Abstract
There is no direct evidence comparing the 2 most commonly prescribed direct oral anticoagulants, apixaban and rivaroxaban, used for stroke prevention in nonvalvular atrial fibrillation (NVAF). A number of network meta-analyses (NMAs) of randomized control trials and real-world evidence (RWE) studies comparing the efficacy, effectiveness, and safety of apixaban and rivaroxaban have been published; however, a comprehensive evidence review across the available body of evidence is lacking. In this study, we aimed to systematically review and evaluate the clinical outcomes of apixaban and rivaroxaban using a combination of data gleaned from both NMAs and RWE studies. The review identified 21 NMAs and 5 RWE studies. The data demonstrated that apixaban was associated with fewer major bleeding events compared to rivaroxaban. There was no difference in the efficacy/effectiveness profiles between these treatments. Bleeding is a serious complication of anticoagulation therapy for the management of NVAF, and is associated with increased rates of hospitalization, morbidity, mortality, and health-care expenditure. The majority of studies in this comprehensive evidence review suggests that apixaban has a lower risk of major bleeding events compared to rivaroxaban in patients with NVAF.
Keywords
Background
Atrial fibrillation (AF) is a common arrhythmia that presents a significant morbidity and mortality risk. 1 Direct oral anticoagulants (DOACs) have been licensed and recommended for the prevention of stroke and systemic embolism in people with AF 2 –5 and are now favored over vitamin K antagonists for stroke prevention due to their superior safety profile. 6 However, strokes and systemic embolism may still occur while receiving therapeutic anticoagulation and major bleeding is the most feared complication of anticoagulant therapy. 7
The 2 most commonly prescribed DOACs are apixaban and rivaroxaban; both are factor Xa inhibitors that have differences in their pharmacokinetic/pharmacodynamic profiles and dosing schedules, with rivaroxaban prescribed once daily and apixaban twice daily. 8 Some real world and retrospective studies have demonstrated that a once-daily dosing regimen leads to better adherence and persistence to therapy; however, other studies have not found increased adherence with once daily regimens and suggest that twice-daily regimens provide greater continuity of action compared to once-daily. 9 Dosing regimen is only one of the variables that should be taken into account when making prescribing choices for AF; efficacy/effectiveness and safety profiles are also important considerations.
Numerous network meta-analysis (NMA) studies of randomized controlled trial (RCT) data and post-trial real-world evidence (RWE) studies have been published comparing the efficacy, effectiveness, and safety of apixaban and rivaroxaban. However, these studies vary in important elements that may affect their results, such as their scope with respect to the population or sample size, the subpopulations or demographics, the geographical location of the RWE, the data source, the analytic methodology for both NMAs and RWE, the outcomes or variables reported, and the length of follow-up. Furthermore, a contemporaneous and systematic review across the available body of evidence is lacking. Therefore, in this study, we have conducted a comprehensive evidence review that includes both a summary of RWE alongside an umbrella review, a standardized and systematic collection of data from systematic reviews or NMAs which summarizes the breadth of the literature for a given topic and brings together a summary of reviews in one place. 10,11 Given their comprehensive overview of the evidence, umbrella reviews are becoming increasingly important to support evidence-based health care.
The aim of this study was to systematically review the range and type of relevant published evidence from both NMA and RWE studies to compare apixaban to rivaroxaban with respect to efficacy, effectiveness, and safety, in nonvalvular atrial fibrillation (NVAF). Although these RWE studies and NMAs do not necessarily represent independent pieces of evidence, the variation in the population, methodology, and the outcomes used means that each of these studies may be considered a sensitivity analysis of the others. Hence, this overview is important in assessing the similarities and differences in the body of literature. The data arising from this comprehensive evidence review would represent an important addition to the published literature and would have the potential to support informed clinical and management decisions for patients requiring anticoagulation for NVAF.
Methods
Systematic Literature Reviews
Two systematic literature reviews were undertaken. The first was an umbrella review of NMAs of RCTs and was carried out following standardized procedures, 10,11 while the second was a review of RWE observational studies. Both reviews were conducted following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guideline, 12 including a 27-item checklist of essential items to be reported in a systematic review (Supplementary Table S1). Data sources were searched for English-language publications on NMAs of RCTs indexed January 1, 2012, to February 7, 2018, and publications on RWE observational studies indexed January 1, 2003, to February 7, 2018. The earlier RWE systematic literature review date was an artefact as we had previously sought information on antithrombotic compounds prior to the advent of DOACs. There were obviously no RWE studies for apixaban or rivaroxaban prior to their approval for use in AF for stroke prevention (rivaroxaban was in the beginning of 2012). Both literature searches were executed in MEDLINE (via PubMed) and Embase (via Embase.com) (Supplementary Tables S2 to S5). The Cochrane library was also used for the NMA search (Supplementary Table S6). Bibliographies of recent systematic reviews identified in the searches and included publications were manually checked for relevant studies not identified in the searches.
Eligibility Criteria
Study selection was based on the population, interventions, comparisons, outcomes, and study designs of interest (PICOS) framework (Table 1), which was defined a priori. The only PICOS criterion that differed across the 2 reviews was study design. Primary outcomes of interest aligned with those reported in ARISTOTLE 13 and included stroke/systemic embolism (composite outcome) and major bleeding (defined as “major” or “modified” by the International Society on Thrombosis and Hemostasis [ISTH] bleeding scale) 14,15 ; these outcomes are considered clinically important for both patients and health decision-makers, as prioritized in the NICE Clinical Guideline on AF management (CG180). 16 Incidence of ischemic and hemorrhagic stroke were secondary outcomes. The systematic review of NMAs included studies that presented results of indirect comparisons between apixaban and rivaroxaban (indirect comparisons of other DOACs were excluded). The systematic review of RWE studies included naturalistic cohort studies (prospective and retrospective) and database analyses from medical health records, registries, and claims. Multiple publications for the same study were considered as only a single study.
The PICOS Framework for the Systematic Literature Review.a
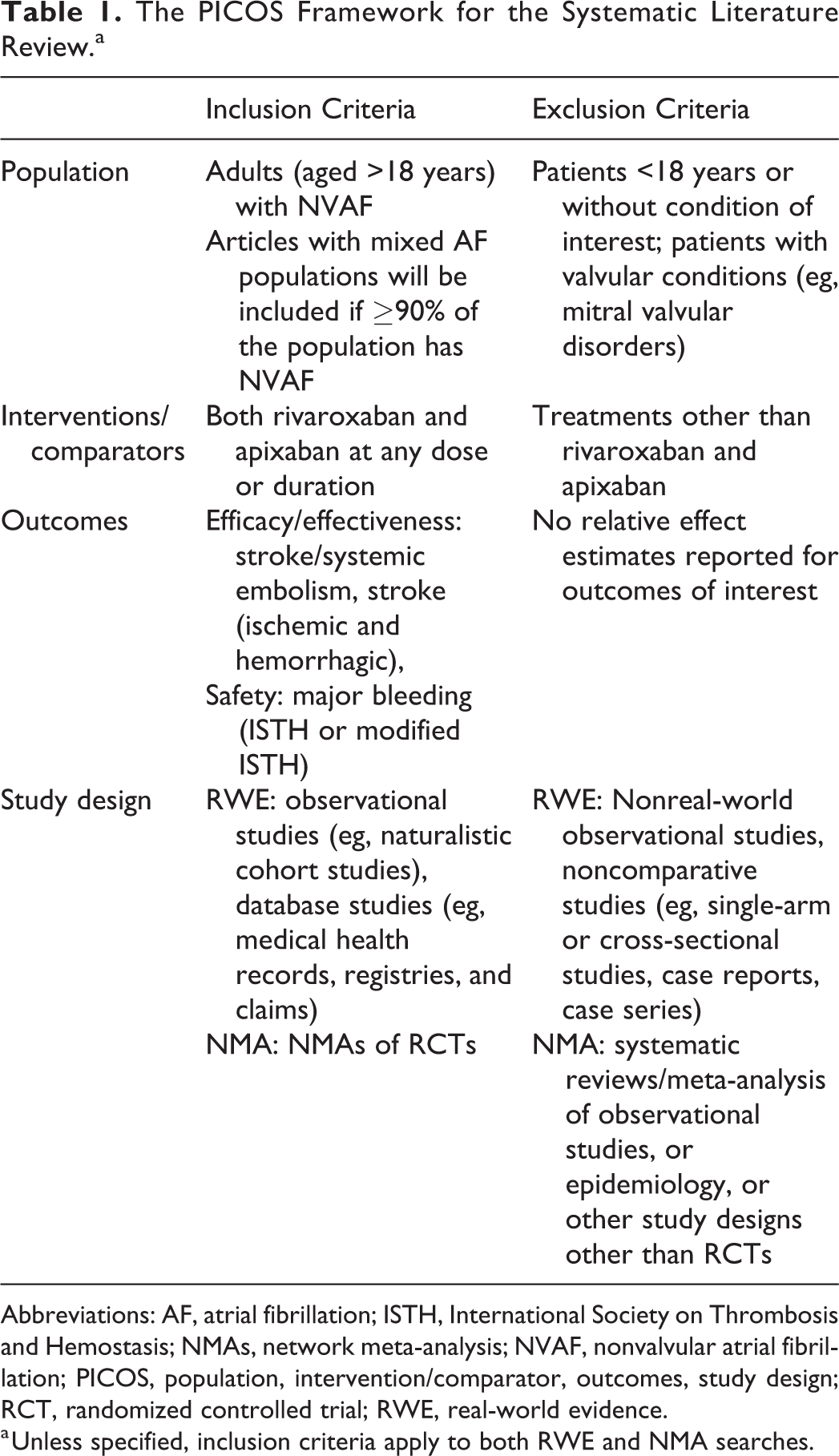
Abbreviations: AF, atrial fibrillation; ISTH, International Society on Thrombosis and Hemostasis; NMAs, network meta-analysis; NVAF, nonvalvular atrial fibrillation; PICOS, population, intervention/comparator, outcomes, study design; RCT, randomized controlled trial; RWE, real-world evidence.
a Unless specified, inclusion criteria apply to both RWE and NMA searches.
Study Selection
Two independent reviewers evaluated titles and abstracts of all citations in line with the PICOS criteria; any discrepancies were resolved by a third investigator. Articles that met predefined eligibility criteria were chosen for full-text screening and were reviewed by the 2 reviewers against eligibility criteria as outlined in the PICOS framework. Full-text articles that met all inclusion criteria and no exclusion criteria were included for data extraction.
Data Extraction and Outcome Measures
One reviewer extracted prespecified data from each included article, and a second reviewer validated its accuracy. Extracted information included study design characteristics, methods, treatment details, patient characteristics/demographics, plus efficacy/effectiveness, and safety outcomes (evaluated as adjusted relative effects, such as hazard ratios [HRs], odds ratios [ORs], and risk ratios [RRs]).
Risk of Bias Assessment
Included NMAs were assessed for risk of bias according to the NICE Decision Support Unit (DSU) checklist, 17 intended for pairwise meta-analysis, indirect comparisons, and NMAs (Supplementary Table S7a). Real-world evidence studies were assessed for risk of bias using the Agency for Healthcare Research and Quality (AHRQ) risk of bias assessment tool. 18 Two reviewers independently undertook the risk of bias assessment and disagreements were resolved by consensus.
Qualitative Assessment
Meta-analysis of relative treatment effect estimates was not undertaken across studies owing to study and patient level heterogeneity that cannot not be statistically measured with this study design; alternatively, a qualitative assessment of the direction and magnitude of reported trends in outcomes across studies was undertaken, with results from individual studies descriptively summarized and presented in forest plots. The forest plots were generated using SAS software (SAS Institute Inc, Cary, North Carolina). 19 Within each identified study, if credible intervals, from NMAs, or confidence intervals (CIs), from RWE studies, did not cross “1,” this presented a statistically significant advantage for either drug, depending on the direction of effect. If credible/CIs crossed “1,” then only a numerical difference was identified, and effect estimates were considered nonsignificant.
Results
Body of Evidence
A total of 485 abstracts were identified for the systematic literature review of NMAs. Following the removal of duplicates, 361 abstracts were excluded, 120 full-text articles were assessed further for eligibility, and 21 met eligibility criteria for inclusion in this review. Reasons for exclusion included study design (76 articles), interventions (10 articles), population (5 articles), and other reasons (8 articles).
A total of 5340 abstracts were identified for the systematic review of RWE. Following the removal of duplicates and studies prior to 2012, 3087 abstracts were excluded, 1370 full-text articles were assessed for eligibility, and 5 met criteria for inclusion in this review. Reasons for exclusion included population (491 articles), intervention (340 articles), study design (168 articles), and other reasons (366 articles; eg, no outcomes of interest, non-English language, duplicate citations).
The study flow diagram is presented in Figure 1. An overview of the NMA and RWE studies, in terms of included trials and outcomes reported, is detailed in Supplementary Table S9.
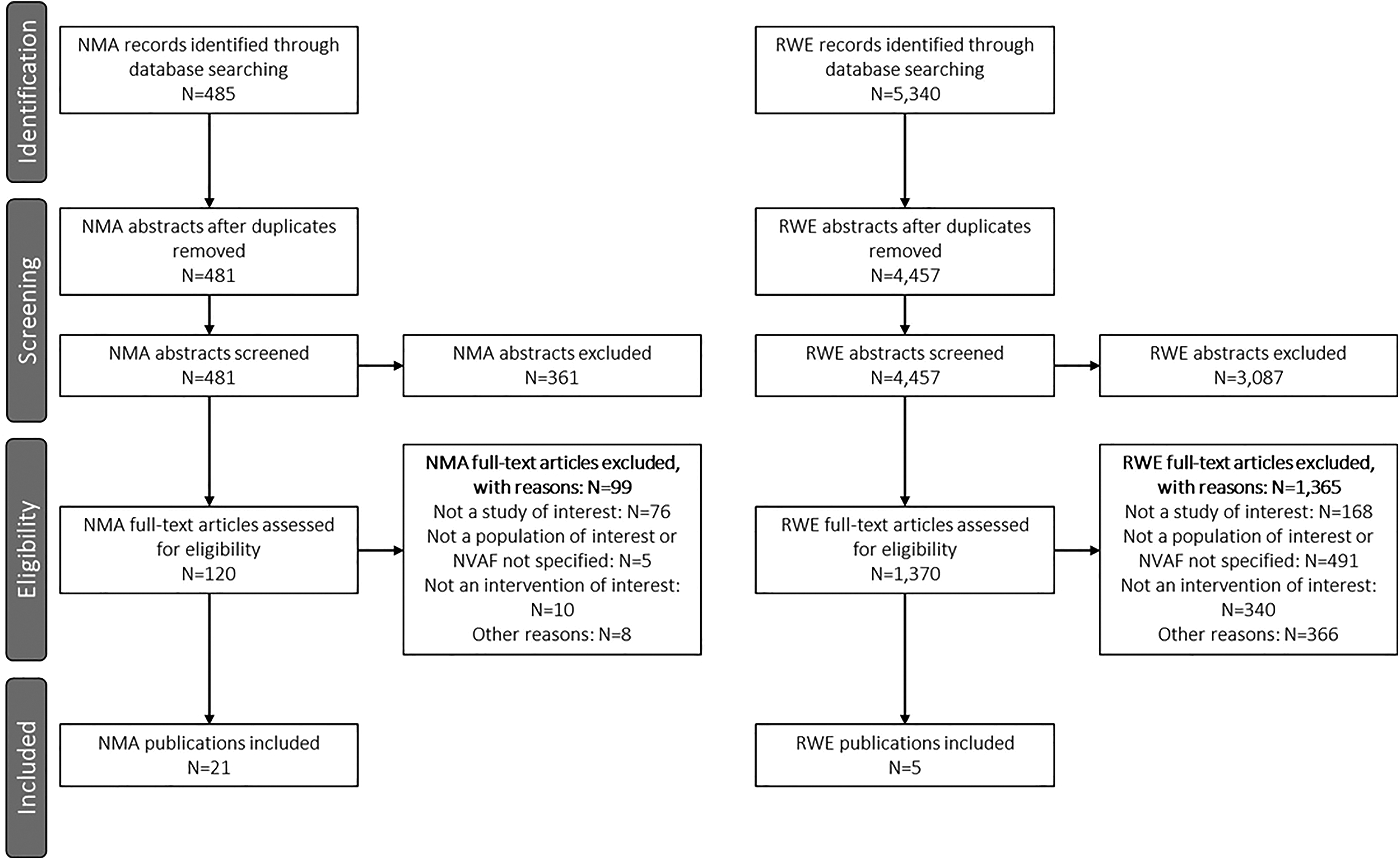
The PRISMA flow diagram of the systematic literature review. NMA indicates network meta-analysis; NVAF, nonvalvular atrial fibrillation; PRISMA, Preferred Reporting Items for Systematic Reviews and Meta-Analyses; RWE, real-world evidence.
Study Characteristics
There was considerable overlap of RCTs included in identified NMAs, as well as heterogeneity in study design characteristics and analytic methodology. The sample size across NMAs ranged considerably, from 13 878 to 897 748 patients. Geographical location was not typically reported, although one NMA considered patients exclusively from Asian countries. 20 Thirteen NMAs used a Bayesian framework with a binomial model, 20 –32 3 the Bucher method, 33 –35 and 1 a frequentist approach. 36 The remaining studies did not report model framework. In addition, the scope of the NMAs differed; some aimed to compare all available DOACs (largest evidence base), whereas others included only a subset of available DOACs. All of the NMAs used indirect comparisons between DOACs. Five of the 21 included NMAs focused on preselected populations, such as AF patients with a previous stroke/transient ischemic attack, 35,37 permanent or paroxysmal AF patients, 24 NVAF patients with renal impairment, 34 or AF patients with chronic kidney disease. 26 Additionally, the publication date of NMAs reflected the availability of RCT evidence when the analysis was conducted. A summary of the 21 NMAs is provided in Supplementary Table S10. To note, many variables were inconsistently reported or not reported by all of the studies.
There was also considerable heterogeneity in study design characteristics and analytic methodology between RWE studies. The sample size across these studies ranged from 12 087 to 22 352 patients for the combined apixaban and rivaroxaban arms. Four of the studies were conducted in North America, 38 –41 and one in Europe (Denmark). 42 One of the RWE studies recruited only NVAF patients ≥65 years of age. 39 All included studies were retrospective cohort studies. Four studies used data from administrative/claims databases, 38,40,41,43 and one from a patient registry. 42 In the 4 studies reporting follow-up time, the mean or median follow-up ranged from 139 to 198 days for apixaban, and from 169 to 195 days for rivaroxaban. A summary of the RWE studies is presented in Supplementary Table S11.
Patient Characteristics
The age range (reported as mean or median) among study participants was broadly similar across the published NMAs and RWE studies, ranging from 69 to 78 years among NMAs, and 68 to 78 years in RWE studies. The proportion of males varied slightly among NMAs, ranging from 56% to 73%, compared to 39% to 62% in RWE studies. Mean CHA2DS2-VASc scores ranged from 2.5 to 3.3 among the included NMAs, and from 2.8 to 4.6 among RWE studies. The baseline risk of bleeding, as assessed by mean HAS-BLED scores, also varied among the RWE studies from 2.0 to 3.1. Time in therapeutic range for warfarin arms, commonly reported in RCTs, varied between 58.5% and 65% in NMAs, with no major differences compared to the RWE studies.
Outcome Assessment
Risk of stroke/systemic embolism
The risk of stroke/systemic embolism reported in the NMAs and RWE studies is presented in Figure 2. Results from 15 of 21 NMAs examining stroke/systemic embolism showed that apixaban and rivaroxaban had a comparable efficacy profile in terms of preventing the stroke/systemic embolism risk; 13 of 15 studies had point estimates suggestive of a potential benefit with apixaban; however, the results were not statistically significant in comparison with rivaroxaban for all studies. Two of the 5 RWE studies reported efficacy: one study, 39 including only AF patients >65 years of age, demonstrated a statistically significant advantage for apixaban compared to rivaroxaban in the reduction of stroke/systemic embolism risk (HR: 0.72, 95% CI: 0.55-0.95). The second study 41 found comparable results between the 2 interventions (HR: 1.05, 95% CI: 0.64-1.72), which did not seem to be affected when only the standard dose of apixaban (5 mg) was considered, but favored rivaroxaban in a dose-adjusting sensitivity analysis that included both the reduced (2.5 mg) and standard dose (main analyses; HR: 1.11, 95% CI: 0.68-1.81). 41
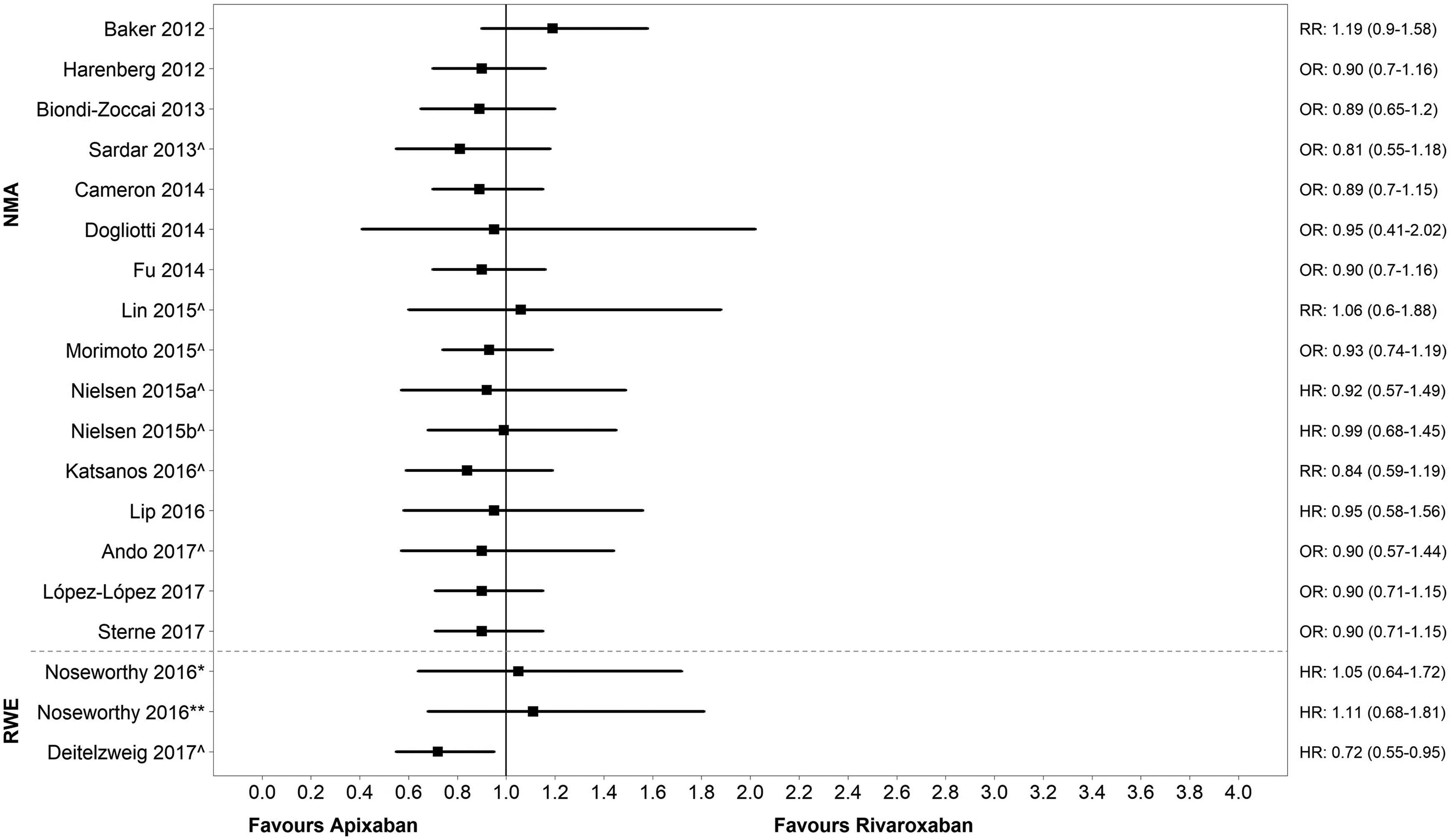
Risk of stroke or systemic embolism: apixaban vs rivaroxaban. Note: the dashed line separates NMA results (top) from RWE results (bottom). Network meta-analysis studies: apixaban dosing was 5 mg and rivaroxaban dosing was 20 mg unless indicated. For example, Lin 2015: rivaroxaban dosing was not reported; Ando 2017: rivaroxaban dosing was not reported. RWE studies: Noseworthy 2016: paper suggests that apixaban and rivaroxaban are a mixture of standard and (unspecified) reduced dosing; Deitelzweig 2017: apixaban dosing was 2.5 mg or 5 mg/rivaroxaban dosing was 10 mg, 15 mg, or 20 mg. ^Notable populations: NMA studies: Sardar 2013 (AF with previous stroke/transient ischemic attack); Lin 2015 (AF patients <65-74 and >75 years); Morimoto 2015 (chronic or paroxysmal AF); Nielsen 2015a: (patients with moderate renal impairment); Nielsen 2015b (patients with mild renal impairment); Katsanos 2016 (AF with previous stroke/ transient ischemic attack); Ando 2017 (AF in chronic kidney disease patients). RWE studies: Deitelzweig 2017 (≥65 years of age). *Main analysis (apixaban 2.5 mg or 5 mg); ** Dose sensitivity analysis (apixaban 5 mg [standard dose]). AF indicates atrial fibrillation; HR, hazard ratio; NR, not reported; NMA, network meta-analysis; OR, odds ratio; RR, risk ratio; RWE, real-world evidence.
No differences were observed for the outcome of ischemic stroke as indicated by the NMAs (range HRs: 0.98-1.09; ORs: 0.75-1.07; RRs: 0.80-1.05). However, contradictory results were found among the 2 RWE studies reporting the risk of ischemic stroke: one reported a statistically significant difference in favor of apixaban (HR: 0.67, 95% CI: 0.49-0.92) 39 ; the other reported a numerical difference in favor of rivaroxaban which was nonsignificant (HR: 1.27, 95% CI: 0.73-2.23) 41 (Supplementary Figure S1).
A numerical difference was found that was suggestive of a potential benefit of apixaban in reducing the risk of hemorrhagic stroke in all 7 NMAs (ie, treatment estimates < 0.80 [HRs: 0.77-0.87; ORs: 0.54-0.83; RRs: 0.55-0.88]), although the differences were not statically significant in any of the studies. Two RWE studies demonstrated a numerical, but nonsignificant difference in favor of apixaban (HR: 0.66, 95% CI: 0.16-2.78; HR: 0.87, 95% CI: 0.50-1.53), and one RWE study favored rivaroxaban (HR: 1.09, 95% CI: 0.89-1.33) (Supplementary Figure S2).
Risk of major bleeding
Sixteen of the 21 NMAs reported apixaban had a significantly lower risk of major bleeding compared with rivaroxaban, with the other 5 demonstrating a numerical difference in favor of apixaban (HR: 0.51-0.74; OR: 0.19-0.71; RR: 0.68-0.80; Figure 3).
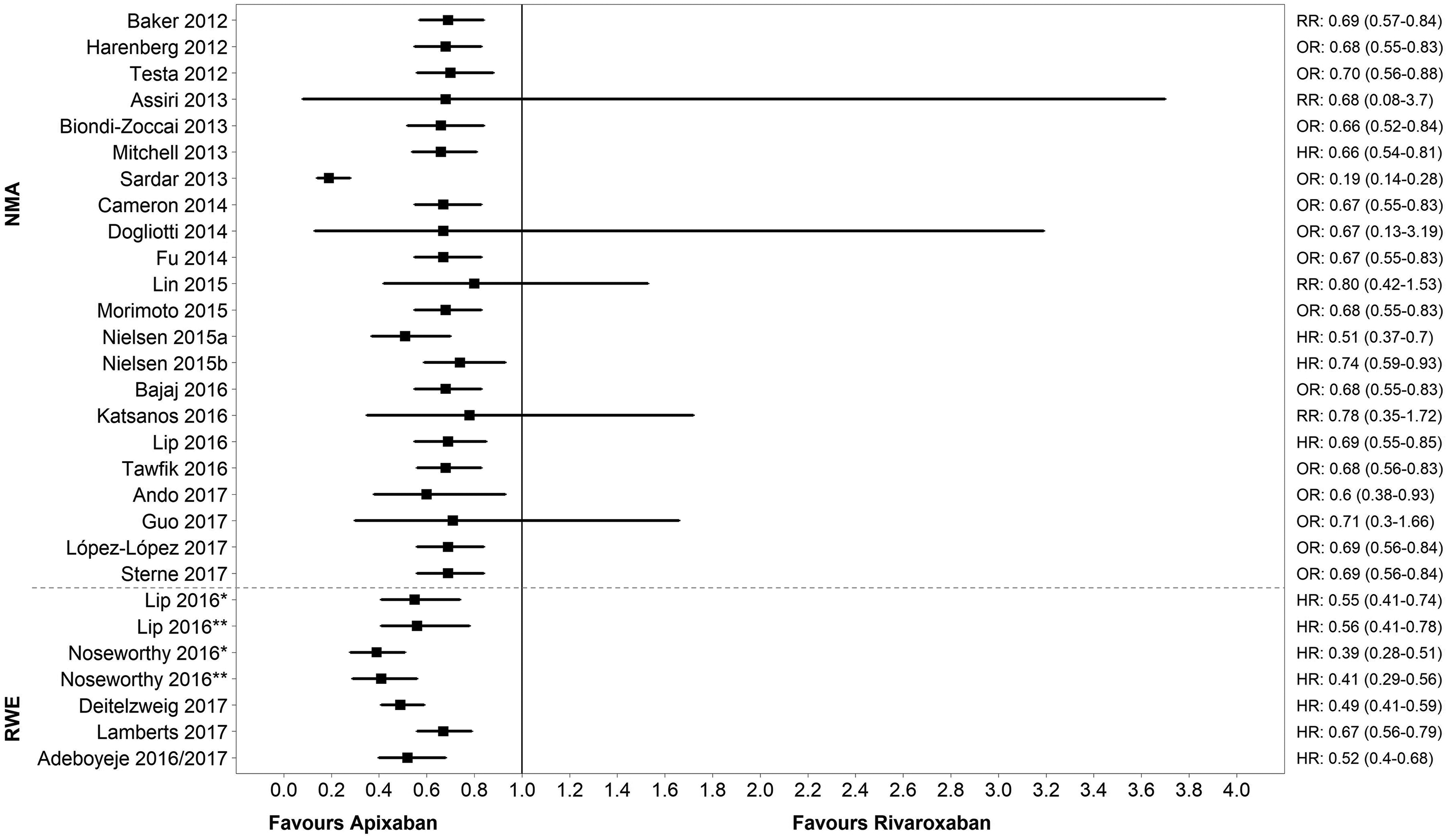
Risk of major bleeding: apixaban vs rivaroxaban. Note: the dashed line separates NMA results (top) from RWE results (bottom). NMA studies: apixaban dosing was 5 mg and rivaroxaban dosing was 20 mg unless indicated. For example, Guo 2017: apixaban dosing was 5 mg or 10 mg, rivaroxaban was 15 mg or 20 mg; Biondi-Zoccai 2013: apixaban dosing was 2.5 mg or 5 mg; Assiri 2013: apixaban dosing was not reported. RWE studies: apixaban dosing was 2.5 mg or 5 mg and rivaroxaban dosing was 15 mg or 20 mg unless indicated. For example, Noseworthy 2016: paper suggests that apixaban and rivaroxaban are a mixture of standard and (unspecified) reduced dosing; Deitelzweig 2017: rivaroxaban dosing was 10 mg, 15 mg, or 20 mg; Adeboyeje 2016/17: apixaban and rivaroxaban dosing was not reported. ^Notable populations: NMA studies: Sardar 2013 (AF with previous stroke/transient ischemic attack); Lin 2015 (AF patients <65-74 and >75 years); Morimoto 2015 (chronic or paroxysmal AF); Nielsen 2015a: (patients with moderate renal impairment); Nielsen 2015b (patients with mild renal impairment); Katsanos 2016 (AF with previous stroke/transient ischemic attack); Ando 2017 (AF in chronic kidney disease patients). RWE studies: Deitelzweig 2017 (≥65 years of age). *Main analysis (apixaban 2.5 mg or 5 mg); ** Dose sensitivity analysis (apixaban 5 mg [standard dose]). AF indicates atrial fibrillation; HR, hazard ratio; NMA, network meta-analysis; OR, odds ratio; RR, risk ratio; RWE, real-world evidence.
All 5 RWE studies reported major bleeding, with all demonstrating a statistically significant advantage in favor of apixaban, with HRs reported in similar ranges as those reported in NMAs (HRs in RWE studies ranged from 0.39-0.67). Two RWE studies performed a dose-sensitivity analysis by restricting to the standard apixaban dose (5 mg). These demonstrated results similar to the main analysis (apixaban 2.5 mg or 5 mg) for the outcome of major bleeding (HR: 0.41, 95% CI: 0.29-0.56 and 0.56, 95% CI: 0.41-0.78, respectively). 40,41
Risk of Bias Assessment
The results from the NICE DSU checklist confirmed that the NMAs provided adequate information on the targeted population, selection of evidence, analytical methods, and presentation of results. However, information on heterogeneity or inconsistency was often inadequately reported. The full results of the risk of bias assessment for NMAs can be found in Supplementary Table S7b and S7c.
The AHRQ assessment scored the quality of the RWE studies as generally high. Selection bias appeared low, and baseline characteristics were similar between treatment groups, with only minimal differences reported. It was unclear whether the study protocol variation compromised the study conclusions (a component of the performance bias assessment) due to limited information available in the publications. The risk from performance and detection bias was rated low for 3 RWE studies, and was considered unclear for the other 2. Risk of attrition bias (as examined by length of follow-up) and reporting bias were considered low for all 5 studies (Supplementary Table S8).
Discussion
To our knowledge, this is the first study to summarize all the published evidence, both clinical trial and RWE, into a comprehensive evidence review comparing clinical outcomes (efficacy, effectiveness, and safety) of apixaban and rivaroxaban for the treatment of NVAF. The summary of evidence demonstrated that apixaban had a significantly lower risk of major bleeding events compared to rivaroxaban in 16 of 21 NMAs with the other 5 demonstrating a numerical difference in favor of apixaban. All 5 RWEs found apixaban to have a significantly lower risk of major bleeding events compared to rivaroxaban. Although the difference in major bleeding in absolute terms is likely to be small, given major bleeding is one of the more common and serious complications of anticoagulation therapy, which often requires medical attention and may lead to hospitalization or fatality, the use of apixaban to treat NVAF patients and the subsequent reduction in bleeding has the potential to positively impact rates of hospitalization, morbidity, mortality, and health-care expenditure.
Since a dose-based interaction effect may be observed with major bleeding, 2 of the 5 RWE studies performed additional dose-sensitivity analyses. 40,41 In the Lip et al study, a sensitivity analysis was conducted to test the robustness of study results for major bleeding among patients prescribed the standard dose (5 mg) which found trends of major bleeding risk to be consistent with the main analyses (apixaban 2.5 mg or 5 mg). 40 A similar finding was also found by Noseworthy et al who performed a sensitivity analysis adjusting for whether patients received reduced doses compared to the main analysis (2.5 mg or 5 mg). 41 The finding that a lower risk of major bleeding with apixaban was independent of dosage is in line with a recently conducted NMA of RWE studies comparing major bleeding risk among patients with NVAF on DOACs or warfarin, 43 and further strengthens the findings of the main analyses.
Regarding stroke/systemic embolism prevention, all 15 NMAs that investigated efficacy found no significant differences between apixaban and rivaroxaban, while in the 2 RWE that investigated efficacy 1 RWE study favored apixaban and 1 RWE showed no significant difference. Apixaban and rivaroxaban demonstrated similar efficacy/effectiveness profiles in risk of ischemic stroke across 15 NMAs and 2 RWE studies. Findings for hemorrhagic stroke in the NMAs demonstrated a numeric difference in favor of apixaban, but observations were nonsignificant. The RWE studies supported these findings, where 2 of 3 RWE studies reported a nonsignificant numeric difference in favor of apixaban associated with wide CIs. The difference between major bleeding and hemorrhagic stroke is thought by hematologists to be due to the different mechanisms underlying these clinical events.
The safety profile of anticoagulants remains a concern for both clinicians and patients. 44 The findings of this review are consistent with those of previous NMAs and meta-analyses of RWE on DOACs in NVAF, 22,43,45,46 demonstrating that apixaban was consistently associated with lower risk of major bleeding compared to rivaroxaban. However, this is the first study to analyze concurrently the entire range and type of peer-reviewed published body of evidence for apixaban versus rivaroxaban. The review of evidence from indirect treatment comparisons of RCTs alongside RWE studies ensured findings were generalizable to the patient population. In addition, the incorporation of RWE studies enabled the reflection of DOAC prescribing patterns in NVAF patients in clinical practice. Since RWE studies can evaluate clinical practice patterns and assess long-term and less frequent safety events (including major bleeding), they have been incorporated into postmarketing approval of DOACs. There is great interest in the RWE studies evaluating DOACs as it is essential that dose selection, treatment adherence, and similar factors be monitored outside of RCTs to determine their impact on key clinical outcomes such as major bleeding or stroke. Furthermore, RWE studies have the ability to assess a broad range of subpopulations, risk factors, and outcomes, including those that RCTs may be unable to assess.
This study has some limitations. Real-world evidence studies are associated with increased risk of inherent selection bias from possible imbalances in patient characteristics between treatment groups. Although such studies may have greater external validity than RCTs, since they reflect real-world clinical experience, there is less certainty about treatment choice bias and less control over data acquisition and quality. Study quality appeared high where domain criteria could be adequately assessed, but many of the included studies reported insufficient information to allow for full assessment of most types of bias; this may reflect the lack of detailed patient care information in some data sources, such as claims data. Although there is a higher chance of data bias and residual confounding among RWE studies as treatments are not randomized (thereby reducing internal validity), careful sampling methodology leads to high external data validity. Additionally, several statistical approaches have been developed that aim to adjust for multiple types of bias (eg, propensity score adjustment, adjusted Cox regression models); these techniques were employed in all 5 RWE studies. However, such methods cannot fully account for potential bias and some residual confounding is inevitable. The NMAs, in their design and scope, included a range of trials and analyses differed between them. Although this was considered when interpreting data, not all studies evaluated all available DOACs; some studies included only a subset. Consequently, there is variation in the evidence utilized within the NMAs, which may have affected the results. However, the definition of major bleeding was similar across RCTs, utilizing the ISTH definition. 15 Despite similarities in the studies and differences in NMA methodology, there was marked consistency in the results. There was also considerable overlap of RCTs included across the NMAs, with the majority of NMAs assessing indirect comparisons from 5 RCTs (ROCKET-AF, J-ROCKET AF, AVERROES, ARISTOTLE, and ARISTOTLE-J). With this degree of overlap across the NMAs studies, the CIs presented are not necessarily unique as the data are redundant across studies. Although these NMAs do not necessarily represent independent pieces of evidence, the differences in methodology and the outcomes used means that studies may be considered sensitivity analyses of the others. It is important to consider these factors when interpreting both the NMA and RWE study results. Hence, this overview is important in assessing the similarities and differences in the body of literature. Further studies that are able to implement a prospective comparison of apixaban and rivaroxaban in NVAF may be considered useful for validating the results of this evidence review.
Conclusion
Direct comparative efficacy and safety of 2 commonly prescribed DOACs, apixaban and rivaroxaban, in RCTs is lacking. This study allows health professionals and decision-makers to review the body of published evidence on apixaban compared with rivaroxaban by drawing conclusions from NMAs of RCTs and RWE studies. This examines how efficacy and safety from RCTs translates into effectiveness and safety among patients with NVAF in real-world clinical practice. The majority of studies in this comprehensive evidence review suggest that apixaban demonstrated comparable effectiveness and a lower risk of major bleeding events compared to rivaroxaban in patients with NVAF.
Supplemental Material
BOE_Manuscript_CATH_Supplementary_20191004 - A Systematic Review of Network Meta-Analyses and Real-World Evidence Comparing Apixaban and Rivaroxaban in Nonvalvular Atrial Fibrillation
BOE_Manuscript_CATH_Supplementary_20191004 for A Systematic Review of Network Meta-Analyses and Real-World Evidence Comparing Apixaban and Rivaroxaban in Nonvalvular Atrial Fibrillation by Nathan R. Hill, Belinda Sandler, Evelien Bergrath, Dušan Milenković, Ajibade O. Ashaye, Usman Farooqui and Alexander T. Cohen in Clinical and Applied Thrombosis/Hemostasis
Supplemental Material
Supplemental Material, Figure_S1 - A Systematic Review of Network Meta-Analyses and Real-World Evidence Comparing Apixaban and Rivaroxaban in Nonvalvular Atrial Fibrillation
Supplemental Material, Figure_S1 for A Systematic Review of Network Meta-Analyses and Real-World Evidence Comparing Apixaban and Rivaroxaban in Nonvalvular Atrial Fibrillation by Nathan R. Hill, Belinda Sandler, Evelien Bergrath, Dušan Milenković, Ajibade O. Ashaye, Usman Farooqui and Alexander T. Cohen in Clinical and Applied Thrombosis/Hemostasis
Supplemental Material
Supplemental Material, Figure_S2 - A Systematic Review of Network Meta-Analyses and Real-World Evidence Comparing Apixaban and Rivaroxaban in Nonvalvular Atrial Fibrillation
Supplemental Material, Figure_S2 for A Systematic Review of Network Meta-Analyses and Real-World Evidence Comparing Apixaban and Rivaroxaban in Nonvalvular Atrial Fibrillation by Nathan R. Hill, Belinda Sandler, Evelien Bergrath, Dušan Milenković, Ajibade O. Ashaye, Usman Farooqui and Alexander T. Cohen in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Authors’ Note
U.F., N.R.H., A.T.C., A.O.A. designed the research study. D.M., E.B., A.O.A., and N.R.H. performed the literature review and analyzed the data. All authors contributed to the writing and editing of the paper and approved the article for submission. Medical writing and editorial support were provided by Dr Angharad Morgan of Health Economics and Outcomes Research Ltd. All data are available in the paper or in the Additional files, or already published online by other researchers.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: N. R. Hill, B. Sandler, and U. Farooqui are employees of Bristol-Myers Squibb Company. E. Bergrath and D. Milenković were employed by Evidera Inc, which provides consulting and research services to pharmaceutical, medical device, and related organizations, during the study and remain employed by Evidera Inc. A.O. Ashaye was employed by Evidera Inc during the study. In their salaried positions, Evidera employees work with a variety of companies and organizations and are precluded from receiving payment or honoraria directly for services rendered. D. Milenković also reports personal fees from UCL CRUK Cancer Trials Centre, during and outside the submitted work. A.T. Cohen reports grants and personal fees from Bristol-Myers Squibb Company and Pfizer Inc during the conduct of the study. Outside the submitted work, he reports personal fees from Boehringer Ingelheim, Johnson & Johnson, Portola, Sanofi, XO1, Janssen, and ONO Pharmaceuticals. He further reports grants and personal fees from Bristol-Myers Squibb Company, Daiichi-Sankyo Europe, Pfizer, Inc, and Bayer AG.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Bristol-Myers Squibb Company and Pfizer Inc.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
