and/or a-β2GPI IgG/IgM levels above the 99th centile. Assays used for aCL and a-β2GPI testing including enzyme linked immunosorbent assays (ELISAs) and automated chemiluminescent assays. A challenge is that lack of universal calibrators for these antibody tests leads to inter-assay variation. Follow up testing is recommended in the laboratory that performed the initial tests and using the same testing platform.14,23
Catastrophic Antiphospholipid Syndrome
CAPS is a life-threatening form of APS and is characterized by the rapid onset of widespread thrombotic events affecting three or more organ systems. CAPS is generally induced by a precipitating acute event such as infection, surgery or malignancy.24,25 Classification criteria for CAPS were proposed in 2002 at the 10th International Congress on aPL, however, the diagnosis of CAPS remains challenging in real-world settings which require an understanding of both the clinical and laboratory features of this rare disorder. 26 A confirmed CAPS diagnosis requires involvement of ≥ 3 organ systems, development of clinical manifestations in less than a week, demonstrating small vessel occlusion on histological sections and positive laboratory tests.27,28 The pathogenesis is poorly understood, with possible factors including the antibody profile, titers and avidity, and underlying genetic, physiological and environmental factors.25,29,30
Due to the rarity of CAPS, and the lack of randomized trials to validate existing diagnostic criteria, recommendations for diagnosis and treatment rely largely on expert opinion. 24 Approximately half of CAPS patients have a history of APS, and in the remainder it is the first manifestation.25,31,32 A high level of suspicion for CAPS is needed for any patient with rapid-onset thrombotic events, progressive multi-organ failure and rapid clinical deterioration. 31 The 2002 CAPS Classification Criteria categorized patients as “definite” or “probable” CAPS, based on the number of organ systems involved, the time of onset, the presence or absence of histological evidence and the laboratory confirmation of aPLs. 33 A confirmed diagnosis of “definite” CAPS hinges on persistent aPL however this is not possible in the 50% of cases with no history of APS. 25 Furthermore, negative aPL profiles at the time of CAPS clinical presentation have been reported. 34 The McMaster RARE-best practice clinical guidelines recommend using the existing APS classification criteria to diagnose CAPS, emphasizing aPL positivity, albeit with the caution of potential false negatives. Tissue biopsy offers highly specific evidence of CAPS, but may not always be attainable in time. 35 In addition, comparative data on the risks and benefits of biopsy are not available, perhaps due to the rarity of CAPS. 36
The differential diagnosis for CAPS with thrombocytopenia and thrombosis in multiple sites includes heparin-induced thrombotic thrombocytopenia (HITT), thrombotic thrombocytopenic purpura (TTP), disseminated intravascular coagulation, scleroderma renal crisis, small vessel vasculitis, and hemolysis, elevated liver enzymes and low platelets (HELLP) syndrome.24,25 Some of these entities have validated predictive scoring systems, typical laboratory and clinical findings and diagnostic laboratory tests which would confirm the alternative diagnosis. For instance, a high 4Ts score, the presence of anti-platelet factor 4 antibodies, positive functional testing and a temporal association with heparin supports a diagnosis of HITT, and a severely low ADAMTS13 levels and anti-ADAMTS13 antibodies support a diagnosis of TTP. Importantly, transient aPL, usually aCL, may be detected in any of these conditions. 25 From an international registry of 522 patients, organ systems most commonly involved in CAPS include renal (73%), pulmonary (60%), central nervous system (56%), cardiac (50%), skin, 37 hepatic (39%), and peripheral vasculature (37%) involvement. 38 Common laboratory findings apart from positive aPL tests, include thrombocytopenia in 65% of cases and schistocytes/red cell fragments on the blood smear in 21.7% of cases. 25
Lupus Anticoagulant Testing During Anticoagulant Therapy
Lupus anticoagulant and other thrombophilia tests should only be performed where results will alter patient management. For patients on therapeutic anticoagulation, false positive or negative LAC tests may occur, 21 and therefore LAC testing on anticoagulation therapy is not advised. 17 Testing should also be avoided in the acute thrombotic phase due to high levels of factor VIII potentially leading to false negative LAC results. 39 Figure 3 depicts the effects of anticoagulant therapies on the coagulation cascade.
Lupus Anticoagulant Testing During Warfarin Therapy
The ISTH 2009 guidelines advocate LAC testing only if the INR is <1.5, 18 however in the 2020 International Society of Thrombosis and Hemostasis (ISTH) guidelines (ISTH 2020) an INR cut-off is not provided. Instead, it is generally advised that LAC testing should be deferred until VKA therapy has been safely discontinued for at least 3 weeks. Alternatively, if essential for clinical management, LAC testing may be performed after three months of adequate VKA therapy by switching temporarily from VKA to therapeutic doses of low molecular weight heparin (LMWH). There is no specific agent available to counteract the VKA effect in vitro. 21 Dilutions of test plasma using 1:1 mix of normal pooled plasma were suggested in the 2009 ISTH Guidelines to correct the VKA effect on factors II, VII, IX and X. 18 However, this practice fell out of favor in the 2020 updated ISTH guidelines due to laboratory variability in the mixing step. 17
Lupus Anticoagulant Testing During Heparin Therapy
Many LAC assays incorporate heparin neutralizing agents such as heparinase, protamine, or polybrene which are generally effective in neutralizing LMWH and unfractionated heparin (UFH) at doses < 1.0 U/mL. However, the effectivity of the neutralizers varies depending on the specific assay and type/brand of heparin used. 39 For example, a recent study demonstrated that enoxaparin only affected LAC testing at supra-therapeutic anti-Xa levels. 40 Samples for LAC testing should be collected as close as possible to the next dose of LMWH.17,39 For both LMWH and UFH, the ISTH recommends verifying the level of heparin that can be neutralized by reagents claiming to contain heparin neutralizers. 17
Lupus Anticoagulant Testing During Direct Oral Anticoagulant Therapy
The dRVVT is particularly susceptible to interference by DOACs, therefore LAC testing should preferably be avoided in this setting.41,42 If essential, LAC testing may be conducted at least 48 h after the last DOAC dose, or longer in patients with renal impairment, and DOAC levels should be measured prior to LAC testing.17,39 In vitro DOAC neutralizers such as DOAC-StopTM and DOAC-RemoveTM are available, however require further validation.39,43 Table 2 provides a summary of the effects of anticoagulants on coagulation tests and proposes strategies for LAC testing in these patients.17,21,39,43–48
Abbreviations: aPTT, activated partial thromboplastin time; CrCl, creatinine clearance; DOACs, direct oral anticoagulants; dRVVT, dilute Russel's viper venom time; FXa, activated factor X; INR, international normalized ratio; LA, lupus anticoagulant; LMW, low molecular weight heparin; PK, pharmacokinetics; PD, pharmacodynamics; RES, reticulo-endothelial system; VKA, vitamin K antagonist; UFH, unfractionated heparin.
Many dRVVT reagents contain a heparin neutralizer able to quench therapeutic doses of UFH and LMWH up to 1 U/mL 39
Though routine monitoring is typically not required, in patients with additional risk factors monitoring of drug levels or drug activity may be beneficial 48
Dabigatran is primarily cleared renally, while rivaroxaban, apixaban and edoxaban are cleared both renally and via hepatic metabolism 45
Confounders in Laboratory Testing
Despite updated ISTH testing recommendations, the practice of aPL and LAC testing remains heterogeneous and variable across individual laboratories, thus creating barriers to standardization of testing. We review and discuss the following sub-topics related to testing and analysis for aPLs.
Lupus Anticoagulant Test Interpretation
Studies of external quality control surveys have demonstrated wide variability in the detection and interpretation of LACs, which may be due to differences in reagents, sensitivity of reagents to LAC, and variability of practices in interpretation.37,49 Nonetheless, a recent international external quality assessment program with approximately 120 participants demonstrated 98% interlaboratory consensus in the correct identification of LAC; in total there were two instances of analytical errors, three instances of interpretation errors, and four instances where the reasons for error were unclear but may be attributed to aPTT-based methods. 49
The ISTH 2020 Guidelines added emphasis on the use of normalized clotting times for all procedures (screening, confirm, and mixing steps), expressed as ratios, which are calculated as follows
17
: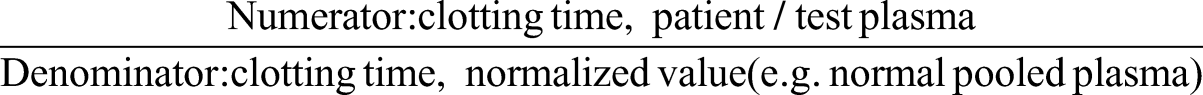
Caveats of the Mixing Study and Mixing Steps
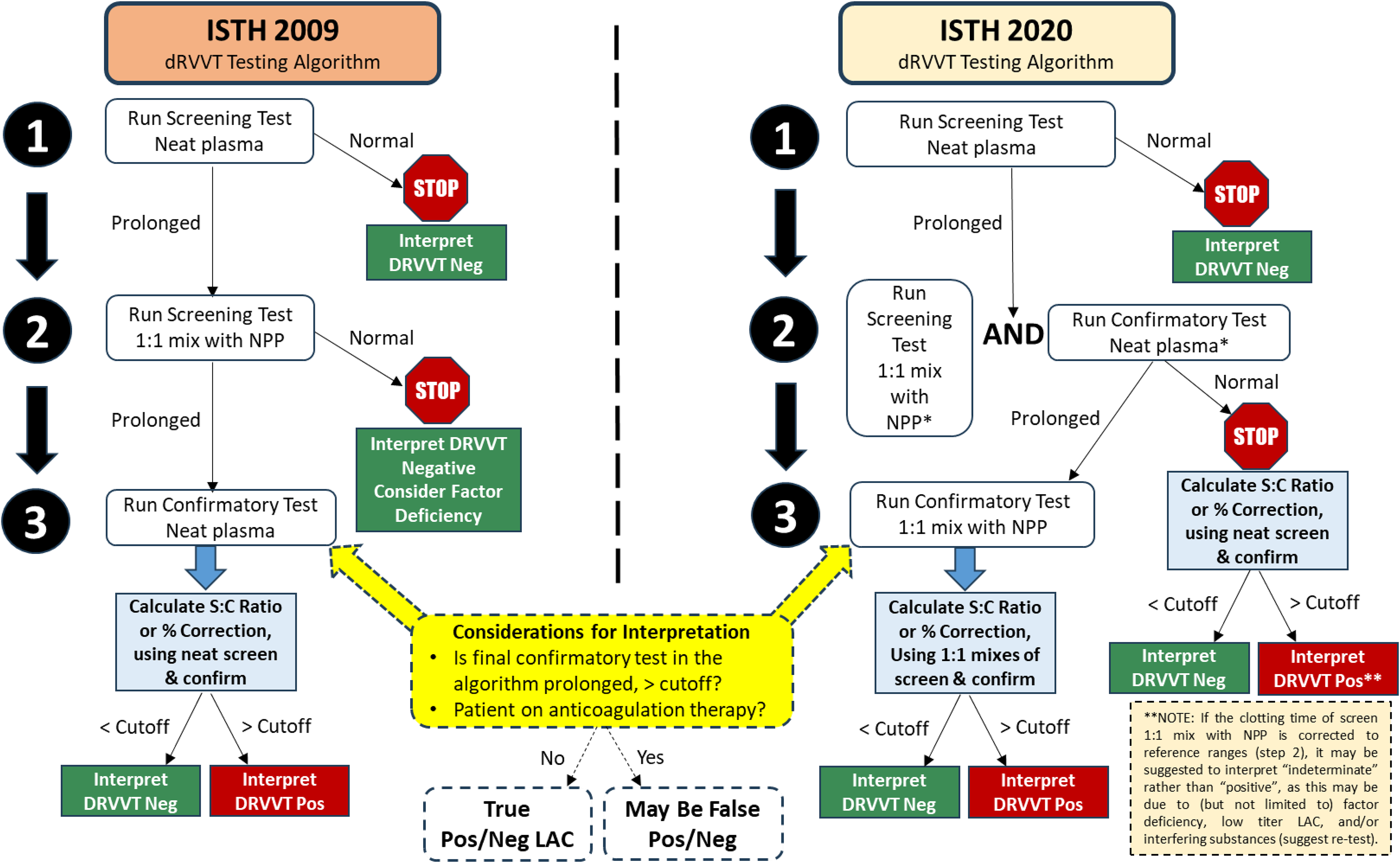
LACs typically act as inhibitors when performing the aPTT mixing test with NPP in a 1:1 ratio, and the prolonged test fails to correct. However, in the case of a weak LAC, the aPTT may correct into the reference range with mixing, leading to misinterpretation as a factor deficiency. 51 In the ISTH 2009 guidelines, a prolonged DRVVT or aPTT normalized screen time should be followed by a 1:1 mixing test with NPP. If the screen is prolonged, but the screen 1:1 mix corrects into the normal range, the interpretation should be negative, and a factor deficiency should be considered. 18 In this scenario, a low strength LAC may be missed.
As highlighted by the ISTH 2020 Guidelines on LAC testing and interpretation, a mixing step is required if the screening LAC assay is prolonged (DRVVT or aPTT).17,18 However, the ISTH 2020 guidelines also advise on performing the confirm test [eg DRVVT confirm or Silica Clotting Time (SCT)] since normalized screening times are prolonged. If the confirm test corrects, test ratios (or percent correction) should be calculated using the unmixed screen and confirm ratios. If the confirm test remains elevated, then the 1:1 mix is also performed on the confirm, and the test ratio (or percent correction) should be calculated using mixed screen and confirm ratios.
Using DRVVT as an example, Figure 4 compares the ISTH 2020 and 2009 guidelines to the use of screen, confirm, and mixing step(s), with interpretation options. The ISTH 2020 guidelines improve the detection of weak LACs, reduce incorrect conclusions of factor deficiencies, and facilitate further appropriate investigation. Use of clinical information to aid interpretation may be helpful or necessary when the pattern of testing appears “indeterminate” (mildly prolonged screen test, corrected screen 1:1 mix test, corrected confirm test, elevated screen : confirm ratio above cutoff indicating a “positive” result, and a normal screen : confirm mix ratio indicating a “negative” result). The clinical significance of such “indeterminate” result patterns should be further investigated, as no standard guideline is provided on these results should be interpreted.
Seronegative Antiphospholipid Syndrome
The entity seronegative APS (SN-APS) has been introduced for patients with clinical manifestations of APS with negative standard laboratory tests. 52 Several studies have explored the presence of other aPL which do not form part of the current diagnostic criteria. These include anti-phosphatidylserine/prothrombin antibodies, antiphosphatidylethanolamine antibodies, anti-annexin A5, IgA isotype a-Β2GPI, antiphosphatidylinisitol, antiphosphatidic acid, and antivimentin/cardiolipin antibodies. One study including 175 consecutive patients with SN-APS, demonstrated that a third of cases showed reactivity to non-criteria aPL. 53 Although there is evidence supporting the role of testing for alternative aPLs in suspected SN-APS cases, these tests are not routinely available.54–56 Furthermore, it is unclear what their prevalence is in healthy controls. To ascertain the clinical significance of non-conventional aPLs, future studies should incorporate healthy controls to better assess their prevalence and clinical significance. 54
“False Negative” Antiphospholipid Antibodies
In addition to advising against testing during therapeutic anticoagulation, The 2020 ISTH guidelines also caution LAC testing following acute thrombotic events, since raised acute phase reactants such as factor VIII and CRP may yield false positive or negative LAC results. 17 Previous studies have described the temporal disappearance of aPL during a thrombotic event, which may lead to a missed diagnosis.57,58 In a prospective study of SLE patients with recent thrombotic events that studied aCL and LACs, aPL previously positive became negative in half the cohort (aCL IgG, by 41%; IgM, by 51%; IgA, by 50%; LAC, by 20%). The majority of those who “lost” aCL IgG and LAC positivity following thrombosis, regained positivity. 57 Other factors which may result in a false negative aPL include pre-analytical factors such as hemolysis, renal loss in nephrotic syndrome, pregnancy, and use of immunosuppressive drugs such as corticosteroids and rituximab.16,59
Management of the Antiphospholipid Syndrome
Universal evidence based therapeutic guidelines are scarce due to the broad clinical spectrum and rarity of APS which precludes well-designed prospective studies. Long term anticoagulation therapy is needed for aPL-associated unprovoked/recurrent/extensive venous thrombosis,60,61 with recent studies supporting moderate intensity dose warfarin (INR 2–3) rather than high intensity dose warfarin (INR 3.1-4). 4 Anticoagulation therapy and treatment should be individualized to ensure that the benefit for each patient outweighs the risk of bleeding. EULAR guidelines suggest low dose aspirin for primary prophylaxis in high-risk patients who do not meet clinical diagnostic criteria for APS.4,15 Treatment and preventive options for obstetric APS include combined low dose aspirin and heparin, low dose prednisolone, intravenous immunoglobulin (IVIG) and hydroxychloroquine.62,63 Due to the high mortality of CAPS, empiric treatment is advised. Treatment options include glucocorticoids, therapeutic heparin, plasma exchange, IVIG, rituximab and eculizumab. Underlying possible precipitants of CAPS also need to be aggressively treated. 64 The combined regimen of immunosuppression, plasma exchange, and therapeutic anticoagulation significantly improved survival and recovery of CAPS in one study (62% vs 23%, p = .014). 64 DOAC therapy should be avoided in triple positive APS, with combined LAC, aCL and aB2GPI positivity, due to high risk of recurrent thrombosis and warfarin is favored in this setting.65–67 Vitamin K antagonists and DOACs are contraindicated during pregnancy. 68
Conclusion
APS is a complex and challenging autoimmune disorder, and appropriate management requires an understanding of its pathophysiology, diagnostic criteria, and therapies. Despite advances in the development of classification criteria, the diagnosis remains complex due to disease heterogeneity and the variability in laboratory testing. While the Sapporo Criteria for APS diagnosis have been the gold standard for two decades, the 2023 ACR/EULAR Criteria aimed to enhance diagnostic specificity, however at the cost of potential over-diagnosis. Prompt and appropriate treatment is needed to improve patient outcomes, particularly in CAPS. As our understanding of the pathophysiology and epidemiology improves, together with refined diagnostic approaches, therapeutic advances will be crucial in improving clinical outcomes in these patients.
Key Points
The latest ACR/EULAR classification guidelines are comprehensive and improves diagnostic sensitivity but may require additional clinical correlation due to decreased specificity, compared to the Sapporo criteria. Catastrophic APS remains a rare entity that is clinically challenging to diagnose and manage, with a high mortality rate. Mitigation strategies for APS testing should consider pre-test variables such as anticoagulation therapy or acute phases of illness which may render false positive or negative results. Despite the most recent ISTH guidelines on laboratory testing for APS, confounders to laboratory testing and result interpretation exist; their significance on patient care is yet to be ascertained.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.


